Abstract
Subsequent to a previous study of border disease virus (BDV) horizontal transmission from a persistently BDV-infected calf to 6 seronegative pregnant heifers, the heifers were slaughtered 60 days after exposure to the infected calf, and their fetuses and placentas were examined. Immunohistochemical examination of fetal organs and placenta showed positive labeling of moderate intensity for pestivirus antigen in 3 of 6 heifers. BDV infection in these 3 animals was confirmed by the detection of BDV RNA in different organs using reverse transcription quantitative polymerase chain reaction. In the placenta, the positive cells were visualized mostly on the fetal side. In those 3 heifers that harbored an infected fetus, the placental tissue in the placentome region showed a moderate to severe mononuclear and fibrosing placentitis and, in severe cases, necrotic areas. The inflammatory population was composed predominantly of T and B cells, a substantial number of macrophages, and, to a lesser extent, plasma cells. This is a novel report of placentitis in persistently BDV-infected fetuses from pregnant heifers that became acutely infected by cohousing with a calf persistently infected with BDV, which extends previous reports on bovine viral diarrhea virus–infected and BDV-infected cattle and sheep, respectively.
Keywords
Border disease virus (BDV), along with bovine viral diarrhea virus (BVDV), belongs to the genus Pestivirus and induces important diseases in small ruminants and bovids. 28,37 Border disease was first described in sheep in 1959, with clinical signs such as tremor and hairy fleece. After vertical transmission, animals can be persistently infected (PI) as described for BVDV in cattle. 16,24,28,37,40 Pestiviruses can infect animals transiently with no or only mild clinical signs such as diarrhea and coughing. 10,18,23 Depending on the time point of infection of the pregnant animal, the fetus is resorbed, aborted, or persistently infected. 37,40
BDV and BVDV are not strictly species specific, 27,30,39 and thus calves can be infected with BDV through contact with PI small ruminants, and conversely, small ruminants can be infected by virus transmission from persistently BVDV-infected animals with possible further spread among small ruminants, as shown in goats. 2,7,8,13,25,30 BDV transmission from cattle to cattle has been assumed after evidence for seroconversion of heifers copastured with a bull persistently infected with BDV in New Zealand, 25 and this has recently been confirmed by cohousing a persistently BDV-infected calf with pregnant heifers in early pregnancy. 7
In PI animals, a wide range of organs become infected, which can be visualized by immunohistochemistry. 9,22,24,41 Undoubtedly, placentation is important in understanding the pathogenesis of a variety of different diseases. The fetal compartment of the fetomaternal unit in the placenta consists of trophoblast cells covered by endometrial epithelial cells. 36,43 It has long been known that infections with pestivirus cause lesions in fetuses such as arthrogryposis, hydranencephaly, hypotrichosis, or hypothyroidism. In bovine fetuses from the fifth month onward, malformations can be seen or normal immunocompetent calves can be born. Although transplacental transmission of most viral agents occurs with no gross tissue destruction, 9,25,36,39,40 minor lesions like hemorrhages and fibrinous ulcerative placentitis together with vasculitis and endarteritis have been reported in cases of BVDV infections. 22,29,30,35 Nevertheless, severe placental lesions have not been associated with pestivirus infection in cattle. The current study complements previous research on BDV transmission 7 by reporting a novel description of severe placentitis from heifers transiently infected with BDV due to cohousing with a persistently BDV-infected calf during early pregnancy.
Materials and Methods
Animals and Samples
To perform this study, samples from 6 heifers used in a previous study were used. 7 Briefly, after an acclimation phase, heifers were inseminated using sperm from a pestivirus-free bull. Afterwards, the pregnant heifers remained housed under quarantine-regulated conditions in an isolation barn together with the PI-BDV calf (BDV strain “BDSwiss R8540/11_ch149”), 7,32 from the 50th to 110th day of pregnancy. Thereafter, the heifers were slaughtered and the uteri as well as fetal membranes with the corresponding fetuses were collected for pathological-anatomical examinations. As controls, placentas and fetal organs from 2 pestivirus-free heifers, as detected by immunohistochemistry, at 3.5 months of gestation were collected from the necropsy room.
Macroscopic and Histologic Examination
The placenta and fetal organs (aural skin, tongue, thyroid gland, thymus, brain, heart, lung, liver, spleen, kidney, umbilicus, forestomachs, and intestine) from all experimental heifers (n = 6) and controls (n = 2) were collected and examined. Three placentomes of the uterus and corresponding intercotyledonary area from different regions (cranial, medium, and caudal) in relation to the uterus position were sampled in each animal. Samples were fixed in 10% buffered formalin for less than 48 hours and conventionally processed through a graded alcohol series before embedding in paraffin wax. Thereafter, 2- to 3-μm sections were obtained from each sample and stained with hematoxylin and eosin (HE), and placental samples were also stained with Van Gieson (VG). All stained slides were examined using light microscopy.
