Abstract
Cytokines are known to play important roles in a wide range of pathologic conditions spanning all organ systems in every species studied. As our knowledge of the physiology of individual cytokines expands and our ability to measure multiple cytokines in smaller biological samples increases, we gain more insight into the significance and function of each cytokine and the importance of cytokine networks. Previous studies that reported measurements of cytokine concentrations from serum or plasma in dogs with infectious, autoimmune, metabolic, endocrine, and neoplastic diseases yield an appreciation for the complexity of cytokine control and potential applications for cytokine measurements in the diagnosis, prognosis, and therapy of a variety of disease conditions. In this review, we highlight the benefits of multiplex cytokine analysis, summarize clinical and experimental reports that have used this technology in dogs, and discuss the strengths and limitations of data analysis for the interpretation of results in these studies. We describe how differences in technical acuity, data reporting tactics, statistical analysis, study population selection criteria, and cross-sectional experimental design methods may affect interpretation of results from this technology. We also suggest methods for analysis in future studies, such as reporting median fluorescence intensity values, analyzing the proportion of patients above population medians, and performing longitudinal studies.
Cytokines directly or indirectly affect every cell in the body. Due to their importance in the balance between health and disease, the biomedical literature contains a plethora of articles describing their function in a variety of species and disease conditions. Most manuscripts highlight the role of a particular cytokine or family of cytokines in the pathogenesis of a specific disease or category of diseases while other reviews offer a broader overview of cytokine function. 5,11,21,48,54,65,67,89 Recent clinical work in cytokine analysis is centered on efforts to understand the interplay between multiple cytokines in what has been previously referred to as cytokine networks 64,87,98 or cytokine signatures. 44,56,69,78 Due to the complexity of each cytokine’s actions 82 and the effects of cytokines on each other, it has been proposed that measuring the concentrations of multiple cytokines in any given health condition would provide more relevant information than the selective measurement of any single cytokine.
Technological advances have significantly improved our ability to measure multiple cytokines concurrently in very small sample volumes, offering great promise for diagnostic, prognostic, or therapeutic conclusions. 15 The most comprehensive, commercially available, multiplex assay that has been validated for the measurement of canine cytokines measures up to 13 proteins (Milliplex MAP Canine Cytokine/Chemokine Magnetic Bead Panel CCYTOMAG kit, EMD Millipore, USA). This assay, heretofore referred to as the Milliplex Canine Cytokine Panel, and other multiplex technologies have already yielded valuable insight in the study of canine diseases. Here we review the advantages and potential benefits of multiplex cytokine analysis, summarize clinical reports that have used this technology in dogs, and discuss the analysis and interpretation of cytokine data obtained from multiplex cytokine assays.
Overview of Cytokine Function
Previous reviews state the importance and clinical implications of cytokine measurements in the diagnosis and management of various diseases. 12,99 This body of knowledge is constantly expanding, as is the discovery of new cytokines and other regulatory molecules that influence cytokine function. In this section, we limit our review to general ideas about the relevant and well-accepted aspects of cytokine function.
Cytokines exert their function on any cell that bears receptors specific to them and may act in autocrine, paracrine, or endocrine fashion. Cytokine production is genetically, epigenetically, and post-transcriptionally controlled, and the net effect of an entire cytokine signaling cascade ultimately determines each cytokine’s effects in the balance between health and disease. Although the absolute quantity of any cytokine would be proportionally related to its effect, cytokine function also depends on the expression and quantity of specific receptors on target cells and the relative activation of downstream signaling proteins, which are all independently regulated.
Traditionally, cytokines have been classified as either pro-inflammatory, anti-inflammatory, chemotactic, or growth regulating. Evidence suggests, however, that many cytokines carry out more than 1 of these functions either directly or indirectly; thus, the overall function of a particular cytokine depends more on the general milieu of all cytokines and their regulators in the immediate tissue environment. Additionally, the functional kinetics of each cytokine, temporal changes in cytokine expression, the presence of cytokine inhibitors, and the variable half-lives of each protein will lead to differences in cytokine effects at specific tissue sites.
Cytokines play multiple, integral roles in the immune defense against infection and neoplasia, but they are also mediators of both inflammatory and autoimmune diseases. Thus, it cannot be said that having measurably increased or decreased concentrations of any cytokine is beneficial or harmful without also knowing much more about the specific clinical conditions under which the cytokine is being measured. Nonetheless, the study of cytokine concentrations in the dynamic phases of health and disease, especially when followed throughout the course of incidence, treatment, or resolution, has already yielded insights into possible diagnostic, therapeutic, or prognostic remove clause for clarity information in human medicine. 94
Similar efforts are currently underway in veterinary medicine. Cytokine analysis has contributed significantly to our understanding of the pathophysiology of sepsis in both human 88 and veterinary medicine. 44 To date, no single assay is used to definitively diagnose or differentiate between stages in systemic inflammatory response syndrome (SIRS), sepsis, severe sepsis, septic shock, and multiple organ dysfunction syndrome (MODS) for any species. 30 However, such studies may one day help identify these clinical presentations more readily and perhaps prevent disease progression in patients suffering from systemic inflammation and its consequences.
Insight from the analysis of cytokine networks also promises prognostic information to inform clinical decisions for patients with infectious and neoplastic diseases. 38,39,74 Currently, cytokine measurements have diagnostic applications as functional assays to measure the effectiveness of immunosuppressive therapy in dogs treated for immune-mediated diseases. 79,85 Furthermore, the therapeutic use of more specific cytokines or cytokine inhibitors has recently expanded in human medicine in lieu of global immunosuppressive therapies. In veterinary medicine, the most successful example of a specific cytokine-targeting therapy may be the use of a Janus kinase inhibitor targeting IL-31 in the treatment of atopic dermatitis. 16,17 Such diagnostic and therapeutic advances will depend on our ability to measure cytokine concentrations. This review will summarize the important findings from canine cytokine analyses that used multiplex cytokine technologies and highlight how these studies have laid a foundation that will lead to additional clinical outcomes.
Methods to Measure Cytokines
A variety of methods have been used to quantify cytokines in biologic samples. Among them, multiplex assays, single immunoassays, and bioassays offer specific advantages and disadvantages. Our current understanding of the complexity of cytokine regulation and function, along with new appreciation for the interplay between cytokines, has demonstrated a need for efficient concurrent analysis of multiple cytokines. 6,8,15,27,41,55 In response, technologies used to quantify cytokines in biological samples have improved to meet these needs. 14,53,86,93 Here we review and summarize the advantages and potential benefits of multiplex cytokine analysis techniques compared with other methods. 14,53,93
Multiplex Cytokine Analysis
There are presently 2 canine-specific multiplex technologies that have been utilized for published canine cytokine studies. Most referenced is the Milliplex Canine Cytokine Panel, a bead-based multiplex technology (akin to flow-cytometry methods) that is commercially validated to measure up to 13 canine-specific proteins simultaneously: GM-CSF, IFN-γ, IL-2, IL-6, IL-7, IL-8 (CXCL8), IL-15, IP-10 (CXCL10), KC-like protein (CXCL1/2), IL-10, IL-18, MCP-1 (CCL2), and TNFα (IL-4 was excluded from the revised canine panel in 2014 to provide optimal accuracy and performance). This panel provides the most comprehensive picture of canine cytokine levels in health and disease. The second commercially available canine-specific multiplex technology is the QuickPlex electrochemiluminescence planar micro-spot (akin to micro-array methods) assay (MSD QuickPlex, Meso Scale Discovery, USA), subsequently referred to as the QuickPlex Assay. This assay is capable of concurrently measuring the concentrations of up to 4 canine cytokines: IL-2, IL-6, IL-8, and TNFα. Both multiplexed cytokine technologies have been found to be generally as reliable as standard individual enzyme-linked immunosorbent assays methods, 24,28 providing quantification by comparison against a standard curve. 53 The major advantage of both assays is that they allow for the simultaneous measurements of more than 1 protein in as little as 25 µL of a biological sample. This advantage is important in veterinary medicine because of the small size of some patients and the potential stress associated with restraint during sample collection. The current limitations of these techniques in veterinary medicine are the availability of validated and commercially available monoclonal antibodies and the relative lack of commercial potential in developing more of these assays.
