Abstract
With the exception of classic functional adenomas in dogs and horses, pituitary lesions are infrequently described in the veterinary literature. Approximately 10% of pituitary glands from asymptomatic humans contain abnormalities, but the equivalent proportion in small animals is unknown. Pituitary glands from 136 dogs and 65 cats collected during routine necropsies were examined to determine the prevalence of pituitary lesions and their histopathological diagnosis. Lesions were characterized in sections stained with hematoxylin and eosin, periodic acid-Schiff (PAS), Gordon and Sweet’s and reticulin stains, and immunohistochemistry for adrenocorticotropic hormone (ACTH), growth hormone, melanocyte stimulating hormone–α, and prolactin. Pituitary abnormalities were identified in 36 of 136 (26.4%) dogs and 10 of 65 (15.3%) cats. Cystic changes were the most common lesion, occurring in 18 (13.2%) dogs and 8 (12.3%) cats. Pituitary neoplasia was detected in 14.1% (12/85) of middle-aged and old dogs; 1 (1.5%) cat had pituitary nodular hyperplasia. PAS and reticulin stains helped differentiate ACTH-immunoreactive adenomas from hyperplastic nodules: adenomas contained PAS-positive intracytoplasmic granules and loss of the normal reticulin network. One dog had a pituitary carcinoma with infiltration into the thalamus. Other pituitary abnormalities included secondary metastases (2 dogs) and hypophysitis (4 dogs, 1 cat). In most cases, the lesion appeared to be subclinical and could be considered incidental, whereas clinical manifestations were apparent in only 4 dogs (2.9%) and none of the cats with pituitary lesions. Pituitary abnormalities are common in dogs and cats, and their clinical relevance requires further investigation.
Keywords
A wide range of abnormalities can occur within the pituitary gland and may lead to various endocrinologic or neurologic signs. Given the increasing availability of advanced imaging in the diagnostic approach of both endocrine and neurologic disease, a better understanding of the range of abnormalities encountered in the pituitary gland is desirable, especially with the progression in sampling or surgical treatment of the pituitary gland through transsphenoidal hypophysectomy in dogs and cats. 22,34,41 At present, there are limited data in the veterinary literature on acquired pituitary abnormalities, and the data that are available are complicated by lack of consensus over terminology and classification.
In dogs, the incidence of Cushing’s disease (pituitary-dependent hyperadrenocorticism) is estimated at 1 to 2 cases per 1000 dogs per year, 34 and pituitary adenomas account for 25% of all intracranial neoplasms in middle-age or geriatric dogs. 18,43 In cats, pituitary tumors are considered uncommon, accounting for 9.3% of all intracranial tumors, 44 often related to hypersomatotropism leading to insulin-resistant diabetes mellitus. 17 No data currently exist on the presence of silent pituitary lesions (so-called incidentalomas in digital imaging) in dogs or cats to contrast with that in humans. This is important given the increasing use of advanced neuroimaging in clinical practice.
Pituitary lesions can be subclinical or cause endocrine dysfunction (hypofunction or hyperfunction) or neurologic signs due to local effects of an expanding mass. 37,46 In humans, approximately 14% of pituitary glands examined at autopsy contain small pituitary adenomas, mainly prolactin (PRL) producing, with the vast majority being clinically silent. 7,19 Other pathologic entities such as pituitary cysts and metastatic tumors are thought to have a similar prevalence in humans. 25,26
The aim of the study was to determine the prevalence of pituitary abnormalities in a general population of dogs and cats and to compare it to that seen in humans. In addition, a further aim was to characterize the histopathologic features of these abnormalities to help with understanding the range of pituitary lesions encountered in dogs and cats.
Materials and Methods
Cases and Histopathology
A total of 201 pituitary glands were randomly sampled from routine necropsies from 136 dogs and 65 cats at the Veterinary Pathology Service at the Veterinary School (SVMS) of the University of Nottingham between 2015 and 2016. This was a convenience sampling study that resulted in a population of dogs with a median age of 6 years (interquartile range: 2–8.5 years; range: 4 days to 14 years) with age distribution as follows: 51 young (<4 years), 43 middle-aged (4 to <8 years) and 42 old-aged (≥8 years) dogs. Cats had a median age of 9 years (interquartile range: 4–14 years; range: 1 month to 20 years), with age distribution as follows: 14 young (<4 years); 12 middle-aged (4 to <8 years), and 39 old-aged (≥8 years). The proportion of males was 57% of the dogs and 55% of the cats. The neuter status for both sexes was 42% and 72% of the dogs and cats, respectively. The most common canine breeds represented were Greyhounds (10%), Labrador Retriever (10%), and Staffordshire Terrier (7%), and 69% of the cats were domestic short/long hair. Cases were submitted as part of routine diagnostic investigations or were donated for teaching purposes. Individual age, sex, breed, clinical history, and final diagnosis were recorded from each animal (Supplemental Table S1).
Pituitary glands were immediately fixed in 10% neutral-buffered formalin before performing a sagittal section of each gland, and both halves were embedded in paraffin wax. Tissue sections (5 μm) were stained with hematoxylin and eosin (HE) for routine microscopic analysis. Measurement of the size of the lesions was performed on the histologic section with the widest lesion diameter (3–9 sections per lesion).
