Abstract
Using light microscopy to describe the microarchitecture of normal and diseased tissues has changed very little since the middle of the 19th century. While the premise of histologic analysis remains intact, our relationship with the microscope is changing dramatically. Digital pathology offers new forms of visualization, and delivery of images is facilitated in unprecedented ways. This new technology can untether us entirely from our light microscopes, with many pathologists already performing their jobs using virtual microscopy. Several veterinary colleges have integrated virtual microscopy in their curriculum, and some diagnostic histopathology labs are switching to virtual microscopy as their main tool for the assessment of histologic specimens. Considering recent technical advancements of slide scanner and viewing software, digital pathology should now be considered a serious alternative to traditional light microscopy. This review therefore intends to give an overview of the current digital pathology technologies and their potential in all fields of veterinary pathology (ie, research, diagnostic service, and education). A future integration of digital pathology in the veterinary pathologist’s workflow seems to be inevitable, and therefore it is proposed that trainees should be taught in digital pathology to keep up with the unavoidable digitization of the profession.
Keywords
Pathologists have been using light microscopes for the diagnosis of diseases in tissue sections on glass slides since the development of modern pathology by Rudolf Virchow in 1858. 73 However, the rapid development of information technologies in the past decades is now also reaching this sanctuary of pathology as it has changed almost all other aspects of modern life. New forms of visualization and storage of histologic sections and network-based communication and exchange of these samples are available and integrated in the workflow of the first veterinary pathology laboratories. These new technologies are usually summarized under the almost interchangeable terms digital pathology (DP) or virtual microscopy (VM), which describe the viewing of microscope slides on a computer screen with an almost equivalent optical resolution and visual magnification of a light microscope. The starting point of the increasing integration of DP was the development of fast and reliable slide scanners, which are able to digitalize entire glass slides. These whole-slide images (WSI) can be viewed, evaluated, and stored potentially forever with constant quality. Viewing of a digital WSI in the sense of DP is, however, not restricted to a static image but includes options to move horizontally (x- and y-axis), to zoom in and out, and sometimes even to fine focus in the slide (z-axis) comparable with light microscopy (LM).
In human pathology, WSI are increasingly used for primary diagnosis (onsite and telediagnosis), secondary consultation (teleconsultation), and archiving. 51,67,75 Also the first, large veterinary pathology labs are using WSI as their primary technology for the evaluation of histologic slides. So far, primary histopathologic diagnoses are not obtained fully automated by image analysis software, but automated quantification, for instance, of human breast cancer tissues stained for HER2 has been approved by the Food and Drug Administration (FDA) for primary diagnosis, and such automated analysis is increasingly applied by (veterinary) pathologists for quantitative analysis in research projects. In addition, VM is a commonly used and much appreciated tool for histopathology training at veterinary schools worldwide.
This review intends to give an overview of the current DP technology and its potential in all fields of veterinary pathology (ie, research, diagnostic service, education).
Hardware
The central step in the workflow of DP that separates it from classical pathology is the digitization of histologic sections into digital images (ie, WSI or virtual slides; Fig. 1). Several types of slide scanners from different vendors are available (for more information on vendors and scanners, see Farahani et al 17 ). Despite several differences in the technical setup, all scanners consist of 4 major components that are more or less equivalent to traditional light microscopes and consist of a (1) light source, (2) robotics to move the slide holder, (3) 1 or several objectives, and (4) an associated camera to capture the images. 17
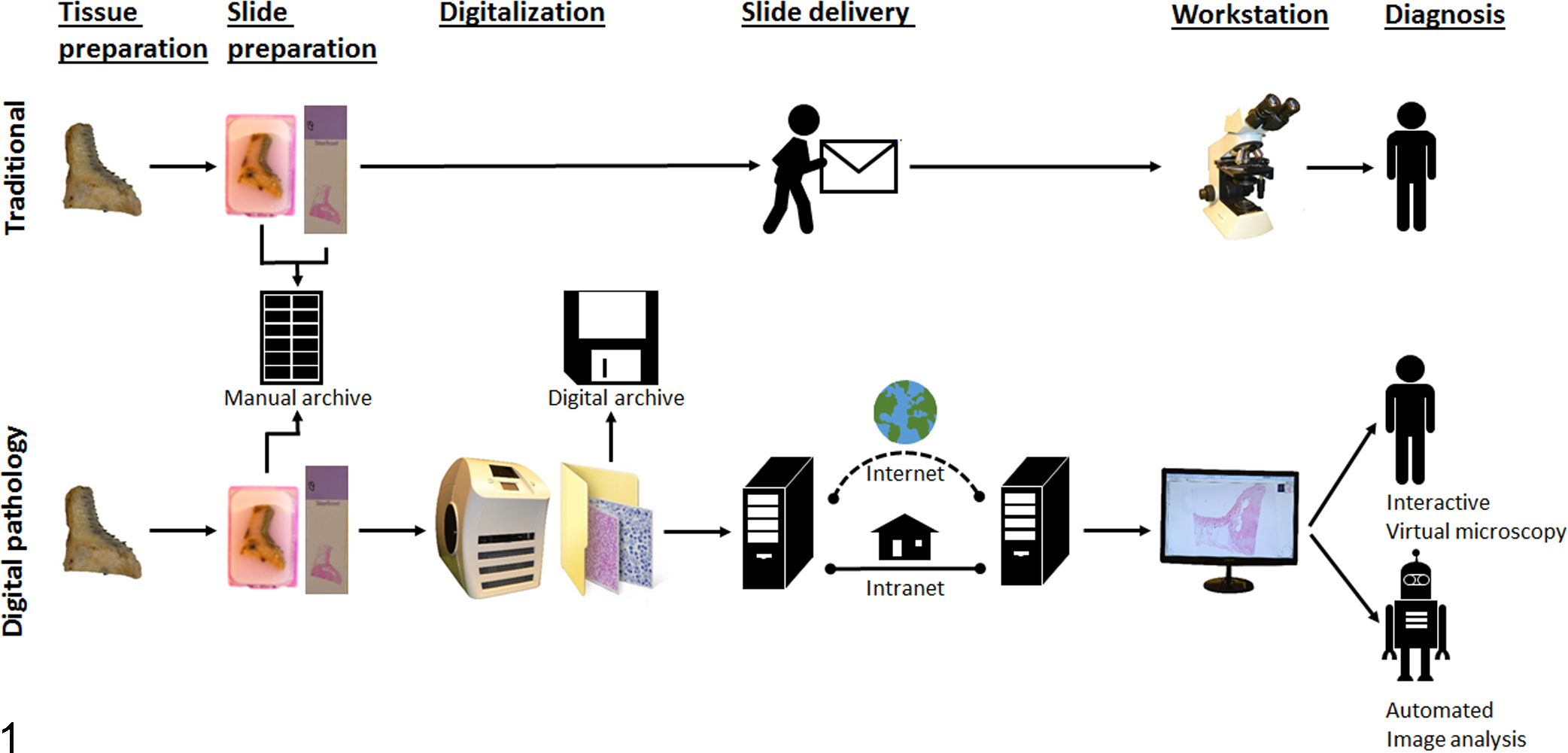
Comparison of the digital pathology and traditional workflow. The central step in the workflow of digital pathology that separates it from classical pathology is the digitization of histologic sections by a slide scanner into whole-slide images, which subsequently can be managed and viewed at a computerized workstation by a pathologist (interactive virtual microscopy) or be assessed by automated image analysis.