Immunohistochemistry
To identify the viral antigen and cell population composition, samples from the same placental areas and identical fetal organs were collected in duplicate as described previously for the histopathological assay. These samples were either fixed in 10% formalin or frozen in liquid nitrogen, the latter fixed by immersion in acetone (10 minutes) followed by cryosectioning. After deparaffinization and rehydration, formalin samples were properly subjected to the adequate pretreatment and antigen retrieval treatment. Thereafter, these samples were processed with a pestivirus-specific antibody (15c5), a BVDV-specific antibody (C42), and different cell marker antibodies (Table 1). Cryosectioned samples, after fixation, drying, and hydrogen peroxidase blocking, were incubated with another pestivirus-specific antibody (C16) and a BVDV-specific antibody (Ca3/34-C42) under the corresponding conditions to localize the pestivirus antigen in fetal organs and placenta. To characterize inflammatory cells present in the placentome, various antibodies were used, as described in Table 1. 20,21,41 For all primary antibodies employed, the secondary antibodies used were the anti-mouse or anti-rabbit EnVision HRP System (code No. K4001/4003; Dako, Agilent Technologies, Glostrup, Denmark) or the Dako Real Detection, anti-rabbit or anti-mouse (code No. K5001; Dako, Agilent Technologies). Visualization was achieved by the Amino Ethyl Carbazole Substrate Kit (AEC; Invitrogen, Paisley, UK) or diaminobenzidine (DAB; Dako, Agilent Technologies; Table 1). Finally, slides were briefly counterstained by hemalaun, mounted, and examined by light microscopy.
Primary Antibodies and Procedures Used for the Immunohistochemical Examination.
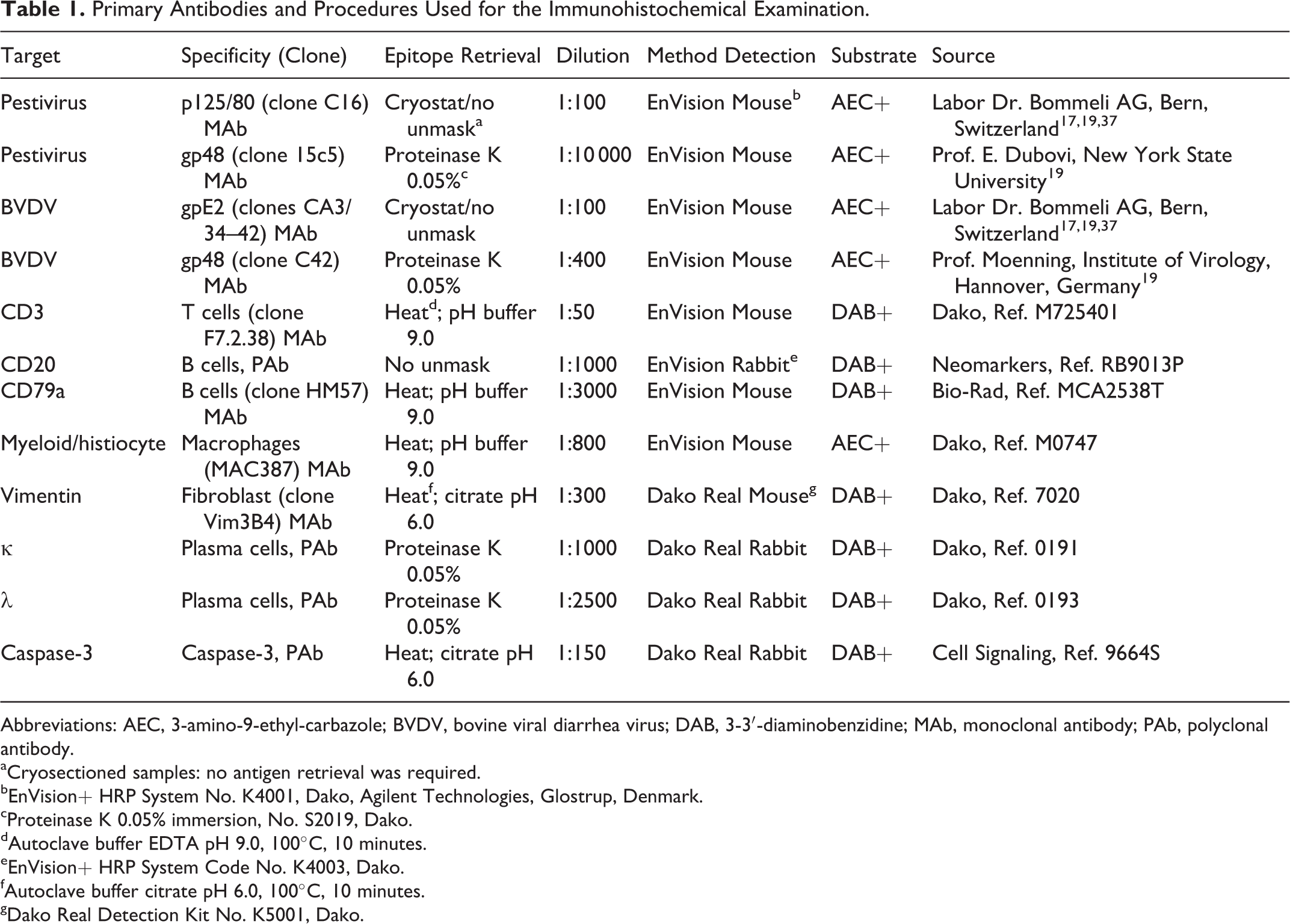
Abbreviations: AEC, 3-amino-9-ethyl-carbazole; BVDV, bovine viral diarrhea virus; DAB, 3-3′-diaminobenzidine; MAb, monoclonal antibody; PAb, polyclonal antibody.