All major endocrine glands were examined in all postmortem cases. These were sampled for histopathologic evaluation only if macroscopic changes were identified or suspected.
Special Stains and Immunohistochemistry
Tissue sections of 13 (12 dogs and 1 cat) pituitary glands presenting proliferative changes and 1 normal control pituitary gland were stained with periodic acid-Schiff (PAS) and Gordon and Sweet’s reticulin stain using appropriate control tissues for validation of the stains. The same samples were immunolabeled for adrenocorticotropic hormone (ACTH). Three canine samples with neoplastic changes that were entire or partially nonimmunoreactive for ACTH were further immunolabeled for growth hormone (GH), α–melanocyte-stimulating hormone (α-MSH), and PRL. Immunohistochemistry was performed using primary antibodies (ACTH, 1:100 monoclonal antibody against ACTH, clone 2F6, Department of Infectious Diseases and Immunology, Faculty of Veterinary Medicine, Utrecht University, the Netherlands; α-MSH, 1:400 polyclonal rabbit antibodies to synthetic α-MSH, MZ111, Biomol International, Exeter, UK; GH, 1:5000 rabbit anti-human antibody STH, N1561, Dako, Glostrop, Denmark; and PRL, noncommercial polyclonal antibody against PRL raised in rabbits, courtesy of Dr B. P. Meij, Utrecht, the Netherlands) in an indirect immunoperoxidase staining procedure, using the avidin-biotin–based technique (Vectastain ABC kit, Vector Laboratories, Burlingame, CA) on 5-μm-thick pituitary sections following previously published protocols. 3,35
Three cases with hypophysitis were additionally stained with Giemsa and Gram to detect potential infectious agents (such as protozoa and bacteria). In 1 of these dogs, immunohistochemistry (IHC) was conducted in a commercial lab using antibodies against canine CD3 and CD20 to characterize the lymphocytic population.
Results
Pituitary lesions were identified in 36 of 136 (26.4%) dogs and 10 of 65 (15.3%) cats. Eighteen (18/136, 13.2%) dogs and 8 (8/65, 12.3%) cats had pituitary cystic changes; 12 (12/136, 8.8%) dogs and 1 (1/65, 1.5%) cat had primary pituitary neoplasms, 2 (2/136, 1.4%) dogs had metastases of an extrasellar carcinoma (salivary adenocarcinoma and nasal adenocarcinoma), and 4 (4/136, 2.9%) dogs and 1 (1.5%) cat presented inflammatory lesions (Table 1). The remaining pituitary samples from 100 dogs and 55 cats were macroscopically and histopathologically unremarkable. The distribution of pituitary lesions in dogs based on age is shown in Fig. 1. All cats presenting pituitary cysts were older than 8 years.
Pathological Findings in Pituitary Glands of 136 Dogs and 65 Cats.
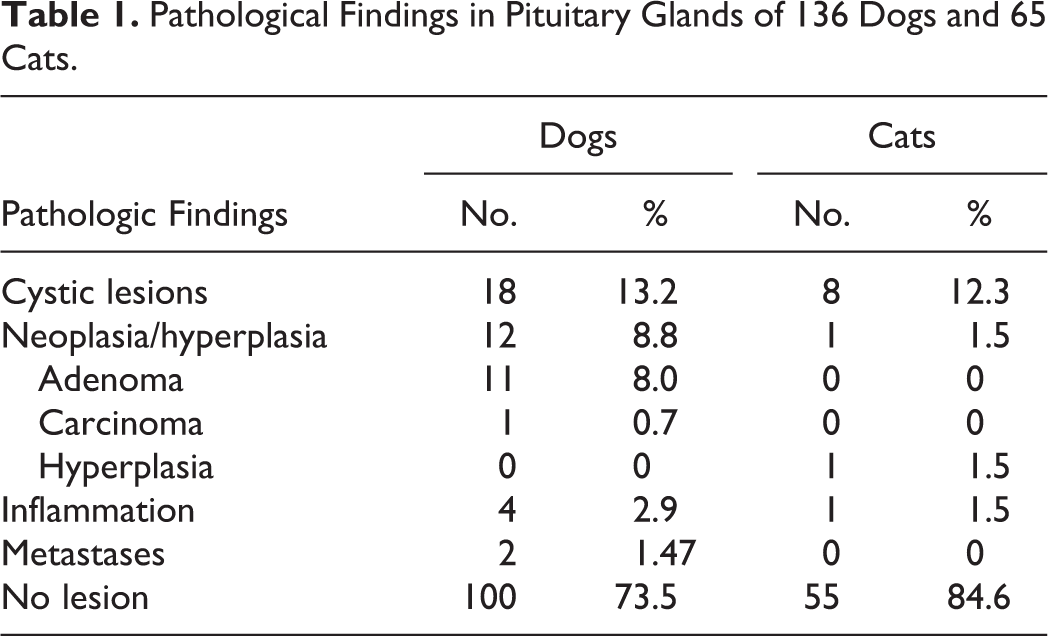
Distribution of pituitary lesions (neoplasia, cysts/cyst-like lesions, inflammation) in dogs of various ages. Only dogs with lesions are included in this figure. Inflamm, inflammation.
