Abstract
Sensitive markers to detect acute kidney injury (AKI) in cats are lacking. Kidney injury molecule-1 (KIM-1) is a promising marker of acute tubular injury in humans, and sequence and structure of feline KIM-1 have been determined. KIM-1 is shed into urine of cats with natural AKI. The objectives of this study were to characterize temporal and cellular expression of KIM-1 in kidneys from cats without and with experimental and natural AKI using histopathology and immunohistochemistry. Tissue sections from 8 cats without kidney disease, 3 to 4 cats with experimentally induced AKI on each day 1, 3, 6, and 12 after unilateral ischemia/reperfusion, and 9 cats with natural AKI were assessed. In sections from cats without kidney disease, patterns of periodic acid–Schiff and aquaporin-1 staining allowed identification of 3 distinct segments of the proximal tubule. KIM-1 staining was absent in segments 1 (S1) and S2, and faint in S3. Injury of S3 in cats with experimental and natural AKI was characterized by cell loss and necrosis, and remaining intact cells had cytoplasmic blebs and reduced brush borders. In experimental AKI, intensity of KIM-1 expression increased in proportion to the severity of injury and was consistently present in S3 but only transiently in other segments. Vimentin was absent in proximal tubules of healthy cats but expressed in injured S3. These findings indicate that S3 is the proximal tubular segment most susceptible to ischemic injury and that KIM-1 is a sensitive tissue indicator of AKI in cats.
Keywords
Chronic kidney disease (CKD) is a common condition in older cats with an estimated incidence of 15% to 49%. 17,39,48 Histopathologic findings typically consist of hyperplastic arteriolosclerosis, interstitial fibrosis, inflammation, and tubular mineralization, which are considered nonspecific terminal changes. 16,27,46,47,52 Affected cats have persistent increases in serum creatinine concentration (SCC) and reduced urine concentrating ability and develop a plethora of additional clinical and laboratory abnormalities associated with reduced glomerular filtration rate, acid-base derangements, ineffective erythropoiesis, and altered blood pressure regulation, all leading to poor quality of life. 13,17,24,27,59 Despite the common occurrence of CKD in cats, the cause remains largely unknown. 17 In humans, it has been recognized that acute kidney injury (AKI) is a common precursor to CKD. 7,18 –20,22,78 Acute kidney injury in humans and animals has been challenging to recognize at the time of occurrence due to the relative insensitivity of traditional tests such as SCC and urine specific gravity (USG). However, in recent years, more sensitive markers indicating injury of specific segments of the kidney have been discovered, and their clinical utility in humans and rodents has been evaluated. 34,57,68,72,74 Kidney injury molecule-1 (KIM-1) is one such marker measurable in urine of humans and rodents with various types of kidney injury. 37,38 KIM-1 is up-regulated 3- to 100-fold in injured tubular cells, and an extracellular portion is released into urine, enabling noninvasive detection and also monitoring of injury progression, resolution, and repair. 71,72 Application of such new and sensitive tests has led to the recognition that many conditions previously considered to be nonharmful are actually injurious for kidneys. Such conditions include prerenal azotemia, hypertension, sepsis, exposure to nephrotoxins, and metabolic derangements leading to reduced glomerular filtration rate. 11,38,57,75 As a result, a fundamentally different understanding of kidney disease (KD) has emerged whereby AKI and CKD are recognized as interconnected risk factors for each other. Furthermore, greater emphasis has been placed on acknowledging that small increases in SCC can indicate AKI in critically ill patients and on implementation of more sensitive assays to detect AKI. 1,8,20,42,53,62
Little is known about sublethal kidney injury in cats and the potential contribution to development of CKD. Several causes of fulminant kidney injury are recognized, such as urethral obstruction, exposure to certain drugs, and ingestion of ethylene glycol and Lilium sp. 23,28,29,43,66 In an effort to more sensitively detect AKI, it was suggested by the International Renal Interest Society (IRIS) to adopt a scoring system for AKI in animals similar to that in humans. 24 The suggested scoring system incorporates small increases in SCC irrespective of reference intervals (RIs) and reduced urine output. However, specific assays to detect AKI in cats remain unavailable.
KIM-1 gene and protein have been characterized in cats, and the protein has been detected in urine of critically ill cats but not those that were clinically healthy. 9 However, precise location of KIM-1 in kidney tissue and changes in expression with injury are unknown. Aquaporin-1 (AQP-1), previously identified in feline kidney tissue, functions to regulate water transport in proximal renal tubules and red blood cells. 14,58 In humans, KIM-1 colocalizes with AQP-1 in proximal tubular cells. 76 Vimentin (VIM) is a cytoskeletal intermediate filament with limited expression in epithelial cells under homeostatic conditions but is increased in renal epithelial cells undergoing dedifferentiation, repair, and osmoadaption. 15,64 VIM was expressed concurrently with KIM-1 in injured proximal tubules (PT) in humans and rats. 76 This study addressed the hypothesis that AKI results in KIM-1 expression in specific segments of the PT of cats. The objectives were to characterize KIM-1 expression in specific areas of the PT in sections without injury and in sections with experimental and naturally occurring AKI.
Materials and Methods
No Kidney Disease
Kidneys were obtained from 8 intact cats (3 male and 5 female) between 1 and 3 years of age with no clinical or laboratory evidence of KD. These cats had SCC < 160 μmol/liter, USG > 1.035, and unremarkable urine chemistry and sediment. The cats were euthanized for reasons unrelated to this study (approved by the Institutional Animal Care Committee, University of Guelph). Cats were sedated by intramuscular injection of ketamine (10 mg/kg) and butorphanol (0.4 mg/kg) 15 minutes prior to collection of blood by venipuncture and urine by cystocentesis. Cats were then euthanized with an overdose of barbiturate; kidneys were removed immediately, sagittally sectioned, fixed in 10% neutral-buffered formalin for 24 to 36 hours, and processed routinely for histopathology.
Experimental AKI
Eight adult male intact cats, aged 15 to 29 months, had SCC < 160 μmol/liter, USG > 1.035, and unremarkable urine chemistry and sediment. In these cats, AKI was induced by unilateral renal ischemia and reperfusion (I/R) similar to that previously described and as approved by the Institutional Animal Care and Use Committee, University of Georgia (AUP A200903-056-Y3-A7). 65 Approximately 10 months prior to study, the cats had been inoculated with Brugia malayi microfilaria but had failed to become microfilaremic. The cats were scheduled for euthanasia since exposure to a human pathogen precluded adoption. Food but not water was withdrawn for 12 hours; cats were premedicated with ketamine (7 mg/kg) and butorphanol (0.4 mg/kg), induced with propofol, and intubated; and anesthesia was maintained with isoflurane. Intravenous lactated Ringer’s solution (10 ml/kg/hour) was administered during anesthesia. A midline laparotomy was performed, and a vascular clamp was placed across the right renal artery and vein for 45 minutes. Reflow of blood was verified after release of the clamp, and the abdominal cavity was closed. Blood pressure was monitored during surgery, cats received intravenous fluids during and after surgery, and postoperative analgesia was provided via transdermal fentanyl patch (25 μg/hour) and oral transmucosal buprenorphine (0.02 mg/kg). Serum creatinine concentration and USG were measured before surgery and on postoperative days 1 and 3. Four cats each were euthanized at 24 hours and 72 hours post I/R with an overdose of barbiturate. Both nonischemic and ischemic kidneys were removed immediately and processed as above.
Kidney sections were also available from 9 male intact cats without clinical or laboratory evidence of KD that had undergone 60 minutes of unilateral I/R. 65 In these cats, cortical wedge biopsies (approximately 2 × 3 mm) were obtained from the body of the ischemic kidney immediately after renal artery and vein clamp placement, from the cranial pole 1 hour after clamp placement (ischemia), and from the caudal pole 1 hour after clamp removal (1 hour each ischemia and reperfusion). Three cats each were euthanized on postoperative days 3, 6, and 12, and nonischemic and ischemic kidneys were collected. Biopsies and kidneys were processed as above.
Natural AKI
Nine cats admitted to the Health Sciences Centre at the University of Guelph had cancer (2), CKD (3), cardiac arrest (1), sepsis (1), Lilium sp. toxicosis (1), or urethral obstruction (1). These cats either presented with AKI or developed AKI during hospitalization or had prior CKD with a recent increase in SCC that was minimally responsive to therapy. Cats were euthanized due to poor response to therapy for KD or due to other untreatable illness and submitted for routine postmortem examination. Kidney sections were fixed in 10% neutral-buffered formalin and processed as above.
Histopathology
All sections were stained with hematoxylin and eosin (HE) and periodic acid–Schiff (PAS) with hematoxylin counterstain. Renal tubular segments, glomeruli, and interstitium of the cortex and outer stripe of the outer medulla (OSOM) were identified by morphologic features as described by Maunsbach and Christensen, 50 Christensen et al, 21 and Ross and Pawlina. 63 Accordingly, the OSOM comprises the region approximately 0.1 cm on either side of the arcuate artery and vein, whereas the cortex comprises the region of approximately 0.5 cm thickness between the capsule and the OSOM (Fig. 1). Changes in the PT of the cortex and OSOM were scored separately, accounting for the frequency of cell necrosis (nuclear pyknosis, karyolysis, karyorrhexis, and hypereosinophilia), loss or coalescence of brush borders, cytoplasmic blebs, apoptotic features, cells with cytoplasmic vacuoles containing KIM-1-immunopositive debris, cells in tubular lumens, and cellular regeneration (cytoplasmic basophilia, flattening of tubules, anisokaryosis, and nuclear or cellular disarray), as proposed in other studies. 32,33,60,77 Possible individual scores ranged from 0 to 5 (Table 1). Inflammation and tubular luminal dilation were also graded on a scale from 0 to 5, and mitoses were counted (Table 1).