Light Source
Most slide scanners are equipped with a bright-field light source. Scanners with an integrated light source for the activation of fluorochromes to digitize fluorescent stains are also available and allow for examination of fluorescence signals at any later time point without the threat of fading. 2
Slide Holders
Maximum slide-loading capacities of the different scanners may vary between 1 and 400 slides. It is a very important feature that should fit to the institution’s digital workload. Especially diagnostic laboratories with a high caseload require large slide holders to avoid repeated and time-consuming manual reloading of the holder.
Objective Lenses
The objective lenses used in the different scanners vary between 2× and 100×, which allows maximum magnifications of 20× to 1000× on the screen, similar to a light microscope. Scanners are composed of various objectives or a single objective, of which the magnification may be doubled by adding an optional 2× lens. Most scanners are, however, equipped with 20× and/or 40× objectives. Only some models, such as the Aperio CS0 from Leica, are also able to scan at 1000× magnification using oil immersion. The decision for the magnification used during the scanning process clearly depends on the intended use. In our experience, WSI captured with 20× objectives are sufficient for the analysis of common entities with mostly very dominant morphologic patterns such as epithelial and mesenchymal tumors. However, at least 40× objectives are necessary for the analysis of hematopoietic tumors and subtle cellular changes on the cellular levels. Especially for cytopathologic diagnoses, 100× objectives may be beneficial. The decision to apply higher magnification is not simple, however, since it has a tremendous effect on the scanning time and the file size. For instance, doubling the magnification from 200× to 400× increases the file size by a factor of 4 from approximately 400 MB to 1.6 GB, and the scanning time for a whole-slide area is also increased by a factor of 4 from approximately 5 to 20 minutes with the Leica Aperio CS2 (see also paragraphs on scanning and storage). 29
Charge-Coupled Device Camera
The visual information of the slide is digitized by a high-resolution charge-coupled device (CCD) camera. Resolution and sharpness can therefore be characterized similar to regular digital photography by number and size of pixels. 17
Scanning
During scanning, the CCD camera takes single pictures of small areas that subsequently undergo complex postprocessing steps to produce the complete WSI. Currently, there are 2 different scanning methods applied. Area scanners produce separate squares or tiles, which are then put together as a WSI by image tilting. A linear scanner produces strip-type images, which are stitched together as a WSI. 17
Scanning time is regarded as an important feature of a scanner, especially when large numbers of slides are scanned every day, such as for routine diagnostics. The required time varies among the different models and is highly dependent on the size of the scanned area. 17
Scanning times of a complete slide with a 20× objective varies between 35 seconds and 3 minutes with the different scanner models according to the vendor’s specifications. 17 Most scanning software automatically selects a scanning area and avoids scanning of blank areas without tissues. This may, however, cause problems, especially for faintly stained slides or tissues such as fatty tissue, which may be not be automatically recognized.