Neoplastic and Hyperplastic Lesions
Affected dogs were of various ages (range, 4–15 years; median, 10 years), with a prevalence in middle-aged and old dogs of 7% (3/43) and 21% (9/42), respectively; these were of both sexes and various breeds (3 Greyhounds, 1 Whippet, 1 Lhasa Apso, 1 Poodle, 1 Cocker Spaniel, 1 German Shepherd, 1 Retriever Labrador, 1 Bull Mastiff crossbreed, 1 Dachshund crossbreed, and 1 Leonberger). Only 1 cat, a 23-year-old domestic short hair, had primary pituitary hyperplasia.
Pituitary Adenoma
Of the 11 dogs with pituitary adenoma, only 1 (a Dachshund crossbreed) had been diagnosed with hyperadrenocorticism ante mortem and had typical associated skin and hepatic lesions. Adrenal gland tissue was available from this dog and 3 others of the 11 dogs with pituitary adenoma, and all 4 histologically presented moderate bilateral adrenocortical hyperplasia. The other 7 (63.6%) dogs had no lesions compatible with hyperadrenocorticism. In a further 33 dogs with no pituitary proliferative change, 10 (30%) dogs also had bilateral adrenal cortical hyperplasia.
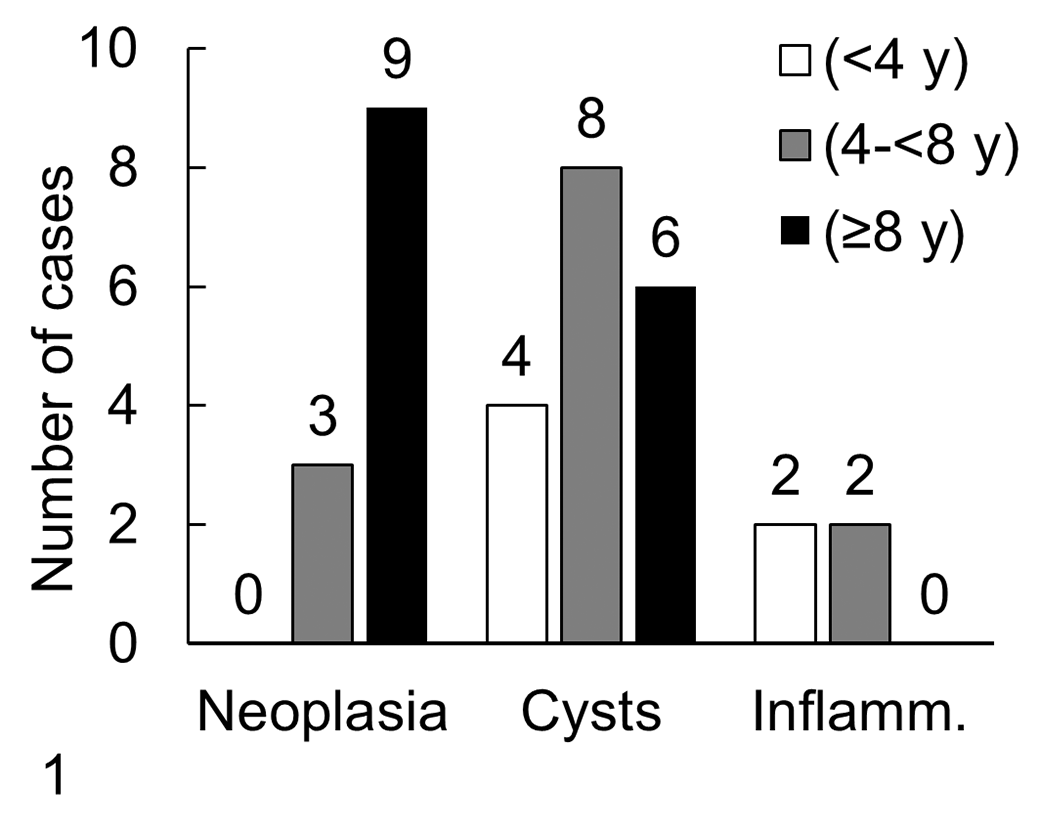
Most adenomas were greater than 2 mm in size (7/11, 63%), with a mean size of 3.2 mm, but only 2 (18%) pituitary adenomas were detected grossly as bulging, soft, nonencapsulated, pale nodules of 7.0 mm and 5.4 mm in diameter, which caused mild pituitary enlargement and loss of symmetry of the dorsal aspect of the gland with no invasion of the adjacent structures (Fig. 2). The adenoma in the dog with hyperadrenocorticism was 1.8 mm in diameter. Microscopically, all adenomas were located in the pars distalis and consisted of variably demarcated, densely cellular nodules of neoplastic cells (Fig. 2a), arranged in sheets (diffuse pattern) with fine supportive stroma and loss of the normal stromal network pattern as shown by the reticulin stain (Fig. 2b). Neoplastic cells had little pleomorphism with small- to medium-sized nuclei, coarse chromatin pattern, and very characteristic abundant, uniformly light eosinophilic cytoplasm with HE stain (Fig. 2a), which contained PAS-positive granules (Fig. 2c), and with consistent ACTH immunoreactivity in those cases examined by IHC (Fig. 2d). No mitotic figures or areas of necrosis were observed in any case. In addition to the described neoplastic cells, the 2 largest corticotroph adenomas contained cells that did not contain PAS-positive cytoplasmic granules; were not immunoreactive to ACTH, MSH, GH, or PRL; had round to oval nuclei with coarse chromatin; and were supported by fine fibrovascular stroma.
