Abstract
Lymphoma has been described in individual cases for goats but not systematically characterized in a larger cohort. This study aimed to subtype caprine lymphoma based on topographic and subgross distribution, immunophenotype, and cellular morphology following the World Health Organization classification system for hematopoietic tumors in domestic animals. Fifteen caprine lymphoma cases were assessed with 6 submitted as biopsy and 9 for postmortem examination. Goats were predominantly young adult (median 3 years) and dwarf breeds (Pygmy and Pygora). The sexes were similarly represented. Nuclear size was measured relative to red blood cells (RBCs) and then adjusted for species-specific differences and designated small (<3× RBCs), intermediate (3–4× RBCs), or large (>4× RBCs). Using immunohistochemistry, 11 of 15 (73%) goats had T-cell lymphoma (TCL; CD3 positive, CD79α negative) and 4 of 15 (27%) had B-cell lymphoma (BCL; CD79α positive, CD3 negative). A multicentric distribution was most common. TCL generally involved the thoracic cavity and/or neck, suggestive of thymic origin or homing. TCLs were further classified as lymphoblastic lymphomas (3/11; 27%), large granular lymphocyte lymphoma (1/11; 9%), diffuse small lymphocytic lymphomas (3/11; 27%), or peripheral/mature T-cell lymphoma (PTCL) not otherwise specified (4/11 [36%], of which 3 were high grade and 1 intermediate grade). In 1 goat with PTCL, lymph nodes had either paracortical expansion or diffuse infiltrates suggesting transition from nodular to diffuse PTLC. BCLs were classified as diffuse large B-cell lymphoma (2/4; 50%) or B-cell lymphocytic lymphoma intermediate type (2/4; 50%). In contrast to dogs and horses, lymphomas in goats are predominantly TCL and frequently involve the mediastinum.
Larger retrospective studies of caprine neoplasms are few. 5,7,11,18,23,25,39 A 40-year survey of pigs and goats in the Republic of South Africa published in 1983 reported that lymphoma was the third most common tumor, accounting for 14% of the total caprine tumors, compared to 19% for each papilloma and malignant melanoma. 5 In a recently published study of 102 tumors in 100 goats obtained through the Oregon Veterinary Diagnostic Laboratory from 1987 to 2011, lymphoma was the most common neoplasm, comprising 17.7% of the assessed tumors. 25 A multicentric distribution has been reported most frequently for caprine lymphoma. 10,12,22,25,32
Clinical signs of caprine lymphoma can mimic other conditions such as lymphadenitis and respiratory or gastrointestinal diseases, which has implications for herd health and potentially public health. Enlarged peripheral lymph nodes are commonly attributed to caseous lymphadenitis, caused by
Lymphoma in goats has been described in a number of individual cases 8,12,13,20 –22,29,32 but not systematically characterized by tumor cell type and topographic distribution. Here we further characterize 15 lymphomas by immunophenotype and morphology. A classification system has not yet been reported for lymphoma in goats. In this study, we apply morphologic features used to classify lymphoma according to the World Health Organization (WHO) classification system adopted for animals to facilitate the description and comparison of neoplastic lymphocytes and lymphoma subtypes in goats. 34
Materials and Methods
Cases
Cases were retrieved from the archives of the Oregon Veterinary Diagnostic Laboratory (OVDL) at Oregon State University (OSU) as previously described.
25
Entries in 2 different database systems, Data General (1987–2001) and Visualab (2001–2011), were analyzed. All diagnostic codes, including the term
Histopathology and Tumor Classification
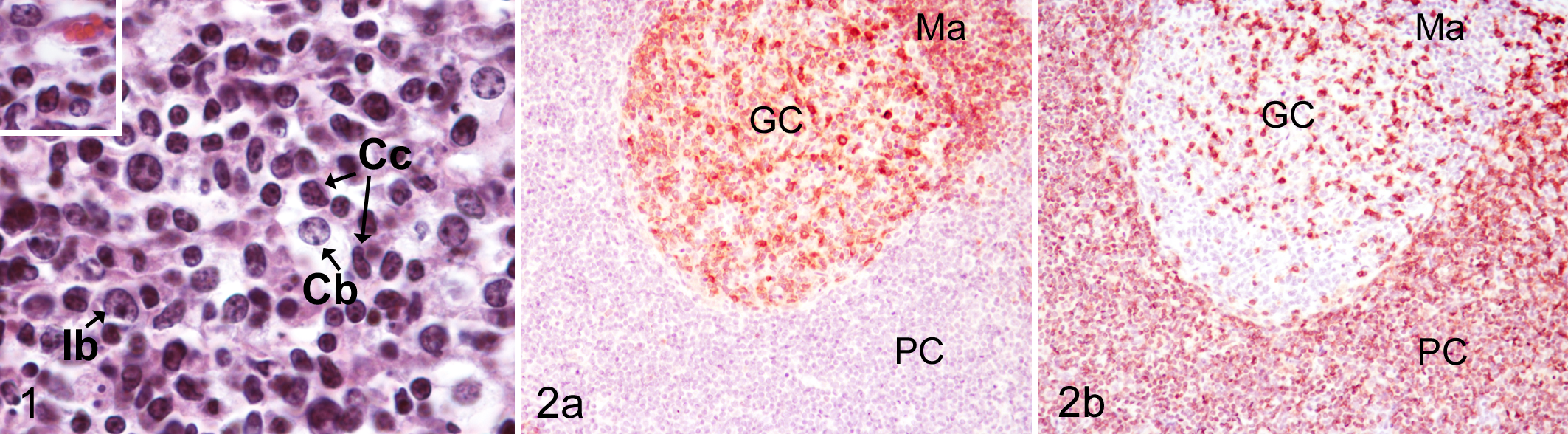