aCryosectioned samples: no antigen retrieval was required.
bEnVision+ HRP System No. K4001, Dako, Agilent Technologies, Glostrup, Denmark.
cProteinase K 0.05% immersion, No. S2019, Dako.
dAutoclave buffer EDTA pH 9.0, 100°C, 10 minutes.
eEnVision+ HRP System Code No. K4003, Dako.
fAutoclave buffer citrate pH 6.0, 100°C, 10 minutes.
gDako Real Detection Kit No. K5001, Dako.
Morphometric and Statistical Analysis
Histopathological evaluation was performed by 2 authors (M.F. and M.H.). All the HE and immunohistochemistry (IHC) samples were scored to assess the presence, distribution, expanse, and cell infiltrate composition in the lesions as well as the pestiviral antigen location and the staining intensity. Lesion severity was evaluated in every placentome sampled by measuring the affected area using the ImageJ software (National Institutes of Health, Bethesda, MD) to estimate the percentage of damage in the heifer. Lesion and pestivirus antigen distribution were semiquantitatively located with the ImageJ program and placed in a coordinate graph. The grade of fibrosis was similarly analyzed, testing the image thresholding with ImageJ software on VG- and vimentin-labeled sections. In addition, cell infiltrate proportion and the level of pestivirus antigen were subjectively allocated in different categories for each cell type on every placental tissue. This was scored as –, not present; +, mild; ++, moderate; and +++, severe.
Statistical analysis was performed using the χ2 test to assess the differences between animals comparing the expanse of the lesions and the grade of fibrosis using the SPSS software (version 21.0; SPSS, Inc, an IBM Company, Chicago, IL).
Reverse Transcription Quantitative Polymerase Chain Reaction Assay
Fetal organs (aural skin, tongue, thymus, thyroid gland, and intestine) and placental samples were used for the identification of pestiviral RNA as described earlier. 7 Samples kept in RNAlater were disintegrated and homogenized mechanically using the Tissue Lyser (Qiagen AG, Hombrechtikon, Switzerland) according to the manufacturer’s instructions. Pestiviral RNA was detected by reverse transcription quantitative polymerase chain reaction (RT-qPCR) (Cador BVDV RT-PCR Kit; Qiagen) with primers in the 5′-untranslated region (5′-UTR) using an ABI 7300 thermocycler (Applied Biosystems, Rotkreuz, Switzerland). 7,21 Sequencing of the 5′-UTR of BDV genome was performed as described. 1
Serological Monitoring
Whole blood for serological assays was collected prior to the introduction of the bull and every 10 days during the entire infection phase (from day 0 until day 60). After serum collection, an antibody enzyme-linked immunosorbent assay (ELISA) test was employed for detection of pestivirus serum antibodies according to the manufacturer’s protocol (Institute of Veterinary Virology, Vetsuisse Faculty, University of Bern, Switzerland). 7,11 In addition, to determine the pestivirus species associated with the antibody titer, a serum neutralization test (SNT) was used. 7 Owning to the phylogenetic proximity, 3 the presence of BDV was only confirmed if its neutralization titers were at least 4 times higher than those toward BVDV. 7,15
Results
At the time of slaughter, the fetuses and placentas from 3 of the 6 heifers were diagnosed as being persistently infected with BDV based on the positive results of the immunohistochemistry and virological identification in fetal organs, as previously reported. 7
Gross and Microscopic Lesions
There were no macroscopical changes observed in the fetuses, uteri, or placentas of any of the BDV-exposed heifers examined on the 110th day of gestation (n = 6) or control animals (n = 2). There were also no histological lesions observed in the fetal organs from either group or the control animal placentas (Figs. 1–3).
