Abstract
In 5 Japanese Black steers (2-2.4 years old) that originated from 5 different feedlots, the livers were found at slaughter to have multiple nodular or cordlike lesions (5 steers) and an extensive fibrotic area (1 steer). Microscopic changes included extensive fibroplasia in the portal tracts and chronic proliferative endophlebitis-like lesions confined to the portal vein branches. Fibroplasia was much more prominent in the macroscopic fibrotic lesion of 1 steer. Portal vein branches presented irregular variciform dilation of the vascular lumen and fibroplastic changes in the subendothelial areas that showed occasional hemorrhage and were simultaneously infiltrated with large numbers of mast cells and moderate to large numbers of eosinophils. Within these subendothelial regions, not only did mast cells exhibit cytologically atypical features, but they also formed multifocal nodules. The venous lesions may represent a variant of mastocytosis with specific involvement of the hepatic portal vein branches in cattle.
Five 2- to 2.4-year-old Japanese Black steers (case Nos. 1-5) were brought to an abattoir from 5 different feedlots. Each animal was conventionally slaughtered on separate days over a 9-month period. Macroscopically, the livers of all 5 steers were mildly enlarged and contained multiple, individual-to-coalescing, widely distributed, firm, white-to-grayish foci, usually < 5.5 cm in diameter. These lesions were unencapsulated, irregularly outlined, and well delineated from the surrounding hepatic parenchyma, exhibiting a nodular or thickened cordlike appearance on cut sections. Frequently, 1 or more angioid lumens with bloody content were embedded in the lesions. In addition, 1 steer (case No. 1) had a 12 × 15 cm relatively well-delineated, white, fibrotic area in the subcapsular region of the right lobe (Fig. 1). This area contained small to medium-sized, convoluted, luminal structures, sometimes containing bloody content (interpreted as blood vessels). Neither vascular thrombosis nor parasites were demonstrated in any sites of the liver. Extrahepatic visceral organs were unremarkable.
Samples were collected from the major organs, including liver and regional lymph nodes. All samples were fixed in 10% neutral buffered formalin, embedded in paraffin, cut at 4 μm, and stained with hematoxylin and eosin. Selected sections were stained with periodic acid–Schiff (PAS), toluidine blue, Gram’s stain, Ziehl-Neelsen stain, and Gomori methenamine silver. Immunohistochemical investigations of the liver samples were performed using an EnVision horseradish peroxidase–labeled polymer immunoperoxidase staining procedure (Dako, Glostrup, Denmark). Rabbit polyclonal anti-human IgG (diluted 1:5000; Dako) and rabbit polyclonal anti-human IgM (diluted 1:5000; Dako) were used as primary antibodies. The immunoreaction was visualized by 3’-3’-diaminobenzidine-tetrahydrochloride (Dako) as chromogen. Sections of bovine tonsil were used as positive controls. Bacteriologic examination was focused on material taken aseptically from each liver. Collected samples were plated on 5% sheep blood agar and MacConkey agar and incubated aerobically and anaerobically.