Pituitary adenoma, dog. Focal nodule in the pars distalis of neoplastic cells arranged in sheets with fine supportive stroma (a, hematoxylin and eosin [HE]), with loss of normal reticulin stromal pattern (b, reticulin stain), abundant, violet, uniform, periodic acid-Schiff (PAS)–positive cytoplasm (c), and strong adrenocorticotropic hormone (ACTH) immunoreactivity (d).
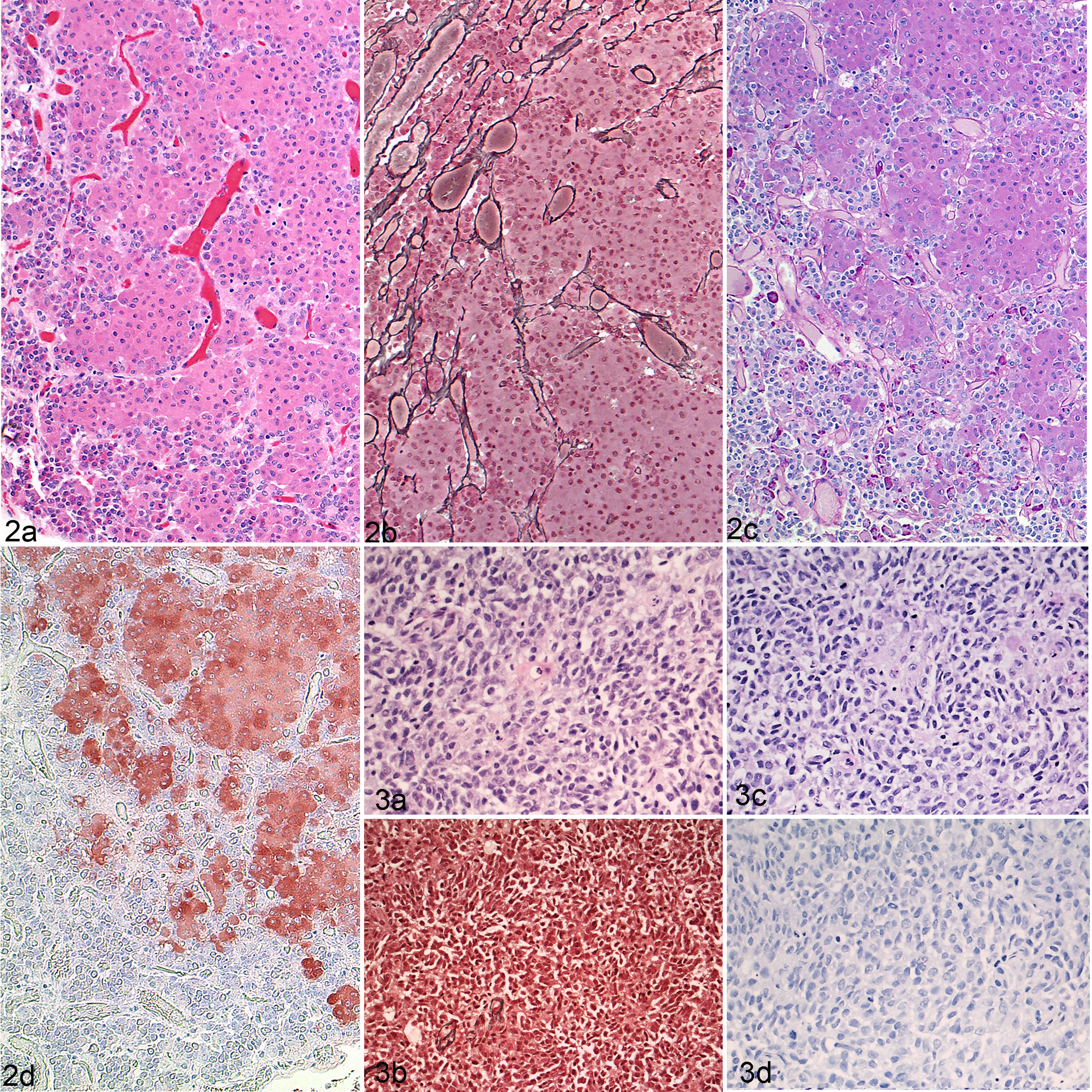
Within the pars distalis and adjacent to 4 of the pituitary corticotroph adenomas, there were small numbers of secretory cells with margination of intracytoplasmic PAS-positive and ACTH immunoreactive granules (Fig. 4a), leaving a clear paranuclear halo. These cytoskeletal changes were immunolabeled with pan-cytokeratin (Fig. 4b).