Single Enzyme-Linked Immunosorbent Assays
Although this review focuses on multiplex assays, the majority of clinical data obtained thus far from any species employed individual single enzyme-linked immunosorbent assays (ELISAs) to measure cytokine concentrations. Some ELISA methods have lower limits of detection, and most are relatively well validated, easy to perform without specialized equipment, and cost-efficient when 4 or fewer cytokines are measured simultaneously. Due to their more common use, ELISAs are still considered the gold standard in cytokine analysis. Their main disadvantage is that multiple assays are needed to obtain information about more than 1 protein. This also requires increasing amounts of the biological sample, reagents, and time. Nevertheless, these assays are frequently used, especially in studies focused on only 1 cytokine or those that include cytokines not measured by modern multiplex technologies. Certainly, many relevant cytokines are still excluded from canine multiplex assays, limited by the availability of canine-specific antibodies and the quantities of each of the proteins of interest in relation to others in the sample. For example, while it would be ideal to include IL-1ß, TGF-ß, C-reactive protein, IL-17, or other well-characterized analytes in the multiplex assay, these have not been validated. Consequently, more than 1 technique is often utilized to evaluate samples from canine clinical studies.
Bioassays
Indirect measures of cytokine activity in canine plasma or serum 20,60,71,91 have also been described. Bioassays make use of inexpensive ingredients and can be evaluated with a standard plate reader, following determination of a standard curve. These techniques have been leveraged to successfully detect cytokines such as TNFα in patient samples that have been diluted more than 400-fold. 1 As a result, several investigators using this method report lower limits of detection for canine TNFα as small as 0.5 ng/mL. 33 Such excellent sensitivity could make the bioassay method more suited for detecting baseline levels of TNFα (or other cytokines found in low abundance) in healthy and diseased canine subjects. Additionally, bioassays have the advantage of reporting only functional activity and therefore may provide a more relevant measure. For example, if cytokine inhibitors are present in the sample, then the combined analysis of cytokine concentrations using ELISA and functional bioassays, will give a clearer picture of the changes occurring in any disease.
In summary, a variety of methods for the measurement of systemic cytokine bioactivity remain in use, including traditional ELISAs, bioassays, and multiplex technologies. Each method has distinct advantages and can provide information complementing data obtained by the other techniques.
Cytokine Measurements in Canine Serum/Plasma During Health
The inclusion of samples from healthy dogs is essential to establishing a baseline or “normal” reference for each study. However, age, 9,34,91 breed, 68,77,80,103 and even brain lateralization 75,76,90 have each been associated with statistically significant differences in cytokine measurements in dogs. Because multiplex cytokine data exhibit such wide variability, these published findings emphasize the importance of having normal samples restricted for inclusion to match the breed, age, and sex of the study population. Focusing the study on a limited age range or specific breed is essential and may reduce variability of the data but also limits the relevance of results to the general population. While matching experimental and control populations is ideal, this can also be challenging in clinical settings.
Published studies of multiplex cytokine analysis have already generated several data sets from “healthy control” dogs. Importantly, the criteria for inclusion (as healthy controls) and the level of description for inclusion criteria in each manuscript vary to some degree between studies. Nevertheless, every small group represents a population of dogs considered to be “healthy” by the investigators of each study. We compared published “healthy dog ranges” for each of the cytokines measured in the Milliplex Canine Cytokine Panel (Table 1) and the QuickPlex assay (Table 2). We found only 3 reports that used the QuickPlex Assay and reported numerical values for healthy dogs (Table 2). 34,49,96 While these results may appear to be more consistent between cohorts, the low number of comparisons should be considered.
Cytokine Concentrations (median and range) Measured in Healthy Control Dogs Using the Milliplex Canine Cytokine Panel (bead-based immunoassay multiplex technology).a
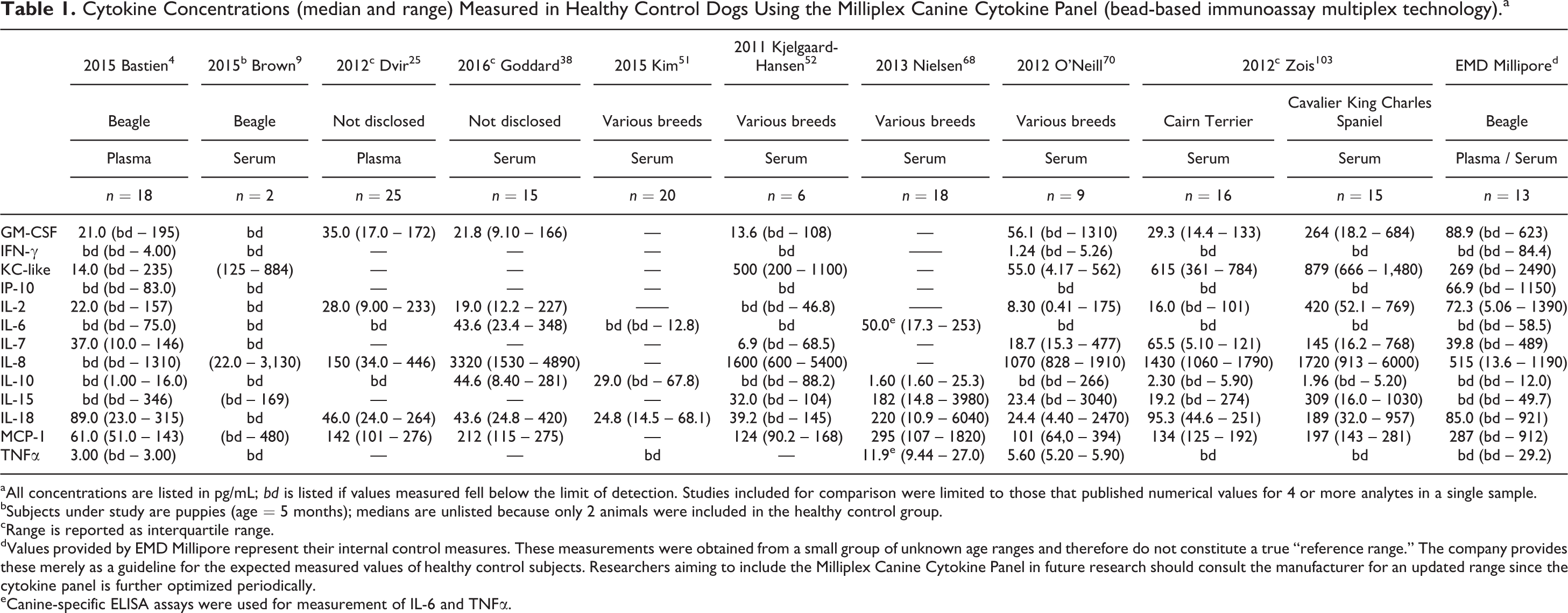
aAll concentrations are listed in pg/mL; bd is listed if values measured fell below the limit of detection. Studies included for comparison were limited to those that published numerical values for 4 or more analytes in a single sample.
bSubjects under study are puppies (age = 5 months); medians are unlisted because only 2 animals were included in the healthy control group.
cRange is reported as interquartile range.
dValues provided by EMD Millipore represent their internal control measures. These measurements were obtained from a small group of unknown age ranges and therefore do not constitute a true “reference range.” The company provides these merely as a guideline for the expected measured values of healthy control subjects. Researchers aiming to include the Milliplex Canine Cytokine Panel in future research should consult the manufacturer for an updated range since the cytokine panel is further optimized periodically.
eCanine-specific ELISA assays were used for measurement of IL-6 and TNFα.
