Abstract
Feline herpesvirus type 1 (FeHV-1) is one of the etiological agents of feline respiratory disease. FeHV-1 is an epitheliotropic and cytopathic virus that mainly causes rhinitis and conjunctivitis, although pneumonia is also occasionally seen. In this study, the authors investigated the pathogenesis of FeHV-1-associated pneumonia, comparing natural cases with viral infection of tracheal ring and cell cultures in vitro, using histology, immunohistology, double immunofluorescence, and transmission electron microscopy as investigative tools. The results confirm that FeHV-1 targets both respiratory epithelial cells and pneumocytes and indicate that FeHV-1 pneumonia is the consequence of continuous cell-to-cell viral spread from the upper airways via the trachea into the lungs. They provide strong evidence that FeHV-1–infected cells die primarily via apoptosis, following loss of cell-to-cell contact, rounding, and detachment. However, virus-induced lesions in vivo are dominated by marked neutrophil infiltration and extensive necrosis with less prominent apoptosis; in the airways, the tissue necrosis can extend into the submucosa. The necrosis appears to result from virus-induced neutrophil influx and release of proteolytic enzymes, such as matrix metalloproteinase-9, from the neutrophils.
Keywords
Feline herpesvirus 1 (Felid herpesvirus 1; FeHV-1) is an alphaherpesvirus of the genus Varicellovirus, with double-stranded DNA and a glycoprotein-lipid envelope. 41 FeHV-1 was first isolated in 1958 and all further FeHV isolates described since have similar antigenic features, thus constituting one serotype. 7,13,44 FeHV-1 infects members of the family Felidae; although the domestic cat is the main host, cheetahs, lions, and pumas can also become infected. 13 Alongside feline calicivirus (FCV), FeHV-1 is the main pathogen of upper respiratory tract disease in which rhinitis and conjunctivitis are the main clinical features, known as cat flu. In the majority of cases, the disease is caused by either or both viruses; however, other etiological agents such as Bordetella bronchiseptica and Chlamydophila felis are also frequently involved. 12
FeHV-1 is excreted in nasal or ocular secretions of infected cats and is naturally transmitted via the oral, nasal, or conjunctival route, resulting in markedly higher FeHV-1 prevalence in multicat establishments. 1,3,9,12,18 Clinical presentation and severity depend on the viral strain as well as the age and immune status of the individual animal, accounting for the generally higher susceptibility of younger animals to infection. 1,3,12,18,52,53 Frequent clinical signs are sneezing and oculonasal serous discharge, which may be accompanied by hypersalivation with drooling, depression, inappetence, and pyrexia. 12 The discharges gradually become mucopurulent, and in severe cases, dyspnea may be accompanied by coughing. 12 Clinical signs usually resolve within 10 to 20 days; however, severely affected animals can develop secondary bacterial infections that lead to chronic suppurative rhinitis and conjunctivitis. 12,43,52 In animals that recover from clinical disease, the virus can become latent in the trigeminal ganglia, rendering the cats prone to spontaneous episodes of upper respiratory disease due to viral reactivation. 12 Occasionally, particularly in young, debilitated cats, fatal primary FeHV-1–induced fibrinonecrotising bronchopneumonia can occur. 5
The histological lesions caused by FeHV-1 have been described in both naturally and experimentally infected cats and include necrosis of the respiratory and alveolar epithelium, which occasionally extends into the submucosa and can induce lysis of the turbinate bones. 4,5,8,10,11,19,20,37,50 In both the upper and lower respiratory tracts, infection is often accompanied by abundant fibrin exudation and suppurative inflammation. 8,19,21,23
Alphaherpesviruses (α-HV), such as herpes simplex virus type 1 (HHV-1), varicella-zoster virus (HHV-3), and pseudorabies virus (SuHV-1), have been shown to spread from cell to cell by releasing new virions at the cell junction, which promotes the infection of adjacent cells. Accordingly, they show a tropism for polarized cells with extensive cell-to-cell contact, such as epithelial cells. 27 So far, information on the mode of FeHV-1 spread is limited, but in vitro studies have also provided evidence of cell-to-cell spread for this virus. 35
Several in vitro tissue culture models have been proposed as alternatives to experimental infection of cats to study FeHV-1 pathogenesis, including tracheal organ cultures, tracheal mucosal explants, and conjunctival and corneal explants. 35,36,42 FeHV-1 replicates in vitro in both respiratory and conjunctival epithelial cells, but inconsistently in corneal explants. 36,42 The aim of the present work was to compare naturally infected cats with FeHV-1–induced pneumonia and suitable in vitro models of FeHV-1 infection (tracheal ring cultures and cell cultures) to examine the mode of viral spread from the upper to the lower respiratory tract and the mechanism of cell death (apoptosis and/or necrosis). The comparison of the lesions seen in FeHV-1 pneumonia with those in FeHV-1–infected tracheal organ cultures was employed to test the hypothesis that the host immunological response significantly contributes to the lesions observed in vivo; a better understanding of how and why a viral infection that is most commonly associated with relatively benign upper respiratory tract disease can progress to fatal pneumonia will assist the prevention and treatment of this infection in susceptible individuals.
Material and Methods
Animals and Tissues
The study was performed on 21 predominantly young cats that had died or were euthanized due to clinical signs of (upper) respiratory disease and had been submitted for a diagnostic postmortem examination. Animals were of various breeds and ranged from 10 days old to adult, with an approximately equal sex distribution. Information on breed, sex, and age, where available, is shown in Supplemental Table 1. A full necropsy was performed and samples from all major organs as well as macroscopically altered tissues were collected, fixed in 10% nonbuffered formalin for 24 to 72 hours, trimmed, and routinely embedded in paraffin wax for histological and immunohistological examinations. In all cases, FeHV-1 pneumonia was diagnosed based on the demonstration of viral antigen within lung lesions by immunohistology (IH); in 11 cases, FeHV-1 was also cultured from the lungs, and in 4 cases, virions were demonstrated in the lung tissue by transmission electron microscopy (TEM; negative staining technique) (Supplemental Table 1). A bacteriological examination of the lungs was performed in 13 cases. Bacteria from the normal commensal population were isolated in 7 cases; in 5 cases, species likely to represent postmortem bacterial growth were cultured; and in the last case, no bacteria were isolated. 34 One lung tested negative for Chlamydia sp by polymerase chain reaction (Supplemental Table 1).