Pituitary adenoma, dog. (a) Clusters of nonneoplastic corticotrophs have intensely periodic acid-Schiff–positive granules in the periphery of the cytoplasm with a clear perinuclear area. (b) Some of these cells have perinuclear pan-cytokeratin immunoreactivity.
Pituitary Carcinoma
A 7-year-old male Greyhound had a pituitary carcinoma manifesting neurological signs, neck pain, and low head carriage without signs of an endocrinopathy. On postmortem examination, a 2× 3× 2cm, nonencapsulated mass arose from the pars distalis and markedly invaded dorsally and discontinuously into the hypothalamus and thalamic region. Histologically, polygonal cells with distinct cell borders and round nuclei were arranged in tightly packed cords, islets, or nests (Fig. 3a, b); separated by moderate amounts of fibrous stroma (Suppl. Fig. S1); and infiltrated the moderately gliotic neurohypophysis and diencephalon (Suppl. Fig. S1). Cellular pleomorphism was moderate, and there were 4 to 5 mitotic figures per 400× field with multiple necrotic foci, which often had a central core of dystrophic calcification. PAS reaction (Fig. 3c) and ACTH, MSH, GH, and PRL-IHC were negative (Fig. 3d).
Pituitary Hyperplasia
One cat presented focal nodular pituitary hyperplasia within the pars distalis, without any previous manifestation of clinical signs or presence of systemic lesions suggestive of hyperadrenocorticism or hyperthyroidism. Histologically, there was a well-circumscribed, 1.4-mm-diameter, focal, nonencapsulated, nodular proliferation of secretory cells, which caused enlargement of the acini, mildly compressing the adjacent pars distalis, with minimal to no visible changes in the normal pituitary stromal network in the reticulin stain (Suppl. Fig. S2). Hyperplastic cells revealed a chromophobic cytoplasm, often vacuolated and poorly stained with PAS, with negative or very weak ACTH immunolabeling.
Secondary Metastases
Two of the 136 (1.5%) unrelated Border Collie dogs aged 10 and 9 years had intracranial metastases from a nasal adenocarcinoma and salivary adenocarcinoma, respectively, which also affected the pituitary gland, the former destroying the gland entirely and the latter infiltrating and replacing approximately 50% of the pars distalis. Clinically, both of these dogs had prominent neurologic signs with no signs of an associated endocrinopathy.
Pituitary Cysts and Cyst-like Structures
Eighteen (13.2%) dogs from various ages and breeds and 8 (12.3%) old cats (mostly Domestic Shorthair), with a median age of 7 years (range, 5–9 years) and 11 years (range, 8.5–14 years), respectively, had pituitary cystic lesions without any related clinical signs reported in the clinical history (endocrine investigations had not been performed in these cases). Eight of the 18 (44%) dogs were brachycephalic breeds.
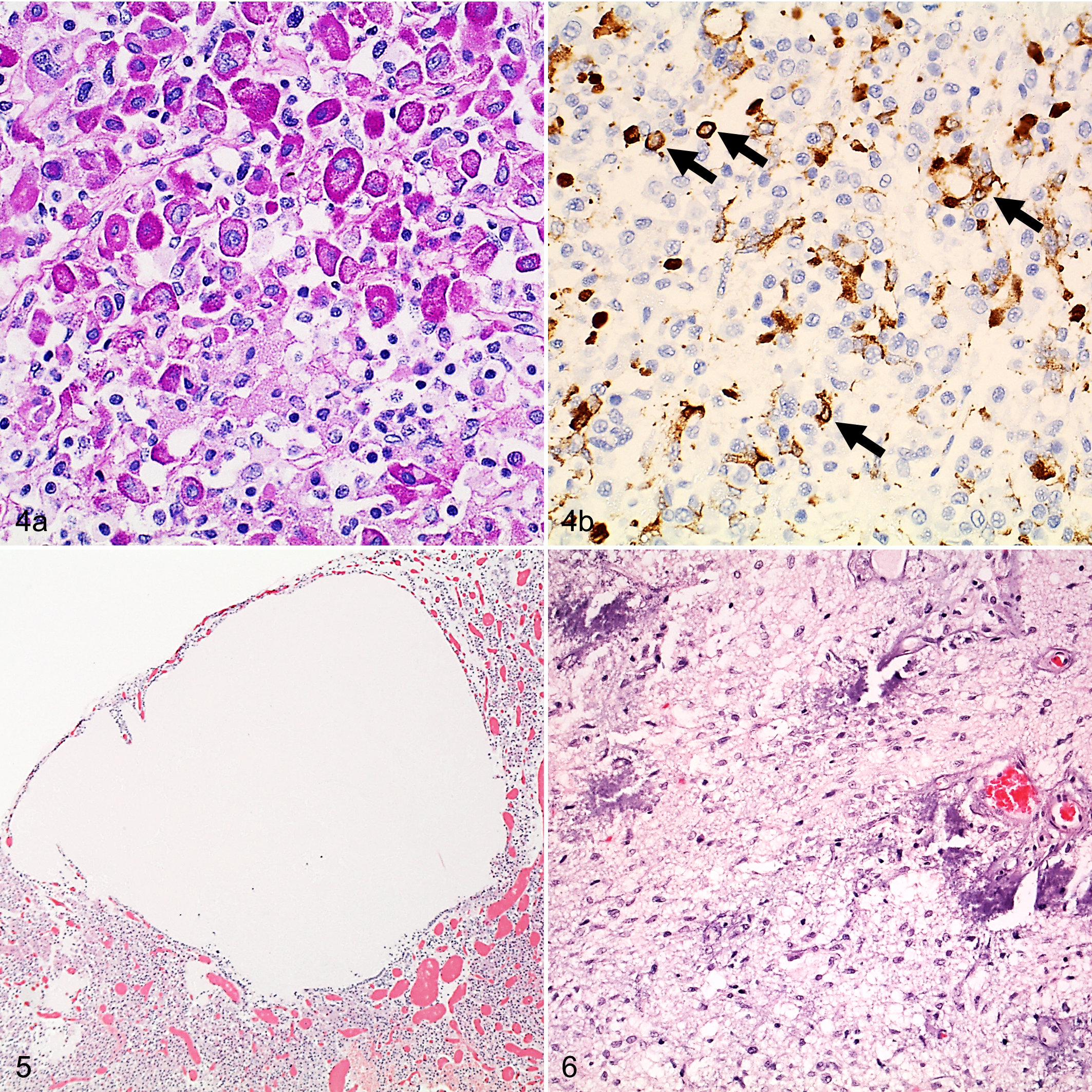
Cysts were variable in size up to 2.7 mm, with a mean diameter of 1.6 mm in dogs, and up to 0.8 mm in cats, with a mean diameter of 0.4 mm. Cysts and cyst-like structures contained PAS-positive, pale mucinous fluid and were morphologically classified based on the lining epithelium and location following a previously proposed system 42 (Suppl. Fig. S3–S6): cysts with epithelial lining were present in 10 dogs and 4 cats, cystic areas without an epithelial lining were present in 7 dogs and no cat (Fig. 5), and enlarged colloid follicles of the pars intermedia were present in 1 dog and 4 cats. The epithelial lining was a cuboidal, ciliated, simple epithelium in 3 dogs and a nonciliated, flat to cuboidal simple epithelium in the other cysts.