Histopathology was performed on archived slides or freshly cut slides from archived paraffin blocks. Histologic sections stained with hematoxylin and eosin (HE) were evaluated for cellular morphology. Classification of the 15 lymphoma cases was based on topography, cell distribution, and cellular morphology based on the updated WHO criteria of classification adapted for specific animal species and, in particular, canine subtypes. 34,36 Distribution was recorded as diffuse or nodular. Nuclear size was initially designated as follows: small (<1.5× the diameter of a red blood cell [RBC]), intermediate (1.5–2× RBCs), and large (>2× RBCs). However, caprine RBCs are approximately half the size (3.4 μm in diameter) of those in dogs (7.01 μm). 2 To account for this species difference, an adjusted nuclear size was determined by multiplying the number of the relative RBC diameter in each size category by 2 (ie, small, <3 RBCs; intermediate, 3–4 RBCs; large, >4 RBCs) and used subsequently. Nuclear morphology was assessed for overall shape, round or oval, and for secondary features, shallow or sharp indentations or irregular contour. Nucleolar and chromatin morphology was described as having dispersed chromatin with indistinct nucleoli, chromatin bands or chromocenters with marked parachromatin clearing and indistinct nucleoli, and distinct nucleoli. Mitotic figures were averaged over 10 fields using a 40× objective (field of view diameter 0.55 mm) and expressed as average mitoses (MA), and they were categorized as low (0–5 mitoses/400× field), medium (6–10 mitoses/400× field), and high (>10 mitoses/400× field). The amount of cytoplasm was categorized as abundant, moderate, or scant to small. The cytologic features of normal caprine lymphocyte subtypes are illustrated in Fig. 1. Brief descriptions of the pertinent features of lymphoma subtypes identified in this series of goats are provided in the Results section.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on an autostainer (Dako Autostainer Universal Staining System; Dako, Carpinteria, CA) according to standard operating procedures. Paraffin sections were high-temperature antigen retrieved with BDTM Retrieval A solution (Dako). Endogenous peroxidase activity was blocked by immersing slides in methanol containing 3% hydrogen peroxide for 10 minutes. The following primary antihuman antibodies (DakoCytomation, Carpinteria, CA) were applied for 30 minutes at room temperature: mouse monoclonal against human CD3 and rabbit polyclonal against human CD79α (Dako M7051 and A0452). These antibodies have been used for the characterization of canine lymphoma 36 and in a previous goat lymphoma case report from Oregon State University. 32
MaxPoly-One Polymer HRP Rabbit or Mouse Detection solution (MaxVision Biosciences, Bothell, WA) was applied for 7 minutes at room temperature, and Nova Red (SK-4800; Vector Labs, Burlingame, CA) as chromagen was used with Dako hematoxylin (S3302) as counterstain. Serial sections of neoplastic tissue incubated with Dako Universal negative serum served as negative controls. Serial sections of a normal caprine lymph node from a goat with no evidence of lymphoma and incubated with CD3 or CD79α or incubated with Dako Universal negative serum were used as positive or negative methods controls, respectively.
Evaluation/scoring of lymphomas was performed based on the proportion of antigen-positive cells (CD3 and CD79α) and used to determine cell type (T- vs B-cell lymphoma, respectively) as follows. Positive staining was defined as red chromagen precipitate limited to the cell membrane. Dark red precipitate was defined as strong and pale red precipitate as light staining. Positive staining between dark and light red was defined as moderate intensity. Averaged over 10 fields at 400×, tumors with a majority of cells (≥75% immunolabeled cells) that were strongly (+++), moderately (++), or lightly stained (+) were considered positive for the target antigen. Tumors with few scattered stained (+/–) or no stained cells (–) were considered negative for the lymphoma phenotype defined by the antigen (Table 1). Distribution of lymphocyte lineages in general and in context of resident lymph node or tissue architecture was recorded and used in the determination of subtype.
Caprine Lymphoma Cases.
