Abstract
A 3.5-year-old, neutered male pit bull dog was euthanized following an approximately 1-year history of intractable diarrhea and weight loss of undetermined cause. At necropsy, the dog was emaciated. The ratio of total intestinal length (duodenum to rectum) to crown-to-rump length was 2.5, in contrast to an average of 5.3 (range, 3.7–6.1) in 10 control dogs examined at necropsy. There was diffuse dilation of the intestinal lumen, consistent with congenital intestinal hypoplasia resulting in short-bowel syndrome. Histologically, the intestinal mucosal was hyperplastic, further supporting the diagnosis of short-bowel syndrome. To the authors’ knowledge, this is the first case of this condition in the veterinary literature.
Keywords
Short-bowel syndrome (SBS) is a clinical syndrome characterized by severe weight loss secondary to a malabsorptive diarrhea from a markedly shortened small intestine. SBS may be congenital or acquired. The 2006 human medical consensus statement on SBS and intestinal failure defines SBS as a “result from surgical resection, congenital defect or disease-associated loss of absorption and is characterized by the inability to maintain protein-energy, fluid, electrolyte or micronutrient balances when on a conventionally accepted, normal diet.” 7 In humans, congenital SBS is a rare neonatal condition associated with high mortality due to difficulties in diagnosis and complications of treatment. 4 Acquired SBS occurs in both humans and animals, primarily as a consequence of resection of large segments of the small intestine. The resection length that results in the development of acquired SBS in animals depends on the age of the patient, intestinal portion resected, presence of the ileocecal valve, and underlying cause of the bowel resection. 13 In the small-animal veterinary literature, SBS is suggested to occur following greater than two-thirds (66%) resection of the small intestine, with involvement of the duodenum and ileocecal valve carrying a poorer prognosis. 3 Canine models of acquired SBS involve surgical resection of 75% of the intestinal length, generally requiring a full jejunectomy. 9 In horses, 60% resection of the small intestine may result in SBS, although resection of >70% of the small intestine has been reported prior to clinical manifestations developing. 5 Regardless of the absolute reduction in intestinal length, SBS is characterized by an intestinal length that is insufficient to allow adequate nutrient absorption. 14 Although acquired SBS is recognized and described in veterinary medicine, there are no reports of congenital SBS in any domestic, exotic, or wild animal species.
A neutered male pit bull dog, aged 3 years 7 months, had a >1-year history of weight loss and intermittent episodes of diarrhea of increasing frequency. On initial clinical presentation at approximately 1 year of age, the owners had noted a 1-month history of mild weight loss and observed parasites in the stool. The dog’s weight at 1 year of age was 28.8 kg. Fecal flotation was not performed at this time, and the dog was treated with a praziquantel (136 mg), pyrantel pamoate (136 mg), and febantel (680 mg) combination dewormer (Drontal Taste Tabs; Bayer). On routine physical examination 14 months following the initial presentation (2 years 2 months of age), the patient’s weight had decreased to 23.4 kg, with no apparent diarrhea. Thirteen months following the second clinical presentation (3 years 3 months of age), the dog was presented for intermittent episodes of vomiting and diarrhea of 3 months’ duration. The patient’s weight had decreased to 22.9 kg. The stool was yellow and liquid and contained a mild amount of mucus. Fecal cytology performed in house at the clinic revealed abundant short rod–shaped bacteria without endospores. No parasite eggs or ova were found on zinc sulfate fecal flotation. Testing of a fecal sample by polymerase chain reaction (Canine Diarrhea RealPCR Panel; IDEXX Laboratories) was positive for canine enteric coronavirus and Clostridium perfringens α toxin gene; Cryptosporidium spp, Giardia spp, Salmonella spp, canine parvovirus type 2, and canine distemper virus were not detected by polymerase chain reacion. The patient was prescribed metronidazole (500 mg twice daily by mouth); amoxicillin (500 mg twice daily by mouth); a kanamycin (100 mg), bismuth (250 mg), and attapulgite (500 mg) product (Amforol; Zoetis); and an oral canine probiotic product (FortiFlora 1 g; Nestle Purina). The patient initially responded to therapy with improvement in stool consistency but did not gain weight. Following 1 month of therapy, a complete blood count (IDEXX ProCyte Dx Hematology Analyzer; IDEXX Laboratories) and serum biochemistry (IDEXX Catalyst Dx Chemistry Analyzer; IDEXX Laboratories) revealed no significant abnormalities. Tylosin (350 mg twice daily by mouth) was added to above therapy and continued until the time of euthanasia. A brief clinical trial of pancreatic enzyme replacement therapy (Panakare 1 tsp twice daily; Neogen) was initiated with no response. Euthanasia was elected.