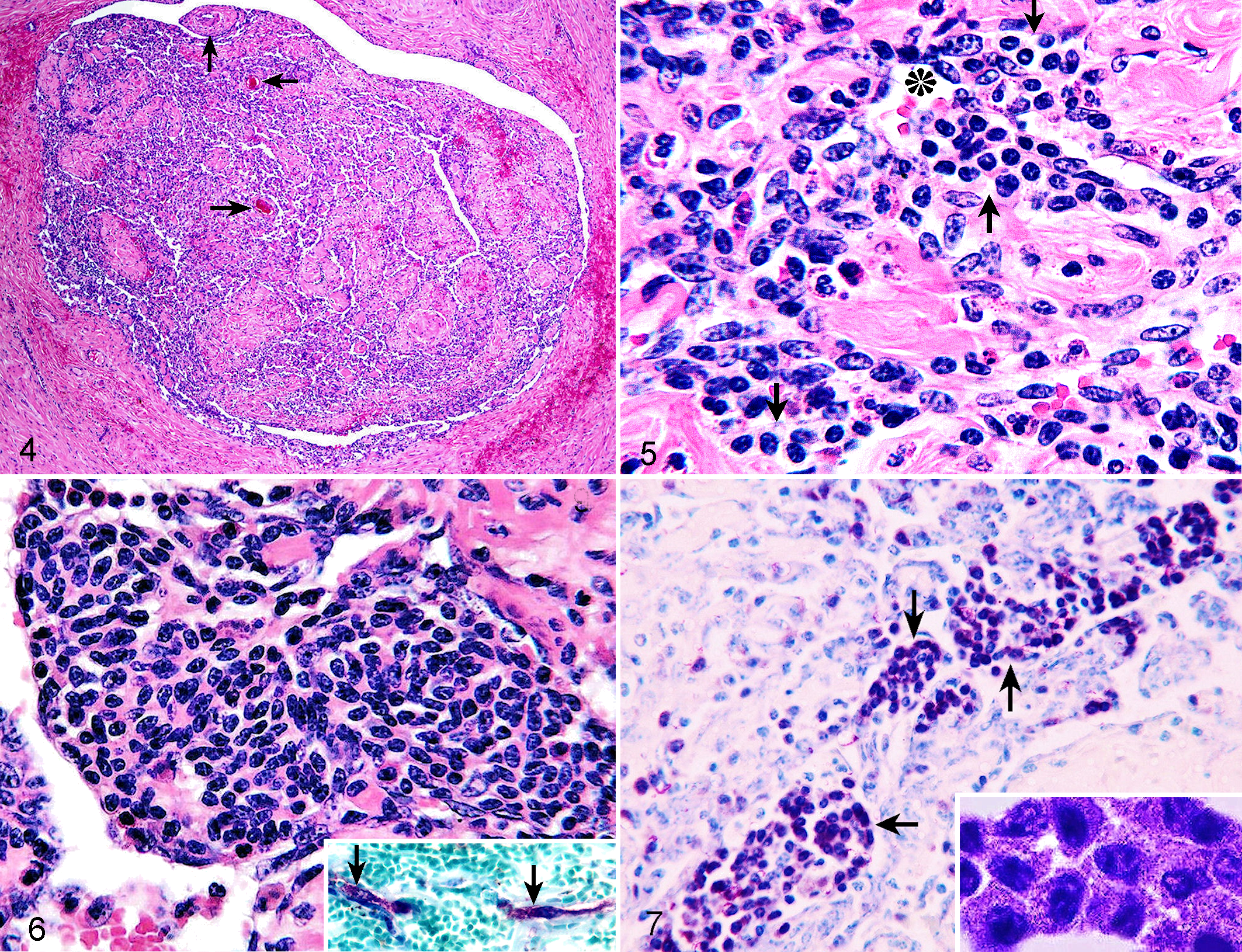
Histopathologically, the livers of the 5 steers revealed qualitatively similar lesions. The most prominent lesions were found in the hepatic portal regions that showed extensive and irregular fibrocollagen deposition, giving rise to the distortion or disappearance of normal portal triad profiles (Fig. 2). In the macroscopic fibrotic area of 1 steer (case No. 1), these fibroplastic lesions were much more severe, to such an extent that little hepatic parenchyma remained. Also prominent in all steers’ livers was disruption of the normal contour of the portal vein branches owing to irregular variciform dilation of the vascular lumens with aberrant intimal proliferation (Fig. 3). Endothelial cells of these veins exhibited occasional swollen nuclei, whereas the tunica intima displayed a unique feature characterized by collagenization and fibroblast proliferation that involved the subendothelial region, forming hemangioendothelioma-like, villous, papillary, or polypoid projections. With increasing severity of fibroplasia, these intimal lesions showed an irregular nodular or folded texture, giving rise to narrowing or total-to-subtotal obliteration of the lumens. Sometimes, recanalization was identified by the presence of small, endothelial-lined, erythrocyte-filled vessels in areas of nodular intimal fibrosis (Fig. 4). In addition, those subendothelial areas undergoing fibroplasia were diffusely infiltrated with large numbers of mast cells and moderate (case No. 1) to large (case Nos. 2-5) numbers of eosinophils (Fig. 5). Mast cells often formed multifocal nodules (0.1-1.7 mm in diameter), which protruded into the vascular lumen (Fig. 6). Cytoplasmic granules of mast cells stained positively with PAS and metachromatically with toluidine blue (Fig. 7). Mast cells, especially those seen within larger nodules, had variably sized, round, oval, or elongated nuclei with 1 to 2 prominent nucleoli and small to moderate amounts of finely granular, pale, or