Focusing is another delicate matter during whole-slide scanning. Currently, most commercial slide scanners do not focus dynamically during the scanning process but rather use a restricted number of focus points on the slide. This often leads to a lack of focus in areas between the focus points. In addition, suboptimal areas in the tissue section such as tissue folds, air bubbles, and incomplete covering by the coverslip strongly influence the focus of adjacent tissue. 50 Newer scanners are capable of focusing continuously during scanning, which reduces focusing problems. 2 Another approach to improve general focusing is z-scanning, which involves the scanning of multiple levels in the z-axis of the slide. This enables the viewer to fine focus digitally, just like in traditional LM. This not only may be helpful for dealing with inadequate focus but also may be necessary for detecting cellular details, especially for cytological specimens. However, z-scanning increases the file size and scanning time significantly with the factor +1 for any additional z-level. A complete failure of scanning in 1% to 5% of all scan processes is reported for the different scanning systems; however, this mainly results from suboptimal slide quality. 44,53,67
File formats of the produced WSI may vary between the different vendors and do not necessarily allow interoperability between different imaging equipment and medical information systems. To avoid the risk of losing data due to a change of the software, some authors have recommended the use of a universal standard image format such as the recently available DICOM standard. 2,29
Computer Workstation
An often-underestimated part of the DP workflow is the actual workstation of the pathologist where the WSI is viewed and evaluated. The pathologists “cockpit” is usually equipped with a personal computer, 1 or several monitors, and different input devices, commonly a computer mouse. Also, laptops, tablet computers, and smart phones may be appropriate for off-site and mobile diagnosis by telepathology, although they are usually of inferior quality because of the smaller size of the monitors and the cumbersome navigation on a touchscreen or keyboard. 9,75 Network connectivity speeds of at least 20 to 100 Mbps are considered sufficient for continuous and smooth exploration of WSI and thus a fast turnaround time. 1,2,74,75
The optimal workstation clearly depends on personal preferences, but new navigation tools are constantly tested to improve handling of the DP workstation. In addition to analysis speed, ergonomics are important since constant false posture may lead to occupational musculoskeletal disorders such as carpal tunnel syndrome. 36 So, common and vertical computer mouse devices, trackballs with scroll wheels, 6 degree-of-freedom navigators (6DOF navigator), touchscreens, gamepads, joysticks, and touchless devices have been used and tested for their efficiency. 37,43,47,56,57,67,68,78 For instance, one study rated a vertical mouse the best input device among 11 tested devices. 43 In another study, the 6DOF navigator was favored by most pathologists over a common computer mouse and a touchpad. 47
The quality of the monitor is another important factor for the quality of the histologic diagnosis using WSI. 36 In general, larger monitors may lead to an increased interpretation speed, because of a larger viewing field and therefore less navigation (ie, panning and zooming). 68,74 Higher resolution (ie, increased number of pixels) may be helpful for fast identification of relevant regions of interest. 56 The guidelines of the American Telemedicine Association recommend that the diagnosis monitor should be color calibrated, for instance, by using a color calibration glass slide, similar to the McBeth color checker. 5,51 However, the relevance of color calibration on diagnostic efficiency and reliability has not been analyzed systematically. 37 Still, color-calibrated WSI seems to be preferred by pathologists and might improve diagnostic confidence and speed. 12,37
Archiving of Digital Slides
Finally, WSI and associated information have to be archived (for a more detailed overview on slide archiving, see Huisman et al 29 ). A great advantage of archiving digital slides is fast and easy remote access, which can be very helpful for reexaminations, retrospective studies, and teaching. Furthermore, digital slides have a constant quality and will not fade or break over time, which is especially appropriate for medicolegal cases. However, storage capacities are still costly for the amount of data associated with DP. 29 A typical file of 1 WSI scanned with a 200× magnification and a single focus plane ranges from 400 to 650 MB after compression. 51 A medium-sized diagnostic pathology lab with 7000 biopsy cases with 4 slides per case on average produces 28 000 sections per year and thus requires at least 11 TB per year (average file size = 400 MB) if all slides are scanned only at 200× magnification. 27,29,67 Scanning of all slides with a 400× magnification, which is certainly required for some cases, would increase the required storage capacity by a factor of 4, that is, 44 TB per year. 27 Z-scanning would increase the size of the files by the factor z for all scanned levels. Although the costs for data storage have decreased tremendously in the past decade and are considered likely to decrease further, the total costs for storage should nevertheless not be underestimated. 27
Software Solutions for Digital Pathology and Automated Image Analysis
When using WSI, 2 applications need to be differentiated: interactive and automated VM. 32 Slide viewer software for interactive VM allows the user to explore the virtual slide actively comparable with LM and is generally provided free of charge by the vendors of slide scanners. In contrast, software for automated VM fully computerizes the analysis and is verified by the pathologist only after finalization of the task. Currently, few software packages are commercially available, and further development is required in this field, as will be discussed below.
Interactive VM
In contrast to LM, interactive VM provides further tools to facilitate the pathologist workflow. Among others, rotation of the image, use of magnification glasses, and thumbnail images for better orientation on the slide may improve handling. Points or regions of interest can be easily and quickly recognized by the use of very low magnifications (<< 20×) and permanently annotated such as by arrows, circles, and rectangles or counted. Distances, length, and areas can be directly and accurately determined by the use of calibrated rulers and individual annotations.
Automated VM
The human eye and its associated visual cortex is a highly efficient and reliable system to extract complex and variable information from images. Histopathologic diagnoses require the identification of highly complex patterns in the section and, at the same time, the ability to exclude irrelevant artifacts, which is currently not possible with any software solution. Pathologists are therefore still the gold standard for histopathologic diagnostic decisions. However, despite all training, significant intra- and interobserver variations are clearly present in human histopathologic diagnoses because of variation in human cognition but are currently accepted because of the lack of nonhuman alternatives. 20,33,70 Complementary automated image analysis may be an approach to increase the accuracy and reproducibility of pathological diagnoses, especially when dealing with quantitative measurements and a large amount of information (“big data”), which cannot be handled by the human brain. Automated image analysis describes all aspects of supervised and unsupervised extraction and processing of information from digital images by computer programs. 63 Currently, there are only very few veterinary studies that use automated VM. 6,10,23,25,69 For example, copper concentrations in the liver were examined with a quantitative positive pixel algorithm in comparison with qualitative manual histopathologic scoring and atomic adsorption spectroscopy (gold standard) and showed diagnostic usefulness of the quantitative positive pixel algorithm and an advantage over the qualitative score. 10 Another study tested canine blood smears using an automated image analysis system, designed to preclassify leukocytes in human blood smears. Results showed good to very poor preclassification of the different leucocytes depending on the type of leucocyte. 69 Although not thoroughly investigated, automatization decreased the required time for the differential cell count. Also, digital image analysis has been used to accelerate differential cell counts of avian blood smears; however, the authors were not able to develop a satisfactory algorithm, presumably because of considerable variability of blood smear preparation. 6 The feasibility of automated analysis of WSI for the analysis of more complex tissue sections has been proven in human pathology, where WSI was successfully used to improve the grading of renal carcinomas and follicular lymphoma. 18,79
Most of the image analysis algorithms used in the currently available automated image analysis programs are inspired by human visual perception. It is a very complex field of computer science and mathematics, and the keywords provided in Table 1 are a very restricted core of topics of relevance for automated image analysis. 7,11
Keywords From the Field of Computer Science and Mathematics With Relevance for Automated Image Analysis.

