Abstract
Histophilus somni is a pathogenic gram-negative bacterium responsible for pneumonia and septicemia in cattle. Sequelae include infectious thrombotic meningoencephalitis (ITME), myocarditis, arthritis, and abortion. These syndromes are associated with widespread vasculitis and thrombosis, implicating a role for endothelium in pathogenesis. Histopathologic and immunohistochemical investigation of 10 natural cases of bovine H. somni myocarditis and 1 case of ITME revealed intravascular H. somni in large biofilm-like aggregates adherent to the luminal surface of microvascular endothelium. Ultrastructurally, bacterial communities were extracellular and closely associated with degenerating or contracted endothelial cells. Histophilus somni was identified by bacterial culture and/or immunohistochemistry. Western blots of the bacterial isolates revealed that they expressed the immunodominant protective 40 kDa OMP and immunoglobulin-binding protein A (IbpA) antigens. The latter is a large surface antigen and shed fibrillar antigen with multiple domains. The cytotoxic DR2Fic domain of IbpA was conserved as demonstrated by polymerase chain reaction. Treatment of endothelial cells in vitro with IbpA in crude culture supernatants or purified recombinant GST-IbpA DR2Fic (rDR2) cytotoxin induced retraction of cultured bovine brain microvascular endothelial cells. By contrast, no retraction of bovine endothelium was induced by mutant rDR2H/A with an inactive Fic motif or by a GST control, indicating that the cytotoxic DR2Fic motif plays an important role in endothelial cell retraction in vasculitis. The formation of biofilm-like aggregates by H. somni on bovine microvascular endothelium may be fundamental to its pathogenesis in heart and brain.
Some gram-negative bacteria selectively target endothelial cells in brain and heart. For example, Neisseria meningitides and Haemophilus influenzae are important causes of human meningitis following bacteremia, with attachment to brain microvascular endothelial cells and violation of the blood–brain barrier. 31 The HACEK group of gram-negative bacteria (Haemophilus spp, Aggregatibacter actinomycetemcomitans, Cardiobacterium spp, Eikenella corrodens, Kingella kingae) causes endocarditis with vascular complications in adults and children. 17,33 Cardiobacterium valvarum is associated with both endocarditis and cerebral vasculitis. 1 Similar to HACEK bacteria, Histophilus somni is a capnophilic pleomorphic fastidious gram-negative bacillus. It is part of the normal flora of the urogenital and, to a lesser extent, upper respiratory tracts of cattle. 9 H. somni disease complex most commonly presents as pneumonia and/or septicemia, followed by myocarditis, infectious thrombotic meningoencephalitis (ITME), arthritis, and/or abortion. 6,23,24,26 –28,32,37,38,46 Vasculitis and thrombosis are morphologic hallmarks of infection, particularly in brain and heart. Early vascular lesions involve capillaries and small venules in multiple tissues, with subsequent involvement of larger vessels. 27 Early in vitro studies with arterial explants showed that H. somni adheres to endothelial cells, leading to cellular contraction and exposure of vascular basement membrane. 45 The role of H. somni lipooligosaccharide (LOS) in apoptosis of bovine endothelial cells in vitro is well characterized. 15 Subsequent studies showed that live H. somni, but not purified LOS, decreased endothelial cell monolayer integrity in vitro. 3 This suggests that additional virulence factors may be involved in endothelial cell contraction. Aggregates of H. somni are evident immunohistochemically in intracardiac capillaries and venules of cattle with fatal myocarditis. 32 These aggregates probably correspond to the cardiac biofilms reported by others, although those studies did not show the cellular location of the biofilms. 36 In the study reported below, the in vivo pathogenesis of H. somni for endothelial cells was investigated by examining lesions and H. somni strains obtained from clinical cases of bovine myocarditis, as well as the effect on brain microvascular endothelium. In vitro pathogenesis for vascular endothelium was determined by examining the role of the secreted virulence factor, immunoglobulin binding protein A (IbpA), in mediating endothelial cell retraction and cytotoxicity. The IbpA large surface and shed protein has 2 direct repeats (DR1 and DR2), each with a cytotoxic Fic motif. 47 Since IbpA enriched concentrated culture supernatant and recombinant IbpA DR2Fic have been shown to cause retraction of bovine alveolar type 2 (BAT2) cells, 48 the role in bovine endothelial cell retraction was studied.
Materials and Methods
Tissues
Samples at necropsy were obtained from lungs and hearts of ten 7- to 9-month-old beef-type cattle from 1 Wyoming property that has a recurrent annual problem of myocarditis due to H. somni. 32 Formalin-fixed samples of lung and heart were obtained from each animal. Cardiac samples included right and left atrial myocardium, both papillary muscles of left ventricular myocardium, papillary muscle of right ventricular myocardium, and interventricular septum. All samples were sectioned and were stained with hematoxylin and eosin. Selected sections of lung and heart were stained with Gram stain. Histologic lesions in heart were recorded as acute, subacute, or chronic based on the presence of bacteria in vessels, the type of inflammation and necrosis, and, where present, fibroplasia-fibrosis and/or mineralization. Sections of lung and papillary muscle from left ventricular myocardium were stained for immunohistochemical detection of antigens of H. somni and bovine viral diarrhea virus using published methods and appropriate positive and negative controls. 12,23 The positive control was from a case of H. somni myocarditis that was confirmed positive by positive aerobic culture. For a background control, the same block was used, omitting the primary antibody. The negative control for H. somni used sections of a normal bovine heart. The rabbit antiserum to H. somni is unreactive with other common gram-negative bacteria found in cattle. 23 Samples of lesioned papillary muscle from left ventricular myocardium of 1 animal (case No. 1, strain No. 08B13317, Table 1) were processed for transmission electron microscopy. 5 The brain of a 10-month-old beef-type steer (case No. 11, strain No. 15B1167, not represented in Table 1) with typical gross and histologic lesions of acute ITME 6 was examined microscopically to establish whether the distinctive relationship that in acute cases exists in heart between H. somni and microvascular endothelium also occurs in brain. The animal originated from a ranch in northern Wyoming, where at least 1 other animal on the property developed signs consistent with ITME.
Clinical Details of 10 Cases of Bovine Histophilus somni Myocarditis.

Abbreviation: IHC, immunohistochemistry.
a(m), moderate; –, negative; +, positive; (s), scant.