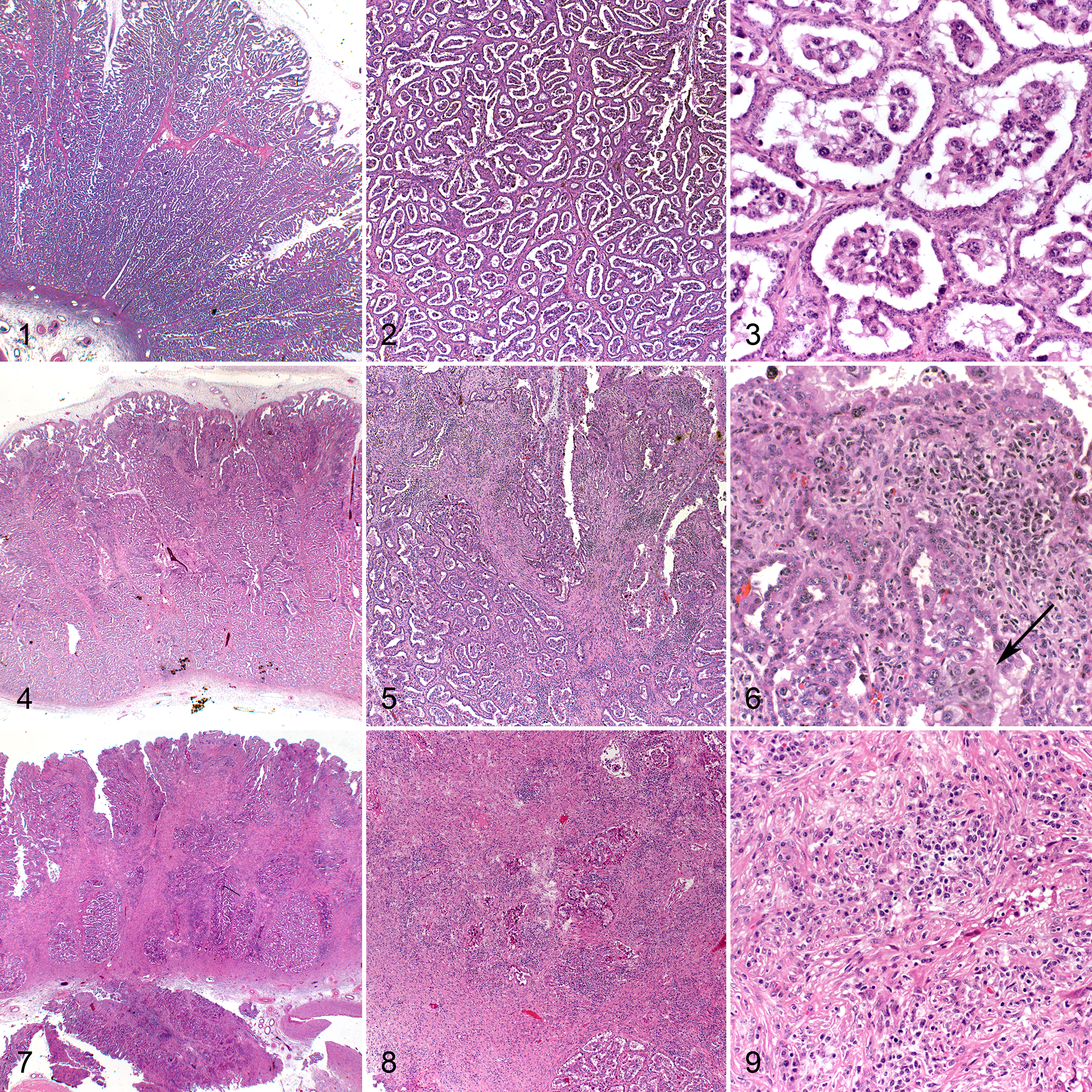
There were, however, histological lesions in the placentas of 3 of the 6 heifers (heifers 3, 4 and 5), shown in Table 2. These lesions appeared to exclusively affect the placentomes while sparing the intercotyledonary regions. Affected placentomes displayed multifocal to coalescing inflammatory infiltrates and moderate to severe fibrosis, which varied between animals, being mild to moderate in 1 animal and severe in 2 animals. Cotyledonary villi were effaced, consistent with a nonsuppurative placentitis involving to varying extents the maternal-fetal interface and the maternal caruncle stalks (Figs. 4–9).
Virologic and Histologic Findings in Pregnant Heifers Infected With BDV.

Adapted from Braun et al. 7
Abbreviations: BDV, border disease virus; BVDV, bovine viral diarrhea virus; RT-qPCR, reverse transcription quantitative polymerase chain reaction; SNT, serum neutralization test quotient = ratio of the serum neutralization titer for serum antibody to BDV and BVDV.
aHistological lesion: rated as none for no lesions; I, inflammation; F, fibrosis; or N, necrosis. IHC, immunohistochemistry staining with C16 and 15c5 antibodies, with –, antigen negative by immunohistochemistry; ++, moderately intense positive labeling for pestivirus antigen.