The availability of image analysis software products is steadily increasing. Each vendor of slide scanners or microscopic systems is currently offering its own system with a wide variety of features and components of automated image analysis with varying capabilities. These systems are usually very expensive but often provide up-to-date image analysis tools such as trainable histomorphology image analysis tools to identify different structures automatically in heterogeneous tissue samples, such as the Genie analysis software from Leica Biosystems. However, several free image analysis software tools are available and can be helpful, even though their capabilities for automated images analysis are often very restricted (Table 2). 31,61
Brief Summary of Selected Image Analysis Software That Is Free of Charge.

Digital Pathology in Diagnostic Service: Why the Investment?
Conventional LM has been an excellent method for the morphologic diagnosis of diseases for more than 100 years. The switch to VM therefore requires strong arguments to supersede this excellent and well-established method and to justify the investment. In the following paragraphs, the pros and cons of DP compared with conventional LM are summarized (Table 3). As with any new technology, technical advancements in DP are fast, and current limitations will progressively reduce with increased acceptance and the consequential increased investments in the larger market. So far, a DP workflow has been implemented in some human pathology laboratories for more than 10 years for routine primary diagnosis, including remote examination of intraoperative frozen sections and secondary consultation. 29,64,67 The published experiences indicate many advantages of the technology, although, admittedly, not all aspects of the currently available technology are satisfying.
Pros and Cons of Digital Pathology in a Diagnostic Service.
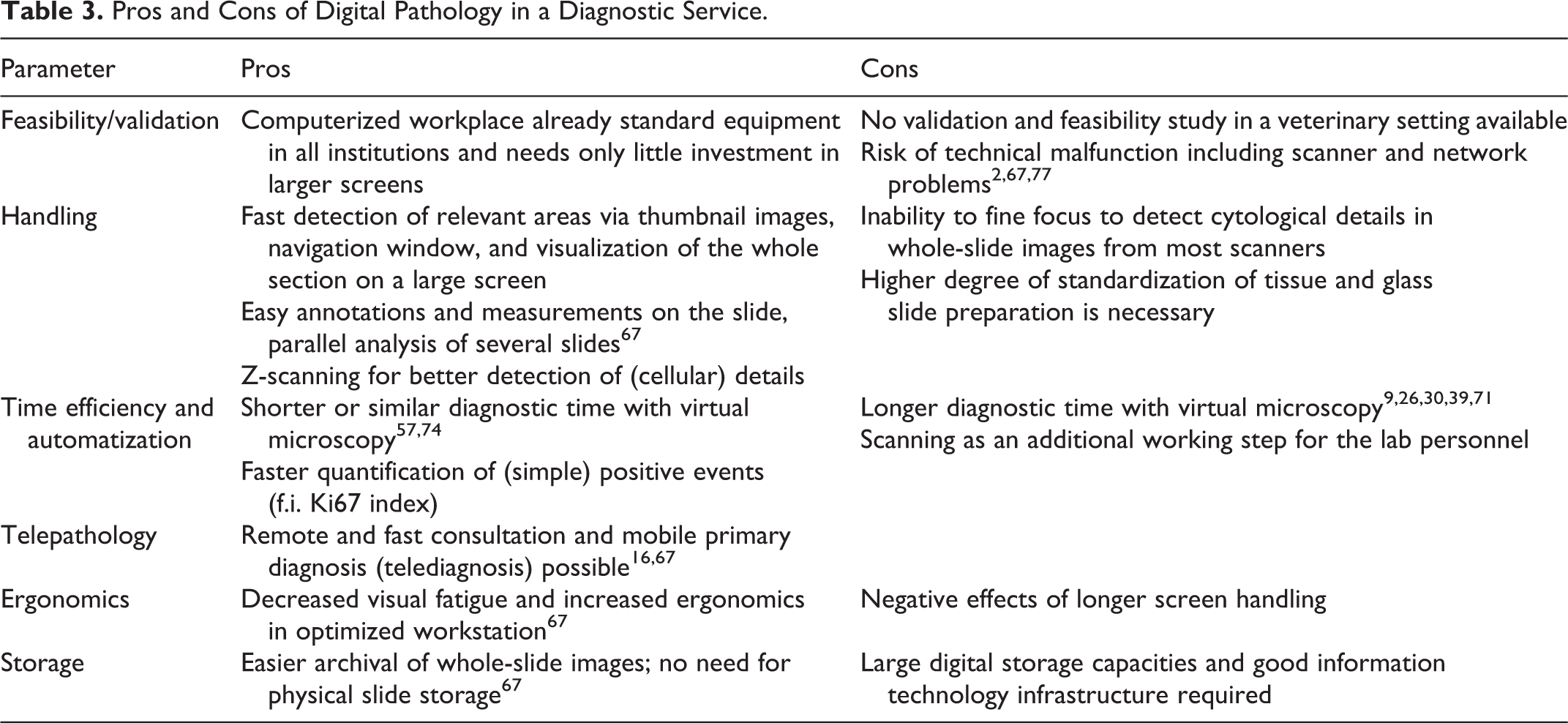
General Mind-set
“Technophobic pathologists” have been described as a major barrier to adopting DP in human pathology. 50,67 This exaggerated proposition is probably a problem of training and habituation rather than unreflected conservativism. The current training of residents in most training programs and the final board examination do not involve WSI as a mandatory topic. Familiarization with the technology and the individual DP system during training and prior to routine use will certainly overcome any negative mind-set that may be present. As will be described below, some veterinary schools, just like human medical schools, have introduced VM into their university curriculum and therefore build a generation of specialists who are familiarized with and confident in VM. However, an implementation of DP into training for students specializing in veterinary pathology and the actual board examination is also required.