At necropsy, the dog was emaciated, with no intra-abdominal or subcutaneous fat stores, and had severe diffuse muscle atrophy with a body condition score of 1.5 of 5. The stomach contained a large quantity of normal ingesta, and the entire small intestine, large intestine, and rectum contained abundant yellow to tan, fluid digesta with undigested particles. The perianal region was stained with dark yellow liquid fecal matter. The small and large intestines were present in subjectively normal proportions but were both grossly shortened (Fig. 1) compared with a dog of a similar crown-to-rump length (CRL) lacking gastrointestinal disease (Fig. 2). The entire length of the digestive tract from the stomach to the anus measured 198 cm. The length of the small intestine from the pyloric sphincter to the ileocecal junction was 165 cm, and the colonic length was 33 cm. The small intestine was diffusely dilated, measuring 1.9 cm in diameter at the ileum and up to 2.9 cm at the proximal duodenum. The ratio of total intestinal length to CRL was 2.5. The small intestinal wall was up to 4 mm thick. The ileocecal junction was grossly unremarkable. Detailed examination of the serosal surface of the gastrointestinal tract from the stomach to the rectum did not reveal any serosal disruption or scar tissue from a prior intestinal surgery, examination of the abdominal wall did not reveal scar tissue from an abdominal incision, and the dog had no reported history of surgery except for routine castration as a juvenile. No significant gross lesions were observed in other organs at necropsy.
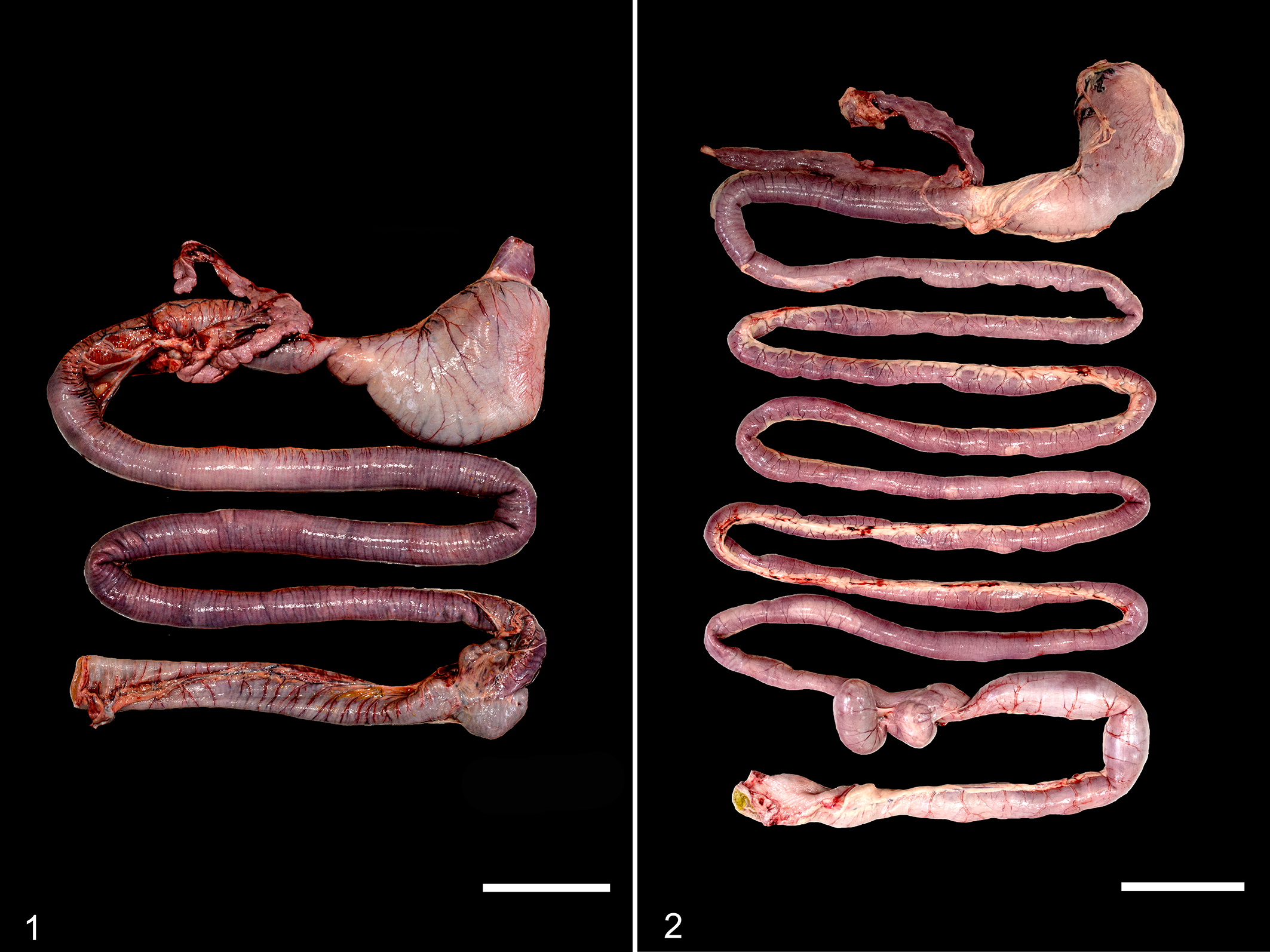
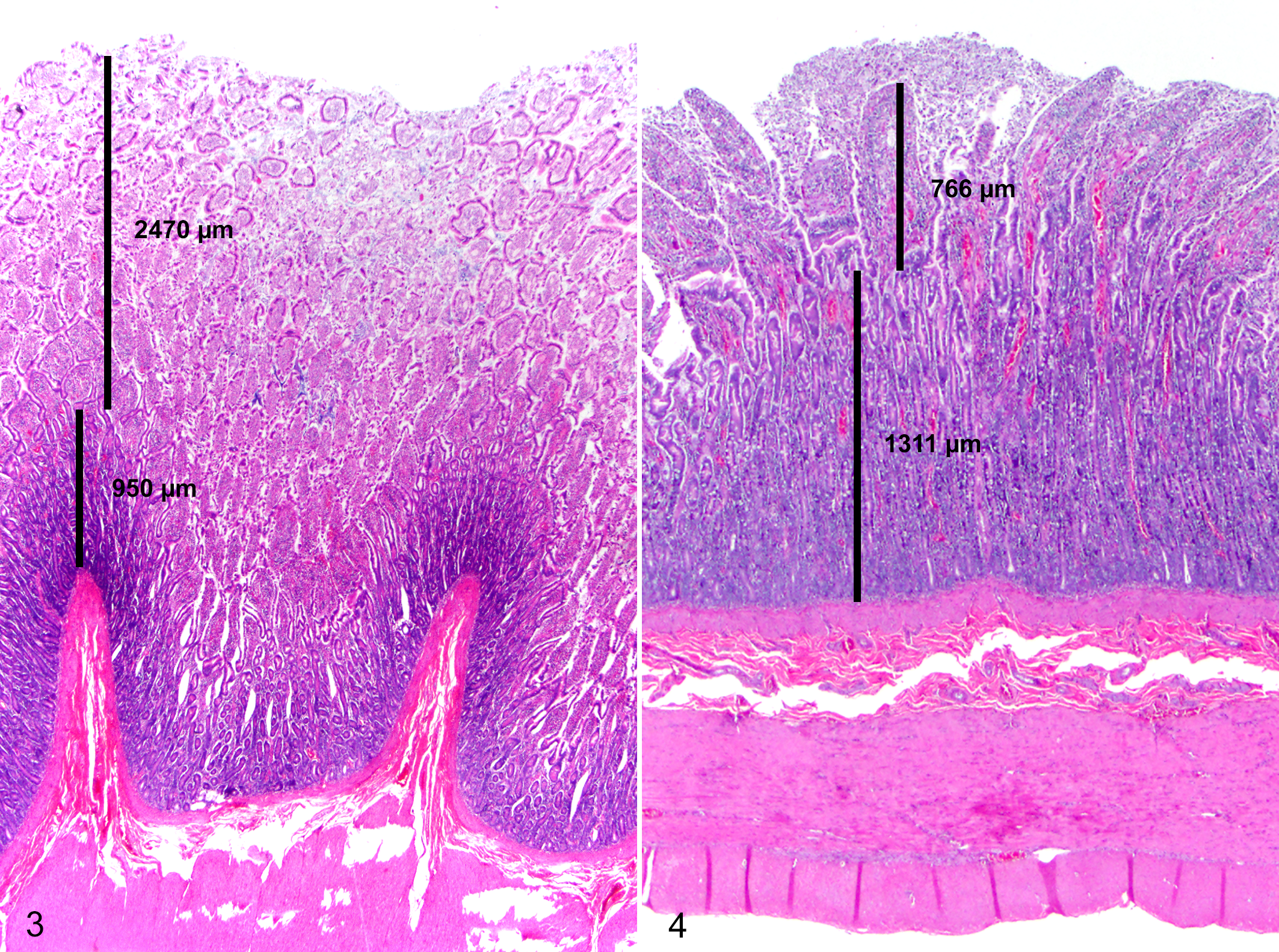
Histologic evaluation of segments of small intestine extending from the proximal duodenum through the ileum showed copious mucus overlying a mildly to moderately hyperplastic mucosa. The crypt-to-villus ratio ranged from 0.8 to 2.5 throughout the duodenum (Fig. 3). Large numbers of individualized, gram-positive, rod-shaped bacteria consistent with Clostridium spp were admixed with the mucus and extended deeply within intestinal glands of all evaluated sections of the small intestine. The submucosa was segmentally infiltrated by moderate numbers of eosinophils and low numbers of lymphocytes and plasma cells. Rare crypts contained eosinophilic cellular debris and low numbers of neutrophils. The colon was unremarkable. No significant histologic lesions were observed in the trachea, lung, liver, kidney, urinary bladder, spleen, skeletal muscle, cardiac muscle, esophagus, adrenal gland, thyroid gland, pancreas, myenteric plexi, spinal cord, or brain. The pathologic diagnosis was diffuse congenital intestinal hypoplasia with small intestinal bacterial overgrowth. The clinical diagnosis was congenital SBS.
With the exception of the low body condition score, the dog was clinically normal for 2.5 years prior to developing clinical signs attributed to SBS. In human neonates, 2 factors that predispose to SBS are absence of an ileocecal junction and presence of <50% of small intestinal length. 11 The intestinal length in this dog was approximately 45% to 50% of the expected intestinal tract length of approximately 420 cm from pyloric sphincter to rectum, for dogs with a CRL length of 80 cm (Suppl. Table S1).
