Abstract
Plexiform vasculopathy refers to an endothelial proliferative disorder affecting cervical or inguinal lymph nodes of cats. The cause of this disorder and the origin of the proliferating endothelial cells are still unknown. In 4 cats with a history of a slowly growing, well-demarcated, nonpainful mass adjacent to the thyroid gland, an enlarged dark brown to red lymph node was removed. Histologically, the lymph nodes showed severe loss of lymphoid tissue with accumulations of erythrocytes. In addition, networks of capillary structures with well-differentiated endothelial cells on a collagen-rich stroma were observed, consistent with benign plexiform vasculopathy. Immunohistochemistry revealed the expression of the vascular endothelial markers CD31 and factor VIII–related antigen. In addition, immunolabeling with a Prox-1 antibody indicated a lymphendothelial origin. With respect to our findings, a lymphendothelial origin has to be considered in cases of intranodal vascular neoplasms.
Plexiform vascularization or vasculopathy of lymph nodes, also known as vascular transformation of lymph node sinuses, has been reported in humans, cats, and in a single dog with a thyroidal carcinoma. 2,4,10,13,14,18 In cats, this uncommon lesion is located in cervical or inguinal lymph nodes. It has been initially described as a distinctive lymphadenopathy characterized by a capillary vasoproliferation with lymphoid atrophy. 10 The pathogenesis of plexiform vasculopathy in humans and animals is unclear. In addition, it is still unknown whether the proliferated endothelial cells are of blood or lymphatic origin. Recently, the possibility of a malignant transformation of plexiform vasculopathy into hemangiosarcomas in feline lymph nodes has been described. 13 However, the histogenesis of the proliferated endothelial cells has not been proven.
This report describes the clinical and morphological findings as well as the immunohistochemical characterization of plexiform vasculopathy in lymph nodes of 4 cats, shedding new light into the histogenesis of this disorder.
A 6.5-year-old, male neutered, European Shorthaired cat (cat No. 1) presented in May 2016 with a marked expiratory dyspnea and a small lump in the area of the neck. Until summer 2016, clinical signs worsened toward acute stridor and obvious difficulties in swallowing. On the right-ventral, paramedian side of the neck, a rigid, nonpainful mass was diagnosed. Computer tomography (Siemens Somatom Emotion 16; Siemens, Erlangen, Germany) with 1-mm slice thickness and contrast application showed a right paratracheally located, dense structure from the mandibular angular process extending to the cranial margin of the fifth thoracic vertebrae with deviation of the trachea and the larynx to the left. Furthermore, a 4-year-old, male neutered, European Shorthaired cat (cat No. 2) presented with a 1.5 cm in diameter, soft, smooth, nonpainful mass in the area of the retropharyngeal lymph node. In addition, a 5-year-old, male neutered, European Shorthaired cat (cat No. 3) and a 12-year-old, female, European Shorthaired cat (cat No. 4) also showed slowly growing nodules in the region of the thyroid gland. There was no clinical evidence for other disorders in any of the 4 cats.
The nodules in all 4 cases revealed well-demarcated, noninfiltrative, encapsulated, dark brown to red, homogeneous masses up to 7.3 × 3.8 × 3.7 cm. The surgically removed nodules were fixed in 10% neutral-buffered formalin and submitted for histological examination. The tissues were trimmed and routinely processed into paraffin wax. Sections were cut at 5 μm and stained with hematoxylin and eosin (HE) and Elastica-van Gieson (EVG) for collagenous and elastic fibers.