In Vitro Studies
Tracheal organ cultures from 6 cats that had been euthanized for reasons other than respiratory disease were prepared and cultured in a submerged, rotary system as previously described in detail. 35 After 24 hours in culture, the tracheal cultures that had maintained complete and consistent cilia movement were infected with FeHV-1 (strain B927) at a multiplicity of infection of ≈ 1, an inoculating dose that ensured continued viral replication for the entire examination period of up to 72 hours, as assessed by both virus isolation and real-time polymerase chain reaction. 14,35 Mock-infected rings from the same trachea were maintained under the same conditions and served as controls.
At the end of the culture period, tracheal rings were collected and either fixed in 4% paraformaldehyde (PFA) for 24 hours and routinely embedded in paraffin wax for histological and immunohistological examination or fixed in 4% PFA with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer and routinely embedded in epoxy resin for TEM.
Feline embryo A (FEA) cells infected with a FeHV-1 strain isolated by Porter et al 43 were maintained for 24 hours in culture, washed with phosphate buffered saline, and centrifuged to form a cell pellet that was subsequently fixed in 4% PFA for 48 hours and routinely embedded in paraffin wax. 26
Histology, Immunohistology, and Immunofluorescence
Consecutive 3- to 5-μm sections were prepared from the paraffin-embedded tissue specimens and stained with hematoxylin and eosin or used for IH and immunofluorescence to demonstrate FeHV-1 antigen, cleaved caspase-3 (as a marker of apoptosis), matrix metalloproteinase (MMP)-2 and -9, and β-catenin. For IH, the peroxidase antiperoxidase and the horseradish peroxidase methods were performed as previously described. 6,31 –33 Briefly, after deparaffinization and rehydration, antigen retrieval, and blocking of endogenous peroxidase, sections were incubated with the primary antibodies at 4°C overnight and the secondary antibodies (30 minutes at room temperature) followed by the specified detection method used to visualize reactions with 3,3′-diaminobenzidine and counterstained with hematoxylin as detailed in Table 1A. Consecutive sections, incubated with nonreactive monoclonal or polyclonal antibodies, as well as the uninfected tracheal rings stained for FeHV-1 antigen served as negative controls. The following were used as positive control tissues: FeHV-1–infected FEA cell pellets (for FeHV-1), feline mammary carcinoma (for MMP-2 and -9), a feline lymph node (for cleaved caspase-3), and an unaltered feline lung (for ß-catenin).
Antibodies, Antigen Retrieval, and Detection Methods for Immunohistology (A) and Double Immunofluorescence (B).
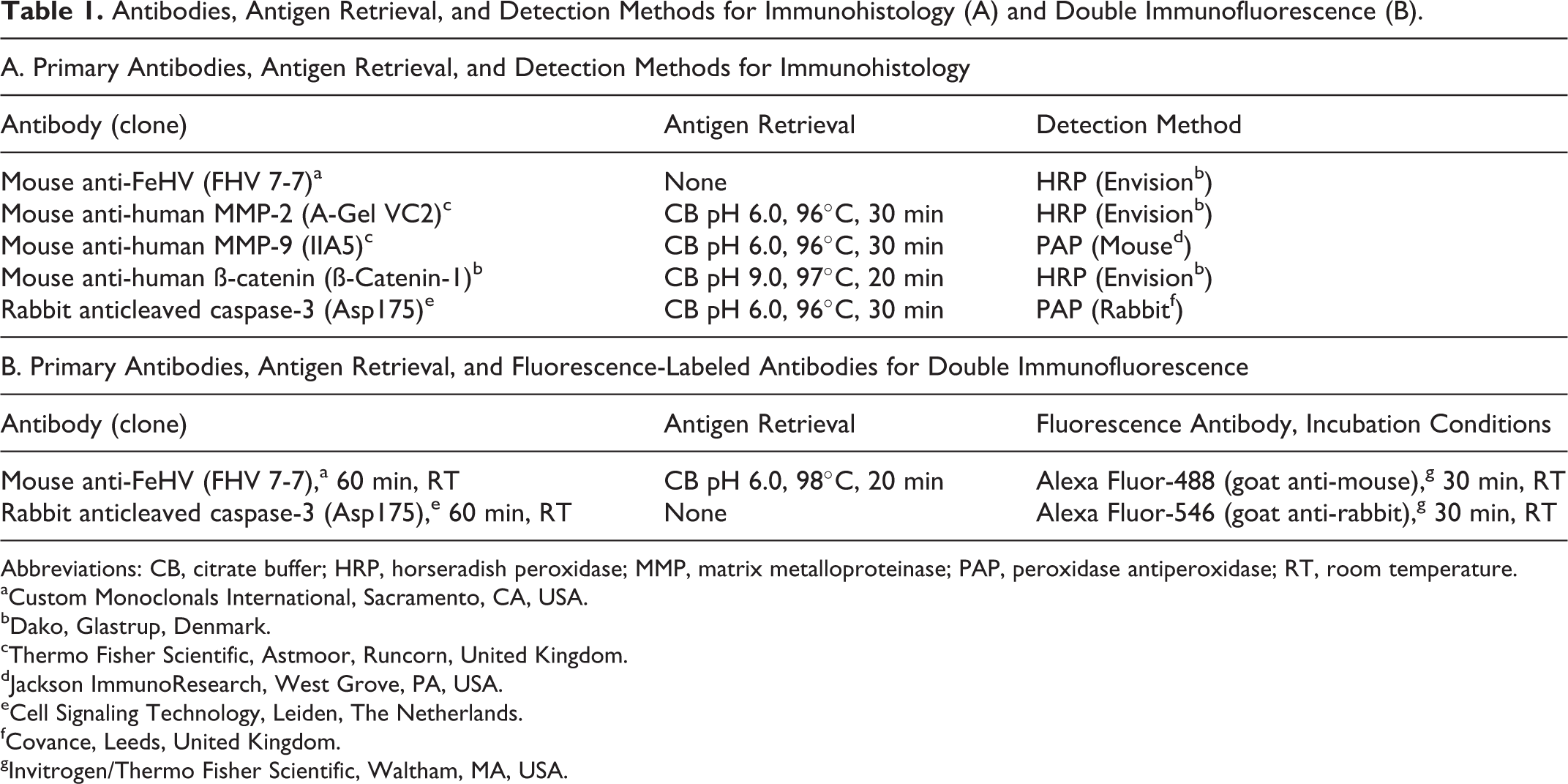
Abbreviations: CB, citrate buffer; HRP, horseradish peroxidase; MMP, matrix metalloproteinase; PAP, peroxidase antiperoxidase; RT, room temperature.