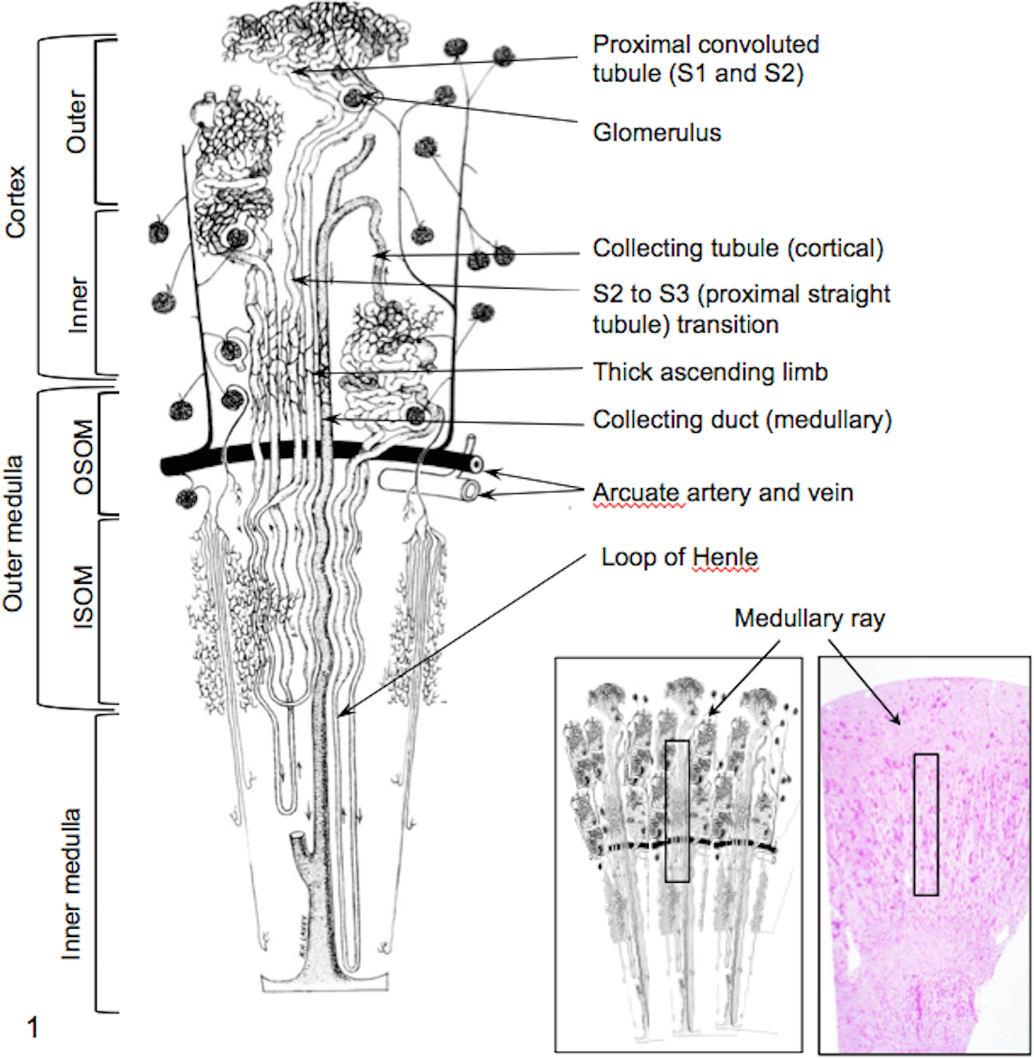
Schematic of nephron showing tubule arrangement in cortex and the approximate division between cortex and outer and inner stripe of medulla. ISOM, inner stripe of outer medulla; OSOM, outer stripe of outer medulla. Loops of Henle in cats are long and extend into the inner medulla. Insets: Schematic and periodic acid–Schiff-stained section of healthy cat kidney outlining medullary ray. Adapted from Beeuwkes and Bonventre (1975). 6
Injury and Immunohistochemical Scores in Sections From Cats Without Kidney Disease.
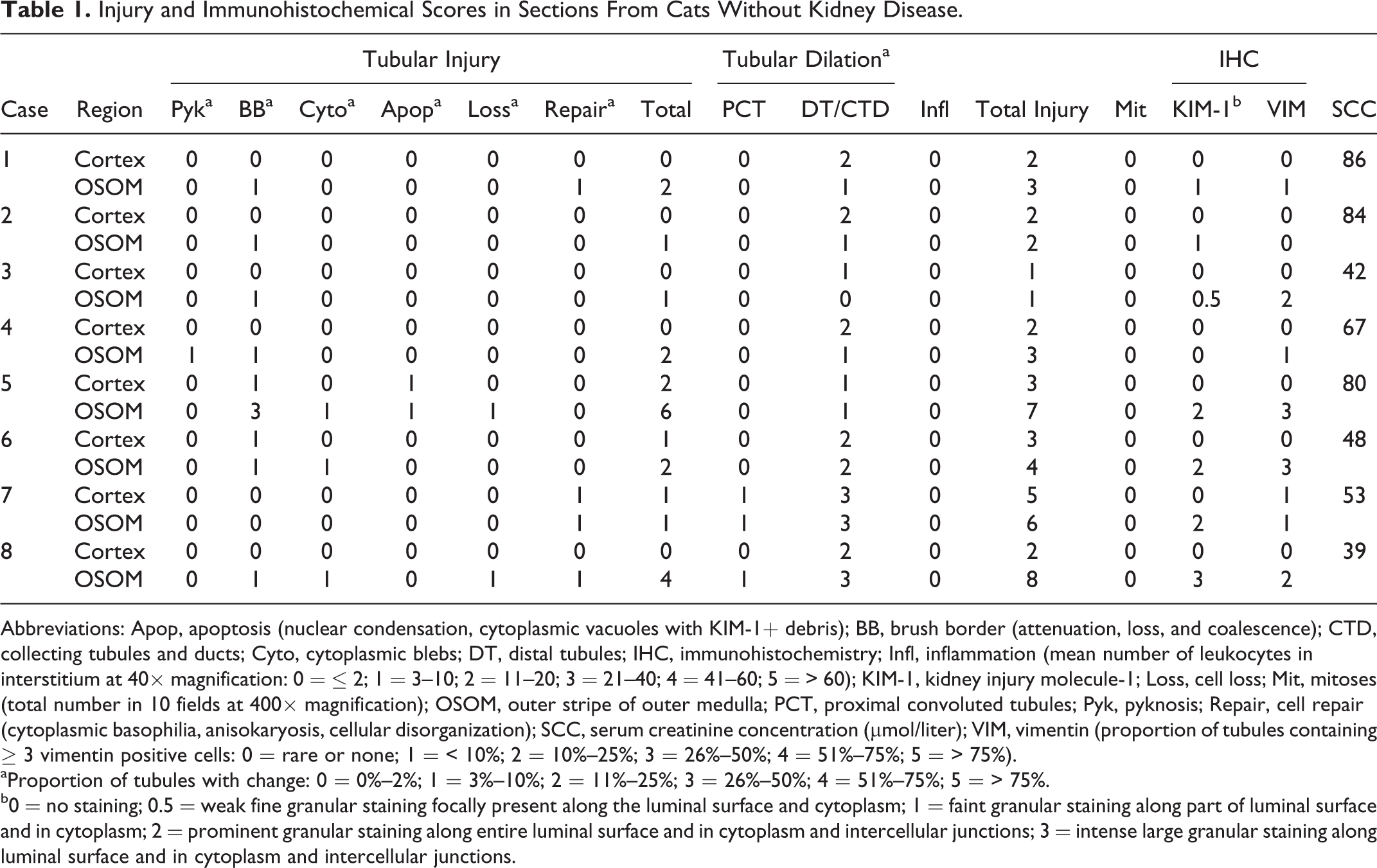
Abbreviations: Apop, apoptosis (nuclear condensation, cytoplasmic vacuoles with KIM-1+ debris); BB, brush border (attenuation, loss, and coalescence); CTD, collecting tubules and ducts; Cyto, cytoplasmic blebs; DT, distal tubules; IHC, immunohistochemistry; Infl, inflammation (mean number of leukocytes in interstitium at 40× magnification: 0 = ≤ 2; 1 = 3–10; 2 = 11–20; 3 = 21–40; 4 = 41–60; 5 = > 60); KIM-1, kidney injury molecule-1; Loss, cell loss; Mit, mitoses (total number in 10 fields at 400× magnification); OSOM, outer stripe of outer medulla; PCT, proximal convoluted tubules; Pyk, pyknosis; Repair, cell repair (cytoplasmic basophilia, anisokaryosis, cellular disorganization); SCC, serum creatinine concentration (μmol/liter); VIM, vimentin (proportion of tubules containing ≥ 3 vimentin positive cells: 0 = rare or none; 1 = < 10%; 2 = 10%–25%; 3 = 26%–50%; 4 = 51%–75%; 5 = > 75%).
aProportion of tubules with change: 0 = 0%–2%; 1 = 3%–10%; 2 = 11%–25%; 3 = 26%–50%; 4 = 51%–75%; 5 = > 75%.
b0 = no staining; 0.5 = weak fine granular staining focally present along the luminal surface and cytoplasm; 1 = faint granular staining along part of luminal surface and in cytoplasm; 2 = prominent granular staining along entire luminal surface and in cytoplasm and intercellular junctions; 3 = intense large granular staining along luminal surface and in cytoplasm and intercellular junctions.