Immunohistochemical investigation was performed with different rabbit polyclonal antibodies and protocols using the avidin-biotin-peroxidase complex (ABC) method (Vector Laboratories, Burlingame, CA) as previously described. 9 Briefly, sections of the lymph nodes were deparaffinized and rehydrated in a descending series of ethanol. Endogenous peroxidase activity was blocked by using 85% ethanol with 0.5% H2O2. Subsequently, antigen retrieval was performed either in citrate buffer (pH 6.0, 30 minutes, 95°C) or by using pronase K (pH 7.4, 20 minutes, 37°C; Merck, Darmstadt, Germany). For Prox-1, a second step for antigen retrieval was performed by using pepsin ready to use (30 minutes, 37°C; ProTaqs Pepsin Digest [Acetic], Quartett, Berlin, Germany). After blocking nonspecific binding activities with inactivated normal goat serum, sections were incubated with rabbit polyclonal antibodies targeting the endothelial marker CD31 (1:100; Acris, Herford, Germany; catalogue no. AP15436PUM) and factor VIII–related antigen (1:500; Dako, Hamburg, Germany; catalogue no. A0082) as well as the lymphendothelial-specific antibody Prox-1 (1:200; ReliaTech, Braunschweig, Germany; catalogue no. 102-PA30S). 3,3′-Diaminobenzidine-tetrahydrochloride (DAB; Sigma-Aldrich, Munich, Germany) was used as chromogen. Sections were counterstained with Mayer’s hematoxylin and mounted. As negative controls, primary antibodies were replaced by an appropriate amount of rabbit serum (Sigma-Aldrich, Taufkirchen, Germany). A normal lymph node of a cat derived from a necropsy case was processed similarly as described above and was used as additional control tissue.