The mild to moderate inflammation was characterized by a mononuclear inflammatory process and fibroblastic component in the endometrial stalk of the placentome that involved almost 35% of the caruncular and cotyledonary villi (Fig. 5) and extended to the arcade or chorionic plate (Fig. 6, Table 3). In severe placentitis, the trophectoderm was severely damaged, and less than 25% of surface trophoblasts could be distinguished and multifocal necrosis was observed (Fig. 8). In these cases, the same type of inflammation was visible, accompanied by a significant degree of fibrosis depending on the extension of the placentitis (Fig. 9, Table 3; P < .005). Fibrosis was confirmed by the VG stain (not shown) and by immunolabeling for vimentin, which was widespread from the base to the tip of the placentome with the intensity ranging from mild to severe (Suppl. Figs. S1, S2; Table 3). The inflammatory cells in the placentitis were predominantly lymphocytes, comprising approximately equal numbers of T and B lymphocytes (Suppl. Figs. S3–S6, Table 3; as demonstrated by CD3 and CD20 immunohistochemistry) with plasma cells in the minority (+; as identified by κ/λ labeling, not shown), disseminated between villi. Activated macrophages were the second most frequently observed cell type (++), as revealed by calprotectin (MAC387) staining, and they were located in the apical zones of villi and around the necrotic areas, depending on the intensity of the lesions (Suppl. Figs. S7, S8). Furthermore, cellular apoptosis, evaluated by staining for active caspase-3, was present in small groups of cells among the cotyledonary villi in the control animals, as well as in mild and severe placentitis (not shown). Statistically, no association could be established between the proportions of inflammatory cell types within the affected area and the severity of fibrosis in the placentome.
Histological Lesions, Extent of Lesions and Fibrosis, Proportion of Different Cell Populations, and Level of Pestivirus Antigen in Pregnant Heifers Infected With BDV.
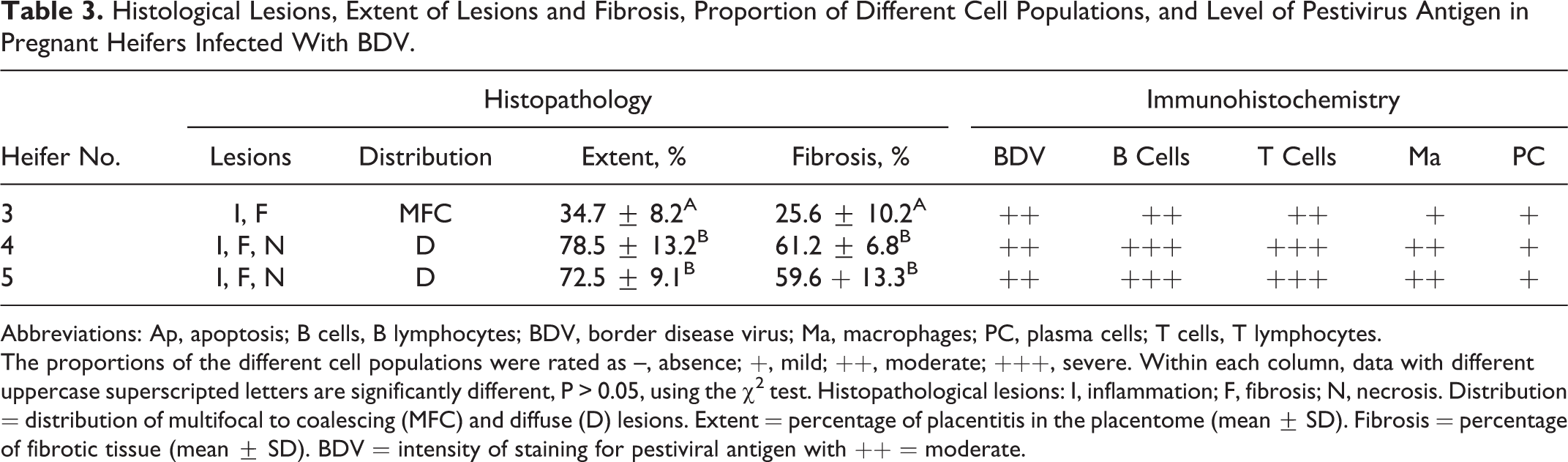
Abbreviations: Ap, apoptosis; B cells, B lymphocytes; BDV, border disease virus; Ma, macrophages; PC, plasma cells; T cells, T lymphocytes.
The proportions of the different cell populations were rated as –, absence; +, mild; ++, moderate; +++, severe. Within each column, data with different uppercase superscripted letters are significantly different, P > 0.05, using the χ2 test. Histopathological lesions: I, inflammation; F, fibrosis; N, necrosis. Distribution = distribution of multifocal to coalescing (MFC) and diffuse (D) lesions. Extent = percentage of placentitis in the placentome (mean ± SD). Fibrosis = percentage of fibrotic tissue (mean ± SD). BDV = intensity of staining for pestiviral antigen with ++ = moderate.
In the control cases (n = 2) and the remaining 3 heifers that did not harbor a persistently infected fetus (heifer Nos. 1, 2, and 6, of which 2 fetuses were most probably transiently infected and 1 remained negative), areas of flat apposition between trophoblast and glandular uterine cuboidal epithelium were observed (Figs. 1–3).