Bacterial Strains
Histophilus somni was isolated by culture from lesions in cardiac papillary muscle in 7 of 10 cattle with myocarditis, from lungs of 3 of the same 7, and from the brain of the steer with ITME. Swabs and homogenates were inoculated onto Columbia blood agar (5% ovine blood) and incubated at 37°C in 10% CO2. Colonies characteristic of H. somni and with bacteria that were gram negative, nonmotile, oxidase positive, and glucose fermenting were further identified by the MicroStation ID System (Biolog Inc, Hayward, CA, USA). The strain from the ITME case was not saved. H. somni strain 2336, which was isolated from the lung of a veal calf that died of severe pneumonia, was used as a positive control for in vitro studies. 23 This strain is extensively used as a challenge inoculum for experimental studies of the pathogenesis of H. somni. 20 –23
Virulence Factor Analysis
The presence of virulence determinants in the clinical isolates of H. somni from myocarditis was determined as described previously. 48,49 In brief, isolates from cases of myocarditis were analyzed by Western blotting after separating whole cell antigens or concentrated culture supernatant (CCS, shed antigen enriched for IbpA and outer membrane blebs) on 10% acrylamide gels (SDS PAGE). Pneumonia challenge strain H. somni 2336 was included for comparison. Blots were reacted against pooled high titer sera from 2 convalescent calves (E5 and E7) from an earlier experimental H. somni pneumonia study in which strain 2336 was used for infection, 21 with murine monoclonal antibody 27 –1 to a 37 kDa outer membrane protein (OMP), kindly provided by Y. Tagawa et al, 42 or with rabbit anti-H. somni IbpA DR2 antigen. 48 The bovine convalescent serum was used because it passively protected calves against experimental H. somni pneumonia 22 and therefore should detect protective antigens. Primary sera were diluted 1:1000 and monoclonal antibodies diluted 1:20 before incubating with blots for 1.5 hours and reacting with secondary alkaline phosphatase-labeled goat antibovine IgG conjugate (Kirkegaard & Perry Laboratories, Inc, Gaithersburg, MD, USA), goat antimouse IgG, IgA, IgM (KPL Labs), or goat antirabbit IgG (Zymed [Invitrogen], Carlsbad, CA, USA) for 1 hour, and developing with 1-StepTM BCIP (5-bromo-4-chloro-3’-indolyphosphate p-toluidine salt) and NBT (nitro-blue tetrazolium chloride). Polymerase chain reaction (PCR) analysis of IbpA DR1 and DR2 domains was done as described. 49 Hemolysis was detected on bovine blood agar plates after 48-hour incubation at 37°C, followed by 48 hours at room temperature in a candle jar.
Toxicity of H. somni IbpA DR2 for Bovine Brain Microvascular Endothelial Cells
Since H. somni IbpA causes bovine respiratory epithelial cells and other epithelial cells to retract, 47,48 the effect of the CCS and recombinant IbpA DR2 GST fusion protein (GST-DR2) and GST control protein on endothelial cells was determined. The role of the Fic motif was confirmed using recombinant GST DR2 with the critical histidine replaced by adenine (GST DR2 H/A). 47 –49 Bovine brain microvascular endothelial cells (BBMECs) 39 were used because microvascular endothelial cells were associated with H. somni in the 10 reported cases of myocarditis and the single case of ITME. Bovine brain microvascular endothelial cells were seeded at 5 × 104 in a 24-well plate. Monolayers were treated with 20× concentrated CS (final concentration) or recombinant purified proteins (GST 4 μg, GST-DR2 10 μg, or GST-DR2 H/A 10 μg) in culture medium. After 4 hours, cells were fixed and actin was stained with phalloidin-rhodamine. The number of retracted cells was counted microscopically and the percentage calculated from the total number of cells in a field. Cytotoxicity of H. somni 2336 CCS and GST-DR2 protein to BBMEC cells was measured also by an ATP-based cell viability assay. Bovine brain microvascular endothelial cell monolayers in a 96-well plate were seeded at 5 × 103 cells per well. Three wells per treatment group were treated with 20× concentrated CS or recombinant purified proteins (GST 4 μg, GST-DR2 10 μg, or GST-DR2 H/A 10 μg) in culture medium for 4 hours, and cell viability was measured by CellTiter-Glo® Luminescent Cell Viability Assay as recommended by the manufacturer (Promega, Madison, WI, USA). In brief, BBMECs were seeded in a 96-well opaque-walled plate (Corning Life Sciences, Acton, MA, USA) at a density of 5 × 103 cells/well and incubated for 24 hours. Then, cells were treated with a final concentration of either 10× concentrated CS of H. somni 2336 or recombinant purified proteins (GST 4 μg/ml, GST-DR2 10 μg/ml, or GST-DR2 H/A 10 μg/ml) in 100 μl culture medium at 37°C. After 4 hours of treatment, plates were equilibrated at room temperature for 15 minutes, 100 μl of the assay reagent in the CellTiter-Glo Luminescent Cell Viability Assay kit was added to each well, and cell lysis was induced on an orbital shaker for 10 minutes at room temperature. Luminescence was read on a SpectraMax® M5 Microplate Reader (Molecular Devices Corporation, Sunnyvale, CA, USA). When added to cells, the assay reagent produces luminescence in the presence of ATP from viable cells. Control wells contain medium without cells to obtain a value for background luminescence. Data were expressed as percentage of ATP concentration in untreated control wells, mean ± SE, 5 replicates for each treatment group. Assays were done in preliminary studies and then repeated. Cytotoxicity data were analyzed for significance with Student’s 1-tailed t-test using GraphPad Prism software (GraphPad Software, La Jolla, CA, USA).
Results
Gross Pathologic Evaluation of 10 Cardiac Cases
Histophilus somni-induced myocarditis was present in the 10 cattle, as established by typical clinical signs, gross and microscopic lesions, aerobic bacterial culture, and/or immunohistochemistry (Table 1). Cardiac lesions were characterized as necrosis of 1 or both papillary muscles in left ventricular myocardium, with additional intracardiac lesions elsewhere in individual cases. Fibrinous valvular and nonvalvular endocarditis affected 2 of the 10 animals. Intralesional gram-negative bacteria were found in the hearts of all 10 animals. They were identified as H. somni based on culture and immunohistochemistry or by immunohistochemistry alone. Gross lesions were assessed as acute (n = 6), subacute (n = 1), or chronic (n = 3). The character of lesions tended to correlate with the duration of clinical signs, when this was known. Cardiac lesions were consistently present in 1 or both papillary muscles of left ventricular myocardium and inconsistent elsewhere in the heart. Acute lesions were 1 to 3 cm in maximum dimensions and dark red in acute cases. Varying degrees of fibrosis, necrosis, and abscess formation attended more chronic cardiac lesions. Other changes were endocardial rupture overlying areas of necrosis, ruptured chordae tendineae, and valvular and nonvalvular endocarditis. Grossly, the lungs either were unremarkable or had disseminated areas of edema and hemorrhage without gross evidence of pneumonia.
Histopathologic and Immunohistochemical Evaluation
Solid or beaded aggregates of intravascular gram-negative bacteria were most numerous in capillaries and veins of papillary muscles of left ventricular myocardium in animals with myocarditis, and this was most evident in the 6 acutely affected animals. Veins were identified on the basis of their diameter, thin walls, and paucity of smooth muscle cells in tunica media. In severe cases, a circumferential layer of intravascular bacteria adhered to vessel walls. These formed a mat < 1 to 20 μm deep (Fig. 1). Larger biofilm-like bacterial aggregates up to 400 μm deep adhered to the walls of veins and to ulcerated endocardium (Fig. 2). Acute myocardial lesions comprised foci up to 300 μm in diameter, with zones of coagulative necrosis (infarction) and myocarditis around capillaries and veins containing intravascular and perivascular bacteria. Perivascular bacteria were present in areas of necrosis, generally in a disseminated pattern consistent with a microvascular distribution (Fig. 3). Individual endothelial cells in capillaries, venules, and veins (100–700 μm diameter) that contained bacteria had dark contracted nuclei consistent with pyknosis or apoptosis. Many affected cardiac vessels contained occlusive and nonocclusive fibrin thrombi. Fibrinoid change was absent. Phlebitis, when present, was generally associated with necrosis in vessel walls. Immunohistochemistry showed that such vessels were full of H. somni antigen that coated the luminal surface of endothelium (Fig. 4). Bacteria were identified in pulmonary alveolar blood vessels by immunohistochemistry in 4 of 10 cattle with acute cardiac histophilosis. The intravascular H. somni bacteria were scant in 3 and moderately numerous in 1. All 10 cattle were negative immunohistochemically for bovine viral diarrhea virus. The animal with acute ITME had a similar relationship between H. somni and endothelium in capillaries and venules of the brain and meninges (Figs. 5–7). Affected venules were partially or completely occluded by fibrin thrombi, with mats of bacteria abutting the vessel wall (Fig. 5).