slightly basophilic cytoplasm (Figs. 5, 6; Fig. 7, inset). In addition, spindle-shaped mast cells were observed in the portal tract connective tissue, indicating atypical morphology (Fig. 6, inset). The mast cells had very few mitotic figures. Neutrophil infiltrates were not associated with lesional hepatic portal vein branches. Occasionally, subendothelial focal hemorrhage was present. Thromboembolic changes could not be seen. In general, the underlying tunica media became thin or distorted depending on the configuration of dilated vascular lumens, showing variable thickness of the tunica media so that it appeared as small muscular islands or peninsulas in many places. This smooth muscle of the tunica media exhibited vacuolar degeneration, manifesting as single, small, clear vacuoles within smooth muscle fibers.
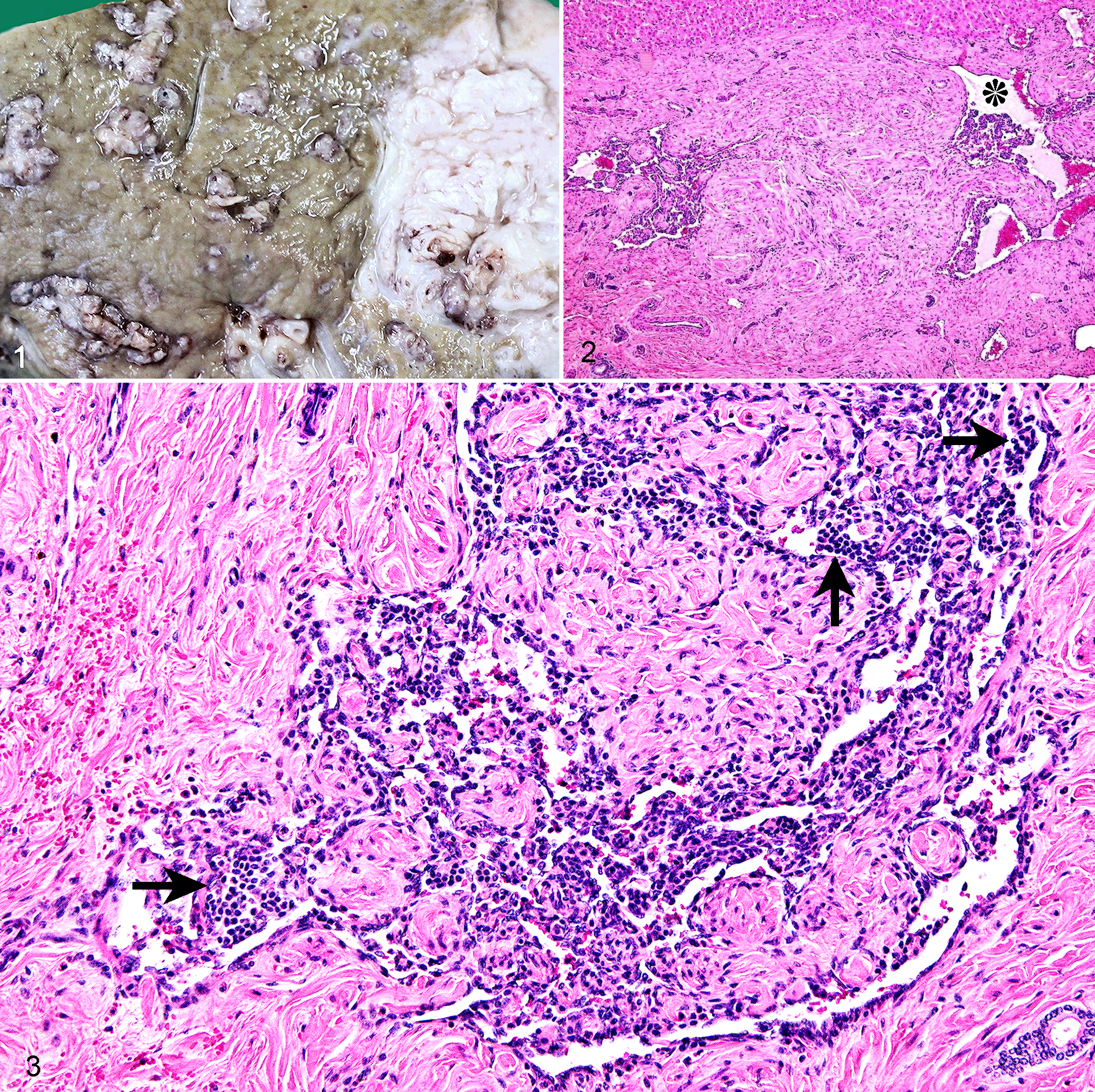
In the fibroplastic hepatic portal regions, neither visible parasites nor other pathogenic organisms, such as fungi and protozoa, were detected within the lumens of the vessels, interlobular bile ducts, or elsewhere. Histochemical stains (Gram’s stain, PAS, Ziehl-Neelsen stain, and Gomori methenamine silver) were negative for microorganisms. Interlobular bile ducts were slightly hyperplastic. Some of the hepatic arterial branches showed an infiltration of the walls by a few mast cells and eosinophils, but others were generally unremarkable. Hemorrhagic foci were noted in the portal tract connective tissue. The terminal hepatic venules (central veins) and hepatic sinusoids were unremarkable. Large numbers of mast cells, moderate (case No. 1) to large (case Nos. 2-5) numbers of eosinophils, and small numbers of hemosiderophages diffusely infiltrated the portal tract connective tissue. Periportal hepatic lobules adjacent to the fibroplastic portal tracts revealed compression atrophy of the hepatic cords (case Nos. 1-5), hydropic degeneration of the hepatocytes (case Nos. 1-5), and dilation of the bile canaliculi with rare bile plugs (case No. 1). Extrahepatic organs and tissues displayed no vascular lesions or any other significant changes, except for splenic hemosiderosis (case Nos. 1-4) and a mild lymphoplasmacytic infiltration in the interstitial connective tissue of the kidneys (case Nos. 2, 3).