aCustom Monoclonals International, Sacramento, CA, USA.
bDako, Glastrup, Denmark.
cThermo Fisher Scientific, Astmoor, Runcorn, United Kingdom.
dJackson ImmunoResearch, West Grove, PA, USA.
eCell Signaling Technology, Leiden, The Netherlands.
fCovance, Leeds, United Kingdom.
gInvitrogen/Thermo Fisher Scientific, Waltham, MA, USA.
For double immunofluorescence, deparaffinized sections from the FeHV-1–infected FEA cell pellet underwent incubation with anti-FeHV-1 followed by anticleaved caspase-3 antibody, with appropriate antigen retrieval and fluorescence-labeled secondary antibodies (Table 1B). Nuclei were stained with 4’,6-diamidine-2’-phenyl-indole dihydrochloride.
Results
FeHV-1 Pneumonia Results From the Extension of Upper Respiratory Tract Lesions Along the Airways
Similar to previously published descriptions, FeHV-1–induced pneumonia was characterized by necrotizing to necrosuppurative bronchitis, bronchiolitis, and alveolitis with occasional intranuclear viral inclusion bodies, neutrophil-dominated leukocyte infiltration, and variable fibrin exudation. 5 The affected cats also exhibited rhinitis and tracheitis of a similar nature (Figs. 1–6). Closer examination confirmed that lung lesions were a continuum of those present in the upper respiratory tract. They represented individual focal lesions that typically contained an affected bronchus and bronchiole from which necrosis and inflammation extended into the surrounding alveoli, with frequent evidence of a direct continuation (Figs. 3, 4).
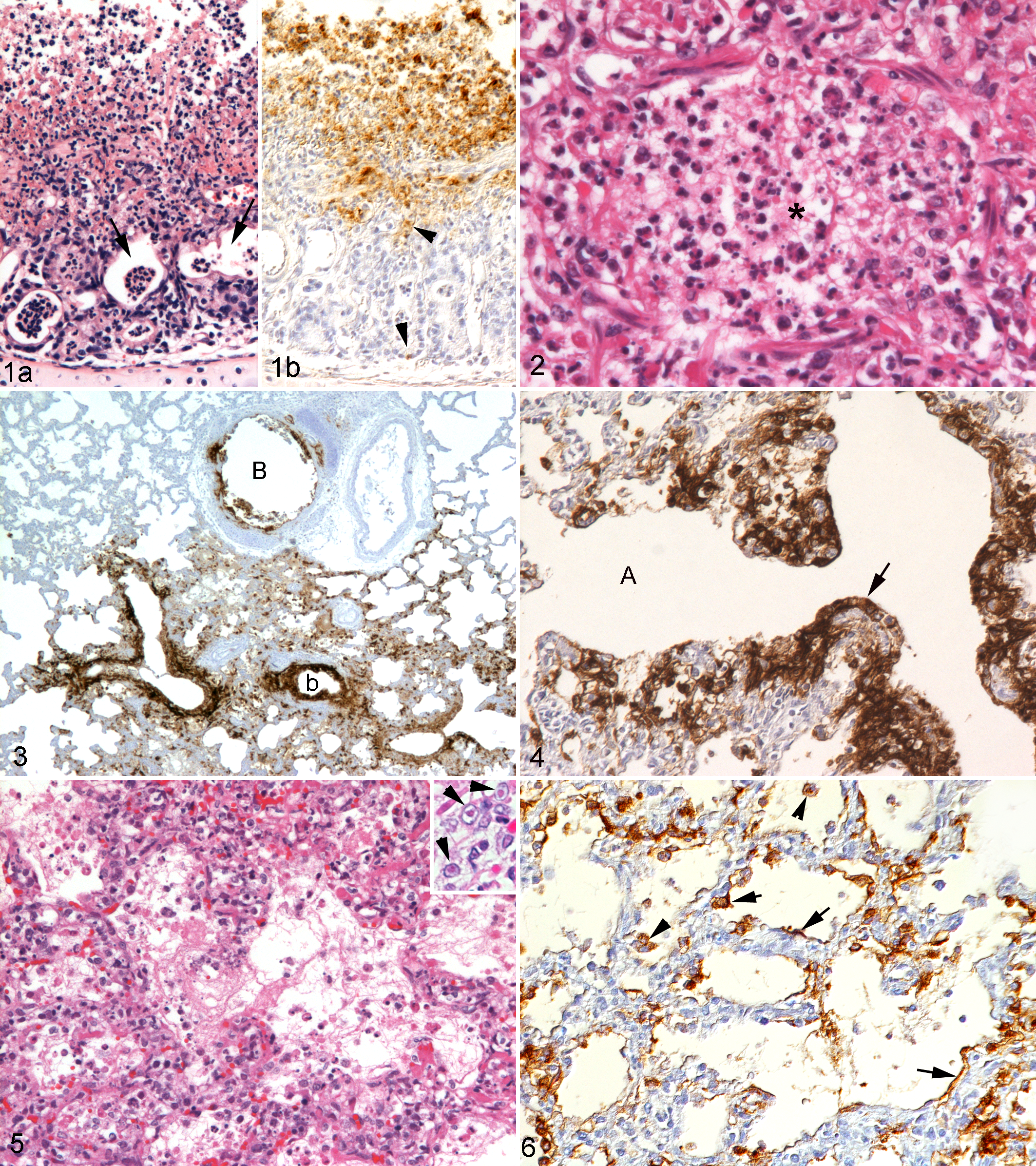
Immunohistology revealed FeHV-1 antigen in the respiratory epithelial cells of the airways, type I and II pneumocytes, and epithelial cells of the bronchial glands. Viral antigen was primarily seen within the cytoplasm but sometimes also in the nucleus of infected cells (Figs. 1b, 3, 4, 6).