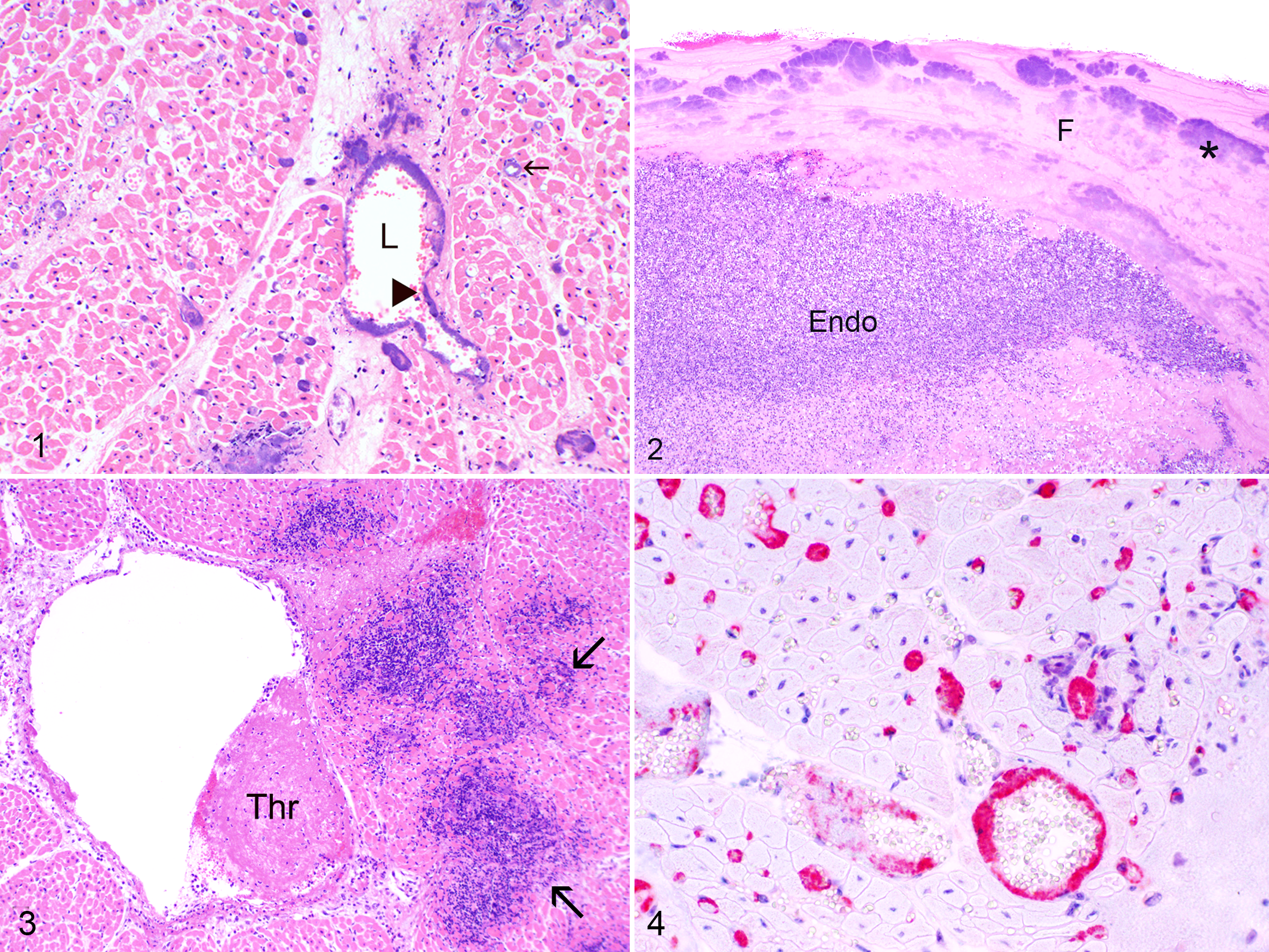
Histophilus somni myocarditis, steer.
Ultrastructural Studies
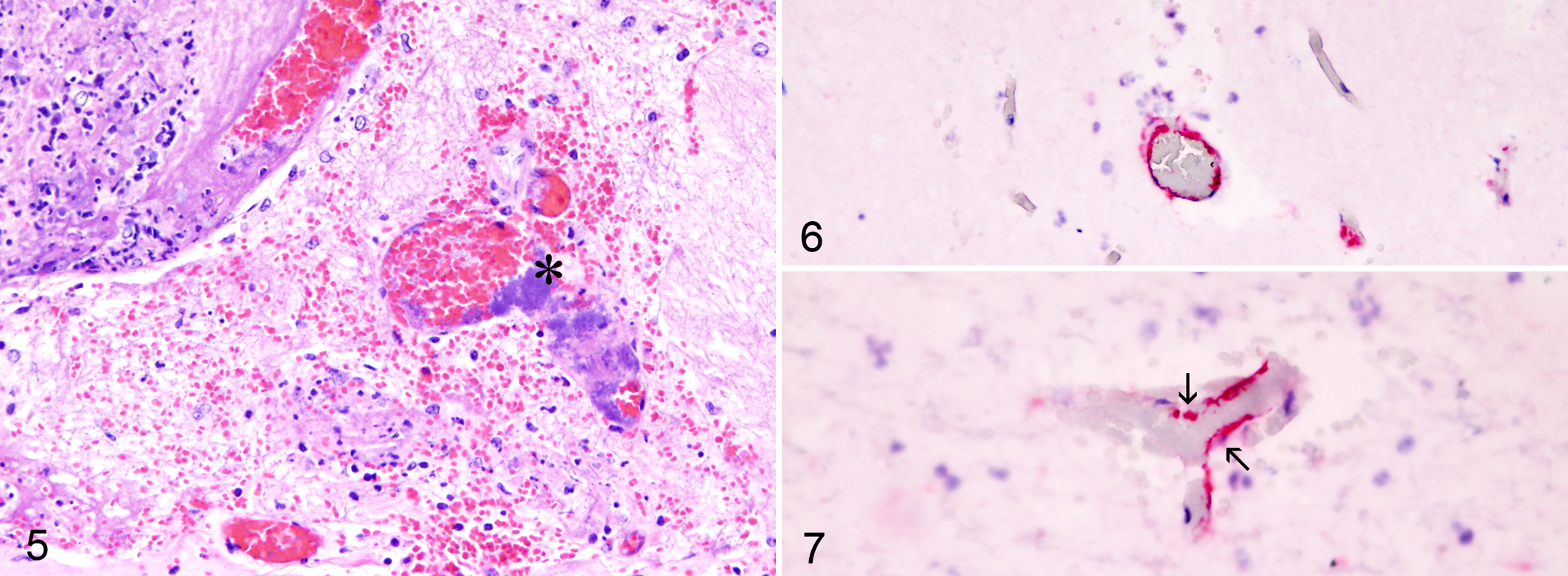
Bacterial communities were closely associated with degenerating endothelial cells (Figs. 8–11). In many vessels, the cytoplasm of endothelial cells was contracted so that bacteria rested on attenuated endothelium or naked vascular basement membrane (Figs. 9–11). H. somni was never detected in endothelial cytoplasm. Some bacteria were located between degenerate endothelium and underlying basement membrane. No extracellular polysaccharide matrix characteristic of biofilm was evident in these conventionally prepared electron microscopy samples, which is consistent with solvent extraction during tissue processing. 34 Extracellular fibrils consistent with fibrin were present in endothelial lesions between H. somni cells.