Immunohistochemistry
Three micron sections were deparaffinized in graded alcohols, rehydrated, and incubated for 10 minutes with endogenous enzyme blocker and 30 minutes with serum-free protein blocker (DakoCytomation, Mississauga, ON, Canada). For AQP-1 staining, antigens were retrieved by heating to 125°C under pressure for 2 minutes and 45 seconds in pH 6.0 target retrieval solution (DakoCytomation). KIM-1 antibody (1:40 dilution, clone 219211; R&D Systems, Minneapolis, MN, USA) and AQP-1 rabbit polyclonal antibody (1:1200; EMD Millipore, Temecula, CA, USA) were applied overnight at 4°C. Signal from bound antibodies was amplified with Envision Dual Link System-HRP (DakoCytomation) and detected with Nova Red chromogen (Vector Laboratories, Burlington, ON, Canada). Slides were counterstained with hematoxylin. Antibody to VIM (1:200 dilution, clone V9; DakoCytomation) and cytokeratin (1:100 dilution, clone AE1/AE3; DakoCytomation) was applied after antigen retrieval at 120°C and pH 6.0 for 2 minutes. Slides were prepared in batches of 6 to 8, and each batch of slides included negative (omission of primary antibody) control slides and sections of feline tissues that expressed the relevant antigen. Additional negative controls for KIM-1 consisted of sections where antibody was preincubated for 2 hours with human KIM-1 peptide (1750-TM; R&D Systems) prior to immunohistochemistry (IHC). Specificity of the KIM-1 antibody was also assessed by Western blotting of urine from cats with AKI, which yielded a prominent band of ∼ 30 kD (data not shown).
Tubule segments were identified by morphology (HE and PAS staining) and expression of AQP-1 (PT and loops of Henle) or cytokeratin (collecting ducts and collecting tubules in cortex and OSOM). 14,69 Serial sections were prepared for all IHC stains, adjoining regions were aligned for scoring and image capture, and parameters in cortex and OSOM were scored separately in all sections. One pathologist blinded to the etiology of underlying disease and duration since I/R scored all sections. KIM-1 IHC staining was scored on a scale from 0 to 3 as proposed by Zhang et al. 84 Immunohistochemistry scores for VIM were based on the proportion of segments containing ≥ 3 positively stained cells. Immunohistochemistry staining for AQP-1 and cytokeratin was used to identify nephron segments and was not graded. Biopsy sections were assessed qualitatively for morphologic changes and KIM-1 staining.
Data Analysis
All data analysis was performed with Prism 6.0 software (GraphPad, La Jolla, CA, USA). Data from different groups were compared with Wilcoxon matched-pairs signed rank tests or 1-way analysis of variance (ANOVA) with repeated measures (Friedman test) for 3 or more group comparisons of nonparametric data, and by linear regression analysis and calculation of nonparametric Spearman correlation coefficient. Significance was set at P ≤ .05.
Results
No Kidney Disease
Serum creatinine concentrations ranged from 39 to 86 μmol/liter, and cats had adequate concentrating ability (USG 1.045–1.072). For histopathologic assessment, nephron organization and terminology as shown in Fig. 1 were applied. The proximal convoluted tubule is composed of S1 and S2 segments, and the proximal straight tubule of the S3 segment. The medullary ray includes the S3 segment, the thick ascending limb (TAL) of the distal tubule, and collecting ducts extending from the inner cortex to OSOM. Thus, the OSOM and inner cortex contain most S3 segments, and the outer cortex consists primarily of S1 and S2 segments.
All sections were stained with PAS and HE (Fig. 2). PAS staining was superior for assessing tubule morphology, brush borders, and basement membranes. In PAS-stained sections, S1 cells were characterized by pale magenta cytoplasm with numerous prominent large vacuoles, whereas S2 cells had fewer and smaller vacuoles and more intensely magenta-colored cytoplasm (Fig. 2a, b). Cells of the S3 segment had a cuboidal to columnar shape, basal location of relatively large nuclei, blue-magenta cytoplasm, and absent or rare cytoplasmic vacuoles. Cells in S3 had less distinct and shorter brush borders than adjacent S2 cells (Fig. 2c). Transitions from S1 to S2 were rarely identified, whereas from the transitions from S2 to S3 were identifiable with PAS but not HE staining (Fig. 2c, d).
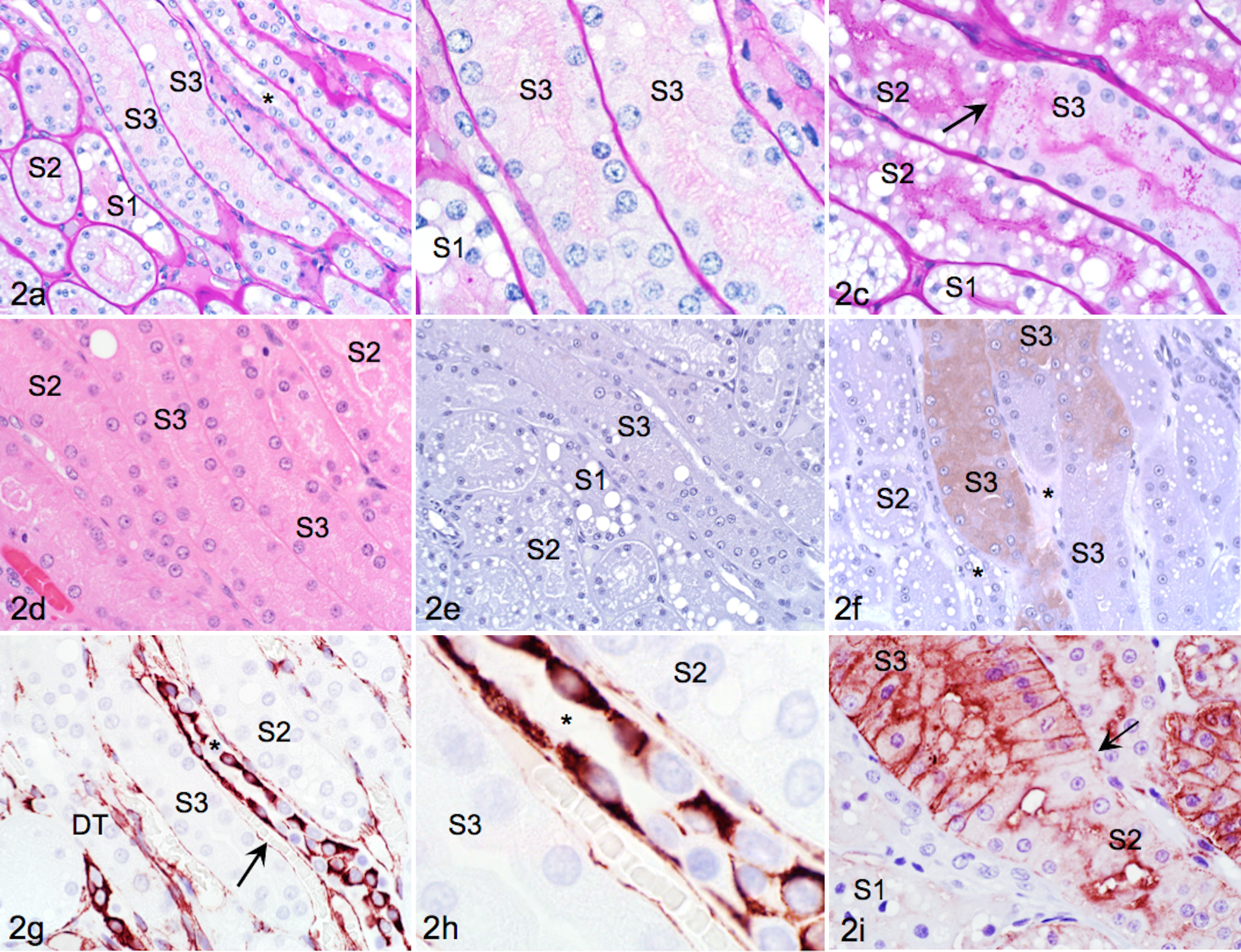
Kidney; healthy cat, adjoining sections of outer stripe of the outer medulla (OSOM). (a) Periodic acid–Schiff (PAS) stain highlights basement membrane and intact and even brush borders. S1 and S2 cells have more intensely stained brush borders and more frequent cytoplasmic vacuoles than S3. Collecting ducts (*) are tubules with flat epithelium running parallel to proximal tubules. (b) Higher magnification shows minimal cytoplasmic vacuolation in S3 cells. (c) Transition (arrow) from highly vacuolated S2 cells to S3 cells lacking vacuoles is highlighted by PAS stain. (d) Different tubule segments are less readily identifiable and have less contrast with hematoxylin and eosin (HE) than PAS stain. (e, f) Kidney injury molecule-1 (KIM-1) immunohistochemistry (IHC). Proximal tubules in the OSOM either were KIM-1 negative or had faint diffuse cytoplasmic staining in some S3 cells. (g) Vimentin IHC. Collecting ducts and endothelial cells (arrow) parallel to S3 are vimentin positive, whereas proximal and distal tubules (DT) are vimentin negative. (h) At high magnification, vimentin IHC yields a mosaic pattern of differentially stained intercalated and principal cells in collecting ducts. (i) S3 stains much more intensely for aquaporin-1 (AQP-1) than S1 and S2. Intense membranous staining of tall columnar cells conveys cobblestone appearance to S3. Transition from S3 to S2 (arrow) is characterized by change in AQP-1 staining intensity and reduction in cell height. There is no AQP-1 expression in collecting ducts or DT.
In cats without KD, there was either none (Fig. 2e) or diffuse faint cytoplasmic (Fig. 2f) KIM-1 staining in some of S3. Immunohistochemical detection of KIM-1 was specific as indicated by lack of reactivity if primary antibody was omitted, or if primary antibody was preincubated with peptide (data not shown).
VIM immunoreactivity was apparent in glomerular and vascular endothelium, collecting tubules (CT), and collecting ducts (CD). Within CT and CD, there was cell-to-cell variably intense mosaic staining, likely attributed to differences in VIM expression by intercalated and principal cells (Fig. 2g, h). 21,69 VIM immunoreactivity was absent in PT.
All segments of PT stained positive for AQP-1 (Fig. 2i). Staining intensity in S1 cells was light and mostly along the luminal surface of cells with occasional fine granular staining at the basilar surface (Fig. 2i). S2 cells had more intense luminal membrane staining, and there was also frequent fine granular staining near the basilar cell border. S3 cells had intense staining along the luminal and basolateral cell membrane, creating a cobblestone effect, and also occasional light cytoplasmic staining. Changes in AQP-1 staining intensity highlighted the transition from S2 to S3, which corresponded to changes in brush border density as identified with PAS staining (Fig. 2c). The descending loop of Henle, vasa recta, endothelium, and erythrocyte membranes were AQP-1 immunopositive, and there was occasional faint staining in glomeruli.