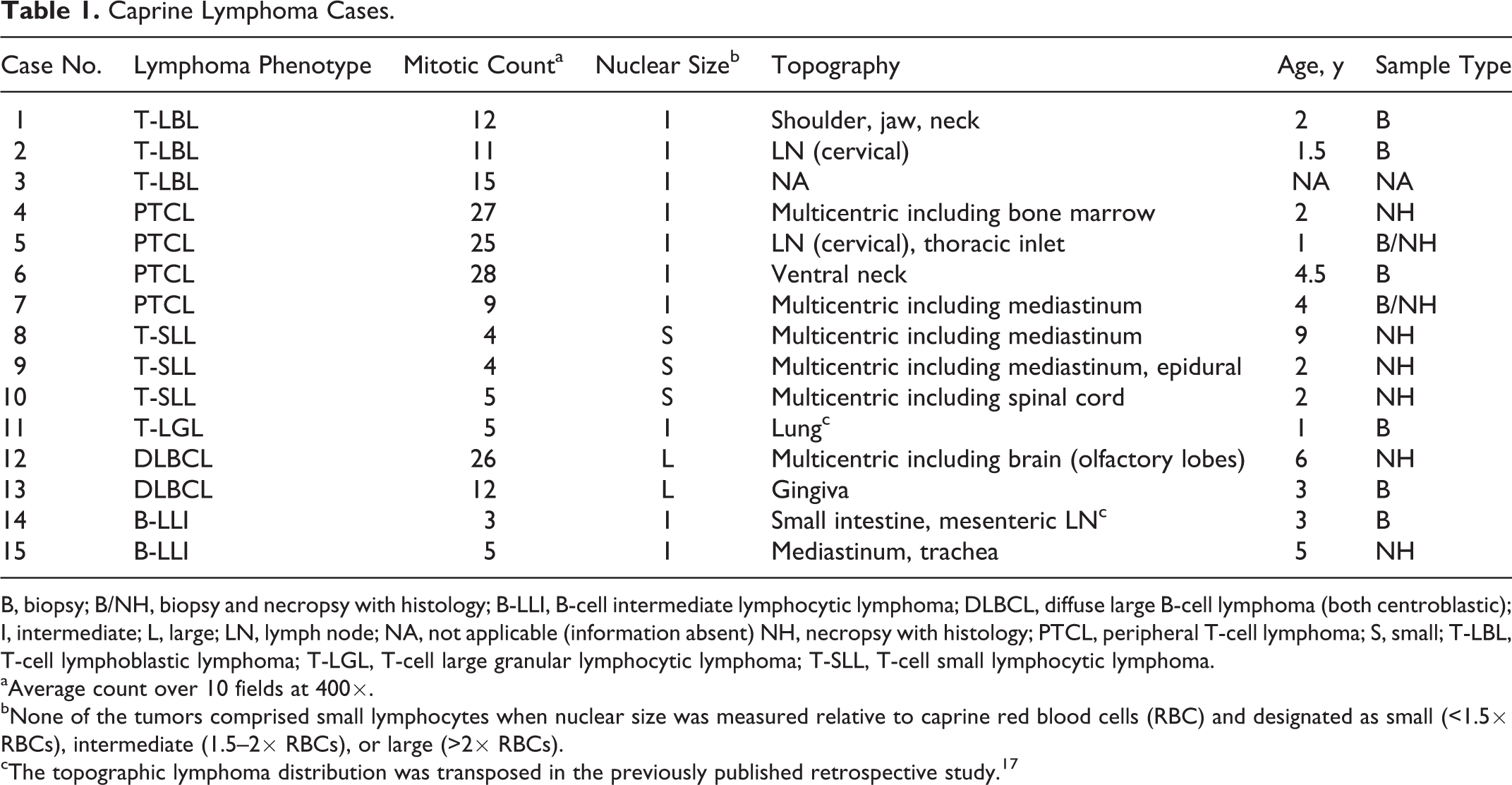
B, biopsy; B/NH, biopsy and necropsy with histology; B-LLI, B-cell intermediate lymphocytic lymphoma; DLBCL, diffuse large B-cell lymphoma (both centroblastic); I, intermediate; L, large; LN, lymph node; NA, not applicable (information absent) NH, necropsy with histology; PTCL, peripheral T-cell lymphoma; S, small; T-LBL, T-cell lymphoblastic lymphoma; T-LGL, T-cell large granular lymphocytic lymphoma; T-SLL, T-cell small lymphocytic lymphoma.
aAverage count over 10 fields at 400×.
bNone of the tumors comprised small lymphocytes when nuclear size was measured relative to caprine red blood cells (RBC) and designated as small (<1.5× RBCs), intermediate (1.5–2× RBCs), or large (>2× RBCs).
cThe topographic lymphoma distribution was transposed in the previously published retrospective study. 17
Cell surface/membrane-associated staining for CD3 and CD79α in the normal goat lymph node control was between +++ and ++ in intensity. Anti-CD3 antibody labeled the membrane of lymphocytes in the T-cell zone as expected with fewer stained lymphocytes in the germinal center and the medulla; lymphocytes in the mantle and marginal zone were largely negative (Fig. 2b). Conversely, lymphocytes in follicular centers, the mantle and marginal zones, and plasma cells and scattered lymphocytes in the medulla, but not in the T-cell zone, showed membrane-associated staining with CD79α (Fig. 2a, respectively).
Results
Animals/Cases
Paraffin blocks were available for 14 of 17 goats with lymphoma from the previous report and were used in this study.
25
One additional lymphoma case was diagnosed during necropsy since the publication of the previous report and was included here, which resulted in a total of 15 caprine lymphomas in this current study (Table 1 and Suppl. Table S1). Complete records were not archived for 1 case. Of 14 cases with complete records, lymphoma was diagnosed in 6 biopsy specimens and 8 necropsies. For 2 of the 14 goats, both a biopsy and necropsy with histopathology submission were available. These cases were included as necropsy to provide the most complete and detailed information about topographic distribution. However, the better preserved tissues from the biopsy submission were used for further analysis in this study. Goats with lymphoma in this study were adults, with reported ages ranging from 1 year to 9 years (median, 3 years). Eight cases were female, 5 were castrated males, and 1 was an intact male. Breeds represented in this study included Pygmy (
Information about topographic location/distribution of the lymphoma was available for 14 cases (summarized in Table 1; reported in detail in Suppl. Table S1). All 8 animals assessed by necropsy with histopathology were diagnosed with multicentric lymphoma, with the majority of tumors identified in the mediastinum/thymus (
Classification of Lymphomas
Of the 15 lymphomas, 11 (73%) were classified as T-cell lymphoma (TCL) by immunohistochemistry (CD3 positive, CD79α negative) and 4 tumors (27%) as B-cell lymphoma (BCL; CD79α positive, CD3 negative) (Suppl. Table S2). None of the tumors were double negative (ie, neither staining for CD3 nor CD79). There was slight variation in overall IHC staining score based on a combination of staining intensity and proportion of positive cells (Suppl. Table S2). One case that had moderate staining intensity for CD3 and light staining intensity for CD79α was categorized as BCL (Suppl. Table S2) based on cell proportions and cytological features (case 15).