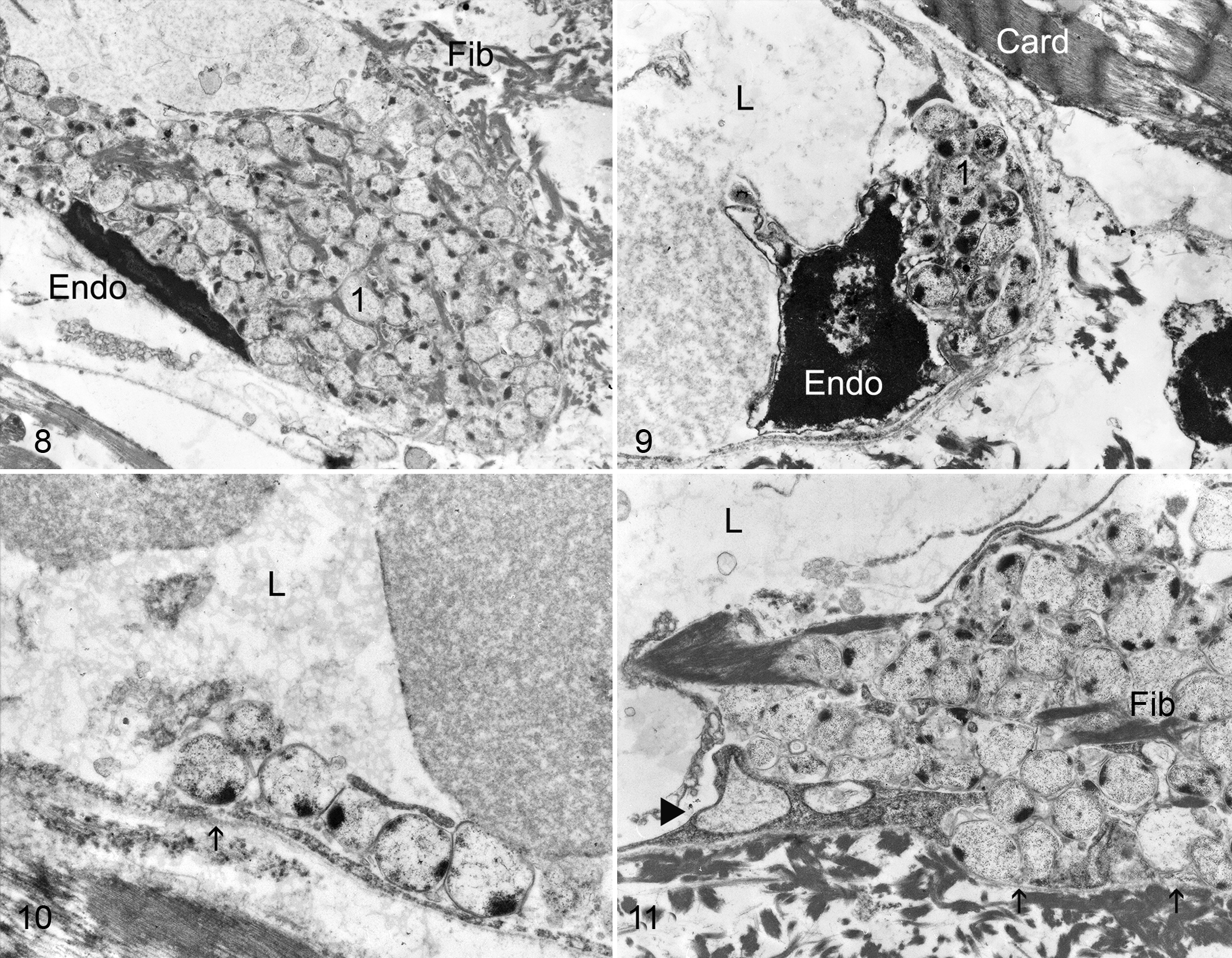
Histophilus somni myocarditis, papillary muscle of left ventricular myocardium; steer. Transmission electron microscopy.
Virulence Factor Conservation
Heart and lung H. somni isolates from 6 of the 7 culture-positive myocarditis cases (Table 1) were characterized in comparison to pneumonia challenge strain 2336. One field isolate was not available for comparison. Two isolates from 2 calves were hemolytic on bovine blood agar (Table 2). Western blotting of H. somni CCS revealed that all myocarditis isolates and the 2336 pneumonia strain had the major antigens recognized by passively protective convalescent phase serum, including those at 78, 76, 60, and 40 kDa (Fig. 12a). Blots of H. somni whole cells revealed similar profiles with minor variations in the size of OMPs in the 37 to 40 kDa range. Where available, isolates from the heart and lung of the same animal were evaluated; no differences were detected between isolates from dual tissue sites. Special attention was paid to antigens in the 40 kDa range since this includes relatively nonimmunogenic 41 kDa major OMP (MOMP), the immunodominant 40 kDa OMP, a 39 kDa OMP, and a 37 kDa OmpA homologue. 11,41,42 Western blots of CCS were used for studies of these OMPs since H. somni releases outer membrane blebs into culture supernatant (AC Ward and LB Corbeil, 1987, unpublished data). Use of a 10% acrylamide gel allowed maximum separation of proteins in this MW range. The 41 kDa MOMP was not detected by convalescent sera at 1:1000 (Fig. 12a) as expected. 41 The immunodominant 40 kDa OMP was strongly detected in all strains and was of slightly variable MW, as described elsewhere (Fig. 12a). 11 Two bands lower than the 40 kDa OMP were interpreted as the 39 and 37 kDa OMPs. The HMW IbpA cytotoxin was not well detected by convalescent phase sera, as in earlier studies. 21 Western blots with specific antibodies were employed to address this. Rabbit antibodies to the IbpA DR2 cytotoxic domain showed that all isolates had the HMW bands of varying intensity and the 76 kDa band characteristic of IbpA (Fig. 12b). Even though HMW bands of some isolates were faint, PCR confirmed that both cytotoxic domains of IbpA (DR1 and DR2) were present in all isolates (Table 2). A monoclonal antibody (27 –1) to the heat-modifiable 37 kDa OmpA homologue 42 strongly recognized the 37 kDa OMP of all isolates, with some variation in relative molecular weight (Fig. 12c).
Characteristics of H. somni Isolates From Hearts and Lungs of 6 Calves With Myocarditis Compared to Reference Isolate.