Cytokine Concentrations (median and range) Measured in Healthy Control Dogs Using the QuickPlex Canine ProInflammatory Panel 3 Ultra-Sensitive Kit (electrochemiluminescence planar micro-spot based multiplex technology).a
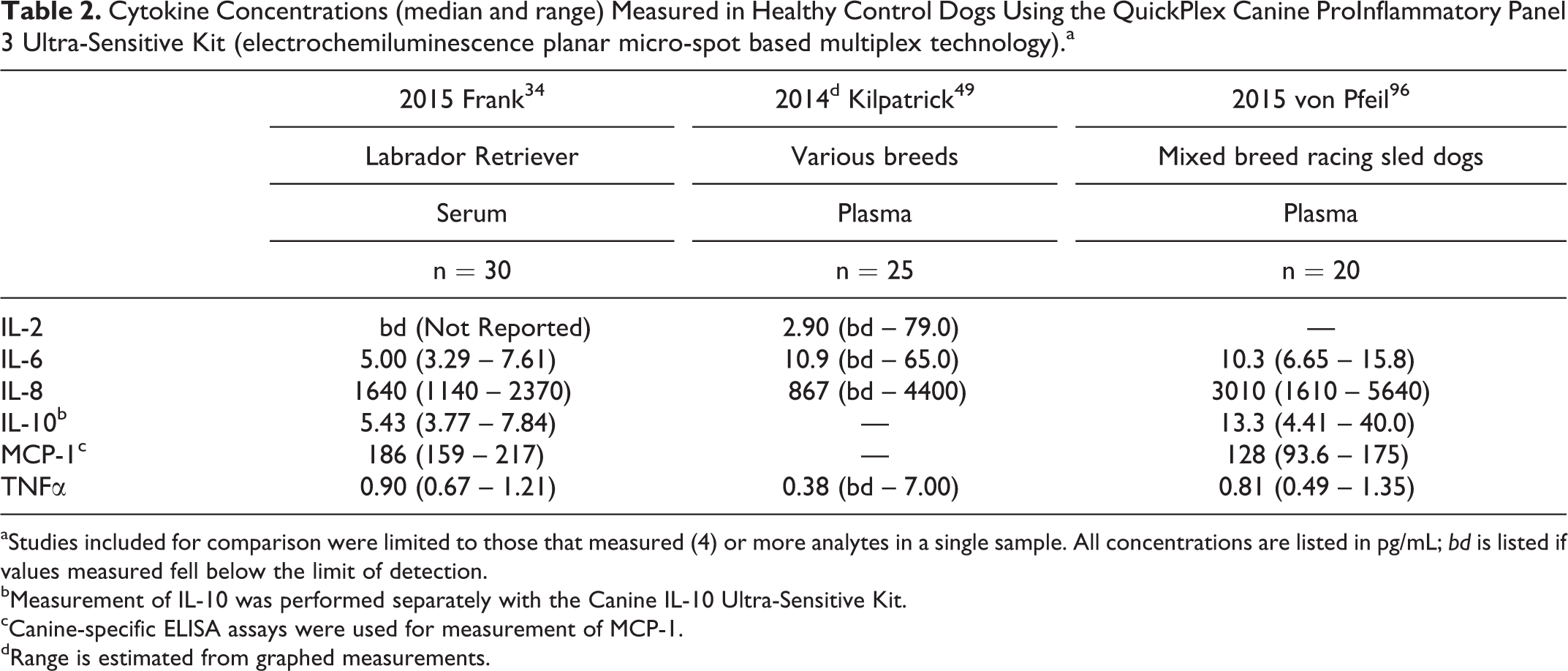
aStudies included for comparison were limited to those that measured (4) or more analytes in a single sample. All concentrations are listed in pg/mL; bd is listed if values measured fell below the limit of detection.
bMeasurement of IL-10 was performed separately with the Canine IL-10 Ultra-Sensitive Kit.
cCanine-specific ELISA assays were used for measurement of MCP-1.
dRange is estimated from graphed measurements.
Analysis of these data has important implications for future work. As shown, the medians and detection limits vary widely among cytokines within and across both methods used, with at least 1 but not all investigators reporting median cytokine levels below detectable limits for all cytokines in some cohorts. Even within a population of healthy dogs, the range of cytokine measurements can vary by 3 orders of magnitude. 44,68 Though exact measured cytokine concentrations were not published, Safra et al 84 tabulated the differences between minimum and maximum concentrations measured for 102 healthy dogs using the Milliplex Canine Cytokine Panel and reported formidable differences within 1 study for multiple cytokines.
While the sensitivity of multiplex assays is often not low enough to detect baseline measurements in healthy dogs, small and statistically significant changes in these baseline measurements are sometimes claimed to distinguish healthy and diseased populations. The broad territory of “healthy” baseline measurements, however, should signal future investigators to expect large (and perhaps not normal) natural distributions in systemic cytokine levels among study or control populations. Moreover, such variation indicates that a wide variety of health factors, including age, breed, prior or subclinical disease, physical fitness, and other variables, may skew study results. This is particularly relevant to veterinary studies that traditionally cannot boast large population sizes. These findings have 2 important implications for future work. First, they help to reiterate the importance and requirement for direct comparison to “normal cohorts” in future studies. Second, they suggest that interpretation of previous “normal” data may in fact deserve a closer look. For example, when results suggest a significant difference between 2 small populations is greater than the difference noted among all the populations considered clinically normal, one would be confident interpreting those results as being clinically significant. Conversely, when results comparing 2 small populations is less than the overall variability among populations considered clinically normal, the interpretation of these results may be more difficult.
Cytokine Measurements in Canine Serum/Plasma During Disease
We conducted a literature review of findings obtained from diseased canine cohorts and summarized important findings obtained using the Milliplex Canine Cytokine Panel. In the process of summarizing these findings, we gained an appreciation for the wide variety of statistical methods and experimental approaches used with this technology. This insight, along with deeper ramifications for future applications and analysis, are described in the next section. For this review, we searched PubMed, using the veterinary science filter, for manuscripts with the keywords canine and each of 13 cytokines that have been validated for measurement using the current Milliplex Canine Cytokine Panel. Here we describe the most significant findings from these manuscripts and when relevant, compare these results to those obtained with other methods of cytokine measurements. Articles excluded from consideration were research efforts that used recombinant canine cytokines as vaccine adjuvants, cytokine treatment, systemic inflammatory changes measured in response to pharmaceuticals or intervention treatments, in vitro production of cytokines for canine cells in culture (eg, macrophages, peripheral blood mononuclear cell, and immortalized cell lines), production of cytokines in species other than dogs, and local inflammatory activity including mRNA expression of cytokines in tissues (eg, the brain, spleen, dermis, endometrium, and mucosa, as well as other isolated sample mediums such as synovial and cerebrospinal fluid). Furthermore, manuscripts were only included if measurements were taken in canine serum or plasma. Twenty-two published manuscripts utilized the Milliplex Canine Cytokine Panel to measure cytokines in the serum or plasma of dogs (Table 3). Salient findings are described in the following sections according to broad categories of disease, including infectious, inflammatory/immune-mediated, endocrine, neoplastic, and metabolic conditions.
Published Studies Using the Milliplex Canine Cytokine Panel for the Measurement of Systemic Cytokine Concentrations in Canine Serum or Plasma.