By immunohistochemical analysis, deposition of the IgG and IgM was not recognized within the wall of the lesional portal vein branches in any of the 5 animals, whereas appropriate staining was found in positive control tissue. Bacteriologic examination performed on liver samples (case Nos. 1-5) failed to identify any likely pathogen.
Distinctive venous lesions associated with eosinophil infiltrates and irregular intimal fibrosis reflective of chronicity were somewhat similar to previously reported descriptions of parasite-associated lesions in the hepatic portal veins of different hosts. 11,19 However, the current steers’ livers did not have evidence of infestations by schistosomes, liver flukes, or other parasites or recognizable changes consistent with parasitic granulomas with worms or cuticle debris, 11,19 excluding the likelihood of parasitism. It has previously been described that infection with Eperythrozoon or Cryptosporidium organisms induces mast cell proliferations in pigs 2 or calves, 9 respectively. In the present 5 steers, histochemical staining procedures and bacteriologic culture for microorganisms did not reveal any agents. Immunopositivity for IgG and IgM was not demonstrated, possibly excluding an immune-mediated mechanism. Although not identified, another factor must have been implicated in the pathogenesis of this hepatic condition, taking into account that mast cells displayed cytologic atypia and formed multifocal nodules in the lesional hepatic portal vessels. Features of such mast cell populations could be distinguished from those recognized in conventional reactive/hyperplastic mast cell proliferations and, thus, were most consistent with a variant of mastocytosis.