Abbreviation: IV Sep, interventricular septum.
aIbpA: determined by Western blotting.
bDR1 & DR2 (IbpA direct repeats with Fic cytotoxic motifs): presence determined by polymerase chain reaction.
cHemolysis: determined on blood agar.
dChallenge strain from a calf with severe pneumonia.
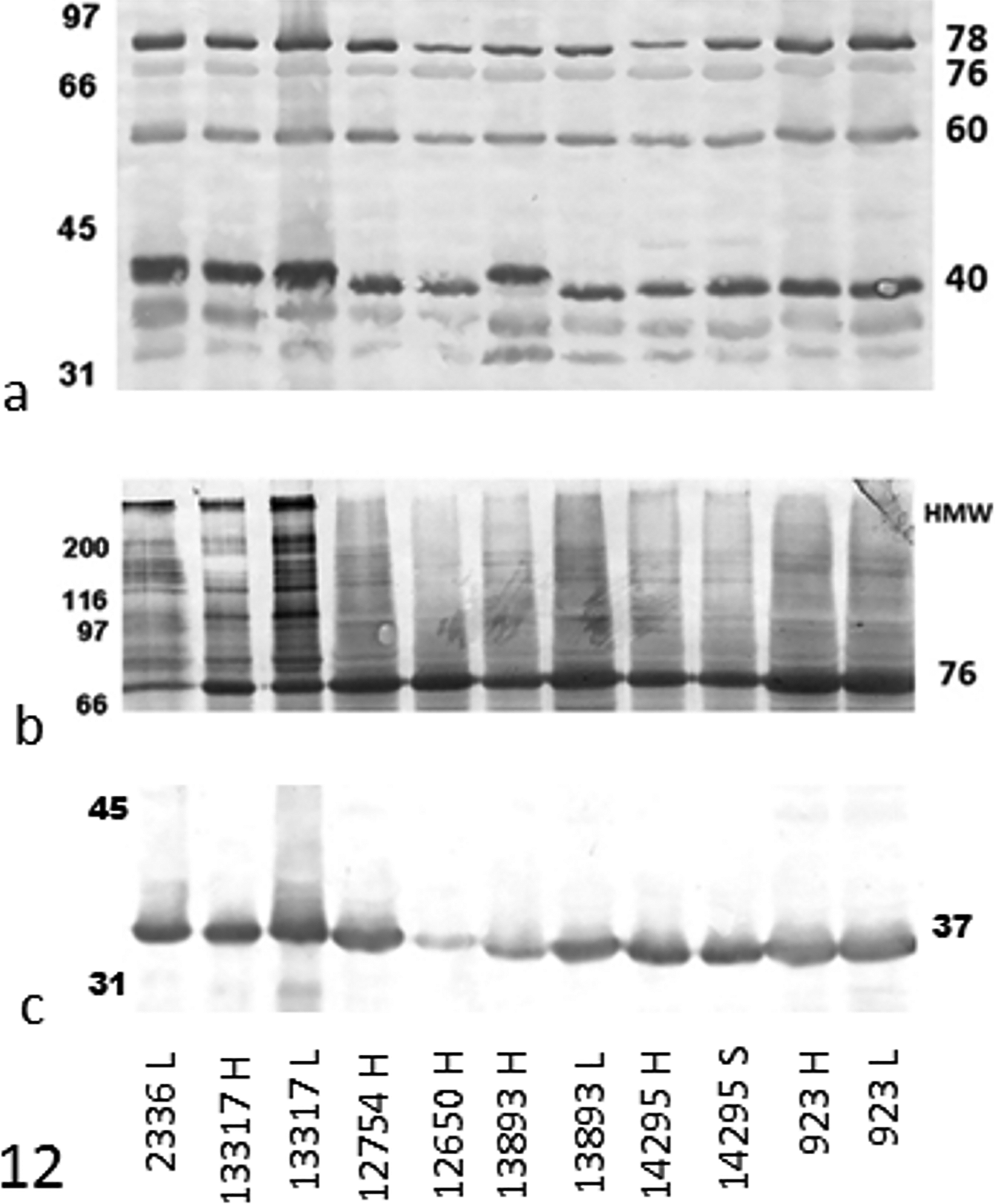
Western blot of H. somni antigens in concentrated bacterial supernatant, compared to pneumonia strain 2336 and probed with 3 different antisera. When available, cardiac and pulmonary isolates from individual calves were compared. Source of isolate: H, heart (papillary muscle); L, lung; S, heart (interventricular septum). (a) Blot reacted with convalescent phase sera from calves with experimental H. somni pneumonia (1:1000). Note that minor variation in the MW of the immunodominant 40 kDa antigen was detected. (b) IbpA virulence factor as recognized by rabbit anti-IbpA DR2. Note that both the p76 and HMW antigenic bands of IbpA are present in all isolates. Even though HMW bands of some isolates were faint, polymerase chain reaction confirmed that both cytotoxic domains of IbpA (DR1 and DR2) were present in all isolates (Table 2). (c) Blot reacted with monoclonal antibody to the 37 kDa OmpA homologue. Note that all isolates are positive. There are minor molecular weight differences among isolates for 37 kDa OMP. Molecular weight (MW, kDa) markers at left and MW of known antigens at right.
In Vitro Studies of IbpA Cytotoxicity for BBMEC
Crude preparations of IbpA in CCS caused cellular retraction when compared with control BBMECs treated with GST (Fig. 13). Fusion proteins of GST-rDR2 also caused retraction, but not GST-rDR2 H/A (with inactivated Fic toxic motif), as compared with treatment by GST alone. When retracted cells were counted, there was no difference between the media-treated control and GST- or GST-DR2 H/A-treated cells. Cells treated with CCS or GST-rDR2 showed significant retraction as compared to controls (Fig. 14). Cytotoxicity analysis by the ATP viability assay confirmed these data (Fig. 15), indicating that CCS enriched for native IbpA with 2 Fic motifs (in domains DR1 and DR2) and rDR2 are cytotoxic for BBMECs. Neither the GST control nor rDR2 H/A (inactive Fic motif) were cytotoxic by this assay, confirming the morphologic data above.