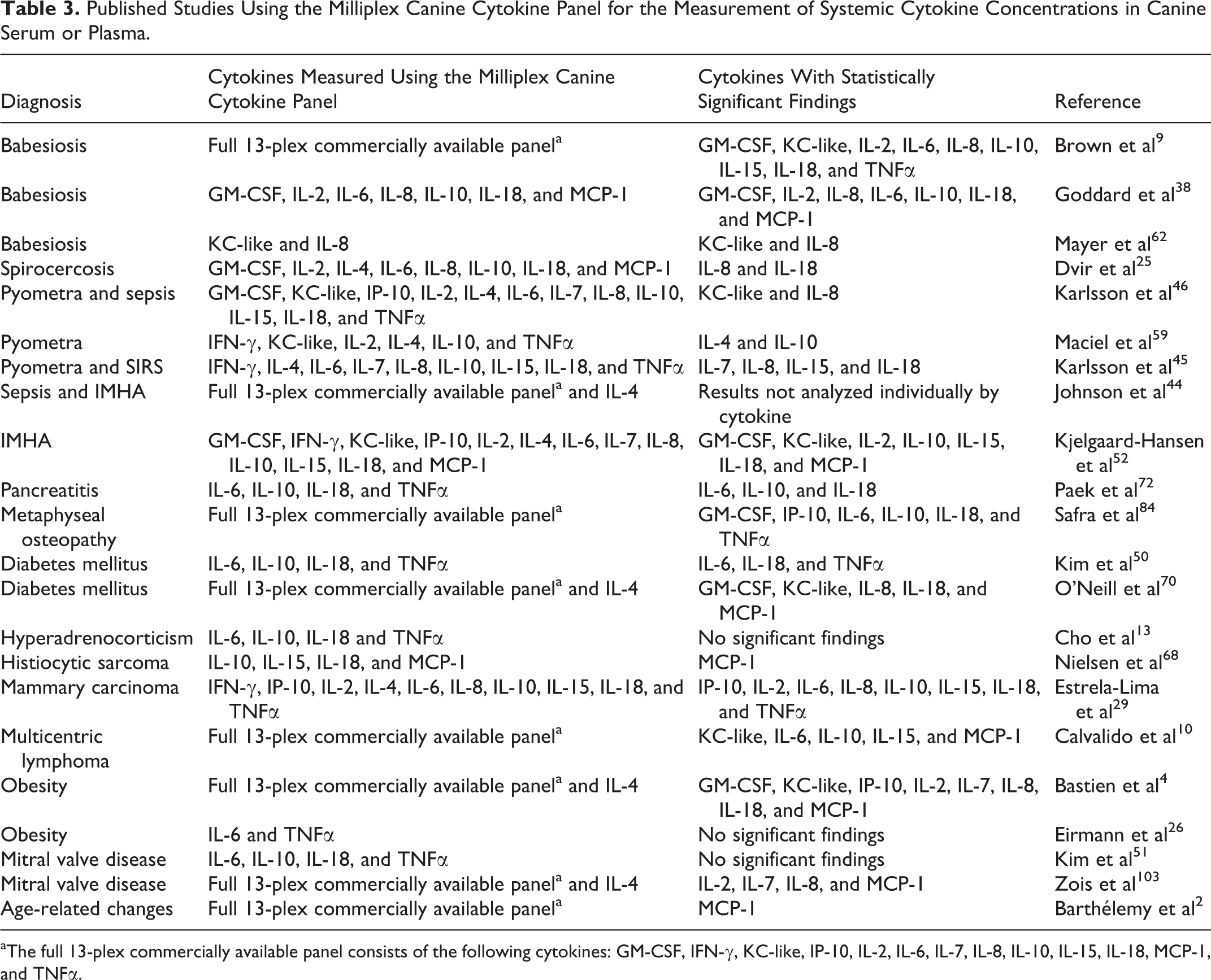
aThe full 13-plex commercially available panel consists of the following cytokines: GM-CSF, IFN-γ, KC-like, IP-10, IL-2, IL-6, IL-7, IL-8, IL-10, IL-15, IL-18, MCP-1, and TNFα.
Infectious Diseases
Systemic cytokine concentrations have been investigated in several infectious diseases of dogs, using a wide variety of methods. Dogs with viral (canine distemper 35 and parvovirus 71 ), parasitic (demodicosis 31 , trypanosomiasis, 35,40 spirocercosis 25 , leishmaniasis, 19,22 and babesiosis 9,38,104,105 ), and several bacterial diseases have undergone sampling for several systemic cytokines. Across these studies and methods, there has been at least 1 significant difference noted for all but 1 (IP-10) of the cytokines measured by the current Milliplex Canine Cytokine Panel. Differences in IL-18, 9,25,38 IL-6, 9,19,38,105 and TNFα 9,40,71,73,104 were the most commonly measured cytokine changes in dogs with parasitic diseases.
Babesiosis
Three reports investigated dogs with Babesia infections using the Milliplex Canine Cytokine Panel. 9,38,62 IL-8 was the only protein measured in all 3 studies, and measurements displayed discordant conclusions, even when using the same assay for a single cytokine. Serum concentrations of IL-8 were increased in dogs infected with Babesia canis canis 62 and decreased in dogs infected with Babesia rossi 38 when compared to respective healthy controls. In cases of Babesia rossi infection, IL-8 concentrations showed a significant correlation with several hematologic parameters, including declining neutrophil counts, indicating possible prognostic value. While some systemic cytokine concentrations were not significantly different between infected and uninfected dogs, serum GM-CSF, IL-2, IL-6, and IL-18 concentrations were found to be significantly higher in dogs with Babesia rossi during more acute presentations (sick for less than 48 hours) than in dogs with more chronic presentations (sick for more than 48 hours). 38 Furthermore, serum concentrations of IL-6 and MCP-1 were statistically different between survivors and nonsurvivors. IL-6 also inversely correlated to serum total triiodothyronine concentrations, suggesting some prognostic value. This finding is consistent with the marked increases in IL-6 concentrations that were noted in 2 dogs experimentally infected with Babesia. 9
In addition to the intrinsic variability posed by the use of different organisms, study cohorts, and experimental methods, these studies have little overlap in the cytokines measured and onset/duration of disease and sampling methods between cohorts. Further studies are therefore needed before applying any clinical conclusions regarding the significance of cytokine measurements in dogs with babesiosis. These studies do, however, provide insight into the pathogenesis of this infection and support the need for careful evaluation of systemic markers of inflammation in response to parasitic infections, suggesting a key role for IL-6 and IL-8 (among other cytokines) in canine babesiosis. The difference between studies points toward the importance of cytokine kinetics and suggests that special attention should be paid to the timing of sampling when comparing clinical populations with infectious diseases.
Spirocercosis
Canine plasma samples from almost 80 dogs infected with the nematode Spirocerca lupi were analyzed in an effort to investigate the cytokine milieu in this disease and its association with neoplastic transformation. 25 Ultimately, only plasma concentrations of IL-8, a potent chemoattractant for neutrophils, were found to be significantly elevated between infected dogs and healthy controls. The high concentrations of IL-8 observed in S. lupi–infected dogs are consistent with previous evidence of neutrophil involvement and a sustained systemic response. Since inflammatory nodules from chronic S. lupi infections undergo neoplastic transformation, infected dogs were further subdivided into those with evidence of neoplastic transformation and those with only inflammatory lesions. Among all cytokines tested, IL-18 was shown to be highest in the non-neoplastic group, measuring lower in the control group (uninfected), and lowest in the neoplastic group. IL-8, in contrast, sequentially increased between control, non-neoplastic, and neoplastic clinical groups, but differences between the 2 infection groups were not statistically significant. Nonetheless, the trend observed mimics findings in human conditions such as adenocarcinoma and Barrett’s esophagus, where increases in concentrations of IL-8 have been linked to the progression of neoplastic transformation. Again, these studies point toward the need for further work in the analysis of IL-8 and possibly IL-18 in parasitic infections.