Histopathological examination in all 4 cases revealed similar morphologic changes. Severe loss of lymphoid tissue was found multifocally and randomly distributed in the outer cortex, paracortex, and medullary cords (Fig. 1). These areas were either replaced by extensive accumulations of erythrocytes or capillary-like proliferations. The latter consisted of unencapsulated areas of proliferated endothelial cells supported by a sparse to moderate fibrous stroma (EVG stain) without evidence of a smooth muscle layer. Areas with proliferated vascular structures measured up to 185 × 60 μm2. Within the vascular spaces, variable amounts of erythrocytes were present. The endothelial cells were characterized by small elongated basophilic nuclei and inconspicuous cytoplasm (Fig. 2). Only scattered endothelial cells showed slightly prominent nuclei bulging into the vascular space, and on average, 1 mitotic figure was seen in 10 high-power fields.
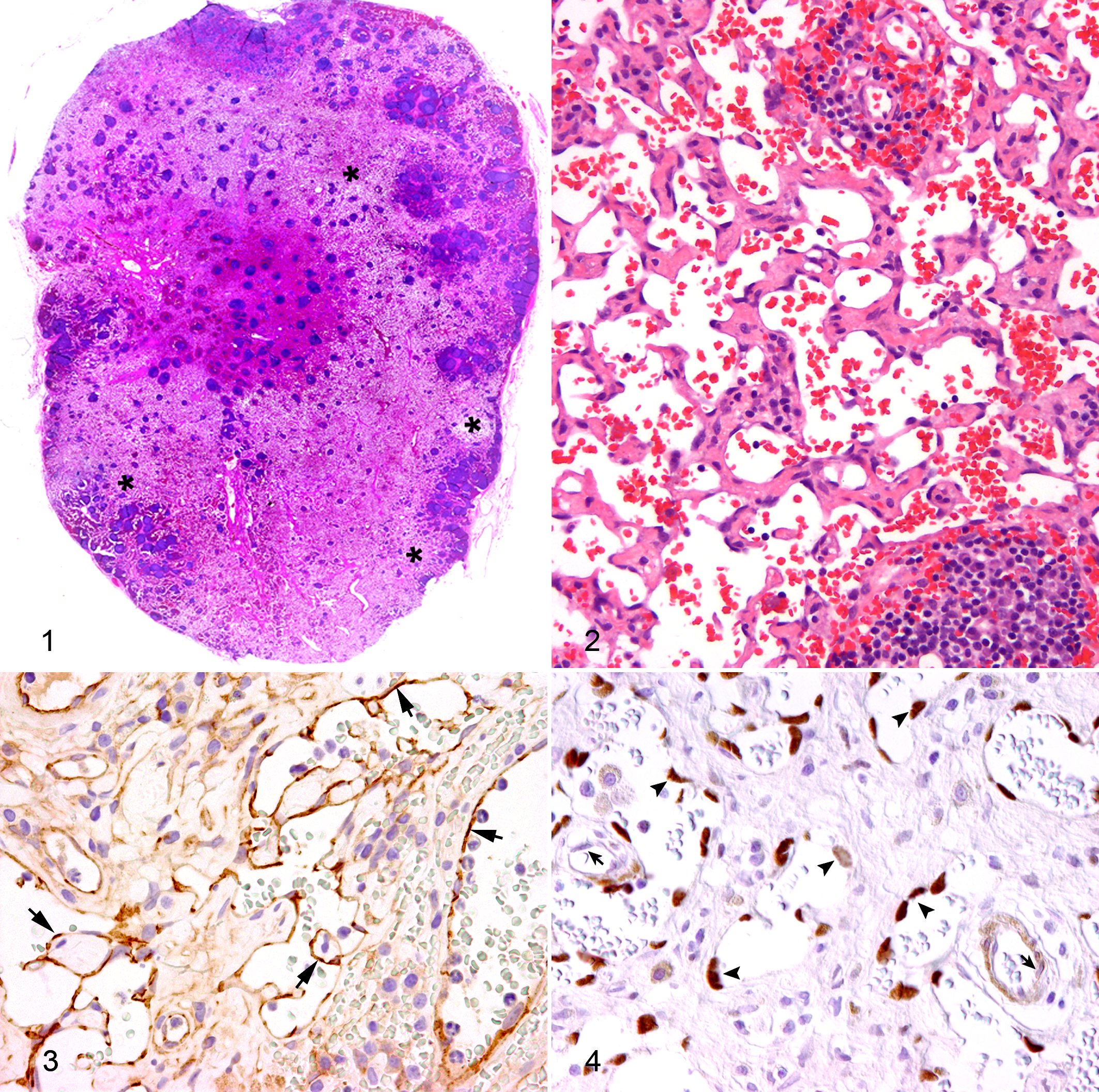
Immunohistochemistry revealed a cytoplasmic expression of CD31 (Fig. 3) and factor VIII–related antigen in all proliferated intranodal endothelial cells. Endothelial cells of intra- and extranodal blood and lymph vessels were stained similarly. In addition, the lymphendothelial cell marker Prox-1 labeled the nuclei of about 80% of the proliferated endothelial cells (Fig. 4), but blood vessel endothelial cells lacked the expression of this marker. This antibody showed also an immunostaining of lymphatic endothelial cells and sinus endothelial cells of the control lymph node.