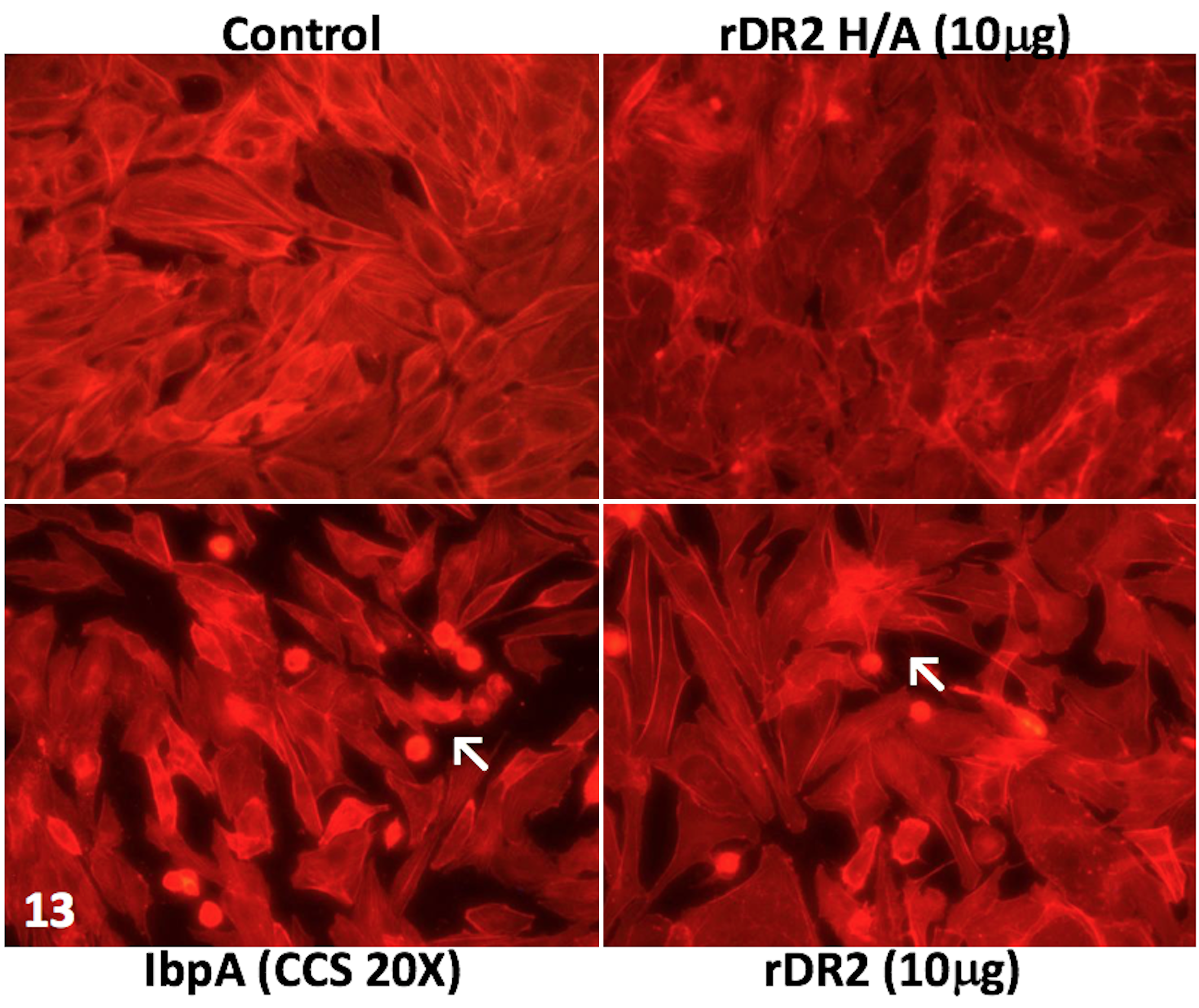
Cytotoxicity of H. somni 2336 concentrated culture supernatant (CCS), the IbpA DR2 domain, and mutant DR2 (DR2 H/A) protein to bovine brain microvascular endothelial cells (BBMECs) in vitro. BBMEC monolayers in a 24-well plate were treated with medium alone (control, data not shown), 20× concentrated CS (final concentration), or recombinant purified proteins (GST 4 μg, GST-DR2 10 μg, or GST-DR2 H/A 10 μg) in culture medium. After 4 hours, cells were fixed and actin was stained with phalloidin-rhodamine and evaluated microscopically for retraction. The (a) GST control cells and the cells treated with (b) GST-DR2 H/A (inactive Fic) were not retracted. Cells treated with (c) CCS (as a source of native IbpA) or with (d) GST-DR2 were retracted (arrows) due to Fic-mediated cytotoxicity.
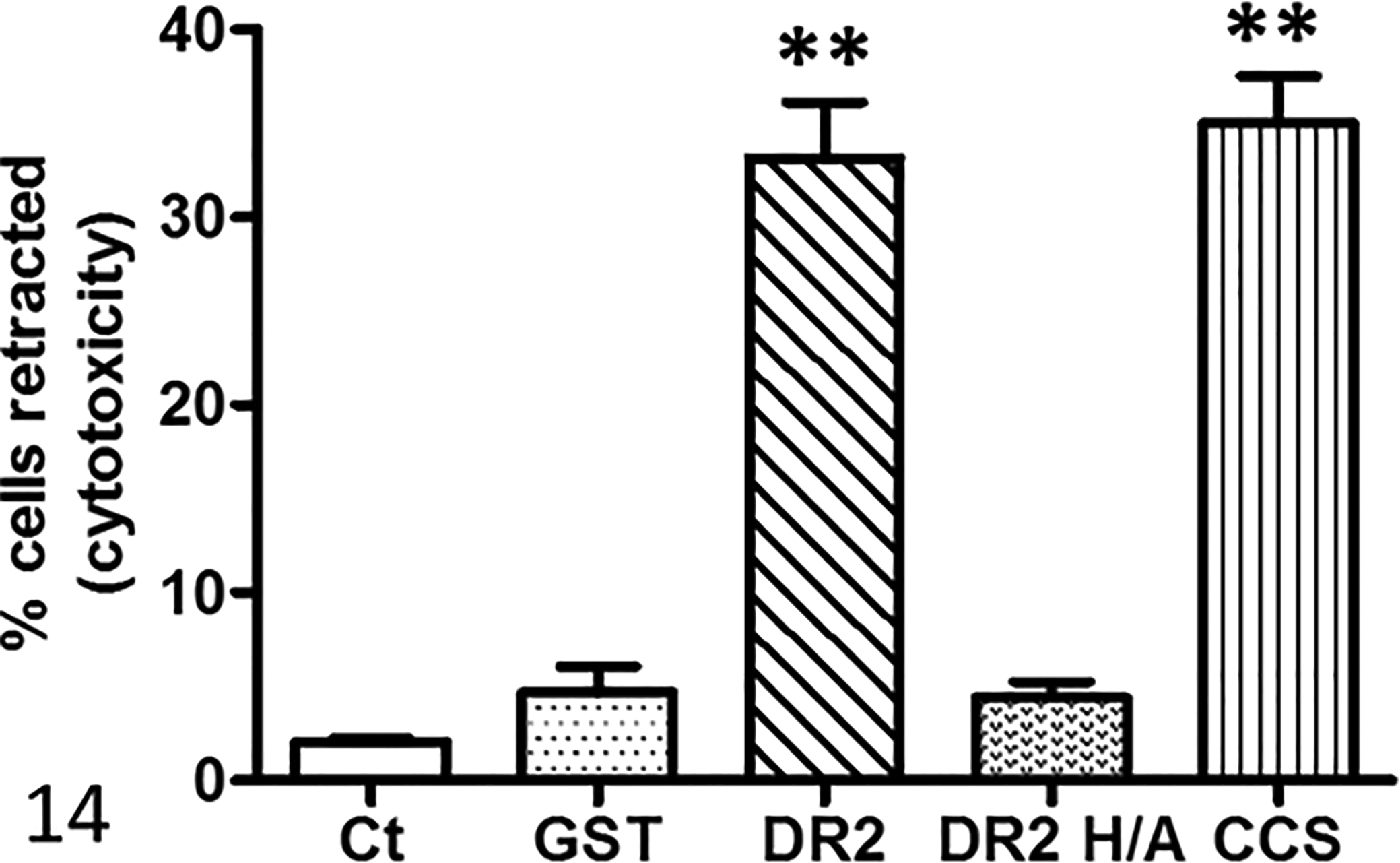
Quantitation of retracted bovine brain microvascular endothelial cells after treatment with GST (negative control), IbpA DR2H/A, DR2, or concentrated culture supernatant (CCS) from H. somni. The number of retracted cells were counted and the percentage retracted calculated from the total number of cells in a field (average of 78 cells per high power field). Bars show the mean ± SEM of 10 microscopic fields. **P < .01 compared to GST or GST-DR2 H/A. Significantly more cells were retracted after treatment with Fic-containing DR2 or CCS than in media controls, in GST controls, or after treatment with recombinant DR2 H/A with the inactive Fic motif. Ct, medium alone.