All caprine lymphomas in this series had a diffuse distribution of neoplastic cells, with the exception of 2 cases: case 15, which had prominent stromal septa resulting in a nodular pattern, and case 4, which had a mixture of diffuse infiltrates and paracortical expansion.
Neoplastic cell morphology was assessed by nuclear size, nucleolar and chromatin features, amount of cytoplasm, and mitotic count of HE-stained slides (Table 1 and Suppl. Table S2). Results for nuclear size, after adjustment for species differences in RBC size to more closely follow the previously published classification scheme, 30,33 are reported in Table 1. Of the 11 TCLs, 3 (31%) had small nuclei (<3× RBCs), 8 (73%) had intermediate-sized nuclei (3–4× RBCs), and none had large nuclei (>4× RBCs). Of the 4 BCLs, 2 had large nuclei, 1 had intermediate nuclei, and 1 had small nuclei. Using caprine RBC as reference (Suppl. Table S2), none of the lymphomas had small nuclei. Four of 11 (36%) TCLs had intermediate-sized nuclei and 7 of 11 (64%) had large nuclei. All 4 BCLs had large nuclei. The adjusted nuclear size was used in subtyping lymphomas.
Counts of mitotic figures averaged over 10 fields using a 40× objective were reported as mitotic figures per one high-power field (MA). Six of 11 (55%) TCLs and 2 of 4 (50%) BCLs were high grade (MA: >10). Three of 11 (27%) TCLs and 2 of 4 (50%) BCLs were low grade (MA: 0–5). Two of 11 (18%) TCLs had an intermediate mitotic count (MA: 6–10).
Lymphoma Subtypes
T-cell lymphomas were subclassified as T-cell lymphoblastic lymphoma (
The diagnosis of T-cell lymphoblastic lymphomas (T-LBL; Figs. 3–5) was based on diffuse infiltrates of neoplastic CD3-positive cells that largely effaced lymph node architecture (Fig. 3) and had intermediate-sized nuclei with dispersed chromatin, indistinct nucleoli, and frequent mitotic figures (Fig. 5). All cases had round to irregularly shaped nuclei with occasional sharp indentations of nuclear membranes. Cortical sinusoids were compressed but neoplastic cells were contained within the lymph node capsule (Fig. 4). Small follicular remnants of CD79α-positive cells were present in 1 case (Fig. 4). The 2 cases, for which the topographic distribution was recorded, involved the cervical lymph nodes, shoulder, jaw and neck (as per history; both cases were submitted as biopsy).
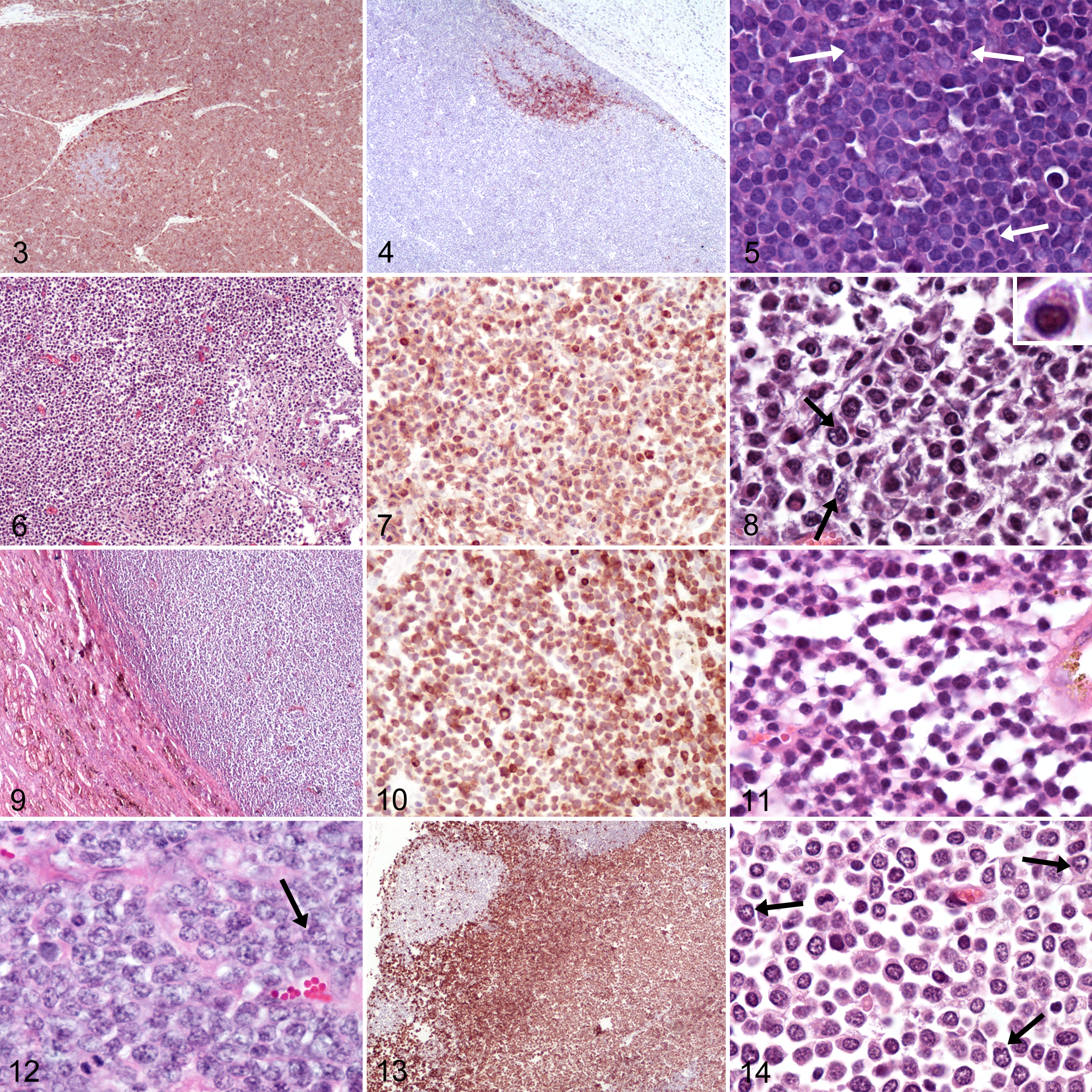
The diagnosis of T-cell large granular lymphocyte lymphoma (LGL; Figs. 6–8) was made in 1 case based on large azurophilic granules within the abundant cytoplasm of large neoplastic cells (Fig. 8, inset). In this case, large granular lymphocytes were CD3-positive cells and had intermediate to large nuclei with occasional deep indentations. This T-cell LGL involved a large portion of the right cranial lung lobes, which were adhered to the pleural surface, and associated intrathoracic lymph nodes.