Bacterial infections and sepsis
One of the most clinically relevant applications for the measurement of systemic cytokine concentrations is their use as potential biomarkers for the diagnosis of sepsis and SIRS. Four clinical studies and 1 experimental study have taken advantage of the Milliplex Canine Cytokine Panel to ask questions about this potential application for dogs. 32,44 –46,59 In 1 study, serum KC-like protein concentrations were significantly increased in dogs with pyometra compared to healthy controls, and concentrations of this biomarker could be measured in more than 90% of dogs tested. For dogs with pyometra, the concentration of KC-like protein was significantly higher in dogs that had criteria of sepsis than those without sepsis, despite the low number of the latter. High concentrations of KC-like protein significantly correlated with elevated CRP and IL-8 concentrations, complete blood count results, and morbidity (days hospitalized), suggesting a potential diagnostic or prognostic application of this protein. 46 Presently, the proteins in canine serum that bind antibodies against murine KC have not been characterized, hence the term KC-like. Sequence analysis suggests that canine CXCL7, CXCL5, and IL-8 have the highest homology to murine KC genes. 46 As such, the application of multiplex technology in canine samples may contribute to the discovery of new cytokines in dogs.
Importantly, in similar studies, other investigators found no difference in KC-like protein concentrations between dogs with pyometra and other healthy or pregnant dogs. 59 Using the Milliplex Canine Cytokine Panel, Maciel et al 59 reported significant differences in the concentrations of IL-10 and IL-4 in dogs with pyometra but not KC-like protein. Similarly, while IL-10 and IL-4 were also measured in the studies by Karlsson et al, 45,46 investigators failed to report significant differences between their cohorts. Instead, IL-10 and IL-4 were detected in less than 28% and 3% of clinical samples (respectively) in 1 study 45 and 74% and less than 10% in the other. 46 These limitations in method sensitivity are in agreement with other manuscripts utilizing the same method. 4,13,25,32,68,70,103
We suggest that differences in the methods for statistical analysis may account for some of the discrepancies between these conclusions; currently, there is no standard approach for the consideration of samples that fall below the limit of detection. In some papers, investigators used zero to represent the concentration of a cytokine that falls below the limit of detection. 46,52,70 In others, a value that is half 4 or the full value 9,38,68,103 of the limit of detection was used to represent these samples for statistical analysis. Still others compared the number of detectable measurements in different cohorts to arrive at conclusions of statistical difference. 50 A standard approach may be useful when comparing measurements of cytokine concentrations given the frequency with which samples fall below the limit of detection in both healthy and affected dogs.
While clinical studies of infectious diseases in dogs have apparently not yielded consistent results, the same is true in human studies. The results suggest that more work is needed to determine the best clinical applications for cytokine analysis in humans and dogs with systemic inflammation. This notion is supported further by experimental models of sepsis, such as those of Floras et al, 32 who showed several cytokines (IL-6, IL-8, IL-10, TNFα, and KC-like protein) were induced to significantly higher levels than baseline within 4 hours of an lipopolysaccharide injection, using the Milliplex Canine Cytokine Panel on canine serum samples. The question is not whether cytokine changes contribute to or reflect the presence of infectious and inflammatory conditions; the question is how to best detect, interpret, and quantify these changes to yield clinically relevant applications. Novel approaches to cytokine analysis attempt to compare cytokine groups, 44 yet much work remains to validate and apply these findings in the veterinary clinic.
Noninfectious, Inflammatory, and Immune-Mediated Diseases
Systemic cytokine concentrations have also been measured in dogs with a variety of other noninfectious, inflammatory, and immune-mediated diseases such as pancreatitis, 72 inflammatory bowel disease, 63,92 brachycephalic syndrome, 77 pulmonary fibrosis, 80,81 and atopic dermatitis. 47,61 As seen in previous sections, the cytokines measured and the methods used for quantification and data analysis were not consistent between studies, making it difficult to make general conclusions about cytokine concentrations in inflammatory diseases of dogs. Four studies used the Milliplex Canine Cytokine Panel to investigate inflammatory signatures in immune-mediated hemolytic anemia (IMHA), pancreatitis, and metaphyseal osteopathy.
Immune-mediated hemolytic anemia
In the first study, Kjelgaard-Hansen et al 52 studied cytokine concentrations in 20 dogs presenting with IMHA and 6 healthy control dogs that were neither sex- nor age-matched. Dogs with IMHA had significantly higher serum concentrations of IL-2, IL-10, KC-like protein, and MCP-1 compared to the controls. When dogs with IMHA were segregated by 30-day survival rates, concentrations of IL-15, IL-18, GM-CSF, and MCP-1 measured at admission were found to be significantly different between IMHA survivors (n = 15) and nonsurvivors (n = 5). Of all cytokines measured, only MCP-1 and IL-18 were independently associated with mortality by multiple logistic regression analysis. Though IL-18 concentrations were not significantly different in diseased animals and controls, the significance of this finding was strong (P = .0001), suggesting a possible prognostic role for IL-18 and MCP-1 in ill dogs. These findings have not been strongly suggested in studies of humans with immune-mediated disease and may represent interspecies variations. 23,101 More recently, a study comparing cytokine changes in sepsis and IMHA suggested individual cytokine changes may be negligible or unable to differentiate between these conditions, but looking at cytokine groups may yield more valuable insight. Clinical applications of these findings await further research, as does our ability to interpret these novel methods for cytokine data analysis. 44
Pancreatitis
Paek et al 72 investigated cytokine signatures in dogs with pancreatitis, another common and clinically important inflammatory condition. Serum concentrations for IL-6, IL-10, IL-18, and TNFα were measured using the Milliplex Canine Cytokine Panel. Median concentrations for IL-6, IL-10, and IL-18 were significantly higher in 25 dogs with acute pancreatitis than 28 healthy controls. No significant differences in IL-6 concentrations were found between dogs with pancreatitis that survived and those that did not survive, although the highest concentrations of IL-6 were recorded in nonsurvivors. Similarly, data from this study also suggested that the differences observed in IL-18 concentrations were due to the presence of a distinct subset of dogs with pancreatitis that had extremely elevated IL-18 concentrations. 72 These observations lead us to question whether dogs with abnormally high cytokine concentrations represent a true subset of patients rather than mere statistical outliers. Regardless, IL-18 concentrations did not differ according to survival and were not found to lower significantly after clinical signs subsided.
Metaphyseal osteopathy
Safra et al 84 compared serum levels of inflammatory cytokines in 26 dogs with metaphyseal osteopathy and more than 100 control dogs. GM-CSF, IP-10, IL-6, IL-10, IL-18, and TNFα levels were significantly higher in affected dogs than controls and not significantly different in dogs in remission than in dogs with active disease. General immune profiles of both affected and recovered dogs were briefly compared to results obtained from children with similar autoinflammatory diseases to highlight the potential One Health link between species and document similarities in the pathogenesis of each disease. These findings may have clinical implications for the therapeutic management of dogs with metaphyseal osteopathy and place the dog as a potential animal model of similar conditions in humans. Prior to this study, dogs with metaphyseal osteopathy had been treated with a variety of approaches, including a wide array of anti-inflammatory strategies. These findings, however, aid clinical decisions by favoring stronger immunomodulatory therapeutic approaches (steroids or cyclosporine over NSAIDs).
In summary, these few studies do not suggest a particular prognostic or diagnostic value for systemic cytokine measurements over standard of care in inflammatory and immune-mediated diseases of dogs. Most support current therapeutic recommendations and advance our knowledge for future work that may confirm or challenge current paradigms. These results also serve to reiterate that there is a wide distribution of cytokine concentrations in the blood of individuals affected by any disease and identify unique clinical data and laboratory parameters that separate “outliers” in certain study populations. These potentially unique subsets within each data set suggest that further analysis of the “outliers” may yield deeper clinical insight in the future. Why do some dogs in each study express such widely different cytokine concentrations than the others? What can this tell us about disease susceptibility or response to therapy? Current data suggest that longitudinal studies and analysis of genetic predispositions are likely needed to answer some of these important clinical queries.