The pathogenesis of SBS is complex, and development of clinical signs is likely multifactorial. Severity and duration of clinical signs may be variable. Experimentally acquired SBS in animals shows a marked enterocyte hyperplastic response within 24 to 48 hours following surgery and can result in a 4-fold increase in mucosal surface area. 10 Grossly, the intestinal wall in this case was thickened up to 4 mm. Histologically, the crypt-to-villus ratio ranged from 0.8 to 2.5 in the duodenum (Fig. 4). The reported canine crypt-to-villus ratio in the veterinary literature ranges from 0.6 to 0.8 depending on the segment of intestine evaluated. 8 In this case, we suspect that the intestinal tract in this animal was adequate to sustain life until unmanageable small intestinal bacterial overgrowth (primarily Clostridium infection) occurred.
Although congenital intestinal hypoplasia has not been documented in veterinary medicine, 3 cases of suspected congenital colonic hypoplasia have been described. 12 In all 3 cases, the diagnosis of colonic hypoplasia was obtained through contrast radiographic imaging rather than with direct ante- or postmortem colon measurements. Colonic hypoplasia in all 3 animals appeared to be of minimal consequence. Congenital malabsorptive disease has been reported in 3 thoroughbred foals euthanized within 72 hours postpartum. 6 In all 3 foals, the mucosa was hypoplastic, characterized by villous shortening throughout all segments of the intestinal tract. In addition, the small intestinal length was reported to be slightly shorter than control foals. However, all foals examined had low birth weights, and intestinal length measurements were not determined in relation to CRL. All 3 foals had failure of passive transfer, with serum immunoglobulin levels of 85 to 280 mg/dL, presumably due to decreased immunoglobulin uptake by a hypoplastic mucosa.
A definitive diagnosis of congenital SBS in veterinary species presents difficulties because of the lack of published normal values expected for the intestinal length and lack of established criteria for intestinal length necessary for normal gastrointestinal function. Veterinary anatomy reference textbooks vary in estimated lengths of the intestinal tract in canines. The Textbook of Veterinary Anatomy by Dyce et al 2 states an estimate of 3 to 4 times the body length (assumed to be CRL) without providing a reference and states that published values should be interpreted carefully because of a lack of adequate sampling populations. Data reported in early references have propagated in veterinary medical literature, but are based on a single publication by Colin in 1871. 1 That publication estimates the average intestinal length–to–body ratio to be approximately 6:1. The intestinal length is assumed to lengthen in the postmortem period because of relaxation of musculature, making correlation and interpretation of intestinal length in live and deceased patients difficult.
Because no reliable intestinal length references are published in the veterinary literature, we measured the total intestinal length and CRL in 10 dogs ranging from 6 months to 12 years of age lacking gross or histologic evidence of gastrointestinal disease. Necropsies were performed within 12 hours of euthanasia or natural death. The CRL was evaluated with the head placed perpendicular to the axis of the spinal column. In this study, the average entire intestinal length (duodenum to rectum) was 5.3 (range, 3.7–6.1) times the CRL (Suppl. Table S1).
In the present case, the evidence of congenital SBS was (1) an intestine that was 45% to 50% of the expected length; (2) emaciation consistent with inadequate nutrient absorption from the intestine; (3) histologic evidence of mucosal hypertrophy and hyperplasia, likely as a compensatory response; and (4) no gross or histologic evidence of a primary malabsorptive intestinal disease. To our knowledge, this is the first report of congenital intestinal hypoplasia resulting in SBS in the veterinary literature. SBS should be considered as a rare cause of chronic unresponsive diarrhea in domestic animals.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985817750455 - Congenital Short-Bowel Syndrome in an Adult Dog
Supplemental Material, DS1_VET_10.1177_0300985817750455 for Congenital Short-Bowel Syndrome in an Adult Dog by Chad S. Clancy, Khrista A. Jensen, and Arnaud J. Van Wettere in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