Diffuse T-cell small lymphocytic lymphoma (SLL; Figs. 9–11) was diagnosed in 3 goats. The key feature of SLL is the uniformly small size of tumor cell nuclei. In the T-cell SLL of this study, lymphocytes had mostly small to rarely intermediate-sized, round nuclei with occasional shallow to rarely deep indentations and dense chromatin obscuring nuclear detail (Fig. 11). All 3 were low grade with 4 to 5 mitotic figures per 400× high-power field (HPF). Two T-cell SLL involved the mediastinum and other sites but not peripheral lymph nodes. The third case had a widespread (systemic), extranodal, and extrathymic distribution; bone marrow showed no evidence of infiltration. In 2 goats, small lymphocytes were present in the vertebral canal.
The remaining T-cell lymphomas were diagnosed as mature (= peripheral) T-cell lymphomas (PTCLs) as the neoplastic infiltrates, and tumor cells lacked characteristic features of other T-cell lymphoma subtypes. One of the 4 PTCLs was intermediate grade with an average of 9 mitotic figures per HPF. Lymphocytes in this PTCL had intermediate-sized, round nuclei with shallow indentations and chromatin branching with parachromatin clearing. Apoptosis was readily identifiable and resulted in a prominent starry sky appearance from frequent tingible-body macrophages containing apoptotic bodies. There was widespread involvement of both peripheral and intracavitary lymph nodes as well as extranodal sites, including the mediastinum. The remaining 3 PTLCs were high grade with over 25 mitotic figures per HPF. Two had diffuse infiltrates of lymphocytes with scant cytoplasm. Nuclei were round to oval with occasional shallow indentations and had branching chromatin patterns without discernable nucleoli (Fig. 12). Both lymphomas were extended over the ventral aspect of the neck and involved the thoracic inlet. The third high-grade PTLC (Figs. 13, 14) had infiltrated lymph nodes, many organs, mucous membranes (eyelids), tonsils, and bone marrow but not mediastinum. Neoplastic cells had moderate to abundant cytoplasm and round to oval nuclei with frequent sharp indentations (Fig. 14, arrows). Most nuclei had chromatin branching with parachromatin clearing and peripheral aggregation. Nucleoli were present but not readily identifiable. In some lymph nodes of this goat, the neoplastic cell infiltrate was limited to the expanded paracortex and compressed but did not obliterate follicular structures and subcapsular sinusoids (Fig. 13). Small foci of necrosis were frequent but vascular infiltration was not identified.
Four lymphomas had a B-cell immunophenotype and were classified as either diffuse large B-cell lymphoma (
Both diffuse large B-cell lymphomas (DLBCLs) were of the centroblastic subtype (Figs. 15–17). This subtype is defined by variable but often large tumor cell nuclei, high mitotic counts, and diffuse infiltrates. Both cases had large round nuclei with occasional shallow indentations and often multiple large nucleoli (Fig. 17). One of the DLBCLs was multicentric with extensive involvement of mucous membranes of the head and extension into the olfactory lobes of the brain. The other DLBCL was diagnosed in the oral cavity with no other site examined. Stroma was delicate and finely reticular in the multicentric DLBCL and more prominent and coarser in the oral DLBCL, consistent with the collagen richness of the resident tissue (Lamina propria).