Infected epithelial cells showed a range of morphological features, from unaltered cells within mildly affected areas to degenerating cells characterized by cytoplasmic swelling and detached cells with morphological features of apoptosis or necrosis (Fig. 2). In addition, viral antigen was occasionally found in alveolar macrophages within alveoli where degenerate infected epithelial cells and free viral antigen were also often present, suggesting that these macrophages had phagocytosed extracellular virions released from dead pneumocytes (Fig. 6).
FeHV-1 Induces Apoptosis of Infected Cells and Spreads From Cell to Cell
Natural pneumonia cases showed a plethora of changes, which sometimes included a significant inflammatory response. Therefore, to assess the direct viral cytopathic effect and exclude any potential effects of an inflammatory response, FeHV-1–infected tracheal ring cultures were examined at defined time points (24, 48, and 72 hours postinfection [hpi]). These suggested a sequence of cytopathic processes. At 24 hpi, the respiratory epithelium showed multiple foci of infection characterized by loss of cilia and rounding up of epithelial cells that had lost their pseudostratified columnar arrangement (Fig. 7a-c). Viral intranuclear inclusion bodies were occasionally seen. At the later time points, larger foci of infection with rounding of cells that appeared to have detached and areas with cell loss were observed. In some cases, a layer of uninfected basal cells remained (Fig. 8a). Detached cells contained viral antigen and often showed shrinkage, blebbing, and fragmentation, with chromatin margination and fragmentation (apoptosis) and expression of the apoptosis marker cleaved caspase-3 (Fig. 8b, c). 55 These changes were associated with an increase in ß-catenin expression along the lateral aspects of the plasma membrane and, in completely detached cells, ß-catenin translocation from the membrane to the cytoplasm (Fig. 8d). Ultrastructural changes included an increase in the nuclear:cytoplasmic ratio, loss of cilia, loss of cytoplasmic organelles, increased electron density of the cytoplasm, nuclear condensation, and apoptotic bodies (Fig. 9a). Detached cells were devoid of desmosomes and hemidesmosomes (Fig. 9b). Viral particles were present in the nuclei and very rarely within the cytoplasm of detaching cells (Fig. 9b).