All samples were immunohistochemically negative for Toxoplasma gondii, Neospora abortus, and Chlamydia abortus. In addition, Coxiella burnetii, Campylobacter spp, and chlamydial infection were excluded on a morphological basis.
Identification of Pestivirus Antigen in Tissue Samples
In heifer Nos. 3, 4, and 5, samples from placentomes, ear skin, thyroid gland, tongue, and internal organs were positive for the pestivirus-specific antibodies C16 and 15c5 in cryosectioned and paraffin-embedded samples, respectively (Figs. 10–13). Interestingly, pestivirus antigen staining was basically limited to fetal epithelial cells of the placentomes and scarce in maternal cells, despite the difficulty of distinguishing the layers in cases with severe lesions. Antigen burden was moderate (++) within lesions of moderate as well as in severe placentitis and showed a multifocal distribution among all the placentomes in the cytoplasm of epithelial cells and trophoblasts (Table 3, Suppl. Fig. S9, and Figs. 12, 13).
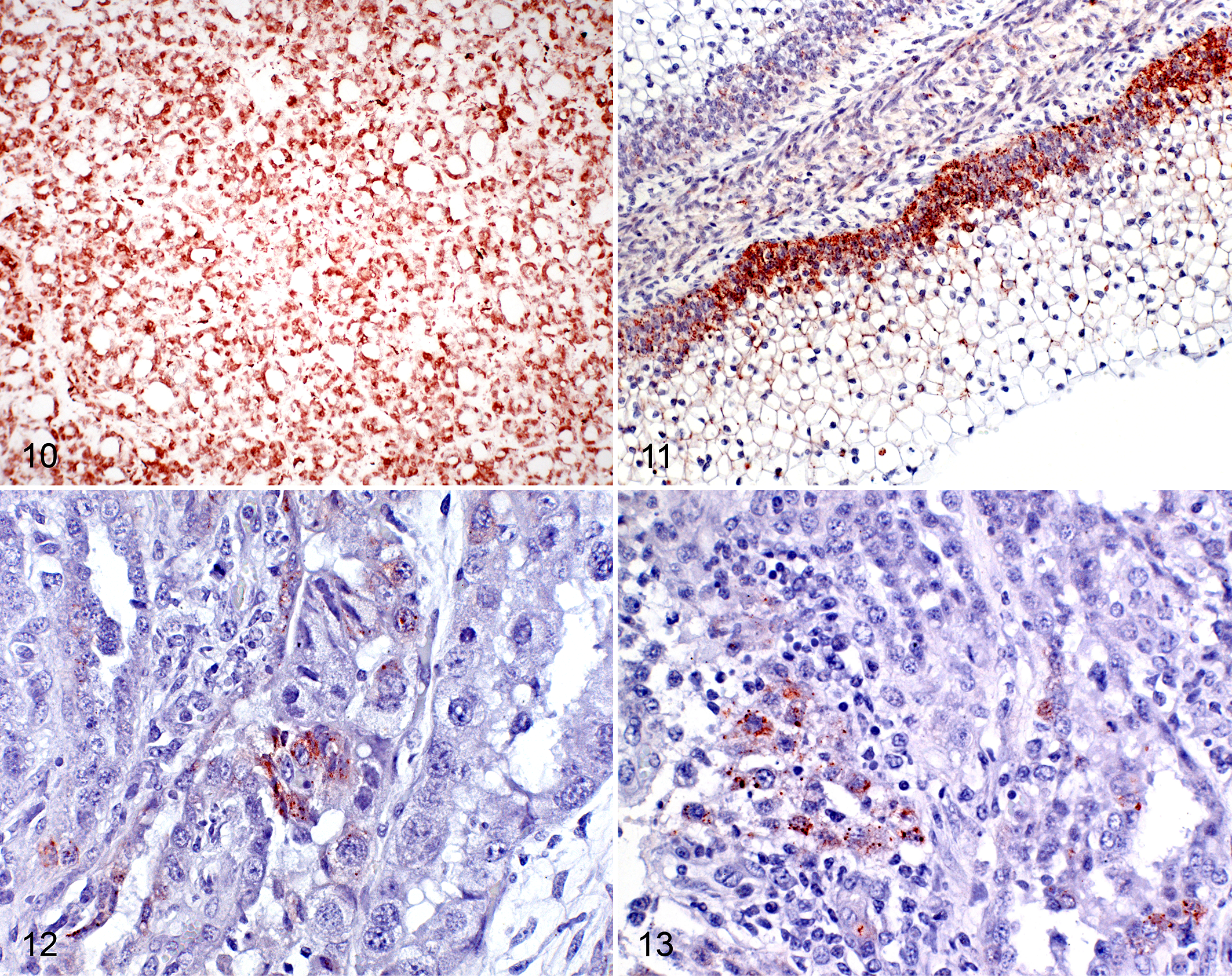
In the skin, antigen was present in the epithelial cells of the hair root sheaths, in epidermal basal cells, and in endothelial cells and medial cells of the vessels. In the thyroid, the follicular epithelial cells and endothelial cells were positive, as were epithelial and endothelial cells in the forestomachs (Figs. 10, 11), as described in previous studies. 2,7,16,21,41 In the internal organs, epithelial and endothelial cells were positive. Furthermore, these samples were negative by the BVDV-specific CA3/34-42 antibody in frozen tissue and also by the C42 antibody in formalin-fixed tissue. Placentas from the remaining heifers without placentitis (heifer Nos. 1, 2, and 6) and those from the control animals (n = 2) were immunohistochemically negative for pestivirus-specific as well as BVDV-specific antigen labeling.