Inflammatory Lesions
Two dogs, a male 6-year-old Scottish Terrier and a male 6-year old Lurcher, had hypophysitis accompanied by moderate bilateral cortical atrophy of the adrenal glands and clinically presenting with hypoadrenocorticism. Both cases had moderate to severe, lymphoplasmacytic inflammatory cell infiltration with loss of pituitary cells (Suppl. Fig. S7). The observed lymphocytic infiltrate was predominantly T-cell rich with low numbers of B cells in the dog in which IHC was performed. No evidence of microorganisms or foci of inflammation were detected in the brain or other tissues.
Secondary hypophysitis was observed in 2 other dogs: one presenting nonsuppurative encephalitis caused by canine distemper virus (extending into the neurohypophysis) and the other as part of an ante mortem–confirmed systemic immune-mediated disease comprising immune-mediated polyarthritis, vasculitis, and anemia. One cat presenting with feline infectious peritonitis had perivascular neutrophilic and lymphoplasmacytic infiltrate within the neurohypophysis.
Other Findings
One (0.7%) 1-year-old Vizsla dog and 8 (15.4%) cats (age range, 2–20 years; median age, 10.5 years) had multifocal areas of mineralization in otherwise unremarkable pituitary glands. The dog presented with chronic renal failure with metastatic calcification affecting various organs, including multifocal, poorly defined mineralization affecting the neurohypophysis (Fig. 6). The cats did not have a clinical history or lesion compatible with hypercalcemia and revealed very few (1 to 3), scattered, up to 200-μm-diameter, round, mineral-laminated concretions within the pituitary capsule, in the neurohypophysis, or within acini in the adenohypophysis.
The adenohypophysis from all 23 puppies and young dogs (up to 9 months old) and a lactating bitch had a uniform basophilic cell appearance with cytoplasm devoid of granules, and large, vesicular nuclei compared with the adult and elderly dogs (Suppl. Fig. S8 and S9). This difference was not observed in 5 kittens and young cats (less than 9 months of age).
Discussion
Within our study, pituitary abnormalities were frequent in both dogs (26.4%) and cats (15.3%); but only 4 dogs (4/136, 2.9%) were diagnosed with a clinical manifestation of pituitary disease; therefore, the presence of a pituitary lesion in most of our cases was unexpected, very similar to the reports in humans with incidentally observed pituitary lesions. The prevalences in our study population might be overestimated for the general dog and cat population, considering that most of the animals were sick animals, with the exception of 8 dogs that were euthanized due to a severe aggressive behavior.
Neoplastic and Hyperplastic Lesions
Pituitary neoplasms were more common in dogs than in cats in the studied population, particularly dogs older than 6 years, in agreement with the literature. 36 In our study, a total of 4 Greyhounds and Greyhound cross-breeds presented pituitary neoplasia, which might suggest a higher susceptibility of this breed to this type of neoplasia, but a further study with a larger dog population is needed to confirm this.