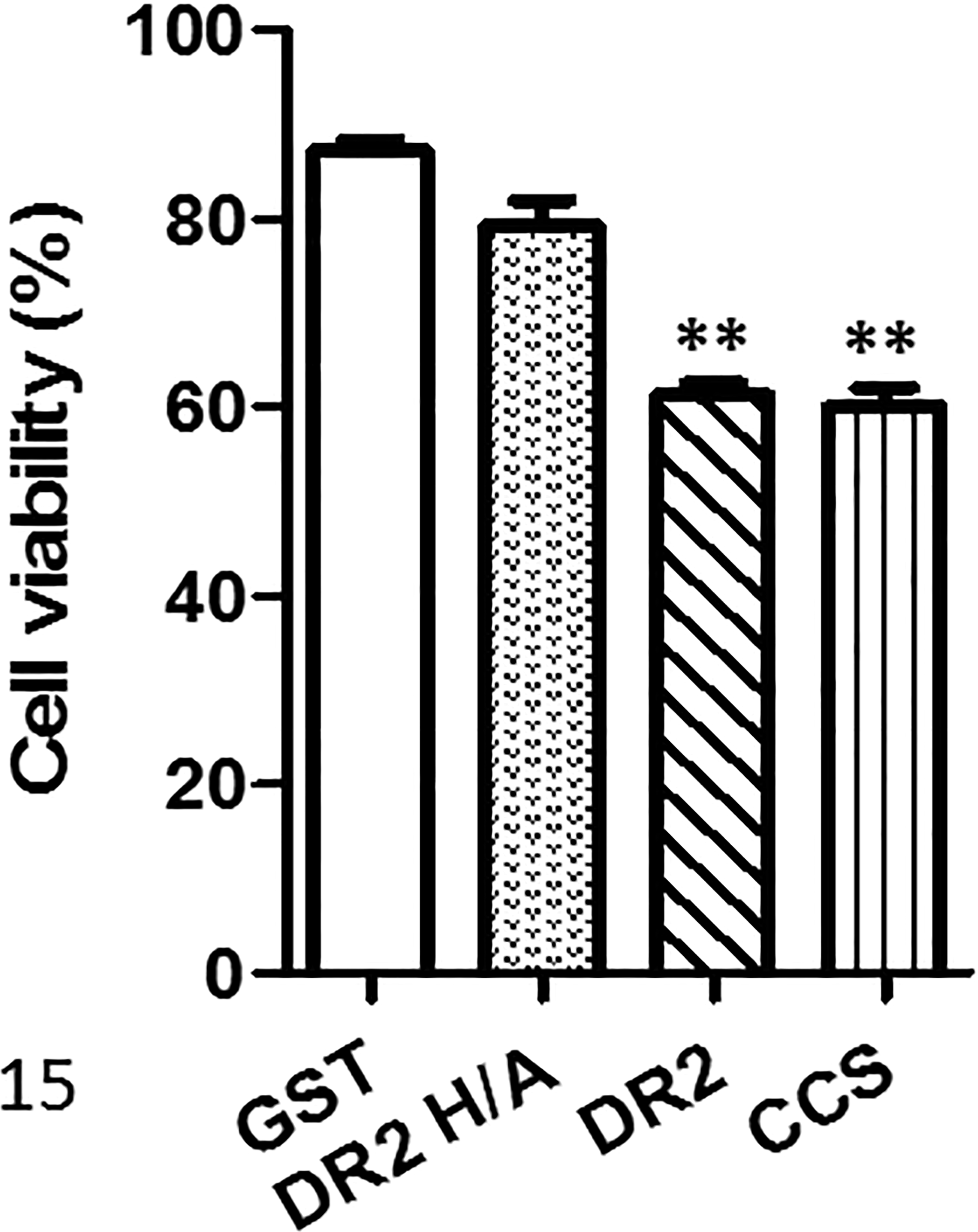
Cytotoxicity of GST (negative control), IbpA DR2H/A, DR2, or concentrated culture supernatant (CCS) from H. somni for bovine brain microvascular endothelial cells measured by cell viability. Bars show mean ± SEM percentage viability compared to control wells. **P < .01 compared to GST or GST-DR2 H/A. Significantly less cell viability was detected after treatment with Fic-containing DR2 or CCS than in GST controls or after treatment with recombinant DR2 H/A with the inactive Fic motif.
Discussion
These studies of in vivo and in vitro interaction between H. somni and bovine cardiac or brain microvascular endothelial cells indicate that complex interactions occur between bacteria and endothelium. This may partially explain the pathogenesis for H. somni-induced myocarditis and endocarditis. The vasculitis characteristic of H. somni infection is associated with plugging of capillaries and small veins by aggregates of H. somni. This is particularly extensive in heart, brain, and spinal cord. Since many such animals tend to die acutely, these early vascular changes can be seen directly. The intimate association between endothelium and H. somni, which also occurs in sheep with ITME, 7 is such that one cannot resolve by conventional light microscopy whether bacteria are near or within endothelium. 25 However, these ultrastructural studies show that the bacteria are extracellular and are not within the endothelial cells. The bacterial communities on endothelium resemble biofilms. Whether H. somni forms biofilms on endothelial surfaces has implications for effective host immune responses and successful antimicrobial treatment. 13 Biofilms are formed in vitro by other major bacterial pathogens of the bovine respiratory disease complex and by H. somni. 35 H. somni biofilms were previously demonstrated in bovine heart following an experimental challenge, although an association with vessels was not reported. 36 The light microscopic features of bacterial aggregates in vessels in this and a previous report 32 suggest that biofilm formation occurs in vivo on endothelium.