Serological Study and RT-qPCR Assay
In the previous study, ELISA tests to detect antibodies to pestiviruses revealed that all the heifers seroconverted within 40 days of cohousing with the PI-BDV calf. 7 Notably, antibody levels in serum of heifer Nos. 3, 4, and 5 were significantly higher than in heifer Nos. 1, 2, and 6 toward the end of the study, strongly indicating that these heifers contained a persistently infected fetus. 7 SNTs performed on serum samples from day 60 postinfection were positive for BDV antibodies in all heifers. The ratio of BDV to BVDV neutralization titers was greater than 4 in all the heifers, which confirmed that seroconversion occurred in response to infection with BDV. 7
Finally, RT-qPCR detected pestiviral RNA in fetal organs (skin, thyroid gland, tongue) and in placentomes from heifer Nos. 3, 4, and 5, and the sequencing part of the viral genome identified BDV as the infecting pestivirus, as described in the previous study (Table 2). 7
Discussion
In this study, we analyzed the main pathological findings in the placentas of heifers cohoused with a PI-BDV calf for 60 days as a complement to our previous description of BDV transmission from cattle to cattle. 7 BDV classically infects sheep, although there have been a number of case reports of calves infected under natural conditions. 13,25,38 As confirmed by the virological results reported earlier (ie, SNT, RT-qPCR, and nucleotide sequencing), 7 in addition to labeling for viral antigen, all the heifers and corresponding fetuses in this study were BDV infected. To confirm the presence of antigen, immunohistochemical staining used in this study provided a highly specific and sensitive method to assess the presence of BDV in placental tissue and fetal organs such as thyroid gland, tongue, and skin. 7,9,20,22,41 All uteri, placentas, and fetuses were macroscopically normal, and no histologic abnormalities were found in the fetal organs. There was histological evidence of inflammation in the placentomes of the 3 heifers in which virus could be identified by immunohistochemical staining, which differs from the placentitis observed in sheep infected with noncytopathic BDV, in BVDV-infected ewes, 29,35,39 and also in BVDV-PI fetuses of pregnant cows, in which placentitis is absent. 16,29 In that sense, despite the fact that seroconversion was observed in this experiment, the BDV infection in the 3 heifers is in accordance with the previous studies in which infection of bovine fetuses in the first trimester of gestation with noncytopathic BVDV could be associated with the birth of persistently infected animals. BDV distribution in the placentomes of 3 heifers was similar to the diffuse BVDV antigen distribution described in epithelial cells on the fetal side and also on the maternal side of BVDV-infected ewes and goats. 22,35 In previous experimental BVDV infection of ewes, no histological changes were observed from day 7 to 14 postinfection, although a severe erosive and fibrinous placentitis on the fetal side of the placenta with abundant fibrin distributed in fetal fluids was observed in a ewe sacrificed at day 36. 35 In another study, ewes were intranasally inoculated with BVDV, and no macroscopic or microscopic lesions could be detected in either the reproductive tract or any of the recovered fetuses. 39 The pathogenesis of placentitis in BDV infection in heifers is not yet understood. However, it might require the presence of the virus in the placenta, which was confirmed in this study by immunohistochemistry, even though cases have been described where viral infections of the placenta occurred without gross or histologically evident tissue destruction. 9,16,25,35,39,40
As the fetus is not immune competent, a persistent infection with large amounts of pestiviral antigen similar to that seen in the present study is expected. The infection of the mothers is transiently characterized by seroconversion without detection of viral antigen except in the uterus and fetal compartment. It is suggested that virus replication occurs through binucleated cells of the allantochorion and that these cells migrate from the allantochorion to the endometrium. 16,40 At this point, the virus spread has no consequence for the mother because an effective antibody response has already been mounted and is measurable. 40
Since the heifers had seroconverted, the placentomes may represent the interface between fetal persisting virus and maternal immune response, and lesions may be the result of an excessive/prolonged immune response. 16,39,40 This correlates well with the fact that antibody levels were significantly higher in those heifers in this study that had persistently infected fetuses and developed lesions in the placentomes.