The most frequent neoplasm in our canine population was the ACTH-immunoreactive pituitary corticotroph adenoma, with a prevalence of 7% in middle-aged and 21% in old dogs. Clinical signs consistent with pituitary disease were not reported ante mortem in 10 of 11 (90.9%) of the dogs presenting with pituitary adenoma, which may be explained by a lack of hormonally active ACTH secretion in otherwise immunohistochemically immunoreactive corticotroph adenomas. In humans, they are thought to arise from corticotrophs that fail to process the ACTH precursor, pro-opiomelanocortin, into the biologically active 1–39 ACTH. 2
The presence of diffuse, moderate adrenocortical hyperplasia that might suggest an increased production of ACTH was present in all dogs with pituitary adenomas where adrenal gland tissue was available. However, adrenal cortical hyperplasia was also observed in 30% of dogs without pituitary adenomas or hyperplasia. Thus, without measuring serum cortisol or ACTH and performing relevant dynamic functional tests, an association between these lesions must be considered with caution in dogs. Nodular adrenocortical hyperplasia is relatively common in older dogs without a detectable pituitary lesion and often attributed to an increased hypothalamic catabolism of dopamine that disrupts negative feedback control in the hypothalamic-pituitary-adrenal axis. 24,28
Only 1 dog had been diagnosed ante mortem with hyperadrenocorticism and had typical lesions of pituitary-dependent hyperadrenocorticism, with a very small (1.8-mm-diameter) ACTH-immunoreactive pituitary adenoma that did not cause any grossly observable enlargement of the pituitary gland. It has been reported that pituitary adenomas less than 1 cm are more likely to be functional than larger ones. 14
In our opinion, the significance of the rather arbitrary term
Every pituitary adenoma had well-defined, strong, uniform, intracytoplasmic ACTH immunoreactivity, with almost identical histological morphology: abundant, granular, eosinophilic cytoplasm that stained uniformly violet with PAS. Human literature reports that corticotroph adenomas have PAS-positive intracytoplasmic granules, attributed to glycolipids, glycoproteins, or mucoproteins in the secretory granules or in their membranes. 31 In diagnostic pathology settings in which IHC is not available, a strong cytoplasmic granular PAS positivity in a proliferative area with a loss of reticulin fibers can support a diagnosis of ACTH-corticotroph adenoma, bearing in mind that cells producing thyroid-stimulating hormone (TSH) and follicle-stimulating hormone (FSH)/luteinizing hormone (LH) also contain PAS-positive granules (but these latter neoplasms have not been reported in dogs). 14 MSH-secreting adenomas share similar microscopic characteristics with ACTH-secreting adenomas but have strong immunoreactivity for α-MSH and its precursor pro-opiomelanocortin, and they are weakly ACTH immunoreactive. 11,21
The ACTH nonimmunoreactive neoplastic cell population with scant cytoplasm observed within 2 corticotroph adenomas might have been corticotroph cells that have already released their secretory granules. However, a preexisting, nonneoplastic cell population (suggesting infiltrative growth) or a second neoplastic population of FSH/LH- or TSH-secreting cells is less likely but cannot be ruled out. 47 Unfortunately, we were unable to identify a source of available antibodies for these additional hormones in dogs and cats.
In humans with functional ACTH-secreting pituitary proliferations, nonneoplastic corticotrophs may have ACTH-immunoreactive secretory granules pushed to the periphery of the cytoplasm by homogeneous, hyaline material consisting of cytokeratin microfilaments arranged concentrically around the nucleus. This morphological pattern is known as Crooke’s hyaline degeneration, and these cells are called Crooke’s cells. It is a characteristic morphological finding in the functional version of ACTH-immunoreactive neoplasms in humans, 5,23,30 which to our knowledge has not been previously described in small animals. The few nonneoplastic corticotroph cells with strong, PAS-positive, peripheral, intracytoplasmic staining adjacent to 4 canine corticotroph adenomas in our study population morphologically resemble Crooke’s cells with peripheral dispersion of PAS-positive granules in the cytoplasm and a strong cytokeratin immunolabeling. However, the peripheral hyaline bands were not easily identifiable on HE, although cytoplasmic accumulation of intermediate filaments could be demonstrated on cytokeratin IHC (Fig. 4b).
The Gordon-Sweet reticulin stain was useful to differentiate adenomas from pituitary hyperplasia: hyperplastic lesions retained the acinar pattern of the preexisting reticulin network, whereas in pituitary adenomas, the reticulin network is minimal. 14,23 However, there appears to be a continuous spectrum of proliferative lesions between focal (nodular) hyperplasia and adenoma, with a similar prognosis and treatment, 36 so distinction between these entities may not be clinically important. The normal pituitary gland of cats is less homogeneous than in dogs and exhibits great variation in cell populations, which apparently correlates with certain phases of the reproductive cycle. 4 Thus, the reticulin stain was especially useful to avoid overinterpretation of proliferative lesions in cats.
Pituitary carcinoma was diagnosed in a dog in this study based on the observed necrosis, mitoses, invasion, and widespread, destructive, discontinuous growth into the brain.