No proximal tubular cells were positive for cytokeratin with antibody A1/A3; however, CD and CT stained positive. Occasional TAL had fine cytoplasmic granular staining (data not shown).
Experimental AKI
Prior to experimental AKI, cats had SCC of 106 to 159 μmol/liter and adequate concentrating ability (USG 1.035–1.058). Kidney injury after 45 minutes of ischemia and reperfusion (I/R) was extensive, and PAS-, AQP-1-, and VIM-stained serial sections were essential to identify type of tubule and segment and to localize KIM-1 staining (Fig. 3). The degree of cell necrosis and injury varied across segments both within and between medullary rays, and tubule identification was difficult with HE alone if there was extensive epithelial sloughing and necrosis (Fig. 3a). Intact tubules or individual cells were considered to be S3 if brush borders and large basal nuclei with single prominent nucleoli were apparent on PAS staining and if cytoplasmic vacuoles were absent (Fig. 3b). Intense membranous AQP-1 expression in some remaining adherent and in sloughed cells identified them as S3 (Fig. 3c). There was prominent KIM-1 staining of individual segments with intensity varying between cells and tubules. Tubules positive for KIM-1 were S3, as evidenced by morphology and AQP-1 staining in adjoining sections. There were occasional faintly labeled S2. Cells of the TAL and loop of Henle in the inner stripe of the outer medulla had vacuolar change, were consistently KIM-1 negative, and occasionally contained luminal KIM-1/AQP-1 positive cells (Fig. 4a, b, respectively).
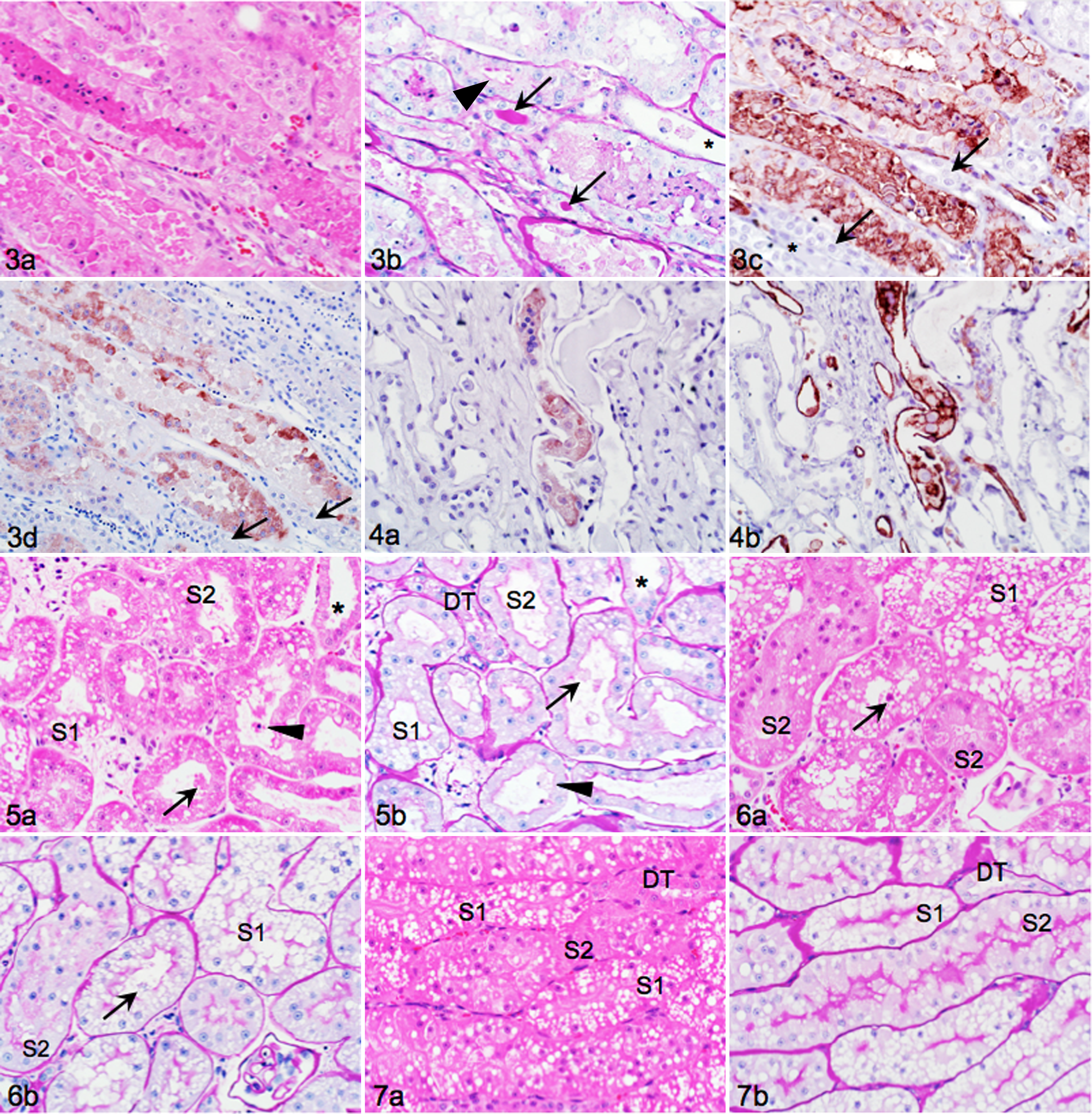
Day 1, I/R for 45 minutes
Cortical S1 and S2 had indistinct nuclei and hypereosinophilia. Tubules had mild to moderate luminal dilation, cellular flattening, loss of brush borders, and occasional cytoplasmic blebs and whole cells in lumens (Fig. 5a, b). In the contralateral nonischemic kidney, there was mild luminal dilation and attenuation of brush borders (Fig. 6a, b). Brush border changes, individual segments, and hydropic changes were more readily apparent with PAS than HE stain. Sections from both I/R and nonischemic kidneys had variably severe changes relative to those in cats without KD (Fig. 7a, b).
In the OSOM, there was extensive tubular injury but marked variability in the severity of injury between adjacent S3 within the same medullary ray: some S3 had relatively intact epithelium with membrane-bound cytoplasmic blebs and loss of brush borders, whereas other S3 lacked intact epithelium and lumens contained extensive cellular casts (Fig. 8a, b). The interstitium was expanded and included congested capillaries between tubules. Within the cytoplasm of intact epithelial cells were occasional apoptotic bodies (Fig. 8a). PAS staining illustrated complete loss of brush borders (Fig. 8b). Strongly PAS-positive homogeneous material characteristic of Tamm–Horsfall protein was evident within the TAL of I/R kidneys (Fig. 8b). 30 Remaining intact S3 epithelium of kidneys with I/R was strongly KIM-1 positive (Fig. 8c). Staining intensity varied between tubules but was overall much greater than in the contralateral nonischemic kidney or in sections from cats without KD. Cytoplasmic blebs attached to the luminal cell membrane and the basilar cell membrane were most intensely KIM-1 positive, whereas luminal cellular debris was variably positive (Fig. 8c).
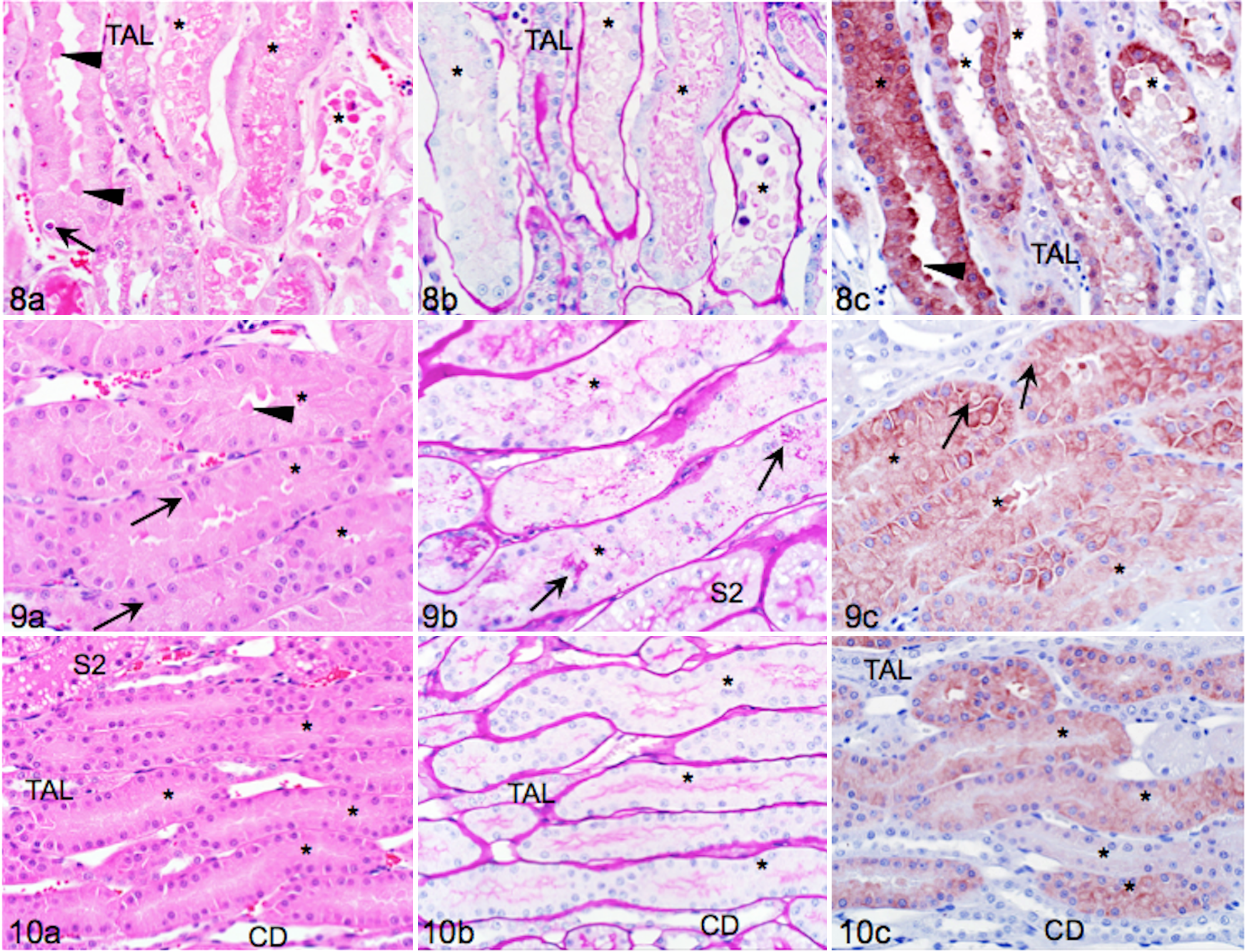
Proximal tubular cells in the contralateral nonischemic kidney were largely intact but swollen with frequent cytoplasmic blebs and luminal cell debris (Fig. 9a). Loss of lateral adhesion of epithelial cells was apparent as intercellular gaps. There were occasional cells with pyknotic nuclei and hypereosinophilic cytoplasm among the intact epithelium or sloughed into lumens (Fig. 9a). PAS staining highlighted that brush borders were attenuated and irregular but less severely affected than in I/R kidneys and that there were cytoplasmic hyaline droplets (Fig. 9b). KIM-1 staining (Fig. 9c) had a different pattern than in sections with I/R. Staining was moderately intense and often most pronounced in the cell periphery (Fig. 9c). There were occasional cytoplasmic vacuoles containing KIM-1 positive material (likely phagolysosomes). In comparison, sections from cats without KD lacked sloughed cells and had even brush borders and faint diffuse KIM-1 staining (Fig. 10a–c).