Validation, General Feasibility, and Handling
So far, no validation study has been published for a veterinary diagnostic service setting to demonstrate the potential feasibility of DP systems. This is, however, indispensable, if DP systems are to be implemented for routine usage, especially in accredited laboratories. This validation must prove that interactive VM consistently has high concordance with LM as the current gold standard.
In contrast, systematic studies are available that address the reliability of VM in the daily routine in human pathology laboratories. They show that interactive VM has a high concordance comparable with LM. 24,60,64,67 The increased use of WSI for routine diagnostics and telepathology in human pathology has led to the development of guidelines for the adoption of DP, which may also be applied in a veterinary setting. 21,40,51,52 These guidelines require that every pathology laboratory implementing VM for diagnostic purposes should validate its DP system, including the scanner, software, network, and workstation (ie, computer, viewing monitors, mouse, or comparable navigation tools) to ensure diagnostic performance and reliability.
In terms of general feasibility, the initial acquisition of a slide scanner and software is usually the biggest hurdle, since it requires investments of about €100,000. Also, continuous costs for updating the software and electronic storage capacities should not be neglected. In contrast, computerized workstations and sufficiently fast networks are already available in most institutions.
After sufficient training, handling of VM may be more convenient compared with LM. One major advantage in our experience is the better overview of the complete section via thumbnail images (navigation windows) and the always-possible visualization of the complete slide on a larger screen with a consequently faster detection of relevant areas in the tissue. In addition, the WSI allow the easy and immediate use of calibrated rulers for accurate measurements, for example, of lesion sizes or distances of tumor cells from the surgical margin. 67,76 Furthermore, it is possible to add annotations to the slides that are saved for later inspection or consulting observers and to simultaneously display and compare several slides side by side on 1 or several monitors. The latter feature may be especially helpful for comparing different staining methods (including immunohistochemistry) of the same specimen or different specimens with the same staining method in diagnostic and in research settings.
The inability to fine focus is regarded as a major disadvantage of WSI using current standard slide scanners. This is most problematic for the diagnosis of cytological specimens, and therefore, feasibility is still controversial for this application. The new generation of scanners, which allow z-stack digital imaging (3-dimensional [3D] digital slides) and with the options of magnification of 1000×, are a potential but still costly solution to this problem, with very large image files to be stored.
Time Efficiency and Automatization
The difference in the time to diagnosis between a DP setting and conventional pathology depends very much on the familiarization with the respective system. For example, some studies have shown a longer diagnostic time with VM. 9,26,30,39,71 However, more recent studies have determined that pathologists familiarized with DP and the individual workstation have similar or even shorter diagnostic times compared with LM. 57,74 The digitization is, however, an additional step in laboratory personnel’s workflow, which requires manual loading of the scanner and very much depends on the speed and the loading capacities of the scanner (Fig. 1).
Automatization using automated image analysis may furthermore increase the time efficiency of DP. Several image analysis software solutions have been developed so far to assist the pathologist in diagnostic services. These algorithms are usually designed to reduce the monotonous, time-consuming, but rather less demanding unidimensional tasks, such as prescreening for fields of interest, counting rare events such as mitotic figures, or prescreening for micrometastases. 13,72 Another special focus in technology development is the quantification of immunohistochemical biomarkers (Table 4). Some of these algorithms for quantitative immunohistochemistry have received clearance for routine diagnostic service by the American FDA, including quantification of human epidermal growth factor receptor 2 (HER2), estrogen receptor (ER), and progesterone receptor (PR) expression. Indeed, it has been determined that automated image analysis of these biomarkers produces a higher diagnostic accuracy than manual scoring when tested against fluorescence in situ hybridization, which is currently the gold standard method. 19,46