Endocrine Diseases
Within the field of endocrinology, systemic cytokine concentrations have been measured using the Milliplex Canine Cytokine Panel in dogs with diabetes 50,70 and hyperadrenocorticism (HAC). 13 Of the diabetes studies, 1 investigation aimed to compare diabetic dogs and healthy controls while another evaluated cytokines as prognostic indicators of diabetic ketoacidosis (DKA). Only IL-18 was higher for diabetic dog groups in both studies, which may indicate the need to further analyze the role of this particular cytokine in the pathogenesis of canine diabetes mellitus. In 1 study, IL-6 was detected in more diabetic than control dogs. Additionally, serum concentrations of IL-6 were significantly higher in diabetic dogs with concurrent diseases than in diabetic dogs without concurrent diseases, and IL-6 was the only cytokine that was significantly reduced with insulin treatment over the course of therapy. 50 It is important to note that despite similar experimental methods, IL-6 was below the limits for detection in all 27 dogs enrolled into the second study. 70 The different conclusions drawn by each investigator may be related in part to differences in enrollment criteria and the variable range of cytokine concentrations between cohorts. As indicated earlier, this wide variation was also noted within “healthy” canine cohorts. The discrepancy between results from these studies highlights how data interpretation can affect clinical conclusions.
Neoplastic Diseases
Systemic cytokines have also been measured in dogs with a variety of neoplastic diseases, including lymphoma, 43,74 mammary carcinoma, 18,29,36,58 histiocytic sarcoma, 68 and hepatic tumors, 66 but only 3 of these studies used the Milliplex Canine Cytokine Panel. Compared to healthy controls, concentrations of MCP-1 measured by this panel were significantly increased in Bernese Mountain dogs with disseminated histiocytic sarcoma. 68 Similarly significant elevations were detected in dogs with lymphoma, as measured by individual canine-specific commercial ELISA kits (R&D Systems, Minneapolis, MN). 74 Additionally, MCP-1 concentrations correlated with disease stage (albeit without statistical significance) and disease-free intervals, suggesting some prognostic value for the measurement of MCP-1 in canine cancer. 74
In contrast, there were discrepancies between bead-based multiplex technology and other experimental methodologies for other cytokines. For example, significant differences in IFN-γ and TNFα were shown to be associated with canine mammary carcinoma when this cytokine was measured using an individual ELISA method with the Milliplex canine-specific commercial kit (Cytomag 90 K, Milliplex), 58 whereas other investigators failed to detect similar differences using the Milliplex Canine Cytokine Panel in dogs with the same malignancy. 29 This lack of statistical significance was attributed to the wide variability between study participants in every cytokine measured, and direct comparisons did not show clear differences between control, malignant mammary carcinoma (MC), and benign mixed tumor (MC-BMT) groups. However, investigators constructed biomarker profiles that reported the percentage of dogs with higher than median levels for every individual cytokine. In this analysis, dogs in the MC-BMT and MC groups without histological evidence of metastasis had markedly lower frequencies of high concentrations of plasma IL-10 compared to those with lymph node metastasis. This finding was so evident that clinical investigators further interrogated IL-10 and its association with survival. Out of all 20 dogs with mammary carcinoma enrolled into the study, only 1 of 9 surviving dogs (11%) had concentrations of IL-10 higher than the median, but 8 of 11 (73%) dogs that died had higher levels. When these data were reorganized by survival rates, measured as number of days between surgical excision and the end of follow-up, Pearson’s correlation analysis confirmed the statistical significance of this finding (P = .0273). 29 In addition to providing keen clinical insight, these comparisons highlight the difficulties posed by the use of different technologies and statistical methods when comparing data from different clinical trials.
The tumor microenvironment is known to contain a unique milieu of cytokines and other signaling molecules that exert complex and contradictory effects—they promote or diminish neoplastic proliferation, recruit inflammatory infiltrates, or aid tumor evasion from the immunosurveillance system. When cytokine production from neoplastic conditions, or even by tumor cells, was demonstrated to markedly alter systemic levels of individual cytokines, multiplex immunoassays became powerful tools to discover new biomarkers that might guide diagnosis or predict postsurgical outcomes. These studies are comparatively in their infancy in veterinary medicine. Importantly, results thus far point toward considerable synergy and some divergence between canine and human diseases. For example, the importance of IL-10 (a prototypical immunosuppressive and anti-inflammatory cytokine) as an important prognostic indicator in canine cancers is not only indicated by the study described previously 29 but also by findings published from dogs with multicentric lymphoma. 10 While the significance of inflammatory cytokines in neoplasia has been well established, these canine studies shed light on a potential role for dysregulation of the anti-inflammatory cytokine IL-10 in neoplasia and highlight differences between species.
In these and many other studies, multiplex cytokine measurements demonstrated large variability within cohorts. Large extremes, representing a mixture of outlier measurements and a numerous remainder of measurements clustered near or below the immunoassay’s lower limit of detection, leave room for several interpretations.
Metabolic Disease/Obesity
There is mounting evidence that obesity is associated with a pro-inflammatory cytokine milieu that contributes to associated comorbidities and insulin resistance. The systemic cytokine milieu has been investigated in dogs with obesity 4,34,37 and during exercise, 96,97,102 but only 2 of these studies used the Milliplex Canine Cytokine Panel to investigate cytokine concentrations in obesity and monitored changes in cytokine concentrations during the course of weight loss. In the first study, only IL-6 and TNFα were measured, and both cytokines were most often under the level of detection. 26 The second study confirmed similar results for IL-6 and TNFα and also demonstrated that weight loss and decreasing body condition scores were associated with decreasing concentrations of GM-CSF, IL-2, IL-7, IL-8, IL-18, IP-10, MCP-1, and KC-like protein. 4 While several inflammatory mediators have already been implicated in obesity, this was the first study suggesting an effect of growth factors (eg, GM-CSF) in this condition. Furthermore, this study reiterated the importance of considering and reporting the body condition scores of animals included in “healthy” and “diseased” cohorts, which may be useful for comparison to infectious, inflammatory, neoplastic, and endocrine diseases.
Analysis and Interpretation of Multiplex Cytokine Data
As previously noted, the literature includes a variety of methods to analyze and interpret data from multiplexed cytokine assays. Here we discuss factors affecting the performance and interpretation of such assays.
Technical Acuity and Study Population
Multiplex analysis offers many benefits, such as lower costs, decreased sample volume, and a more detailed analysis of cytokine levels in clinical samples. Even with these positive attributes, multiplex assays such as the Milliplex Canine Cytokine Panel are technically complex, and their results can be influenced by a variety of factors such as variability between manufacturers, storage conditions of the kit reagents, sample collection parameters, experimental preparation of analyte standards, operator error in assay execution, instrument calibration, and differences in data analysis and interpretation. 3 When evaluating systemic cytokine measurements in any species, one must consider not only the potential disease, chronicity, and treatment but also variables such as body condition score, nutrition, breed, and age for certain cytokines. Studies thus far have only begun to scratch the surface when it comes to measuring potential physiologic differences in cytokine levels between groups of healthy and diseased dogs, but the data obtained can help us determine and hopefully avoid possible pitfalls in studies going forward.