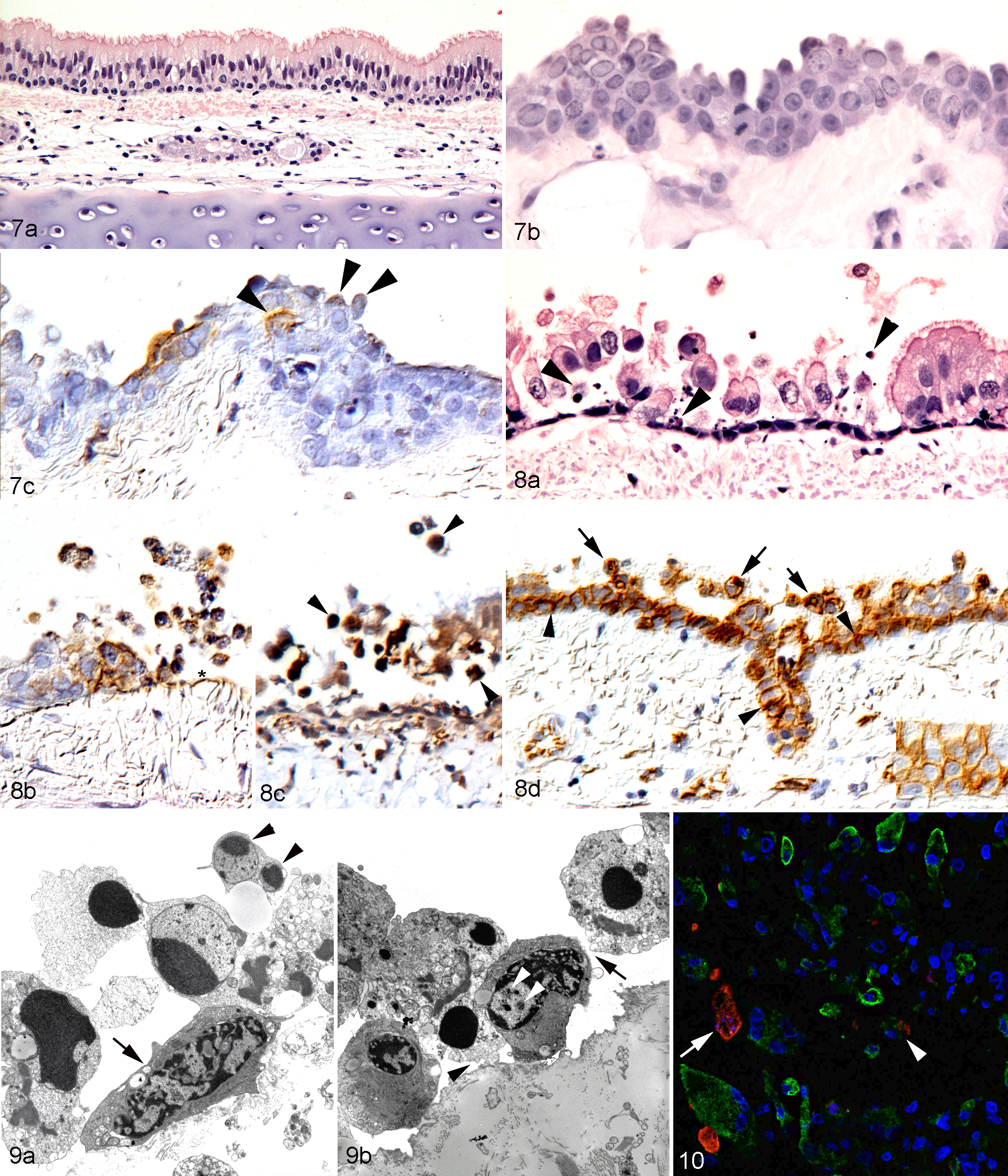
To confirm that FeHV-1 induced apoptosis of infected cells, double immunofluorescence was applied to FeHV-1–infected cultured FEA cell pellets. At 24 hpi, a few infected cells were found to express both viral antigen and cleaved caspase-3, that is, to undergo apoptosis (Fig. 10); some of these cells also exhibited typical morphological features of apoptosis (shrunken cells with condensed nuclei).
Similar features were seen in naturally occurring cases of pneumonia; FeHV-1–infected respiratory epithelium exhibited ß-catenin translocation and underwent apoptosis (Fig. 11a–d). In both the pneumonia cases and tracheal ring cultures, there was abrupt transition between infected and noninfected, and between degenerate and morphologically unaltered, epithelial cells (Fig. 11b, inset). Furthermore, in tracheal organ cultures, the increased size of the infected foci at 72 hpi indicated that cell loss appeared to expand circumferentially. In FeHV pneumonia, there was evidence of similar cell-to-cell spread of viral infection from the airways to adjacent alveoli, where infected cells were in close contact with each other, and also from the respiratory epithelium to the underlying bronchial glands (Fig. 1).
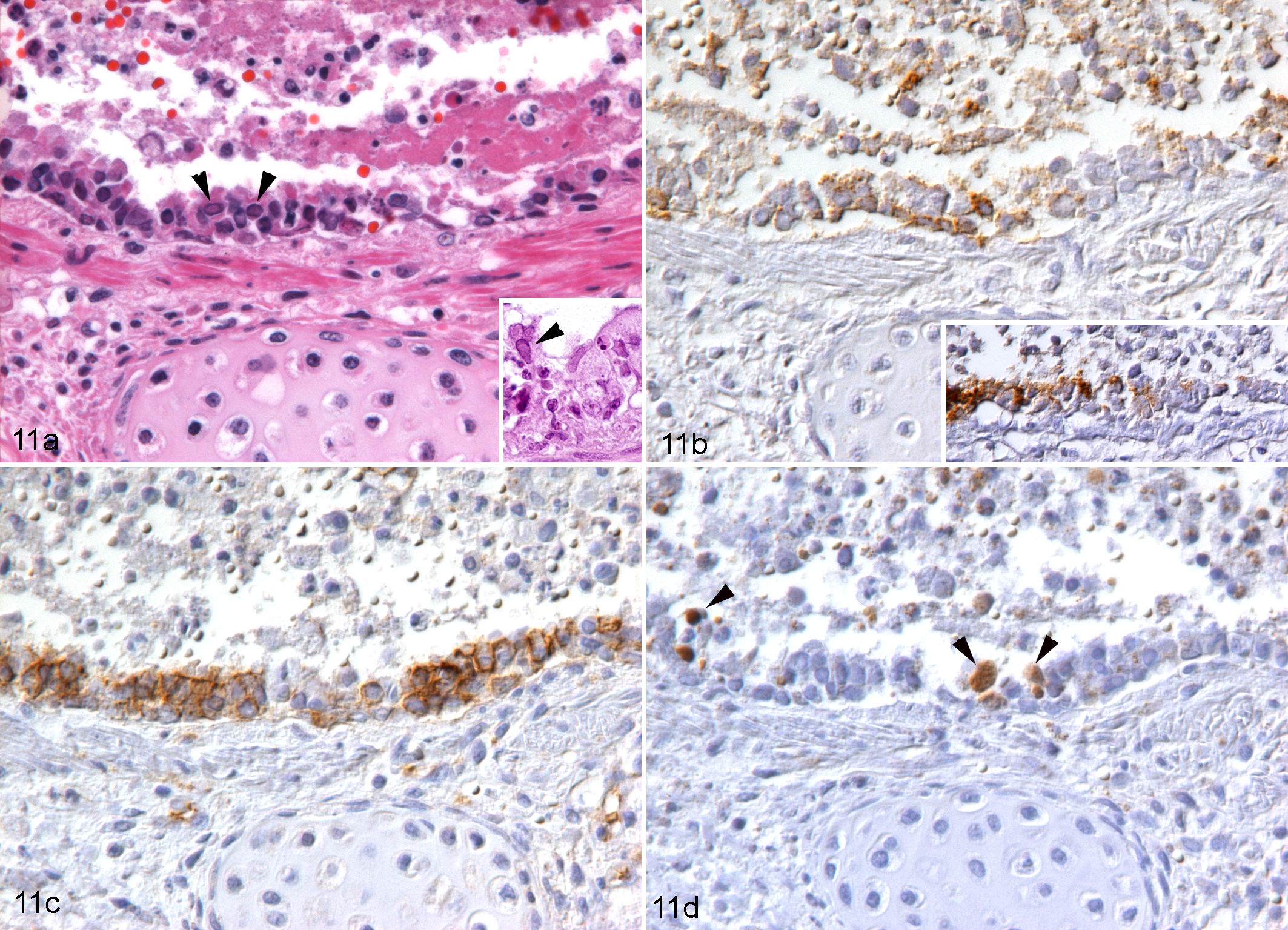
FeHV-1 pneumonia, cat, lung. Necrotizing bronchitis, case No. 1. (a) Epithelial cells appear shrunken and hypereosinophilic and often slough into the lumen, where they are admixed with fibrin and few inflammatory cells. Some epithelial cells exhibit intranuclear inclusion bodies (arrowheads, also in inset). Hematoxylin and eosin. (b) Numerous respiratory epithelial cells, aligned and detached, are found to be FeHV-1 infected. Inset: In another bronchiole, abrupt transition is seen between FeHV-1 infected and noninfected epithelial cells. Immunohistochemistry (IHC) for FeHV-1 antigen. (c) Epithelial cells in the affected area exhibit increased β-catenin expression along the lateral aspect of the plasma membrane or β-catenin accumulation in the cytoplasm. IHC for ß-catenin. (d) Several rounded and shrunken epithelial cells in the affected area are confirmed to undergo apoptosis, based on the expression of cleaved caspase-3 (arrowheads). IHC for cleaved caspase-3.