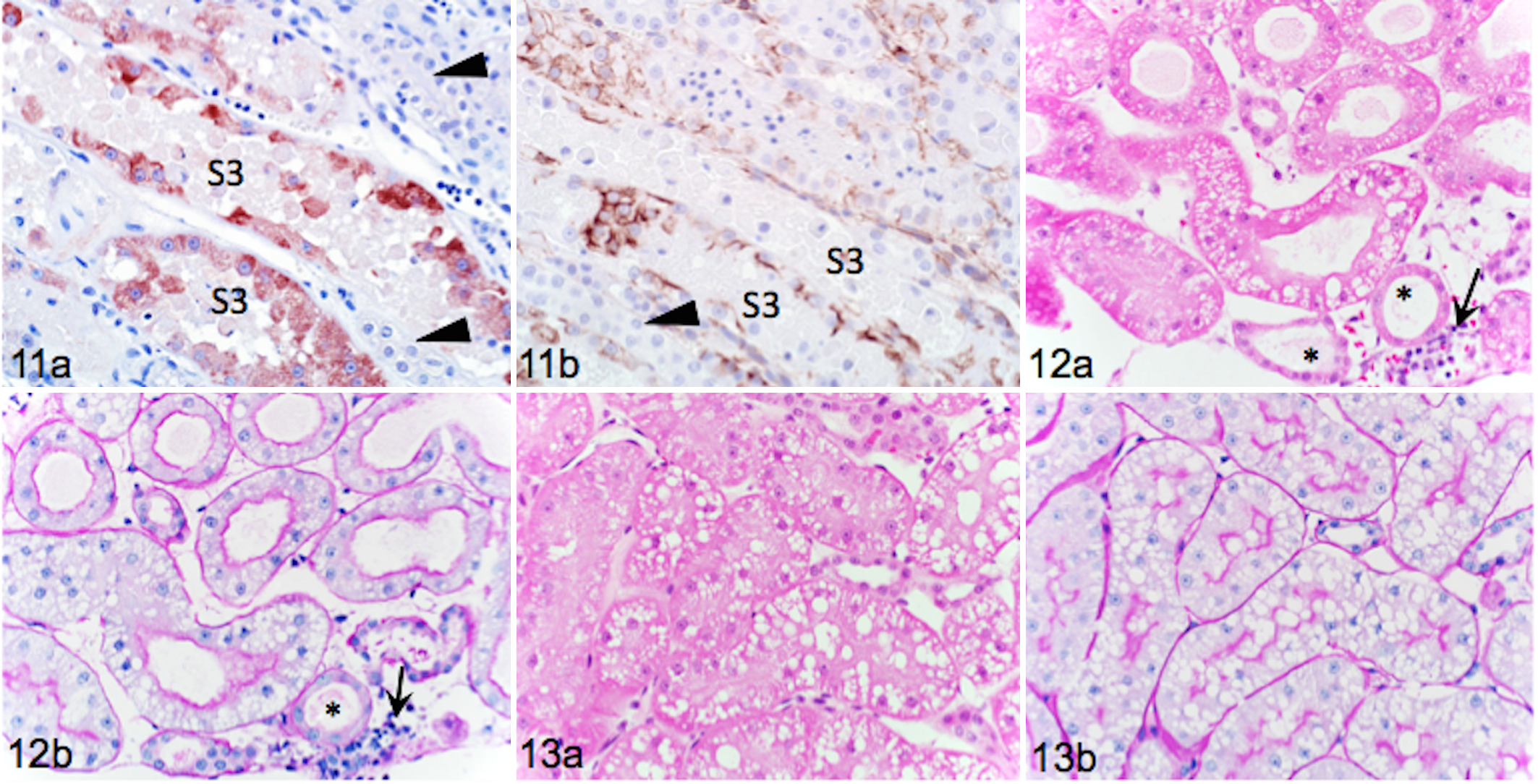
KIM-1 positive S3 cells attached to basement membranes of I/R kidneys stained positive for VIM, suggesting focal dedifferentiation (Fig. 11a, b). Only rare faint cytoplasmic VIM-positive filaments were noticed in contralateral kidneys (not shown).
Day 3, I/R for 45 minutes
By day 3 after I/R, a greater proportion of S1 and S2 tubules in the cortex had luminal dilation and cellular flattening (Fig. 12). Interstitial edema was more extensive and there was accumulation of leukocytes (neutrophils, lymphocytes, eosinophils, and plasma cells) in subcapsular veins with capillary extravasation (Fig. 12a, b). Luminal debris, necrotic cells, and cytoplasmic blebs were less prominent, and brush borders were short but present. A few cells had very fine VIM-positive filaments (not shown). In contralateral kidney sections, luminal dilation, cell flattening, and leukocyte accumulation were absent, and interstitial edema, cytoplasmic blebbing, and cell loss were less frequent than I/R kidneys (Fig. 13a, b). Brush borders were thicker and more even on day 3 (Fig. 13b) than day 1 (Fig. 6b). Cortical KIM-1 expression was not detected.
In the OSOM, S3 had moderate to marked luminal dilation, luminal cell debris, attenuation of brush borders, and cell flattening (Fig. 14a, b). Cell repair as indicated by cytoplasmic basophilia, anisokaryosis, anisocytosis, cellular disorganization, and nuclear crowding was evident but varied markedly between segments (Fig. 14a). Single cell necrosis (hypereosinophilia, pyknosis, karyolysis) and loss of cells into lumens were still present, but tubular casts were less prominent. Leukocytes (lymphocytes, neutrophils, eosinophils) were present in dilated capillaries between S3 segments and within the vasa recta (Fig. 14a). Intact S3 cells were KIM-1 positive (Fig. 14c), but expression was overall less intense and more variable than on day 1. There were intensely stained individual epithelial cells and luminal cell debris was strongly positive, but intact tubular cells undergoing repair had less intense and more diffuse immunopositivity (Fig. 14c). Sections from I/R kidneys with intense KIM-1 staining were much more extensively and strongly VIM positive (Fig. 14d) relative to day 1.