These 4 cats presented with a cervical lymphadenopathy displayed clinical signs and morphological findings similar to those described in the literature as plexiform vascularization. 10,13,18 The hallmark of this disorder is a vascular proliferation characterized by well-differentiated endothelial cells attached to a fibrous stroma. To avoid the distinction between blood or lymphatic vascular channels, the term vascular has been used widely in the literature. 2 Histologically, capillary-like channels containing erythrocytes are suspected to be either true blood capillaries or lymphatics showing lymphaticovenous anastomosis. Channels containing only lymphatic fluid are considered of lymphatic origin. 2 In human and veterinary medicine, the histogenesis of the endothelial cell proliferation in plexiform vasculopathy has not been determined yet. Identification of normal vascular structures and vascular proliferations can be supported by immunohistochemical evaluations. Cell markers like CD31 (platelet endothelial cell adhesion molecule 1 [PECAM-1]), a transmembrane glycoprotein expressed by platelets, megakaryocytes and endothelial cells, and factor VIII-related antigen (FVIIIRa) present in endothelial cells and megakaryocytes, have been described for the immunolabeling of normal and neoplastic endothelial cells of blood and lymphatic vessels. 8,12 Frequently used markers for differentiation between vascular and lymphatic endothelial cells include Prox-1 (prospero-related homeobox gene 1), a transcription factor involved in lymphendothelial differentiation, 12 and Lyve-1 (lymphatic vessel endothelial receptor 1), a receptor for the extracellular matrix glycosaminoglycan hyaluronan (HA) that has been identified almost exclusively on lymphatic vessels. 3,15,17 The immunohistological results of this study using the lymphendothelial-specific marker Prox-1 indicate a lymphatic origin of the proliferated cells. The origin of the varying number of erythrocytes within the lumina of the vascular channels remains undetermined. A lymphaticovenous anastomosis cannot be excluded. In human lymph node biopsies, the presence of erythrocytes within lymphatic vessels is described, and a communication between venules and lymphatic vessels has been suggested similar to lymphaticovenous communications in canine lymph nodes. 19 Drainage disturbances may be associated with an inosculation of the lymphatic and venous circulation, resulting in distension of vascular spaces separated by the endothelial cells or widening of preexisting mixing chambers or even the opening of venous and lymphatic capillaries into certain sinusoids. 4,5,14
In a recent study about benign and malignant vascular lesions in feline cervical lymph nodes, it was stated that benign vascular proliferations may undergo malignant transformation toward hemangiosarcoma. 13 However, the blood vascular origin of these neoplasms has not been proven. With respect to the results described above, it has to be considered that these intranodal malignant transformations may represent lymphangiosarcomas. Lymphangiosarcoma is an uncommon malignant neoplasm in cats, which typically causes severe subcutaneous swelling of the ventral thoracic region, of the ventral abdominal wall, or in the mediastinum, mesentery, and omentum. 6
The pathogenesis of plexiform vasculopathy is still undetermined. Possible pathogenetic mechanisms include drainage disturbances caused by tumors in the lymph node or in close vicinity, thrombosis, severe congestive heart failure, or angiogenic factors released by a neoplastic process. 2,7,10,11,13 In addition, occlusion of the efferent lymphatic vessels and/or veins has to be considered, and indeed a comparable lesion of lymphatic proliferation has been experimentally reproduced in cervical lymph nodes of rabbits by incomplete occlusion of veins combined with complete occlusion of lymphatics, or by complete occlusion of lymphatics alone. 5,14,16 The presented cases did not reveal any evidence for the underlying cause of this disorder. In all animals, there was neither a suspect of a neoplastic process nor evidence of a lymphatic/venous obstruction.
As differential diagnoses, other intranodal vascular proliferations have to be considered. Angiomatous hamartoma is characterized by a replacement of lymphoid tissue by haphazardly arranged vessels and smooth muscle bundles with or without adipose tissue within a fibrous stroma. 1 Nodal lymphangiomatosis has been exclusively described in women and consists of smooth muscle cells forming fascicles and sheets around anastomosing ectatic vascular spaces. 1 Hemolymph nodes that resemble morphologically lymph nodes contain blood within the sinuses. 5,20 Finally, nodal hemangiomas have to be considered. 1 None of these differential diagnoses have been described in cats.
In conclusion, immunohistochemistry of the plexiform vasculopathy in cervical lymph nodes of 4 cats pointed out a lymphatic origin of the intranodal vascular proliferations using Prox-1 as a specific marker for lymphendothelial cells. This finding may contribute to the still unclear pathogenesis of plexiform vasculopathy in cats.
Footnotes
Acknowledgements
We thank Bettina Buck, Petra Grünig, Kerstin Rohn, Christiane Namneck, and Claudia Hermann for excellent technical assistance. The authors are further grateful to Uta Rönneburg, Sabine Zeibig-Biehle, Malgorzata Bielecki, Dr. Bettina Habenbacher, Dr. Birgit, and Jörg Keßler for their cooperation in sampling and examining the animals and their assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