Examples of Image Analysis Software in Human Pathology in Evaluation of Breast Cancer Available for Routine Diagnostic Service.a
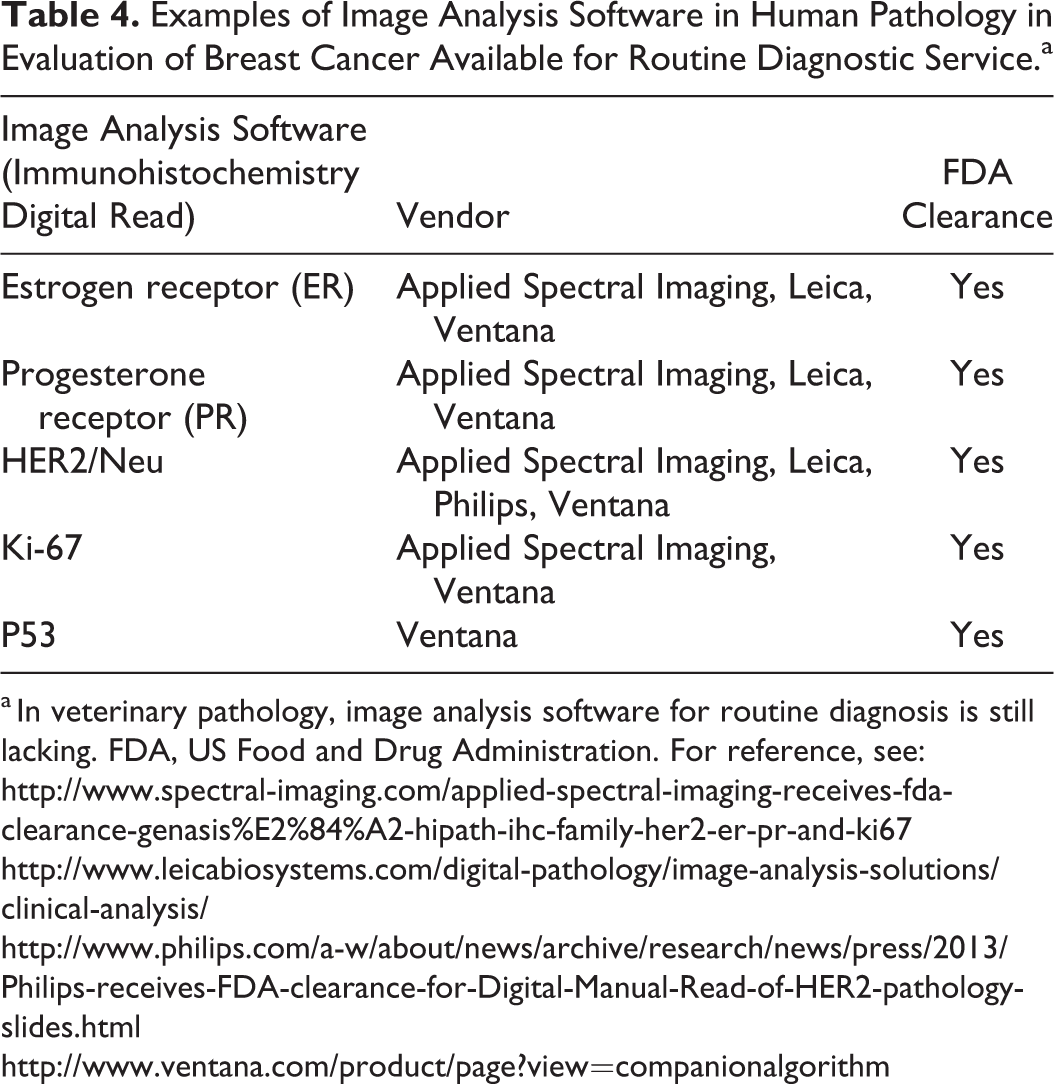
a In veterinary pathology, image analysis software for routine diagnosis is still lacking. FDA, US Food and Drug Administration. For reference, see:
Another field that is currently investigated but still in its fledgling stage is computer-aided diagnosis and prognosis. In human pathology, 41 just like in veterinary pathology, 33 tumor grading depends on examination of a specific number of high-power fields. Manual selection and analysis of these fields is vulnerable to selection bias and intra- and interobserver discordance. 33,41 Automated analysis of a WSI, however, can aid this determination and is somewhat feasible for some types of tumors in human pathology. 18,79 The first studies suggested that automatic WSI analysis may facilitate morphologic grading of renal carcinomas and follicular lymphoma and reduce variability compared with manual grading. 79 Similar studies are so far not published for applications in veterinary pathology.
Telepathology
Telepathology is another benefit of WSI. First, it allows off-site access to digital slides for primary diagnosis, which enables the pathologist to work away from the laboratory or from home. Second, the ability to easily and instantly forward WSI to colleagues and subspecialists at a distance enables rapid second opinion teleconsultation and teleconferencing between pathologists at different locations. 16,77 WSI are considered superior to other telepathology systems (ie, static or dynamic telepathology) because they allow the pathologist to explore the whole slide independently at various magnifications and in high quality. 50 In contrast to DP, static telepathology consists only of single images taken manually with a camera mounted on a light microscope in a quite time-consuming manner, while dynamic telepathology describes a quite cumbersome real-time streaming of visual fields by 1 active observer to a remote passive observers. 42 In veterinary pathology, static telepathology has been shown to achieve acceptable diagnostic accuracy and may currently be more common because of the affordability of the technical equipment. 42
Ergonomics
Improved ergonomics may be another reason to integrate DP systems into the routine pathology workflow, as reported by Thorstenson et al. 67 Based on a questionnaire, they showed that almost all pathologists experienced an improvement of ergonomics after implementation of DP into the diagnostic routine.
Storage of WSI
Storage of WSI on servers has many advantages. They can be accessed almost instantly even from remote places without the need to physically retrieve and deliver the glass slide from the manual archive to the histopathology laboratory and back. 67 However, digital slides require enormous amounts of cost-intensive storage capacities, especially if 400× or higher magnifications are used, which in our experience is often required for an acceptable image quality (see previous paragraph on hardware requirements). 1,67,74,76
Digital Pathology in Research
Scientific studies aim to obtain information that is objective and quantitative and if possible does not include any “human factor.” Pathology, in contrast, is still a mostly subjective, descriptive, and qualitative or semiquantitative scientific discipline that is performed by well-trained but still very human pathologists and is nevertheless still the gold standard. 38,76 The reason for this rather archaic approach to analyze histologic or cytologic slides is the unique ability of the human visual and neuronal system to process complex visual information, which has not been reached by any image analysis software so far. Nevertheless, the human factor introduces major drawbacks into histopathology, such as questionable reproducibility, reliability, slow and insufficient quantitative measurements, and considerable intra- and inter observer variability.