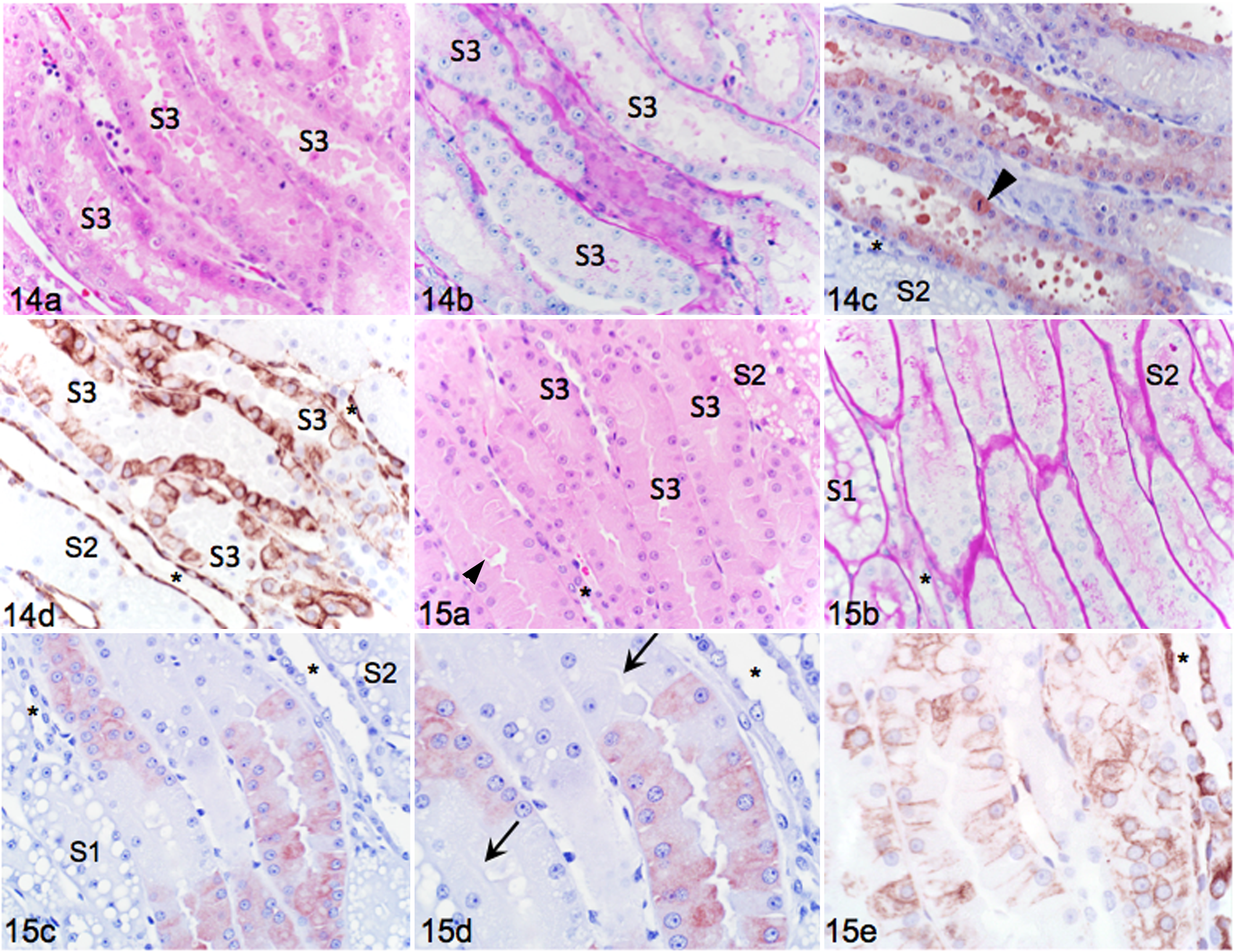
In contralateral sections, epithelial cells in the OSOM were less swollen (Fig. 15a, b) than on day 1 (Fig. 9a), but there was still occasional cytoplasmic blebbing and luminal debris. Pyknotic, hypereosinophilic, and luminal cells were rare, and brush borders were clearly identifiable (Fig. 15b). Some S3 sections remained KIM-1 immunopositive, but individual cells and portions of tubules were distinctly negative (Fig. 15c, d). VIM staining (Fig. 15e) in cells positive for KIM-1 was less intense than with I/R, and mostly along the cell borders.
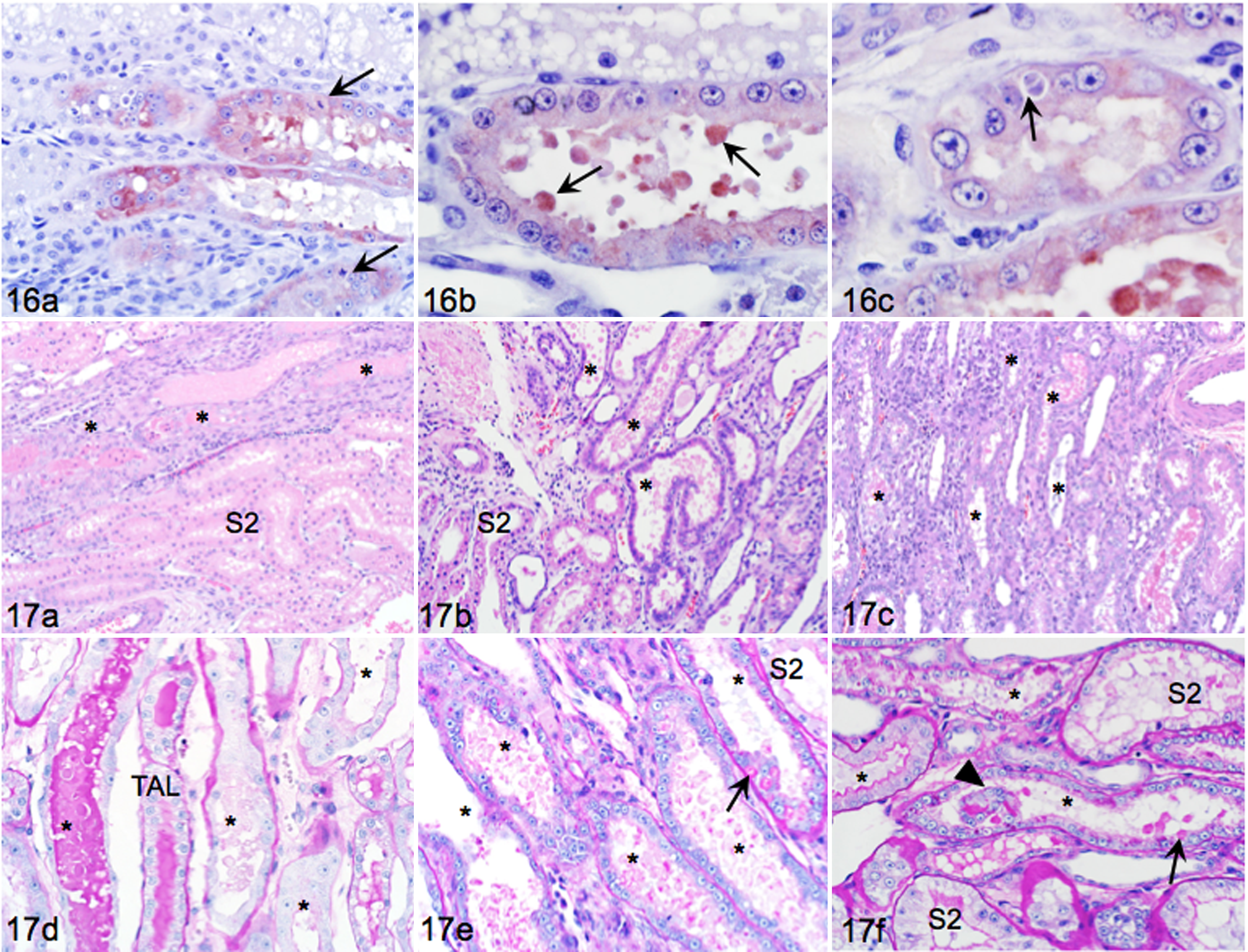
At higher magnification, disorganized repairing epithelium, mitoses, binucleation, anisokaryosis, and nuclear crowding were prominent in the OSOM in I/R (Fig. 16a). In other areas of the same section, KIM-1 positive S3 were composed of more evenly sized cells with frequent KIM-1 positive luminal blebs (Fig. 16b). KIM-1 positive S3 cells sometimes contained cytoplasmic structures suggestive of phagocytic vacuoles (Fig. 16c).
Days 3, 6, and 12, I/R for 60 minutes
The type of injury to S3 on day 3 was of a similar nature but more severe than that after 45 minutes I/R and affected a greater proportion of S3 (Fig. 17a). On day 6, tubules were profoundly dilated, epithelial cells flattened, and many tubules had intensely basophilic epithelium (Fig. 17b). Luminal debris was less on day 6 than day 3, but interstitial leukocyte infiltrates were more extensive. On day 12, interstitial leukocyte infiltrates were widespread, collagen deposits were apparent, and tubules were fewer per field with distorted tubular arrangement (Fig. 17c). Tubule and segment identification was difficult. There was marked variability in the degree of injury within and between components of the medullary ray.
Staining with PAS showed complete loss of brush borders and coalesced brush borders amid luminal cell debris (Fig. 17d). Short stretches of basement membranes were denuded, and anisocytosis, anisokaryosis, binucleation, and occasional mitotic figures were present among epithelial cells (Fig. 17d). Day 6 sections had prominent PAS-positive cytoplasmic globular material in S3 cells, and amorphous aggregates persisted among luminal debris (Fig. 17e). Some S3 had prominent but short brush borders, whereas others entirely lacked brush borders. Epithelial crowding and binucleation were common. Day 12 sections had thicker and more even brush borders in some S3 but irregular brush borders and presence of PAS-stained aggregates among luminal debris in other tubules (Fig. 17f). Persistence of individual cells and portions of tubules in some S3 lumens reflects ongoing injury.
KIM-1 was expressed with variable intensity on days 3, 6, and 12 after I/R. Throughout all sections and at each time point, KIM-1 was expressed only in S3 in the OSOM. On day 3, KIM-1 staining was most intense in cells dislodged into S3 lumens (Fig. 18a). Flattened epithelial cells that remained attached to basement membranes had greater KIM-1 immunoreactivity along the luminal aspect than in the cytoplasm. KIM-1 positive cells were intensely VIM positive (Fig. 18b) and expressed AQP-1, confirming S3 type (Fig. 18c). In day 6 sections, strongly KIM-1 positive cell debris persisted in lumens, and attached epithelial cells had more uniform cytoplasmic staining (Fig. 19a). Cuboidal epithelial cells with faint KIM-1 staining were intensely VIM positive, whereas columnar epithelial cells with greater KIM-1 expression had relatively less intense VIM staining (Fig. 19b). Cuboidal epithelial cells were also AQP-1 positive, confirming S3 origin (Fig. 19c). This pattern of VIM expression is suggestive of initial injury followed by recent reinjury. By day 12, S3 cells had faint cytoplasmic KIM-1 staining (Fig. 20a) that still corresponded to intense VIM staining (Fig. 20b). AQP-1 (Fig. 20c) expression assisted with identification of S3.