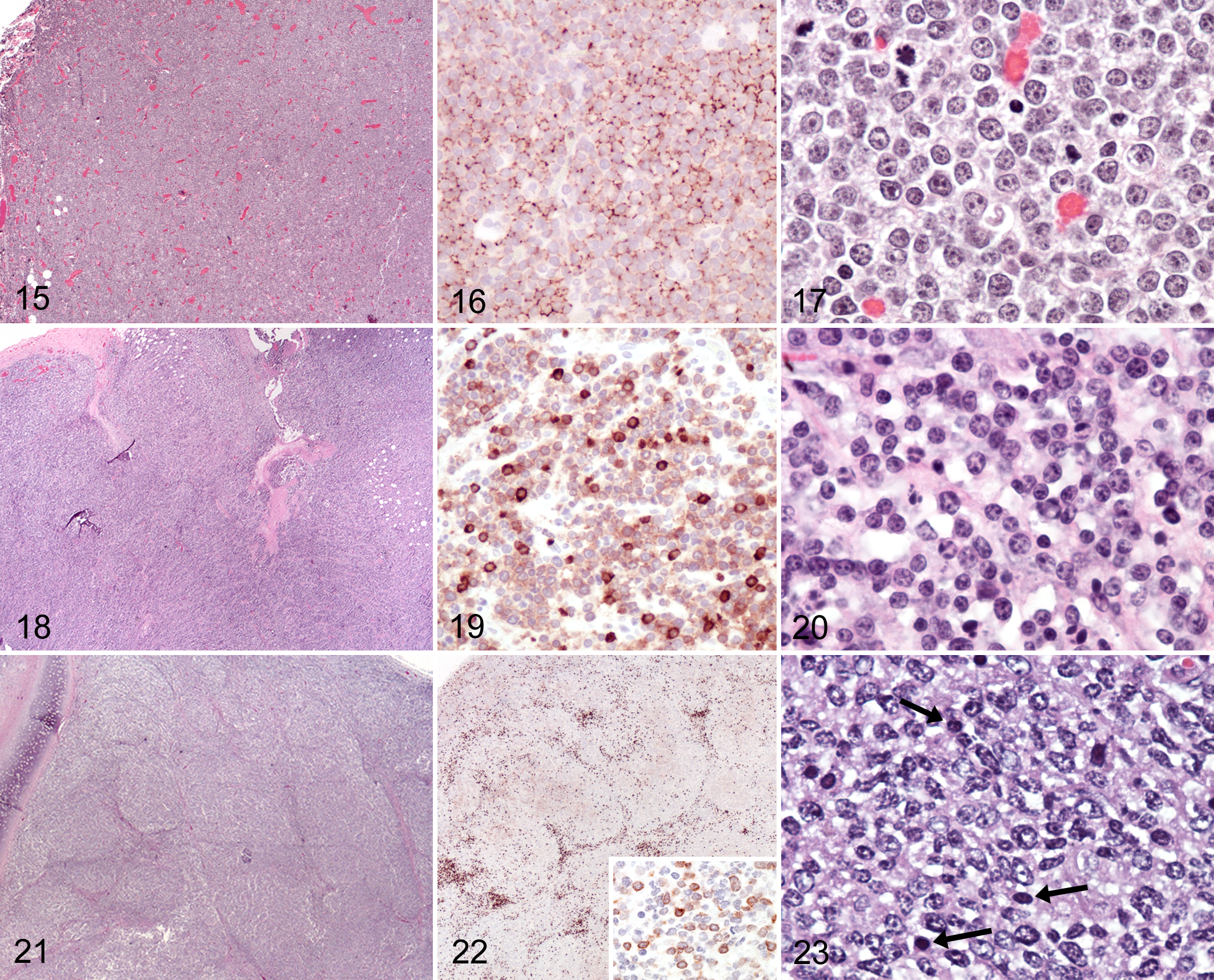
B-cell lymphocytic lymphoma intermediate type (LLI) was diagnosed when tumors had intermediate-sized nuclei and could not be assigned to another lymphoma subtype. One goat had B-cell LLI in the intestine and mesenteric lymph nodes (case 14). Round cells with moderate amounts of cytoplasm formed diffuse infiltrates that were contained within the lymph node capsule and had rare mitotic figures (Figs. 18–20). Small- to intermediate-sized, round nuclei with shallow indentations had branching chromatin or prominent chromocenters and indistinct to multiple small nucleoli. A few CD3-positive cells were scattered throughout the neoplastic infiltrate, supporting stroma, capsule, and perinodal tissue. The cellular features of this single intestinal lymphoma most closely resembled cells of the marginal zone, and mucosa-associated lymphoid tumor (MALT lymphoma) or MALT-associated marginal zone lymphoma (MZL) was considered. However, neoplastic cells in the mesenteric lymph node lacked the typical subgross distribution into bands connecting follicles.
The remaining B-cell lymphoma was from the mediastinum and trachea of the single Alpine goat (case 15). A nodular subgross appearance was imparted by prominent stromal septa separating dense aggregates of intermediate sized CD79α-positive lymphocytes (Fig. 21). Lymphocytes immunolabeled for CD79α had intermediate-sized nuclei with oval to irregular contours, prominent chromatin centers, and occasional nucleoli (Figs. 22 [inset] and 23). Interspersed were fewer smaller cells with small nuclei, dense chromatin, and inconspicuous nucleoli that stained positive for CD3. Most T cells were concentrated at the periphery of nodules (Fig. 22). Some of the largest cells within the nodules did not stain for either CD3 or CD79. Small numbers of plasma cells were present in stromal septa. This lymphoma did not neatly fall into a category, possibly due to suboptimal preservation of cytologic features at postmortem examination. This neoplasm was provisionally classified as B-cell LLI. Primary differential diagnoses included T-cell/histiocyte-rich B-cell lymphoma (TCRBCL) and follicular and MALT-associated lymphoma.