In a survey of 25 national and international laboratories performing Luminex bead-based cytokine assays, the NIH/NIAID-sponsored External Quality Assurance Program Oversight Laboratory (EQAPOL) at Duke University reported that approximately 41% of participating sites required remediation training for accuracy and precision problems during the 18 months of external proficiency testing it scored. 57 Marks for poor-performing sites were improved by focusing on careful and consistent laboratory techniques and following uniform guidelines for data analysis. 83 Investigators newly considering multiplex cytokine analysis may initially experience many of these same pitfalls and are encouraged to strictly adhere to immunoassay kit protocols and familiarize themselves with their data analysis software. Though the proficiency testing in this study surveyed only the older filter plate technology, EQAPOL plans to migrate to the newer magnetic kits in the future to better fulfill their mission of identifying experimental variables that significantly impact the outcomes of Luminex systems.
Data Reporting and Statistical Analysis
It was common practice among all canine studies for cytokine data to be reported and analyzed as absolute concentration values. During multiplex analysis, measurement of each cytokine in a clinical sample is produced by mixing samples with analyte-specific antibodies conjugated to specially dyed magnetic beads, with a distinct dye mixture coded for every analyte tested. After sample incubation and subsequent addition of detection antibodies, quantities of bound analyte are determined by correlating the correct dye signature to a nonspecific fluorescent reporter signal, such as streptavidin-conjugated phycoerythrin. A median fluorescence intensity (MFI) measurement is calculated after fluorescent signals are tabulated for each event of the unique bead type. When more than 100 bead events are counted, EMD Millipore asserts that this MFI measurement is characteristic of the “true” presence of an analyte within an experimental sample, later coupling this value to an absolute concentration through the help of a standard curve. Particularly for “normal” or unstimulated clinical samples, this conversion of experimental measurements into a well-understood quantification adds a confounding effect to data analysis with low-abundance analytes that may present a MFI value below the lower limit of detection (LLOD) assigned to the standard curve, measuring at or even below the intensity of the assay blank. Further statistical analysis of these data points (once converted to pg/mL) cannot be accomplished without assigning an absolute value to these unknown samples (eg, the LLOD, LLOD/2, or 0) or removing them entirely from consideration. Before the 2015 study by Breen et al, 6 no scientific attempt had been made to clarify whether MFI or absolute concentration values was the most appropriate data set for multiplex analysis. Comparing the expression of 14 analytes in the plasma of 177 patients, Breen et al 6 demonstrated that while both techniques resulted in similar findings, using MFI values only produced a greater statistical power. Additionally, although almost all measured readings for IL-6 and TNFα were clustered near or below the lowest standard reading for these analytes, the distribution of these fluorescence measurements was still essentially within the linear range of their concentration curves. MFI-based statistical analysis provided meaningful comparisons between clinical groupings, while absolute concentration value analysis censored these values as out of range. In fact, the strongest justification of an MFI-based statistical analysis is that there are no out-of-range problems to this approach. In comparison, several researchers have published peer-reviewed critiques outlining how modifying out-of-range values biases statistical outcomes. 42,100 A follow-up analysis by Breen et al 7 in 2016 quantifying the presence of 37 analytes in the plasma, saliva, serum, and urine samples from 169 patients further demonstrated these findings by emphasizing the unpredictable statistical effects that were introduced by background subtraction and mathematically transforming MFI results into pg/mL. Importantly, the MFI values may vary significantly between individual assays, and reporting of MFI values may be more relevant when a single assay contains all of the samples measured than when multiple individual assays are compared.
In our review of studies using the Milliplex Canine Cytokine Panel, cytokine concentrations measured from clinical samples often failed to display a Gaussian distribution. Data may not be normally distributed for all or almost all cytokines measured in different clinical cohorts, based on analysis with 1 or sometimes several normality tests such as the Shapiro-Wilk 2,4,10,38,70,84,103 or D’Agostino-Pearson Omnibus test. 13,26,45,50,51,72 Therefore, statistical analysis of multiplexed clinical data will rely on nonparametric tests or mathematical transformations to produce a normal distribution of the data. Researchers unfamiliar with the Milliplex Canine Cytokine Panel should expect large variability between individuals within the same clinical groups and treat their data accordingly.
Growing appreciation for the extremely varied nature of cytokine measurements has also led to the development and success of new analytic techniques, such as frequency analysis, 50 panel scoring, 44 and biomarker signatures. 29 The key element behind each analysis is to tease apart systematic differences between time points, treatments, or clinical groupings that may be obscured by either large variability or poor sensitivity in the lower range. Instead of a direct comparison of measured fluorescent intensities or concentration values, frequency analysis counts the number of patients within a clinical grouping whose measurements meet a well-defined criterion. For every cytokine tested, clinical measurements can be designated to represent either ones or zeroes depending on benchmarks, such as whether they were nonzero or fell above the LLOD or even if they were greater than the median measurement or above the 90th percentile for this particular cytokine (either within their clinical grouping or against a standardized control group). These totals can then be expressed as fractional values or percentages (ie, frequencies) and compared across groups to discover significant differences by cytokine. Alternatively, individual cytokines can be summed across cytokine categories, such as pro-inflammatory, anti-inflammatory, growth factors, and T-cell–associated, to provide several profile scores for individual patients (ie, panel scoring). When there is excellent division of experimental cohorts, such as grouping selectively by disease severity, duration of illness, or diagnostic staging/scoring, frequencies for every cytokine can be graphed for a particular time point or experimental grouping to represent pictorial “biomarker signatures.” Small changes in cytokine concentrations may translate to frequency shifts that are more pronounced when viewed in this manner. They can also emerge as trending patterns when biomarker signatures are further divided and compared by clinical findings or survival data. For further background, please refer to the excellent example by Estrela-Lima et al 29 for how biomarker signatures can deepen the interpretation of experimental data or to earlier examples of how this analysis was first employed in human medicine. 56,95
Cross-sectional Versus Longitudinal Analysis
To interpret the results of future studies and determine the likely clinical significance of their findings, investigators are urged to compare their results not only with their own “control” cohorts but also with the ranges of cytokine concentrations previously observed in other studies of healthy dogs. While comparing values obtained across experiments has limitations (quality control and repeatability issues), it is our opinion that studies reporting clinical significance outside of the range of variability that is found in the larger cohort of healthy dogs would be informative. Otherwise, differences between small cohorts of healthy versus sick patients may risk being overstated, leading to Type II errors.
There is a paucity of studies measuring systemic cytokine levels in healthy controls over time, leaving unanswered questions regarding the variability of systemic cytokine levels within healthy and diseased cohorts. The majority of studies in dogs compared cross-sectional data at a single time point between a small number of “patients” and a small number of “controls.” In most cases, whether the mean, median, or standard deviation remains static throughout health or disease remains to be investigated. The assumption that a statistically significant difference in cytokine concentrations between cohorts at a single time point is or is not associated with disease may be confounded by differences measured over time or differences associated with disease onset, severity, and staging within each cohort. In this regard, failing to detect a significant difference in systemic cytokine concentrations may inappropriately lead to the conclusion that a particular cytokine is not playing a role in the disease, even though similar measurements with greater numbers or more specifically defined disease states could yield opposite results. On the contrary, one can imagine a scenario where a significant difference in cytokine levels would be assumed to play a significant role in disease onset or progression, only to find that it is the differences in baseline and not disease that lead to this conclusion. Figure 1 depicts 2 hypothetical scenarios that describe the possible effects of variability within cohorts across time. In the first example, the control population chosen for comparison would yield significantly different results even though there is no significant difference in the average cytokine levels between the 2 control populations. The “healthy” control-1 population is significantly different than either study group-1 or study group-2; however, if the “healthy” control-2 population is compared to the study groups, investigators would fail to recognize a difference attributable to disease. In the second example, different conclusions would be attributable to variability between the control populations, but depending on which control and study population is analyzed, investigators may or may not report significant differences. In both examples, there are no significant differences between the study groups, which would add an additional layer of uncertainty. Since the vast majority of studies published to date do not compare systemic cytokine concentrations across time or in more than 1 control or study cohort, further studies are needed to determine if either of these hypothetical errors actually affect our current understanding of cytokine measurements. The wide range of normal cytokine measurements reported in “healthy” control populations as seen in Tables 1 and 2 further supports the need for such longitudinal studies and appropriate (nonparametric) statistical analysis in future studies.