FeHV-1–Induced Lesions Are Associated With a Neutrophil-Dominated Inflammatory Response and Can Extend Beyond the Epithelium
The FeHV-induced cytopathic changes in pneumonia cases were generally accompanied by a variably intense acute inflammatory response, ranging from abundant fibrin exudation with only a few inflammatory cells to a densely cellular infiltration (Figs. 1a, 2, 5, 12, 13a). The inflammatory infiltrate was predominantly neutrophilic, with a variable number of foamy macrophages as well as occasional lymphocytes and plasma cells. Neutrophils infiltrated the airway respiratory epithelium and submucosa, including the glandular structures, and were present in abundance within alveolar lumina (Figs. 1a, 2, 12). Neutrophils were most prominent in areas with severe tissue destruction and less frequent in older lesions, which exhibited marked thickening of the alveolar septa, type II pneumocyte hyperplasia, and a predominantly mononuclear infiltrate.
Inflammatory processes were accompanied by activation of endothelial cells in pulmonary vessels, indicated by their plump appearance and intense MHC class II expression (data not shown). Numerous band neutrophils and occasional monocytes were often seen adhering to the endothelial surface, indicating cell recruitment into the tissue. Frequently, marked perivascular edema was seen within and around the lesions.
Within inflammatory lesions, numerous epithelial cells, most of which were infected, showed morphological features of necrosis (cellular swelling, disruption of the plasma membrane, pyknosis, and karyorrhexis) and apoptotic cells were rare (Figs. 2, 12). In several cases, necrosis of the airways extended beyond the respiratory epithelium into the submucosa, occasionally associated with thrombosis or necrosis of submucosal blood vessels (Fig. 13). Four cases (case Nos. 4, 15, 18, 21) exhibited excessive necrosis that completely effaced all preexisting structures and extended to the surface of the bronchial cartilaginous rings (Fig. 13a). This was accompanied by an intense suppurative inflammation and the accumulation of abundant extracellular viral antigen admixed with cell debris above the cartilage surface (Fig. 13b). In contrast, cell death in FeHV-1–infected tracheal rings never extended beyond the respiratory epithelium (Fig. 8a–c).
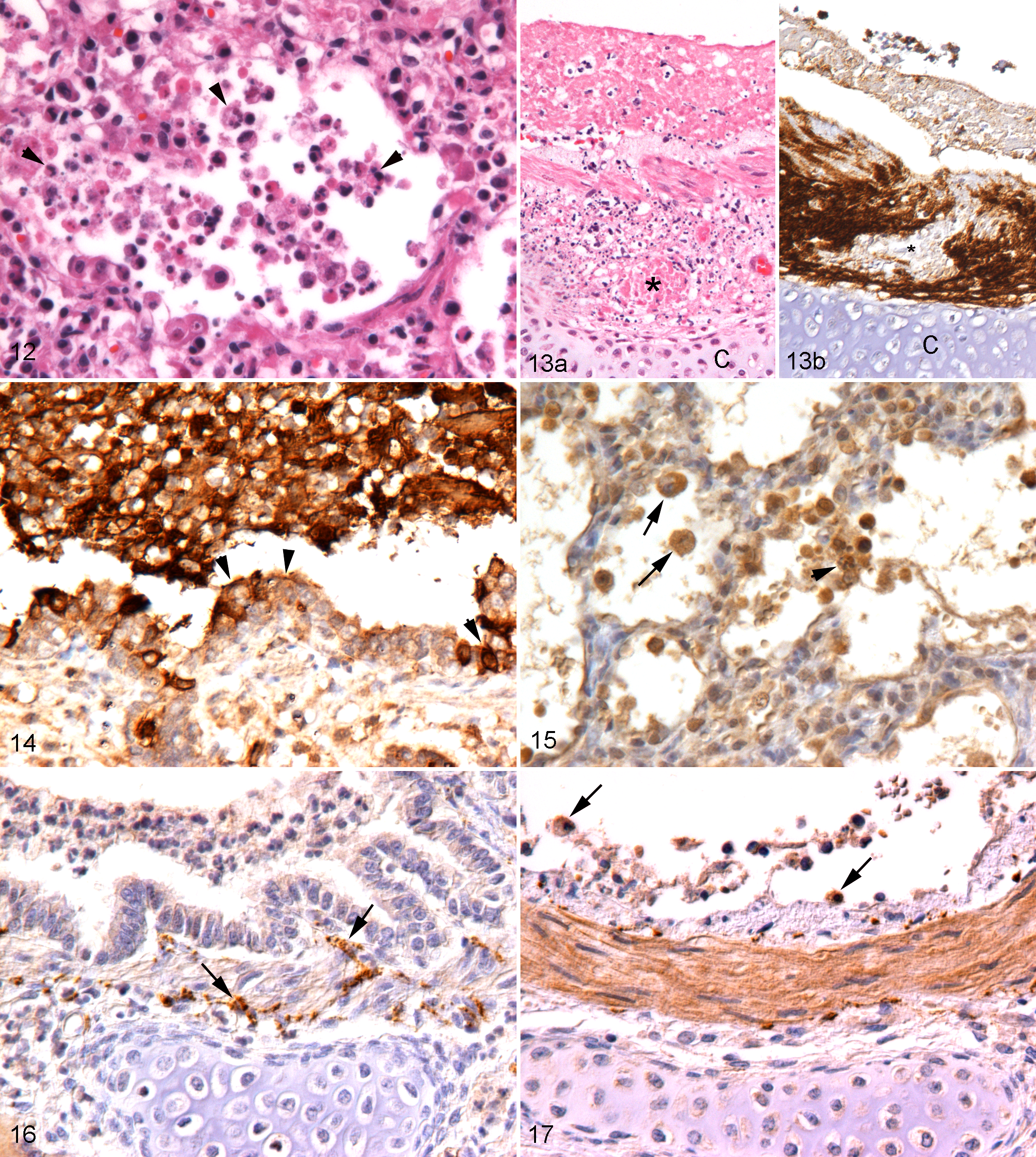
Expression of the gelatinases MMP-2 and -9 was investigated as a potential mechanism of the extensive tissue destruction. In both infected and control tracheal rings, respiratory epithelial cells, fibrocytes, and chondrocytes exhibited variably intense MMP-2 expression. Lymphocytes and plasma cells, present in low numbers both in vessels and in the submucosa, were negative or weakly positive. In severely damaged areas, detached epithelial cells, submucosal fibroblasts, and, to a lesser extent, chondrocytes exhibited mild MMP-2 upregulation. MMP-9 expression was minimal in both uninfected and infected tracheal rings with no significant variation after FeHV-1 infection. In contrast, pneumonia cases exhibited strong MMP-9 expression in neutrophils and macrophages and mild to moderate expression by respiratory epithelial cells, pneumocytes, and chondrocytes (Figs. 14, 15). MMP-2 was less intensely expressed, but epithelial cells, submucosal fibrocytes and chondrocytes in the airways, and fibrocytes and epithelial cells, mainly in areas of severe necrosis and inflammation, exhibited a moderate reaction (Figs. 16, 17). Within areas of complete tissue destruction, staining for both MMPs was seen in the necrotic debris.
Discussion