Discussion
A recently published large retrospective study of caprine neoplasms obtained through the Oregon Veterinary Diagnostic Laboratory reported that lymphoma represented 17.7% of goat tumor submissions and was the most common tumor of goats in this population. 25 In the current study, 15 goat lymphomas were further characterized by topographical distribution, immunophenotype, and histomorphologic features in accordance with the WHO classification system of hematopoietic neoplasms in domestic animals, 34 which has not yet been reported.
The population of goats in the current study had a median age of 3.0 years (SD ± 2.5) when diagnosed with lymphoma. The younger age of goats with lymphoma in this study, compared to the median age (7 years) of goats with neoplastic disease in general previously reported, 25 suggests a predilection for lymphoma to develop in younger animals. In a 25-year survey of cancer in young human adults by the National Cancer Institute, lymphoma accounted for 19% of all cancers in 15- to 29-year-olds in the United States and is the most common cancer in this age group. 6 Lymphoma in young cats has largely been associated with feline leukemia virus (FeLV) infection. 31 However, most of these studies were conducted prior to widespread vaccination. A recent study from the United Kingdom found that most young cats with mediastinal lymphoma are FeLV and feline immunodeficiency virus (FIV) negative (48/55, 87%). 15 To date, there is no clear evidence for a role of retroviruses in caprine lymphoma pathogenesis.
A multicentric distribution of caprine lymphoma at the time of diagnosis is most common in this and previously published reports. 10,12,22,25,32 In comparison, lymphoma in alpacas is reported to present as disseminated disease, with a biphasic age distribution, either in crias (≤2 years of age) or in adults (median age = 6 years), with an overall median of 3.1 years. 1 Similar to lymphomas in adult llamas (median age = 7 years), 1 the lymphomas in our caprine study had a predilection for the thorax, specifically the mediastinum (Table 1), but were diagnosed at an earlier age (median age = 3 years). The location and T-cell phenotype of most of the caprine lymphomas suggest thymus as either the organ of origin or homing as reported for mediastinal lymphoma in cats and dogs. 19,31 Residual thymic tissue may be located along the entire length of the ventral neck and may have given rise to some of the extrathoracic lymphomas located to this area. Localized caprine lymphoma in topographic areas outside the chest and neck was limited to individual cases of intestinal (including mesenteric lymph nodes) and gingival B-cell lymphoma in this study, as well as an additional nasal (cavity/sinus) lymphoma in the previously published study, 25 for which archived material was not available. However, 2 of these 3 cases and the 4 of 5 lymphomas localized to the neck and thoracic inlet (including a case from the previous study for which no archived tissue was available) 25 were diagnosed on biopsy samples only. It is possible that the reported localized topographic distribution was biased by limited sampling. However, the mediastinal distribution noted in this study may be suggestive of the underlying origin or homing of caprine lymphoma and could benefit from further study.
The immunophenotype of caprine lymphomas in this study was predominantly of T-cell lineage (73%). We are only aware of 2 previously reported immunophenotyped caprine lymphomas: 1 case was diagnosed as T-cell lymphoma, 29 the other as B-cell lymphoma. 32 The predominance of T-cell lymphomas in goats is similar to that documented in alpacas and cats. 1,33 In contrast, most lymphomas described in llamas, dogs, and horses are of B-cell lineage. 1,14,26,36 –38
Classification of the 15 lymphoma cases in this study was based on a combination of topography, immunophenotype, and cellular morphology and distribution as described in the WHO classification of hematopoietic tumors in domestic animals and adapted for specific canine lymphoma subtypes. 36 Neoplastic cell morphology was assessed by nuclear size, nucleolar features, and mitotic count, which have not yet been reported for goats. Since RBCs were used as reference, species differences had to be considered. Specifically, goat RBCs are approximately half the diameter of dogs. In a study directly comparing RBCs between different species, the mean RBC diameter for dogs at 7.01 μm is double that for goats at 3.40 μm. 2 In contrast, neutrophils or mature small lymphocytes are more consistent in size across species and reported as 7 to 8 μm in diameter for the latter. Therefore, the nuclear size categories for goat lymphomas in this study were scaled to reflect this difference by multiplying the number of relative RBC widths in each size category by 2 (Suppl. Table S2). The adjusted nuclear size results are more consistent, from a comparative aspect, with other species where the predominant type of T-cell lymphomas is small and intermediate sized. Thus, we recommend the adjusted RBC reference when characterizing caprine lymphoma.