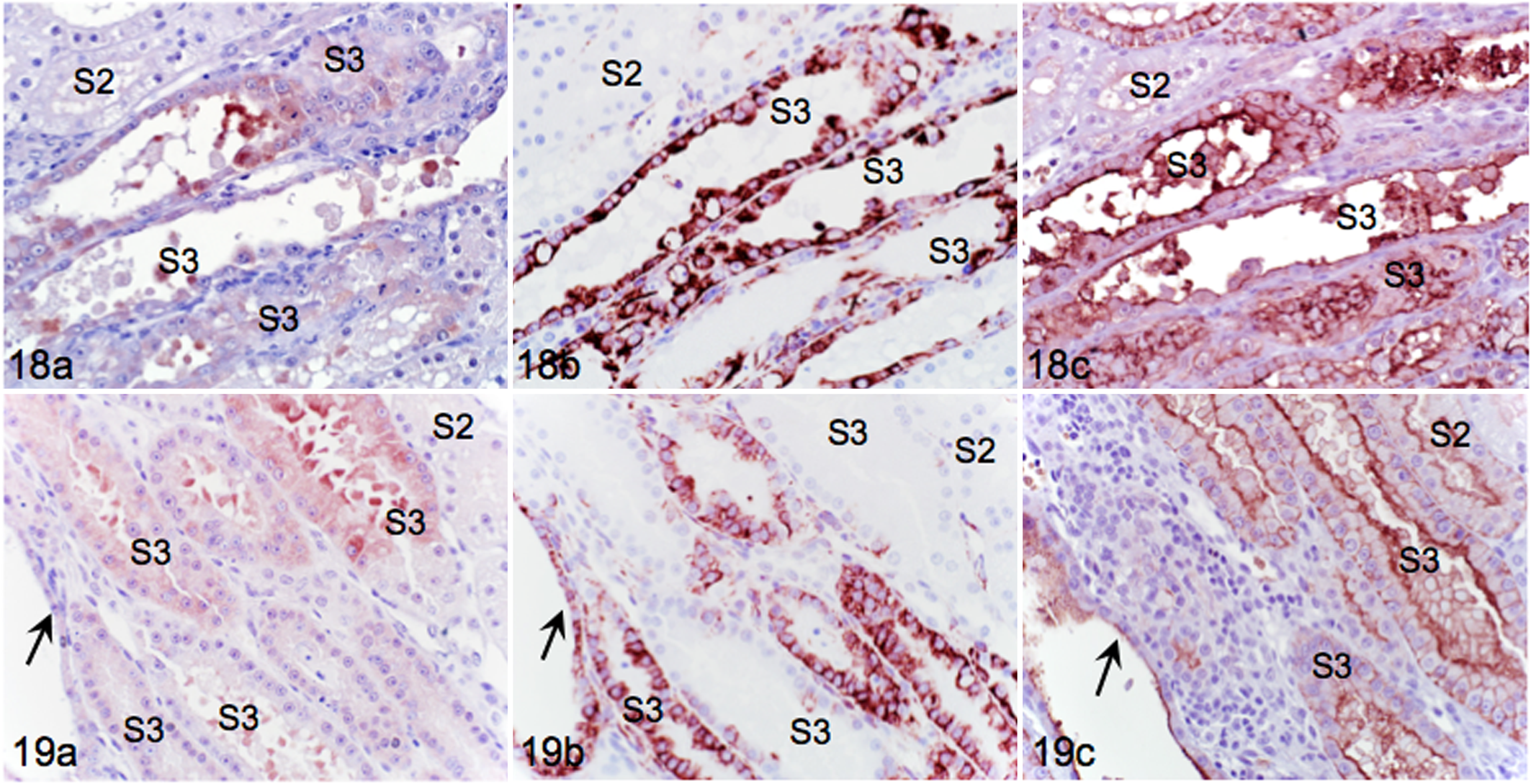
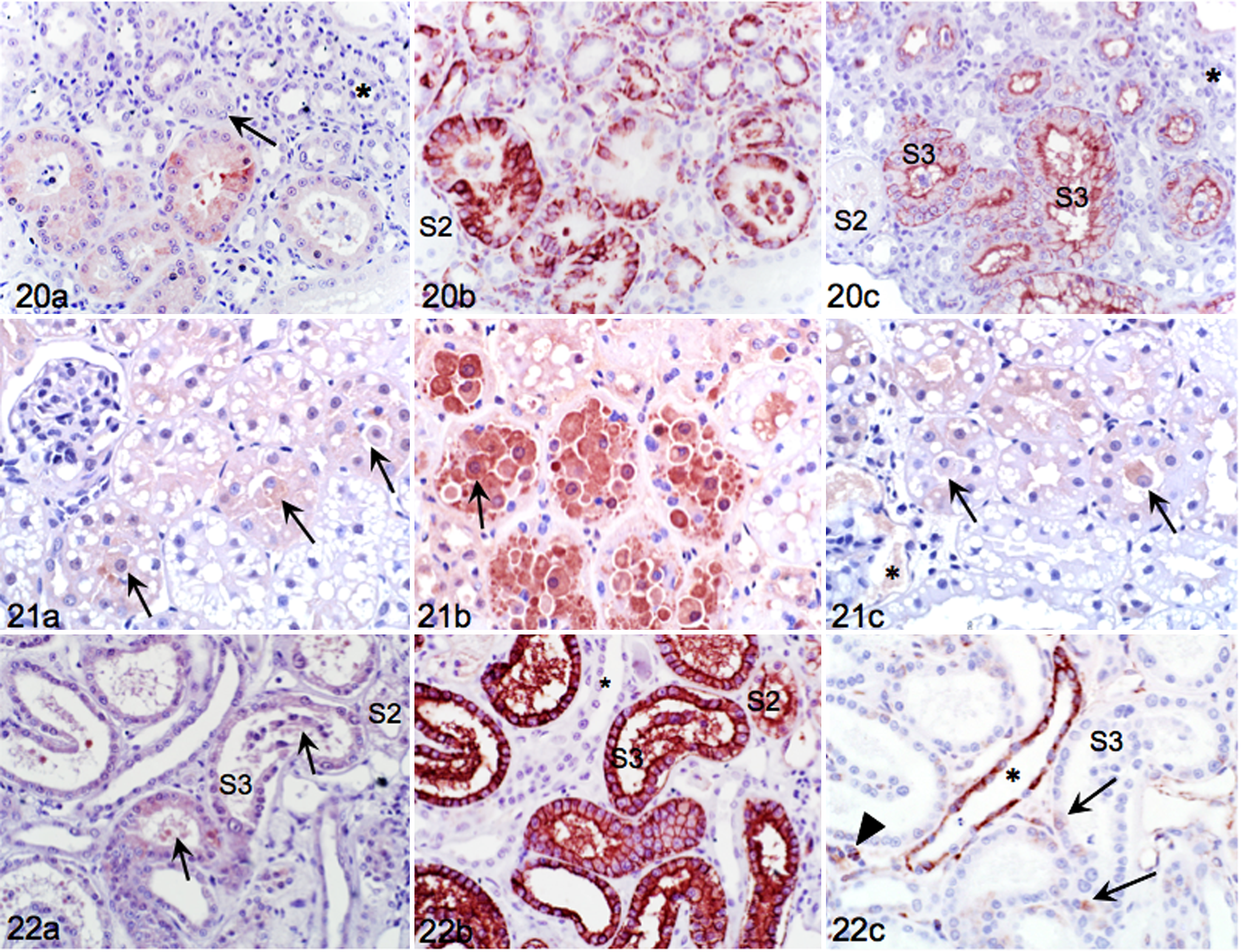
Biopsies obtained immediately after vascular clamping were made up of cortex with S1 and S2 but lacked OSOM with S3. Sections showed light but widespread KIM-1 staining of swollen cells in tubules with extensive but poorly demarcated vacuoles (Fig. 21a). Staining was most pronounced at the luminal aspect of cells, and cells dislodged into lumens were also KIM-1 positive. In sections from 1 hour after onset of ischemia, cell swelling, pyknosis, ballooning, and dislodgement into tubular lumens were extensive, and affected cells were intensely KIM-1 positive (Fig. 21b). In sections from 1 hour after clamp removal (reperfusion), cell swelling and KIM-1 staining intensity were reduced but pyknotic nuclei and dislodged KIM-1 positive luminal cells persisted (Fig. 21c). There was occasional KIM-1 positive fluid in Bowman’s capsule (likely filtrate regurgitation).