The synepitheliochorial placenta is formed by interdigitations between the maternal layer, the caruncle, and the fetal portion with uninucleated and, to a lesser extent, binucleated trophoblasts. In our study, this was replaced by an inflammatory infiltrate composed mainly of lymphocytes and macrophages with fewer plasma cells in addition to varying amounts of fibrous tissue. The lymphocytic component, composed of roughly equal numbers of T and B lymphocytes, would be related to the adaptive immune response against pestiviruses as demonstrated in previous studies with BVDV. 12,17 The presence of macrophages in the placentomes in this study was significantly increased in contrast to that in other studies, where macrophages were lacking in cases with fetuses less than 6 months old. 31,36 Thus, the macrophages may trigger an adaptive immune response, leading to injury of the maternal-fetal junction with harmful consequences for the fetus. 10,12 In fact, it could be assumed that the placentitis observed in this study was a consequence of the infection of the placentomes in utero with BDV, as indicated by immunohistochemistry, PCR, and SNT results.
A role for the adaptive intrinsic apoptosis pathway has been reported in persistent pestiviral infections, as described for mucosal disease–associated lesions. 20 During normal pregnancy, this pathway is also known to be activated, as the number of apoptotic cells increases significantly in the maternal crypt epithelium, maternal stroma, and fetal chorionic epithelium. 5 We were unable to detect any enhanced activation of the pathway in infected animals compared to controls, suggesting it is unlikely to be significantly involved. It cannot, however, be entirely excluded that previous apoptosis had itself led to the inflammatory process and tissue replacement by fibrosis.
Whether apoptosis would be advantageous to the virus in this scenario is unclear; depending on the virus tropism and multiplication pathway, apoptosis may enhance viral spread or conversely be detrimental by reducing availability of target cells. 20,34
Pestivirus antigen has also been described inside endothelial cells, causing vascular lesions and, as a result, tissue hypoxia. 17 –20 Nevertheless, in this study, there was no histological evidence of vasculitis.
The experimental animals were kept under controlled conditions and therefore not exposed to infection by other abortogenic agents as may be observed in field cases. Macroscopical lesions were not observed, and histological lesions were not compatible with those induced by known agents. C. abortus and C. burnetii are characterized by a fibrinosuppurative placentitis and fibrinous exudates in the placentome as well as vasculitis, 4,6 while Brucella abortus and Bacillus spp in cattle are characterized by a severe and diffuse fibrinonecrotic placentitis 44 and necrosis between villous trophoblasts and the crypt epithelium, respectively. 26 The placentitis described in this study is also different from that observed in abortions caused by the protozoans Toxoplasma and Neospora. 12,14,42 C. abortus, T. gondii, and Neospora caninum were also excluded by immunohistochemistry.
Although placentas and fetuses were collected from the heifers at day 110 of gestation and no abortion had occurred by then, it can be assumed, taking into account that all the examined placentomes (in PI-fetuses) were affected, that the structure of the placentome toward the end of gestation would be moderately to severely impaired. This would make it unlikely that the fetus would have remained viable to term but rather would probably be aborted. The severity of the placentitis, measured by the affected area in the placentome, could not be related to the level of pestivirus antigen, as no differences could be observed between the intensity of staining for viral antigen and the level of placentitis. With this in mind, the development of the lesions would appear to depend more on the extent of host response and likely the virulence of the strain rather than on viral load. 10,17,33 Therefore, interaction between T-lymphocytes and macrophages might play a role in the development of a T-helper 1 response, which has been suggested as being deleterious to fetal survival. 17,33
The development of fibrosis in placental tissue is difficult to evaluate because to our knowledge, no literature is available for cattle. No gross lesions were observed to suggest chronic placental inflammation, demonstrating the importance of histologic examination for detection of this lesion. In the literature, examples of placentitis with other characteristics have been reported with pestiviruses; this may be due to the time and duration of infection. 16,39,40 The changes in the placenta may well explain the intrauterine growth retardation described for BDV in goats and sheep and BVDV in cattle. 2,9,37 Initially, the mothers (heifers/ewes) are infected, followed by infection of placental tissues and finally the fetus. 40
In summary, the results presented in this study reveal that BDV is able to produce a severe mononuclear and fibrosing placentitis in cattle. The damage exerted by viral infection of endothelial and epithelial cells in the placenta may consequently compromise placental function. To our knowledge, this is the first description of placentitis caused by a transient BDV infection in heifers during gestation that has led to a persistent infection in their fetuses. However, more studies are required to understand the pathogenesis of BDV in depth and to characterize the immune response in the bovine placenta.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985817754123 - Border Disease Virus Infection of Bovine Placentas
Supplemental Material, DS1_VET_10.1177_0300985817754123 for Border Disease Virus Infection of Bovine Placentas by Miguel Fernández, Ueli Braun, Sandra Frei, Matthias Schweizer, and Monika Hilbe in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the dedicated assistance of the technical staff of the Institute of Veterinary Pathology, University of Zurich, for the laboratory work carried out in this research and Dr. Alex Malbon for language corrections.
Declaration of Conflicting Interests
The author(s) declare that they have no conflicts of interests with either the research, the authorship and/or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The University of Zurich financed the experimental part of this study.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