Biofilms are matrix-enclosed, metabolically cooperative bacterial populations in which cells adhere to each other and/or to surfaces or interfaces. 13 The biofilm matrix consists of extracellular polysaccharide (EPS), proteins, and nucleic acids. 18 H. somni forms EPS characteristic of biofilms. 35,36 As expected, ultrastructural examination of intravascular aggregates in heart did not reveal EPS in the current study, due to the use of routine processing for conventional electron microscopy. 18 Demonstration of the extracellular polysaccharide ultrastructurally requires special processing that is most often used for bacteria in vitro. 34 Protein components of the biofilm matrix are often fibrillar. 18 An earlier ultrastructural study of H. somni did not detect material external to the outer membrane. 38 However, heavy staining of the organism revealed a fibrillar surface network that binds bovine IgG2, identifying the fibrils as immunoglobulin protein A. 10,43 Such fibrils may be involved in biofilm formation since strain 129Pt, which lacks IbpA, forms little or no biofilm in vitro. 35 Bacterial biofilms occur in human valvular endocarditis and resemble those of H. somni on bovine endocardium. Emboli are mentioned in reports of human valvular endocarditis. 31 Since biofilm-like aggregates of H. somni form on bovine myocardial microvascular endothelium in the absence of endocarditis (8 of 10 cases reported here), septic emboli originating from endocarditis are unlikely to be involved. H. somni appears to form a biofilm-like aggregate directly on microvascular endothelium de novo. This is analogous to microcolonies of Neisseria meningitides, which form on the apical surface of capillary endothelial cells throughout the body, including those of brain and heart. 14 Retraction of endothelial cells and luminal thrombi are reported in meningococcal purpuric lesions. 14 The formation of microcolonies, endothelial retraction, and thrombus formation are similar to this report of H. somni pathogenesis in vivo and in vitro but are uncommon in reports from other infections of the heart and brain. Bacterial colonization of bovine microvascular endothelium and release of fibrillar IbpA with cytotoxic motifs at endothelial surfaces may explain the endothelial retraction, exposure of basement membrane, and subsequent thrombus formation in H. somni myocarditis.
It appears that the distinctive relationship that develops in the heart between H. somni and microvascular and venular endothelium also occurs in thrombotic meningoencephalitis, since similar beaded aggregates of H. somni form within vessels. Infectious thrombotic meningoencephalitis can occur concurrently with cardiac histophilosis or independently. Although only 1 case of ITME was examined in the current study, it is likely that ITME also begins with the formation of biofilm-like masses on endothelium. That relationship is masked when hemorrhage, necrosis, and vasculitis supervene.
An array of known virulence factors was examined in H. somni isolates from calves with myocarditis to address the possibility of strain differences and to determine whether protective antigens were conserved. Hemolysis is associated with virulence in some bacterial species, but only 2 of 6 calves with H. somni myocarditis yielded hemolytic isolates on bovine blood agar. The reference challenge isolate 2336 is nonhemolytic and experimentally induces both pneumonia and myocarditis. 19,21 –24,36 This suggests that hemolytic activity by H. somni is not essential to induce myocarditis or pneumonia. Myocardial strains had the same virulence factors/antigens identified by Western blotting as the pneumonia isolate. No new antigens recognized by convalescent phase sera were detected. We previously detected no differences between TME and pneumonia isolates. 11,41,49 This is consistent with the absence of specific virulence factors that might be associated with cases of H. somni infection in which the dominant or sole lesions are myocarditis, ITME, or pneumonia. The 37 kDa OmpA homologue is conserved among many gram-negative pathogens. 8 OmpA has 4 hypervariable domains in other bacterial species. 16 Mannheimia haemolytica OmpA contributes to adherence to bovine bronchial epithelial cells. 29 The H. somni 37 kDa OmpA homologue from different strains reacted with the monoclonal antibodies used in this study and varied in molecular weight from strain to strain, similar to the OmpA of Haemophilus parasuis. 44 The hypervariable domains of OmpA in other gram-negative pathogens are thought to be involved in mammalian host and tissue specificity. 16 The role of the 37 kDa OMP of H. somni in virulence and species/tissue specificity merits additional investigation.
The conservation of both the 40 kDa OMP and IbpA virulence factors of H. somni in these myocarditis strains has implications for protective vaccination. The IbpA DR2 domain and the sequence of the cytotoxic DR2Fic motif were conserved in all pathogenic isolates tested previously, including several myocarditis isolates from the current study. 49 Monospecific antibodies to the 40 kDa OMP protected calves against experimental H. somni pneumonia and recombinant IbpA DR2 protein actively protected calves against H. somni pneumonia. 19,22 Since both the 40 kDa and IbpA antigens are conserved in myocarditis and are protective, it is a reasonable inference that vaccines containing these virulence factors will protect against myocarditis and pneumonia.
One mechanism for the pathogenesis of H. somni infection is likely to be endothelial cell retraction caused by IbpA DR2 (and DR1). Both the DR1 and DR2 “Fic”-containing domains of IbpA caused retraction in vitro of bovine alveolar type 2 (BAT2) epithelial cells. 48 All heart and lung isolates from the myocarditis cases expressed IbpA, including both the DR1 and DR2 domains as determined by PCR. Three of these myocarditis isolates were previously shown to have identical Fic motifs, as did isolates from other H. somni syndromes. 49 In vitro studies with BBMECs demonstrated that H. somni strain 2336 CCS (enriched for IbpA DR2) causes cytotoxicity as measured by the ATP viability assay, and retraction as determined morphologically. Since the sequence of the DR2 domain was conserved in all isolates tested, 49 it was concluded that this CCS would be representative of other strains. The mechanism of cytotoxicity for endothelial cells is likely to be complex. Although H. somni thickly coated the inner walls of some blood vessels in vivo, bacteria were never found in the cytoplasm of morphologically intact endothelium, which is consistent with previous in vitro experiments using epithelial cells. 48 The associated endothelium was retracted and degenerate, as occurs in vitro in endothelial cell cultures treated with H. somni. 3,4,15,40 Histophilus somni lipooligosaccharide is involved in endothelial cell apoptosis after long incubation periods but does not account for all the cytotoxicity observed in vitro, especially at early time points. 3 Heparin-binding proteins appear to be involved in attachment of H. somni to endothelial cells, perhaps due to the IbpA heparin-binding motifs that have been reported previously. 4,43 IbpA DR2 Fic activity results in adenylylation of Rho GTPases and disruption of the actin cytoskeleton, causing retraction of HeLa cells or BAT2 cells by IbpA DR2. 47,48 The same mechanism may account, at least in part, for IbpA DR2-induced bovine endothelial cell retraction. The current studies demonstrated retraction of myocardial microvascular endothelial cells covered by aggregates of H. somni in vivo and of H. somni-treated brain microvascular endothelial cells in vitro. Thrombosis and vasculitis are characteristic of H. somni infection in many tissues during infection. 9,23,26 Ultrastructural studies of the myocardial microvascular endothelium in this study revealed that intraluminal H. somni were typically associated with fibrin rafts indicative of thrombosis. It was previously shown that H. somni-activated platelets increased pulmonary artery endothelial production of tissue factor, with increased expression of proinflammatory and procoagulative cytokines by BBMECs. 30 Increased release of tissue factor plus procoagulative cytokines promote coagulation and thrombosis. 2 Retraction of BBMECs and deposition of fibrin detected in and around biofilm-like clusters of H. somni in myocardial microvasculature are consistent with in vitro investigations of H. somni interaction with bovine endothelial cells from several tissues. This may have broader implications for the role of biofilms in endocarditis and vascular thrombosis in general.
Footnotes
Acknowledgements
We thank Paula Jaeger and Mercedes Thelen for histology preparations, Amy Boerger-Fields for assistance with bacteriology, Lauren Crum for help with the cell culture graphs, and Carol Hearne for ultrastructural preparations. We are grateful to Dr Yuichi Tagawa (National Institute of Animal Health, Japan) for the monoclonal antibody to the H. somni 37 kDa OMP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the USDA Animal Health Program WYO-397-05, an undergraduate EPSCoR grant for T. Allen while a preveterinary student at the University of Wyoming, and USDA NIFA grants 2005-35204-16257 and 2011-67015-30177 to L. B. Corbeil.