37
However, we are aware of the discrepancy with other references in which metastatic behavior of the tumor is needed to diagnose a pituitary carcinoma
14,24,32,36
and the term
In the 2 cases of pituitary metastases of a salivary adenocarcinoma and nasal adenocarcinoma of the current study, the observed neurologic signs were mainly attributed to other coexisting intracranial metastases from the same primary neoplasm. Ante mortem clinical signs of pituitary dysfunction were not detected, similar to what is reported in humans, where only 7% of the metastases to the pituitary are symptomatic, with diabetes insipidus, visual field defects, headache/pain, and ophthalmoplegia being the most commonly reported symptoms. 8 Distant metastases from extrasellar carcinomas to the pituitary gland, such as lymphoma, melanoma, transmissible venereal tumor, and adenocarcinomas, have been previously reported in dogs. 36
Pituitary Cysts
Pituitary cysts or cyst-like lesions were the most common finding, affecting dogs and cats similarly (12%–13%), and they appear to be more common in middle-age to old dogs and elderly cats. The cysts with epithelial lining, sometimes ciliated, are thought to be remnants of the distal craniopharyngeal duct inside Rathke’s pouch, while the cystic areas without epithelial lining appear to arise from degeneration of pituitary tissue or blockage of secretion. 42 Enlarged colloid-containing follicles of the pars intermedia were more often found in cats. All pituitary cysts found in this study were considered subclinical, but occasionally cysts may become large enough to exert pressure on adjacent structures or even disrupt pituitary function. 36
In the current study, brachycephalic breeds appeared to have a higher prevalence of pituitary cysts than non-brachiocephalic breeds as previously identified. However, the prevalence we observed in dogs in general is markedly lower compared with a previous survey in which cystic craniopharyngeal duct remnants were detected in approximately 50% of the dogs. 36,42 This previous survey limited the studied population to 6 breeds (Dachshunds, Terriers, Schnauzers, Boxers, French Bull Dogs, and German Shepherd; no data are available about the proportions of each breed), whereas we included dogs from 58 breeds. If in the previous study the proportion of brachycephalic dogs was higher, this might explain the differences in prevalence.
Inflammatory Lesions
Two dogs and 1 cat presented hypophysitis secondary to systemic disease: canine distemper virus infection, clinically confirmed autoimmune disease with polyarthritis and vasculitis, and feline infectious peritonitis. The other 2 canine cases of hypophysitis appeared to be primary, and we hypothesized an immune-mediated origin, because no other focus of inflammation or infectious agents was detected in these cases. Only a few cases of autoimmune lymphocytic adenohypophysitis have been reported in dogs, 1,20,22,33,38,45 usually related to panhypopituitarism and secondary hypoadrenocorticism with adrenocortical atrophy, similar to the cases identified here.
Other Findings
Multifocal mineralization of the pituitary gland seems to be more common in cats (15.4%) than in dogs (0.7%), and in all 8 cats, it was considered to be an incidental finding. 7 In the affected dog, the mineralization was considered to represent metastatic calcification due to chronic renal failure. To our knowledge, calcification in the pituitary gland has only been described previously together with neoplastic lesions or cysts, 15 similar to the dystrophic calcification reported here in the necrotic areas of the pituitary carcinoma.
The uniform, more basophilic appearance of the pituitary gland in puppies, young dogs, and a lactating bitch was interpreted as diffuse, physiologic hyperplasia in highly active hormonal situations such as growth or lactation. Additional immunohistochemical evaluation of the cell populations in the pituitary glands of these cases would be needed to substantiate this interpretation. In humans, there is a progressive change in the appearance of the pituitary gland during the first years of life, according to the ongoing change in hormonal secretion. 6 In humans, aging changes such as fibrosis, deposition of amyloid, iron pigment, and a decrease in acidophilic and chromophobic cells have been reported, 12,39 but none of these changes were observed in geriatric dogs or cats we examined.
To summarize, given the presence of pathological changes in 26% of the dogs and 15% of the cats in our study, pituitary abnormalities should be considered common in both species. This is in keeping with that described in humans. Most of these lesions were not identifiable macroscopically, so postmortem sampling and histopathological examination of the pituitary gland is needed. Within our population, we identified a prevalence of adenomatous pituitary lesions in middle-aged to old dogs of 14%, similar to the 10% to 15% reported in humans. 9,25 The prevalence in cats appears to be much lower. Given that these lesions were incidentally observed in this cohort of dogs and cats, further investigations are needed to establish which are subclinical and which cause undiagnosed functional or clinical effects.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818784162 - Pathological Findings in the Pituitary Glands of Dogs and Cats
Supplemental Material, DS1_VET_10.1177_0300985818784162 for Pathological Findings in the Pituitary Glands of Dogs and Cats by Laura Polledo, Guy C. M. Grinwis, Peter Graham, Mark Dunning, and Kerstin Baiker in Veterinary Pathology
Footnotes
Acknowledgements
We thank the technical diagnostic team of the University of Utrecht, the pathology team of SVMS of the University of Nottingham, and Alan Lasslett for excellent technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