The term hepatic mastocytosis would be appropriate for the condition described in this report. As is the case with the current steers, human systemic mastocytosis in the World Health Organization classification is primarily based on the histologic demonstration of multifocal and compact mast cell infiltrates in the tissue, 7 which can be highlighted by either tryptase immunohistochemistry or another special stain (ie, toluidine blue or Giemsa). Furthermore, the 4 minor criteria recommended for this entity include (1) > 25% of mast cells with cytologic atypia, (2) KIT mutation at codon 816, (3) CD117-positive mast cells that aberrantly express CD2 and/or CD25, and (4) serum tryptase persistently > 20 ng/ml. 6,7 According to the World Health Organization classification, either 1 major and 1 minor or 3 minor criteria have to be fulfilled to establish the diagnosis of systemic mastocytosis. 6,7 Atypical morphology was recognized in those mast cells that formed multifocal nodules at the intimal regions of the hepatic portal veins, and mast cells in the portal tract connective tissue displayed spindle-shaped morphology that is frequently seen in mastocytosis, 18 giving sufficient evidence to meet 1 of the 4 minor diagnostic criteria for mastocytosis.
Of particular interest was fibroplastic change in the subendothelial areas of the intrahepatic portal vein branches. Mast cells may have stimulated the subendothelial fibroplastic lesions (mast cell–fibroblast interaction), 4,20 accounting for extensive fibroplasia in the regions of the portal tracts as well. Mast cells secrete transforming growth factor-β and tryptase, which stimulate fibroblast growth and fibronectin/collagen I synthesis. 4,20 An extensive fibrotic area observed in 1 steer (case No. 1) was considered the most advanced stage of such fibroplasia. However, it remains to be explained why, topographically, the development of mastocytosis lesions affected the subendothelial region of the tunica intima and was restricted exclusively to the intrahepatic portal vein branches while relatively sparing other hepatic regions or other organ systems/tissues.
The intralesional infiltration of eosinophils is a significant feature of the lesions, considering that eosinophil infiltrates or eosinophilia (with or without organ involvement) has almost consistently been documented in mast cell lineage disorders (both mastocytosis and mast cell neoplasms) in humans and domestic animals. 1,2,5,8,10
In domestic animals, most reported cases of mastocytosis were sporadic or isolated, published descriptions being centered on the skin, bone marrow, liver, spleen, kidney, intestine, or lymph node of dogs, pigs, horses, or cattle. 2,3,10,14,16 Even if mastocytosis may affect different organs, selective vascular system involvement is uncommon. There are few relevant descriptions in domestic animals, with only 1 report of a variant of systemic mastocytosis in a cow that involved almost all arterioles in the skeletal muscles. 15 In humans, whereas the majority of cases with mastocytosis are confined to the skin (urticaria pigmentosa), approximately 10% involve other organs or tissues, including the bone marrow, gastrointestinal tract, spleen, lymph node, or liver. 8,12,13,17 Similar, but not identical, to the observations made in the current bovine cases, some studies have previously described that some human patients with hepatic mastocytosis demonstrate hepatomegaly and exhibit a variety of lesions in the hepatic portal regions, including mast cell infiltration, inflammation, fibrosis, venous lesions, or veno-occlusive disease, with or without intrahepatic cholestasis, portal hypertension, or ascites. 13,17,21 The venous lesions such as those that occurred in the livers of the present steers have never otherwise been described in this species, representing a newly recognized variant of mastocytosis with specific involvement of the intrahepatic branches of the portal veins in cattle. The underlying etiopathogenic mechanism remains unresolved for this disorder.
Footnotes
Acknowledgement
I wish to thank the veterinary meat inspectors of the Hokkaido Meat Hygiene Inspection Center for collecting the material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