T-LBL was one of the most common subtypes in the goats of this series, in contrast to subtype distribution in other domestic animals. 1,14,26,36,37 The distribution was limited to lymphoid tissue of the head, neck, and thoracic inlet. Thymus is the most common primary site of T-LBL in mice. 36 Thymus may be affected in human patients at presentation and is a common site of relapse. 26 In domestic animals other than goats, the thymus is infrequently involved. In dogs, T-LBL is an aggressive disease. Limited to lymph nodes in early stages, a multicentric distribution is common in late stages and may include spleen and any other tissue, including mediastinum/thymus. 9,16
This series included a single lymphoma of large granular lymphocytes. LGLs are of natural killer (NK) or (cytotoxic) T-cell lineage and have been reported in dogs, cats, horses, birds, and rarely ruminants (cow). 30 Distribution is generally multicentric and highly variable; LGL typically involves the intestinal tract and liver (cat and horse) or presents as leukemia with colonization of the spleen (dog and rat). 34 Only lung and lymph nodes were reported as affected in this young-adult female Pygora goat and had extensive infiltration by the LGL. A leukogram had not been obtained. The LGL in this goat was of T-cell lineage. Primary non-Hodgkin lymphoma of the lung is uncommon in any species, including humans. Most primary pulmonary lymphomas are of marginal zone B-cell lineage, presumably arising from bronchial-associated lymphoid tissue (BALT) formed in response to local antigenic stimuli. 3,4,27 A few individual cases of non-Hodgkin, non–B-cell lymphomas of the lung of humans are published and most commonly classified as extranodal NK/T cell lymphoma (ENKL) based on staining for CD3, granzyme B, and Epstein-Barr virus RNA; other subtypes such as CD3-positive, CD57-negative, EBV-negative PTCL not otherwise specified (NOS) are rare. 17 Testing for NK-specific markers was not performed on the LGL in this series as markers are currently not available for use on formalin-fixed, paraffin-embedded material.
T-cell lymphoma of peripheral or mature lymphocytes can be subdivided based on cell size and topographic distribution. 34 Three low-grade T-cell lymphomas of small lymphocytes were subclassified as SLLs. Interestingly, 2 of 3 T-cell SLLs had infiltrated the spinal canal or spinal cord. In contrast to domestic animals, humans are rarely diagnosed with T-cell lymphoma involvement of the central nervous system (CNS), usually at treatment relapse of lymphomas of immature lineages. 28 A third lymphoma in this series with CNS involvement had infiltrated the brain, likely by extension from the nasal cavity, and was of B-cell lineage.
T-cell lymphomas of peripheral lymphocytes (PTCL) that cannot be further subtyped are classified as “not otherwise specified” (PTCL-NOS). Outside of goats, cats are the only domestic animal species in which PTCL is frequently diagnosed. 38 One of the high-grade PTCLs showed a mixed distribution pattern in lymph nodes with diffuse involvement in some and paracortical expansion in others. This latter pattern is typical for nodal PTCL and overlaps with that of T-zone lymphoma; however, T-zone lymphoma comprises small lymphocytes. 35 In advanced disease, PTCL forms diffuse infiltrates, and we speculate that this case was in transition from nodal to systemic PTCL. In dogs, extranodal PTCL is less common than nodal PTCL, typically presents as cutaneous form or as rapidly progressing disease, and is often characterized by vascular invasion. 36 Neither involvement of the skin nor vascular invasion were features in this caprine PTCL.
DLBCL is the most common lymphoma in dogs and often involves single or multiple peripheral lymph nodes, with abdominal lymph nodes and organs, skin, spleen, and mediastinum less frequently affected. 36 One of 2 DLBCLs in this series received a full necropsy and presented with multicentric disease.
One of the B-cell LLIs was from the mediastinum and trachea and provided a diagnostic challenge. Neither frequent T cells nor a nodular subgross appearance are typical for B-cell LLI. The mixture of cell phenotypes and the prominent stromal component supported TCRBCL. However, the largest B cells present in the tumor were only of intermediate size. Similarly, primary mediastinal B-cell lymphoma characteristically has prominent stromal septa. 34 Over large areas, nuclei showed irregular contours and indistinct nucleoli similar to centrocytes, and scattered T cells may be encountered in follicular lymphoma (FL). 34,36 However, extranodal FL has only been described in the equine liver and not in association with epithelium or the mediastinum/thymus. 14 MALT-associated mantel-zone lymphoma (MZL) has been described in the respiratory tract of human and veterinary patients. It is defined by epithelial infiltration by intermediate lymphocytes with irregular nuclear contours, 34 which could not be confirmed because of diffuse mucosa ulceration in this case. Furthermore, neither prominent chromocenters of neoplastic cells nor mantle cuffs connecting lymphoid follicles were a common feature in this goat. Thymus-associated MALT lymphoma has been reported in humans but not domestic animals. 24 They are characterized by cells with small irregularly shaped nuclei resembling centrocytes that form broad bands, plasma cells around sclerotic stromal vessels, and accompanying small lymphocytes and large lymphoid cells. However, at low magnification, the mixture of lymphocyte lineages imparts a variegated rather than nodular appearance. Furthermore, neither interspersed reactive lymphoid follicles with a preserved mantle zone nor epithelial infiltration by neoplastic lymphocytes with monocytoid B-cell features were identified in the current case.
In summary, we have shown that the WHO classification system for lymphomas in domestic animals can be successfully applied to lymphomas in goats. Following this system, we identified a predominance of T-cell phenotype and multicentric distribution in goat lymphomas of this series, with almost equal frequency of PTCL, T-LBL, and T-cell SLL, and a single case of T-cell LGL. Only 2 subtypes of B-cell lymphomas were identified, DLBCL and B-cell LLI. Future studies on larger case cohorts are needed to further characterize caprine lymphoma subtypes and to identify possible correlations of lymphoma subtypes with clinical outcome.
Footnotes
Author Note
Portions of this study were presented at the 2015 ACVP meeting in Minneapolis/St Paul, MN.
Acknowledgements
We thank the histology laboratory personnel and information technologist Matthew McClain at the Oregon Veterinary Diagnostic Laboratory for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding of the study was provided by the Department of Biomedical Sciences in the College of Veterinary Medicine at Oregon State University. Additional support was provided by the National Institute of Environmental Health Sciences Center P30 ES000210 at Oregon State University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