Feline herpesvirus 1 is an important cause of upper respiratory disease in cats and occasionally also causes pneumonia, but always in association with upper respiratory tract lesions (rhinitis and tracheitis). 5,11,37,45,50 This was demonstrated in older experimental studies where upper respiratory disease was followed by lung involvement. 8,23,29 Our results are in accordance with these findings and suggest that FeHV-1 pneumonia is the consequence of viral spread along the respiratory tract. There was strong evidence that FeHV-1 first infects lower airways and then spreads to adjacent alveolar pneumocytes, resulting in a distinct multifocal, airway-centered lesion pattern that differs from that seen with naturally occurring FCV pneumonia and the pneumonia that develops after experimental FCV infections using aerosols or in response to other viruses that reach the lungs with the blood, such as cowpox virus, together representing the various potential routes of viral pulmonary infection. 22,40,49 These findings indicate that, like other α-HV, FeHV-1 spreads from cell to cell. 27 Members of the α-HV family commonly show a tropism for polarized cells with extensive cell-to-cell contact, which allows the release of virions at the lateral cell junctions and subsequent infection of the adjacent cells. 27 In accordance with previous studies, our in vitro results from FeHV-1 infection of tracheal ring cultures provide further evidence that FeHV-1 behaves in a similar manner. 35,36 At 24 hpi, we observed several foci of infection with cell degeneration and death, which appeared to expand circumferentially with time; the virus was always found in morphologically intact cells at the border with the uninfected epithelium. This also suggests that the virus is released prior to cell degeneration and death, a feature described for another α-HV, Marek’s disease virus (GaHV-2). 46
The present study confirms the epitheliotropism of FeHV-1. In the tracheal rings, only respiratory epithelial cells and submucosal tracheal glands became infected. In some cases, there was evidence that viral replication and production were most intense in the upper layers, suggesting that FeHV-1, like other α-HV, has a preference for well-differentiated and polarized epithelial cells. 27 In naturally infected cats, the virus was found in respiratory and glandular epithelial cells of the airways and alveolar type I and II pneumocytes. Viral antigen was mainly seen in the cytoplasm and only occasionally in the nucleus of infected cells, corresponding to the replication site of α-HV (nucleus) and subsequent transport of the progeny virus to the lateral domains of the cells. 27,48 FeHV-1 antigen was also found in alveolar macrophages in areas of extensive virus-associated necrosis. Other α-HV such as bovine herpesvirus 1 (BoHV-1) and SuHV-1 have been shown to infect macrophages; however, we and others observed macrophages in naturally infected animals admixed with cellular debris and cell free virus, which suggests that FeHV-1 antigen in the alveolar macrophages is the result of phagocytosis. 2,5,25
The in vitro part of the present study illustrates that FeHV-1–infected cells undergo apoptosis. Using cultured tracheal rings, we could show a sequence of morphological changes: after loss of cilia, (hemi)desmosome-mediated polarization, and cell contact, cells detached from neighboring cells and basement membrane and sloughed off. This was accompanied by ultrastructural changes consistent with apoptosis. In FeHV-1–infected FEA cells (the cell line commonly used for FeHV-1 isolation), double immunofluorescence for cleaved caspase-3 and virus antigen confirmed apoptosis as the form of direct virus-induced cell death. 26 Comparable changes have been described in other in vitro models of α-HV infection, that is, HHV-1 induced similar rounding up and detachment of infected epithelial cells in human nasal mucosal transplants; however, these changes were not clearly associated with apoptosis. 15 We also found apoptosis in the natural FeHV-1 pneumonia cases, where sloughed respiratory epithelial and alveolar cells with morphological evidence of apoptosis and cleaved caspase-3 expression were present.