The recent technical advancements in automated image analysis pose a great potential to increase the reproducibility of histopathologic data. At its core, automated image analysis is thought to reduce the workload of the monotonous and repetitive task of counting, to increase reproducibility by elimination of intra- and interobserver variability, and thus to increase the degree of objectivity. 28,58 For instance, in a recent study, we used WSI of porcine liver sections after bipolar radiofrequency ablation in vivo to objectively quantify different zones of tissue destruction and cell death. With a self-designed algorithm, we were able to compare objectively the extent of cell damage in different tissue sections of different animals (Fig. 2). 23 Another more simple pixel count approach was used to quantify myocardial fibrosis in a murine model of pressure overload–induced cardiac hypertrophy (Fig. 3). 25
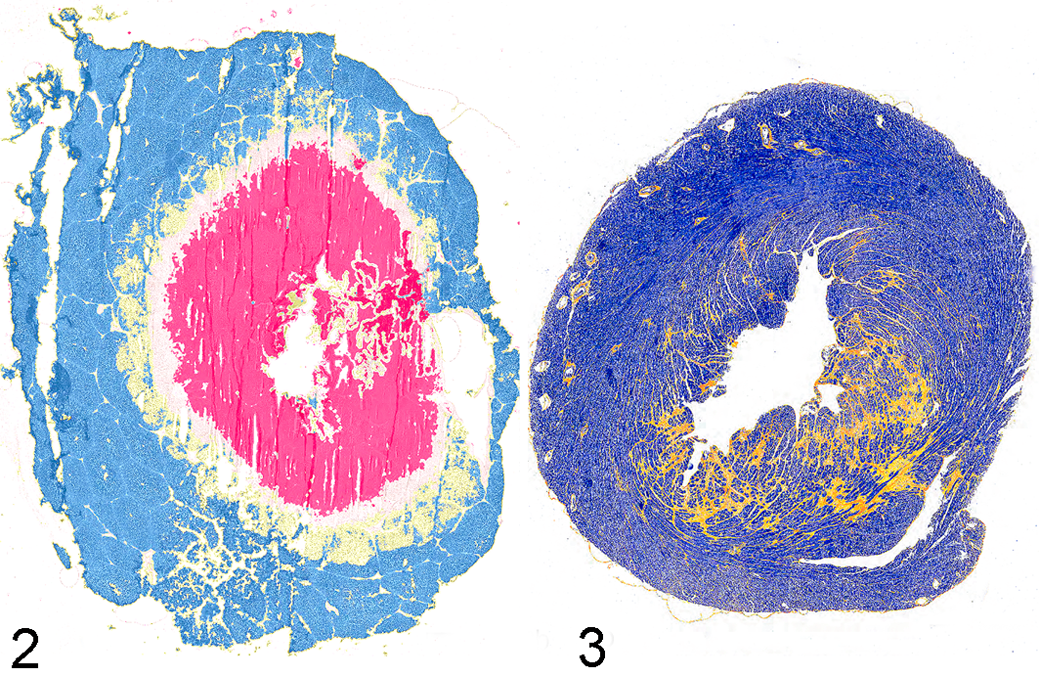
Another recent study used automated image analysis for scoring mouse models of colitis in comparison with the pathologist’s visual analysis as the gold standard. 59 Automated image analysis of hematoxylin and eosin–stained tissue sections failed to provide reproducible results because of the variation in the staining intensity of the slide. Variability in the staining intensity and other steps of tissue collection and preparation significantly influence the outcomes of automated image analysis, whereas pathologists are able to effectively integrate these artifacts and differences between slides in their decision process. 28,58,76 On the other hand, this sensitivity may, however, allow detection of slight and consistent differences that might be missed by a tiered, subjective, and semiquantitative measurement made manually by the pathologists, thus increasing the opportunities for statistical evaluation of slides with constant staining quality. 4,19,76 The analysis of immunohistochemical staining is a more common application of automated image analysis. It is based on rather simple yes/no decisions on the presence or absence of an immunohistochemistry signal (ie, brown pixel), with strong contrast to the background. In a study on automated image analysis of a murine colitis models, automated detection of the CD3-positive cells was strongly correlated with the pathologist’s score and was considered suitable for standardized assessments. 59 Objectivity is nevertheless an issue also for automated image analysis, because it is still the pathologist who develops or trains the algorithms and thus integrates subjective decisions during this process. The high reproducibility that comes with automated analysis should not be confused with reliability or accuracy of the analysis. For example, a new source of discordance is intra-algorithm variations. 66,76
Digital Pathology for Educational Purposes
Traditionally, histopathology and cytopathology have been taught using LM and glass slides for every student, static and dynamic images, multiheaded microscopes, or video microscopy. 15,34,62 All of these educational tools have disadvantages: access to light microscopes and glass slides is restricted to specific time and places; in static images, it is impossible to zoom in or out and explore the surrounding tissue; multiheaded microscopes allow the examination of slides only in small groups; and video microscopy often is of inferior quality and handling is cumbersome. 15,62
More recently, VM has been identified as an excellent alternative method to teach and examine in histopathology and is increasingly implemented in many veterinary faculties in the curriculum of histopathology, histology, and parasitology as a tool for lectures, self-directed learning, and practical examinations.* VM is usually offered complementary with conventional LM. Some medical faculties even completely replaced LM with VM for their education with an overall very positive feedback. 35,49 However, despite the positive perception of VM, veterinary students highly appreciate being trained additionally by the means of traditional LM. 34 This is in accordance with some pathologists, who fear that a strong focus on VM in the education of veterinary students may lead to a lack of transfer of skills in the analysis of glass slides by conventional light microscopes, which is still almost exclusively used in routine veterinary practice, especially for cytopathology analysis. 22,48,62,65 A survey about the use of microscopes in veterinary practices revealed that 93% of practitioners use microscopes in at least every 10th appointment for the analysis of fecal samples, fine-needle aspirates, skin scrapings, and vaginal smears. 65 Knowledge transfer of LM techniques should thus still be considered as substantial “day 1 skills” of veterinary students. 34,48
Table 5 summarizes the main advantages and disadvantages of the integration of VM into the veterinary curriculum. Students often mention accessibility at any time and location and the annotation of key features in the slide as the main advantages of VM. 15,22,34,45 In addition, they appreciate that VM allows easier orientation in the slide via thumbnail images and low-magnification whole-slide views. In addition, the current generation of well-trained students usually appreciates the available WSI viewers as easier to handle than a traditional microscope. 14,62
Advantages and Disadvantages of Virtual Microscopy in Veterinary Education.