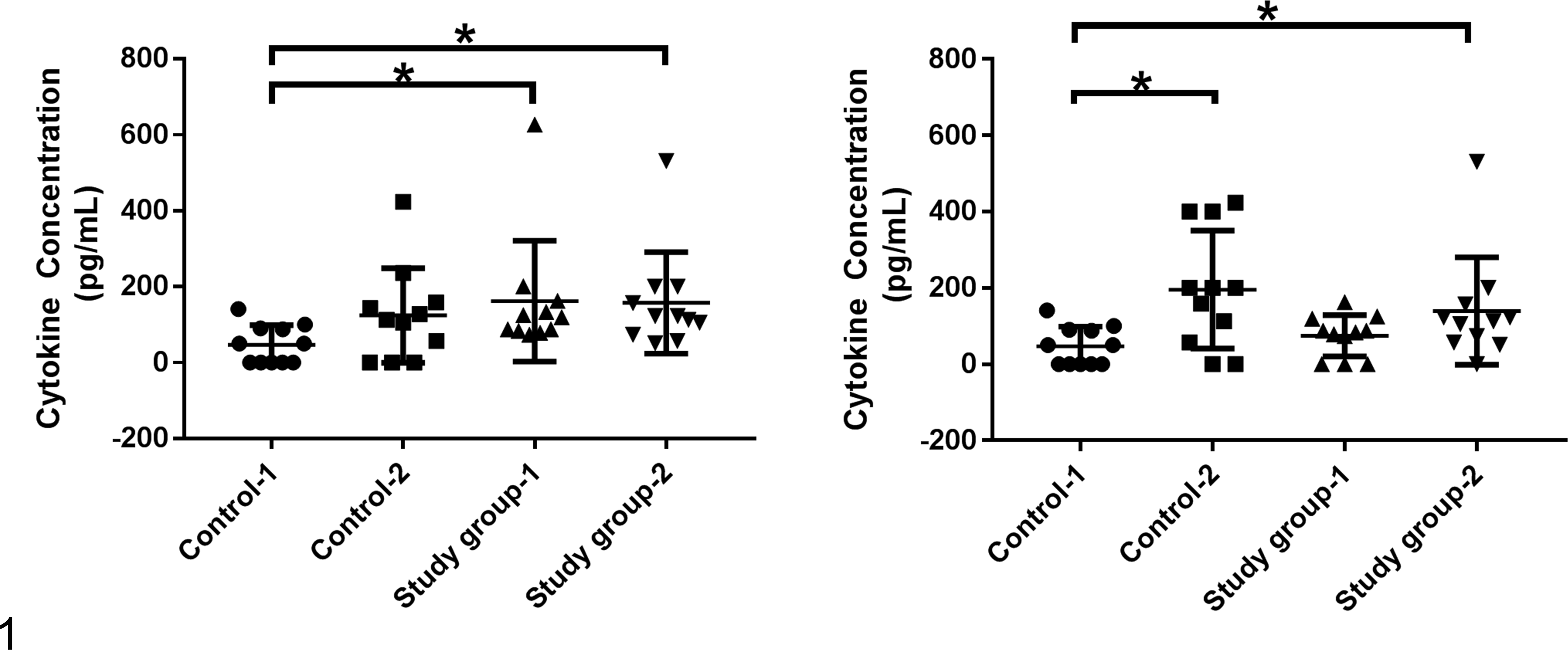
Hypothetical example of the potential effect of longitudinal variability within cohorts. Control 1 and Control 2 represent 2 baseline values taken either from the same population at different time points or from 2 different populations in cross-sectional analysis. Study group 1 and study group 2 represent 2 disease cohorts measured either from the same population at different time points or from 2 different populations in cross-sectional analysis. The figure shows how analysis of only 2 of these populations at any given time would yield potentially different conclusions depending on which 2 populations were analyzed. Statistical analysis was performed using Mann Whitney tests and differences were considered significant at P < .05.
Sampling, Causality, and Autoregulation
Whether disease is the cause of increases or decreases in cytokine concentrations has yet to be definitively determined for most canine diseases and cannot be inferred from even the best cross-sectional studies. Additional experimental studies and larger longitudinal studies are needed because studies of measured cytokine levels in serum or plasma are relatively limited in veterinary medicine. In addition, the biologic activity of proteins detected by monoclonal antibodies in peripheral blood samples cannot be inferred from these types of assays. Despite the convenient accessibility and clinical advantages of measuring cytokines in peripheral blood, much work remains to be done to determine if local tissue concentrations correlate with cytokine levels in serum or plasma. Such results, therefore, may not be indicative of the functional activity of any cytokine at a particular tissue site. Furthermore, given the important autocrine and paracrine nature of cytokine function and regulation, increases in cytokine concentrations may be irrelevant if these cannot exert biologic activity due to the presence of inhibitors, regulation of cellular receptors, or changes in downstream molecules that may not be concurrently measured.
In fact, the recognition of the intricate and complex effects of cytokine activities on one another is one of the driving factors that increased interest in measuring multiple cytokine networks simultaneously using multiplex technology, but much remains to be elucidated in regard to the sequence, degree, and magnitude of these interactions in any given patient or disease condition. Nevertheless, significant cytokine differences associated with a particular disease or group of diseases may suggest which cytokines play more crucial roles in either development, progression, or control of pathologic conditions in dogs and will help to advance our understanding of these interactions, prioritize therapeutic interventions, and develop innovative diagnostic tools, one step at a time.
Conclusions
Individual cytokines can perform a plethora of functions based on concentration, receptor availability, secondary messengers, transcription factors, and the presence of synergistic and/or antagonistic cytokines in the microenvironment. All of these factors are independently regulated, and therefore, the concentration of any particular cytokine in a test sample is typically an insufficient indicator of the patient’s health or disease state. Specifically, the function of any particular cytokine depends on the balance of concurrent synergistic and opposing cytokine effects. Therefore, the simultaneous measurements of multiple cytokines in a given test sample may be better predictors of pathophysiologic, therapeutic, and prognostic significance.
Multiplex systemic cytokine concentrations in the dog have already been investigated in a wide array of disease conditions. Many investigators have also contributed to our understanding of canine cytokine regulation by measuring systemic cytokine concentrations in different cohorts of healthy dogs. These studies have laid a solid foundation for multiplex cytokine analysis and will likely lead to clinically useful conclusions that will improve our current medical practice.
We aimed to produce an organized literature review of the canine studies that have employed the Milliplex Canine Cytokine Panel to date. This review may aid in the appreciation of previous efforts, the design of new cytokine studies, and the interpretation of their results. Our review highlights the notion that cytokine concentrations can be influenced by a multitude of technologic, physiologic, and pathologic factors. In addition, careful statistical analysis is needed to interpret the results of multiplex cytokine analysis. In most cases, further studies are still needed to determine the true clinical significance of differences in cytokine concentrations. In order to strengthen the value of such findings, future goals in the use of multiplex technologies should include the derivation of a consensus for the methods used to analyze data obtained from veterinary studies.
Footnotes
Acknowledgments
The authors wish to thank Jennifer Uurtamo and Alex Ko from Millipore Sigma for their expert advice and consultation in the use of the multiplex assay. In addition, we wish to thank Ashlesh Murthy and Michelle Hanke for careful review and editorial assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors (A.M.M.) and (K.R.) are supported by intramural funds from Midwestern University for the production of scholarly work that advances knowledge and improves the health of animals and people. Specifically, this study was supported by MWU’s Vice President for Research & Strategic Initiatives’ One Health Research Award.