We used immunohistological detection of β-catenin to further characterize the FeHV-1–induced cytopathic changes. β-catenin is a multitask protein involved in both maintenance of the cell structure (ie, epithelial polarization) and cell cycle signaling and is expressed on the cytoplasmic aspect of the cell membrane. 38 In vitro and in vivo mouse models have demonstrated that β-catenin regulates the epithelial repair after lung injury, showing an increase of signaling in epithelial cells during the transmigration of neutrophils across the alveolar epithelium. 56 It has also been suggested that β-catenin overexpression can induce apoptosis, rendering it part of a physiological mechanism to eliminate cells from the population. 30 We sometimes observed an increase in β-catenin expression at the lateral borders of epithelial cells that had started to round up and an accumulation of the protein in the cytoplasm of completely detached cells. Under physiological conditions, β-catenin is constantly phosphorylated and kept at a low concentration in order to stabilize the cell-to-cell junction. Following cell damage, phosphorylation of β-catenin ceases and the protein accumulates in the cytoplasm and then translocates to the nucleus, where it promotes cell division. 39 In the current study, the overexpression of β-catenin was associated with cell damage, suggesting that this may represent part of the apoptotic pathway in infected cells.
All FeHV-1 pneumonia cases exhibited substantial neutrophil-dominated inflammation. This was seen without evidence of secondary bacterial infection. Similarly, an initial neutrophilic response was previously described in experimentally infected germ-free cats, suggesting that neutrophil recruitment is a direct consequence of the viral infection. 23 This is supported by a study using BoHV-1, where infected epithelial cells secreted IL-1 and TNF-α, which induced the transmigration of neutrophils across the bronchial epithelial cell layer. 47 Furthermore, upregulation of acute-phase inflammatory cytokines, including neutrophil chemoattractants, has been reported in cats with FeHV-1–induced rhinitis. 28 In our in vitro infected tracheal cultures, we saw infection of the respiratory epithelium, but the virus never spread beyond the basement membrane. Equid herpesvirus 1 (EHV-1) shows a similar behavior, whereas HHV-1, SuHV-1, and BoHV-1 have been shown to cross the basement membrane. 15,16,51,54 For FeHV-1, the behavior seems to vary depending on the in vitro system, since viral spread across the basement membrane was reported in conjunctival and tracheal explants using the FeHV-1 strain C27, grown in Crandell Rees feline kidney cells for an unknown number of passages prior to inoculation. 36 More recent, authors have attributed these variations in behavior to the virus strain and the temperature at which the tissue was cultured. 42 Furthermore, the tracheal organ cultures in the present study were complete tissue rings, as opposed to the small explants used by Li et al, 36 which has been suggested to be a factor in the differences in infection in corneal explants observed in different models. 42 For SuHV-1, destruction of the basement membrane was associated with the presence of a trypsin-like serine protease; however, whether the enzyme was of cellular or viral origin was not clear. 17 In our study, there was clear evidence of a direct association between viral infection, the neutrophil-dominated inflammation, and tissue necrosis, suggesting that cellular enzymes, such as MMP-2 and -9, were involved in the processes. 39 It is interesting that we did not see significant MMP-9 and MMP-2 expression in the tracheal in vitro model, although there was evidence of mild upregulation in the infected epithelium. In contrast, there was consistent strong MMP-9 expression in FeHV pneumonia, predominantly in neutrophils and macrophages. In human periodontal disease caused by HHV-1, it has been demonstrated that opsonization of the virus leads to neutrophil recruitment, which in turn amplifies the tissue destruction by increasing the upregulation of MMP-9. 24
In FeHV-1 pneumonia, the necrotizing lesions were seen in association with a neutrophilic inflammatory response. The necrosis was often extensive and partly extended into the underlying tissues, reaching the bronchial cartilage in severe cases. This differs substantially from the FeHV-1 lesions seen in the in vitro tracheal cultures, where they were mainly centered on the epithelium and characterized by apoptosis. The neutrophilic response in the natural cases might play a role in the development of necrotizing lesions. This is indicated by the increase of MMP-9 expression. What induces the suppurative inflammation is still not completely understood; infected intact and necrotic epithelial cells, alveolar macrophages, and the virus itself could all participate by releasing acute inflammatory cytokines. Likewise, the mechanism of cell destruction needs to be further understood as other intracellular substances (ie, lysosomal enzymes) released from neutrophils and macrophages are probably involved. The differences between the natural in vivo cases and in vitro models highlight the advantages of a model in which the systemic immune response is absent (to allow the study of direct viral effect) and the limitations, as this response can be a significant influence in the progression and outcome of the viral infection.
Footnotes
Acknowledgements
We are grateful to the technical staff in the Histology and Electron Microscopy Laboratories, Veterinary Laboratory Services, Shirley Bonner in the Infection Biology Department, School of Veterinary Science, University of Liverpool, and the Histology Laboratory, Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich, for excellent technical support. This work is dedicated to the memory of Dr Anne Vaughan-Thomas, who contributed significantly to the project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