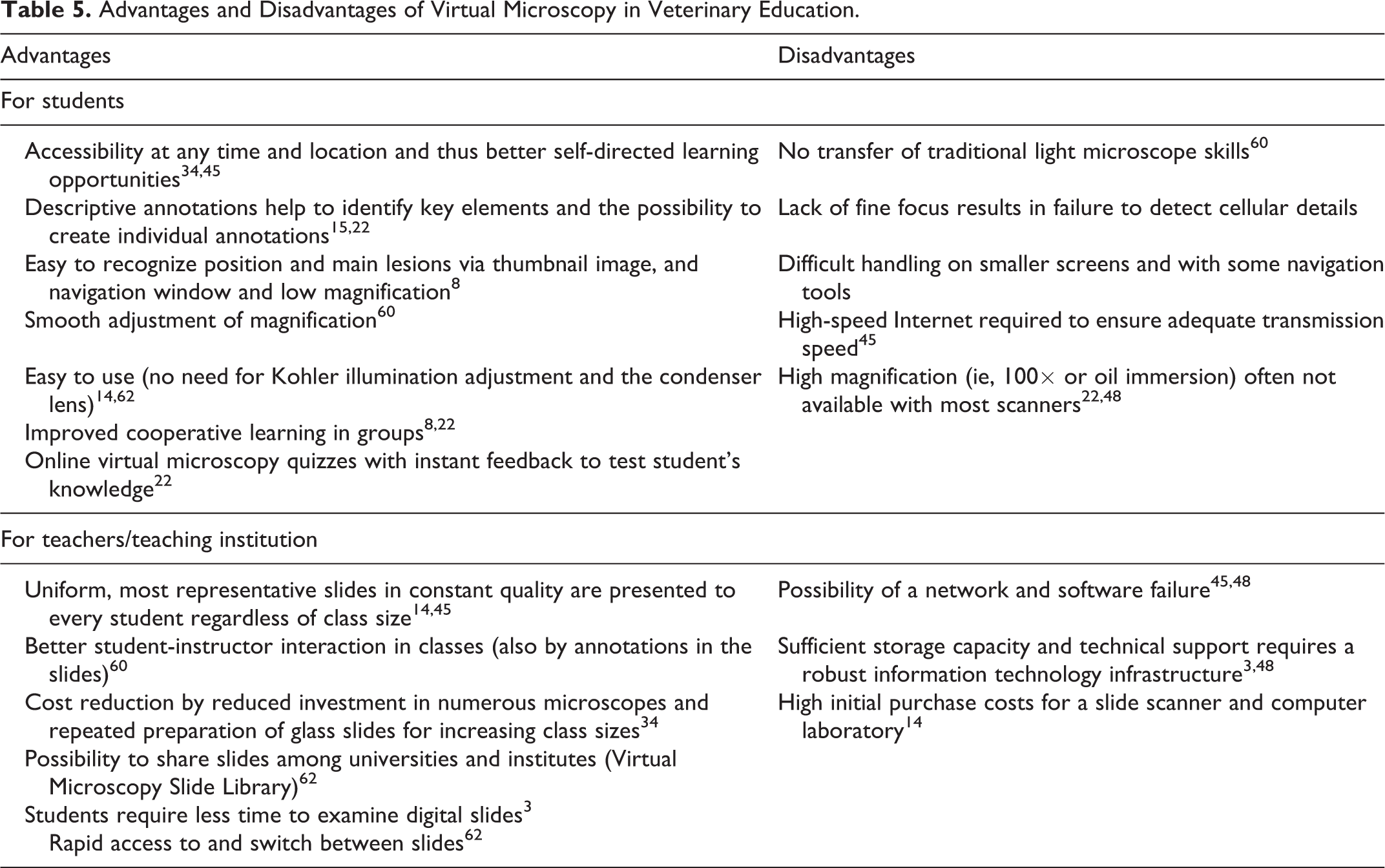
The transfer of histopathologic skills seems to be similar in VM compared with traditional microscopy. The overall accuracy and examination performance of students trained by VM or LM did not differ significantly in first studies, despite the warm appreciation of VM by veterinary students. 8,49 Still, lecturers consider DP a great supplement and excellent didactic tool for teaching cytopathology and histopathology. 34,48,62 It allows the presentation of uniform and most representative cases in constant quality to every student regardless of class size. 14,45 The presentation of WSI is usually more interactive and flexible and supported by short-term or permanent annotations in the WSI. 60 WSI can also be shared between institutions and may finally lead to a VM slide library, which is contributing to a more standardized interinstitutional education of veterinary students and veterinary pathology residents. 62 One of the major hurdles to integrating DP into the curriculum is certainly the high investment in expensive slide scanners. However, the reduced need for investment in light microscopes and the concurrent application of WSI for diagnostic and research purposes may justify the costs to stakeholders.
Conclusion and Future Prospects
Virtual microscopy is increasingly applied in all fields of pathology and poses a unique chance to improve the performance of pathologists as diagnosticians, researchers, and teachers, despite all current minor weaknesses. Major advantages of DP and WSI include remote and off-site access to digitalized slides, easy handling, improved ergonomics, and quantitative measurements. In addition, although digital veterinary pathology is in its fledgling stage, automated image analysis will improve in the next decade and will certainly facilitate and broaden the pathologist’s work by providing higher reproducibility and reliability of qualitative and quantitative diagnoses. These innovations will inevitably influence the pathologist’s routine workflow regardless of whether we are skeptics or promotors of digital pathology. Therefore, we should wonder less if we like the technology and instead consider how we can integrate it in our current workflow in order to contribute to its further development, rather than leaving it completely to information technology specialists. Current and future pathologists should therefore learn and be taught information technologies and image editing to keep up with the inevitable digitization of their profession.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