Natural AKI
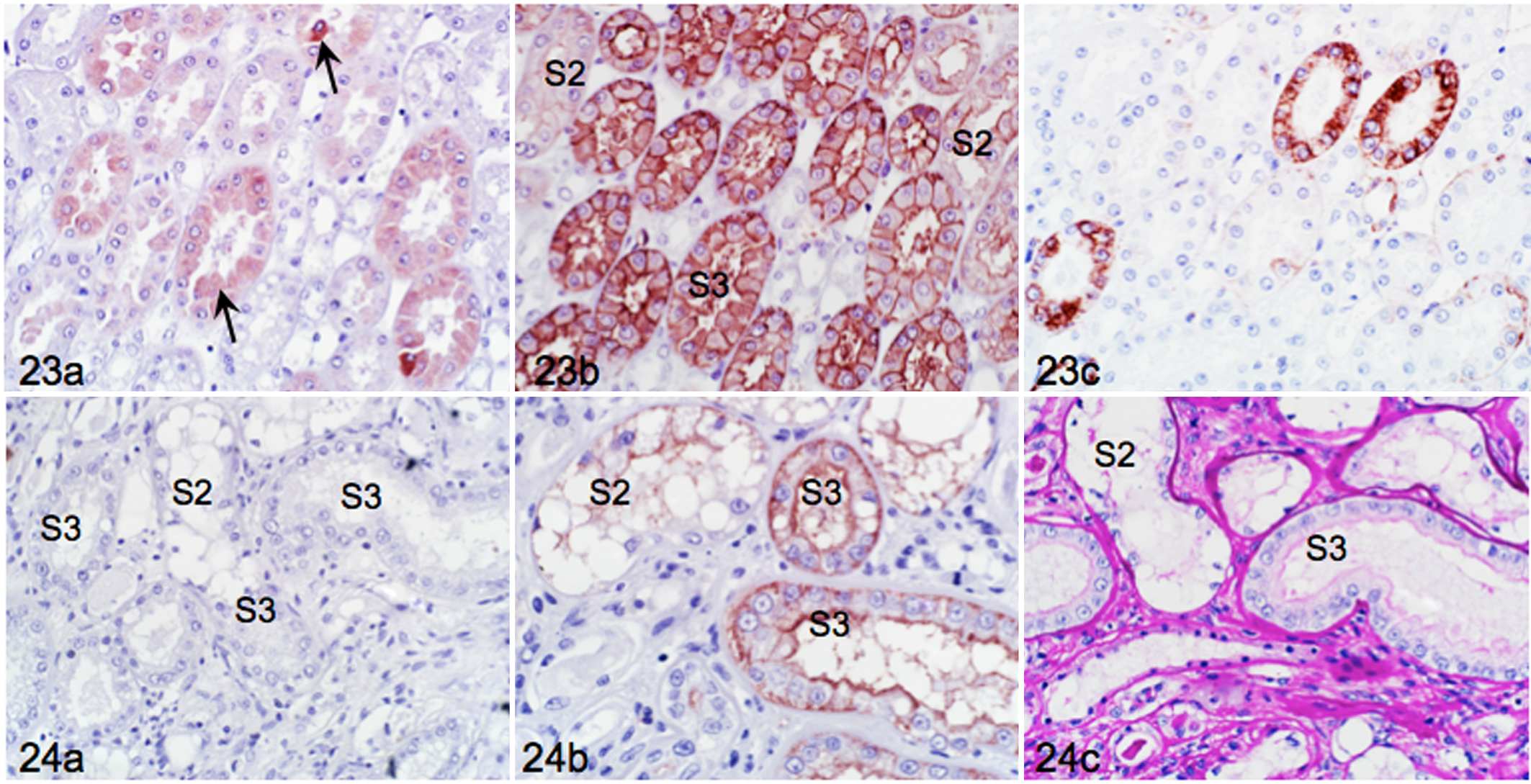
In cats with natural AKI, SCC on admission ranged from 126 to 1702 μmol/liter, and USG from 1.009 to 1.060. Affected cats had various illnesses, and the time frame from euthanasia until tissue collection was variable. KIM-1 was expressed in S3 of most but not all cats. Cat 1 had CKD and was treated with multiagent chemotherapy for transitional cell carcinoma. The cat developed AKI and died 24 hours later, presumed to be from tumor lysis syndrome. Kidney sections showed dilation of PT with extensive loss of brush borders, sloughed luminal cells, and moderate KIM-1 expression (Fig. 22a) in S3 that were positive for AQP-1 (Fig. 22b). There was no expression of VIM in S3 except for rare, very fine filaments (Fig. 22c). Glomerular sclerosis and interstitial nephritis indicated pre-existing CKD with superimposed AKI (not shown). Cat 7 had AKI concomitant with trauma and sepsis 3 days prior to euthanasia. KIM-1 was expressed in S3 that also stained strongly positive for AQP-1 in adjoining sections (Fig. 23a, b). KIM-1 positive tubules had flattened epithelium, cytoplasmic blebbing, luminal dilation, and positive cell debris in lumens. Cytoplasmic filaments identified by VIM IHC were apparent throughout most S3 with occasional strongly positive tubules (Fig. 23c), suggesting that injury was acute but not as severe as experimental I/R. Cat 9 had historically severe CKD, a recent increase in SCC, and interstitial inflammation, tubular loss, and atrophy. KIM-1 expression was absent in S3 cells (Fig. 24a) identified by intense membranous AQP-1 staining (Fig. 24b). With PAS staining, basement membranes were noted to be intact, but brush borders were attenuated and thin (Fig. 24c). Lack of KIM-1 and VIM expression in S3 cells (not shown) suggested that the increase in SCC was unlikely due to recent injury of PT.
Statistical Analysis
In cats without KD (Table 1), histopathologic and IHC assessment yielded mean cortical total injury scores and KIM-1 IHC scores of 2.5 and 0.1, respectively, and mean OSOM total injury score (TIS) and KIM-1 IHC scores of 4.3 and 1.4, respectively. Scores for total injury (P = .031) and KIM-1 IHC (P = .003) were significantly different between cortex and OSOM.
In sections from cats with experimental AKI (45 minutes I/R), scores for total injury, KIM-1, and VIM IHC were consistently higher in kidneys with I/R than in contralateral sham-operated kidneys (Table 2). Injury scores for OSOM were significantly higher than for cortex. For example, day 1 mean total injury scores were 3.8 and 12.3 in cortex and OSOM, respectively, of nonischemic kidneys (P = .008), and 16.0 and 24.5 in cortex and OSOM, respectively, of kidneys with I/R injury (P = .043) (Fig. 25a). Mean KIM-1 IHC scores in cortex and OSOM of nonischemic kidneys were 0.25 and 2.00, respectively (P = .001), and 0.38 and 3.00 in cortex and OSOM of kidneys with I/R (P = .001), respectively (Fig. 25a). Injury and KIM-1 scores in the OSOM relative to cortex were consistently and significantly higher in I/R than nonischemic sections.
Injury and Immunohistochemical Scores in Sections From Cats With Unilateral Ischemia/Reperfusion (45 minutes).
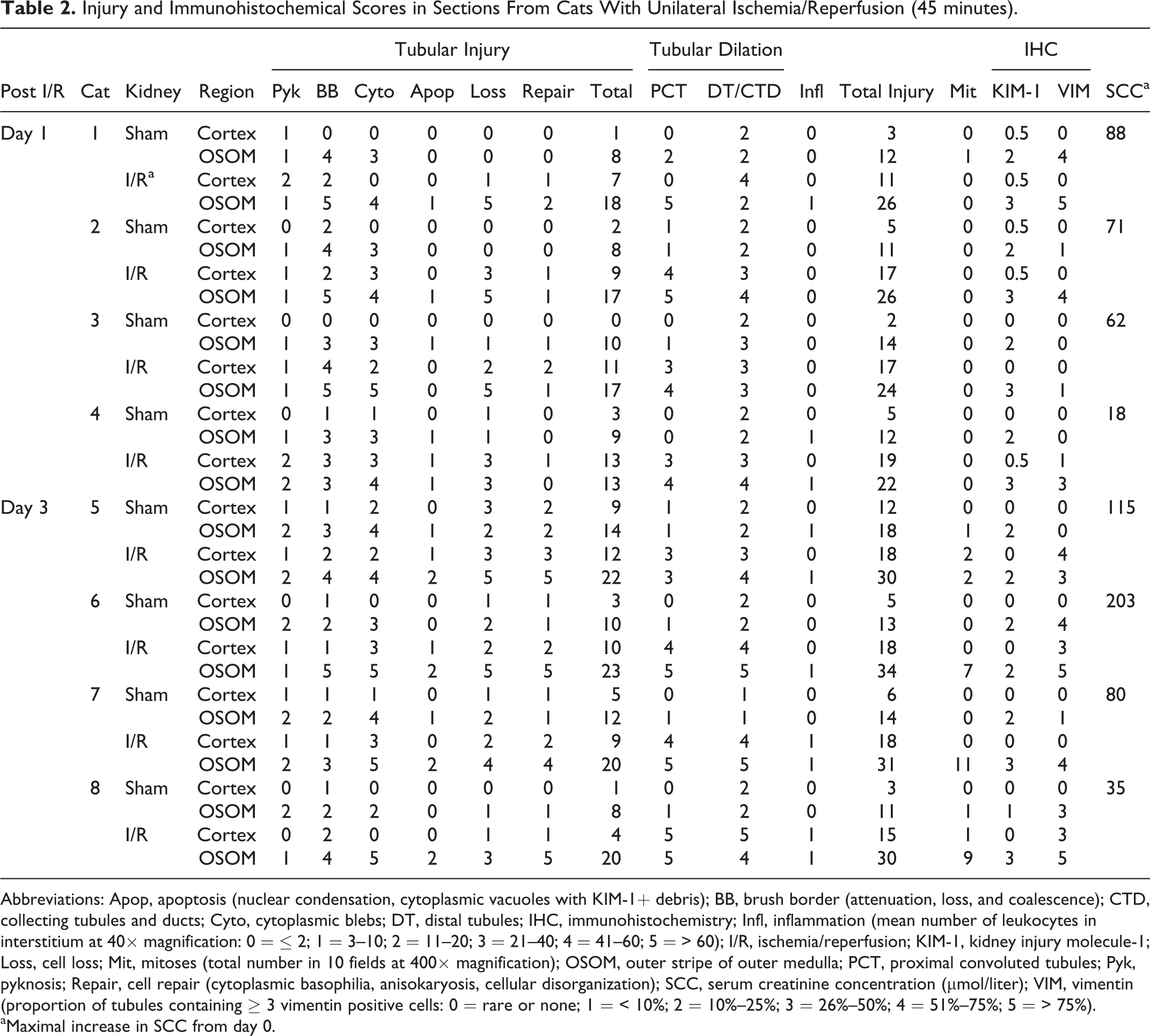
Abbreviations: Apop, apoptosis (nuclear condensation, cytoplasmic vacuoles with KIM-1+ debris); BB, brush border (attenuation, loss, and coalescence); CTD, collecting tubules and ducts; Cyto, cytoplasmic blebs; DT, distal tubules; IHC, immunohistochemistry; Infl, inflammation (mean number of leukocytes in interstitium at 40× magnification: 0 = ≤ 2; 1 = 3–10; 2 = 11–20; 3 = 21–40; 4 = 41–60; 5 = > 60); I/R, ischemia/reperfusion; KIM-1, kidney injury molecule-1; Loss, cell loss; Mit, mitoses (total number in 10 fields at 400× magnification); OSOM, outer stripe of outer medulla; PCT, proximal convoluted tubules; Pyk, pyknosis; Repair, cell repair (cytoplasmic basophilia, anisokaryosis, cellular disorganization); SCC, serum creatinine concentration (μmol/liter); VIM, vimentin (proportion of tubules containing ≥ 3 vimentin positive cells: 0 = rare or none; 1 = < 10%; 2 = 10%–25%; 3 = 26%–50%; 4 = 51%–75%; 5 = > 75%).
aMaximal increase in SCC from day 0.
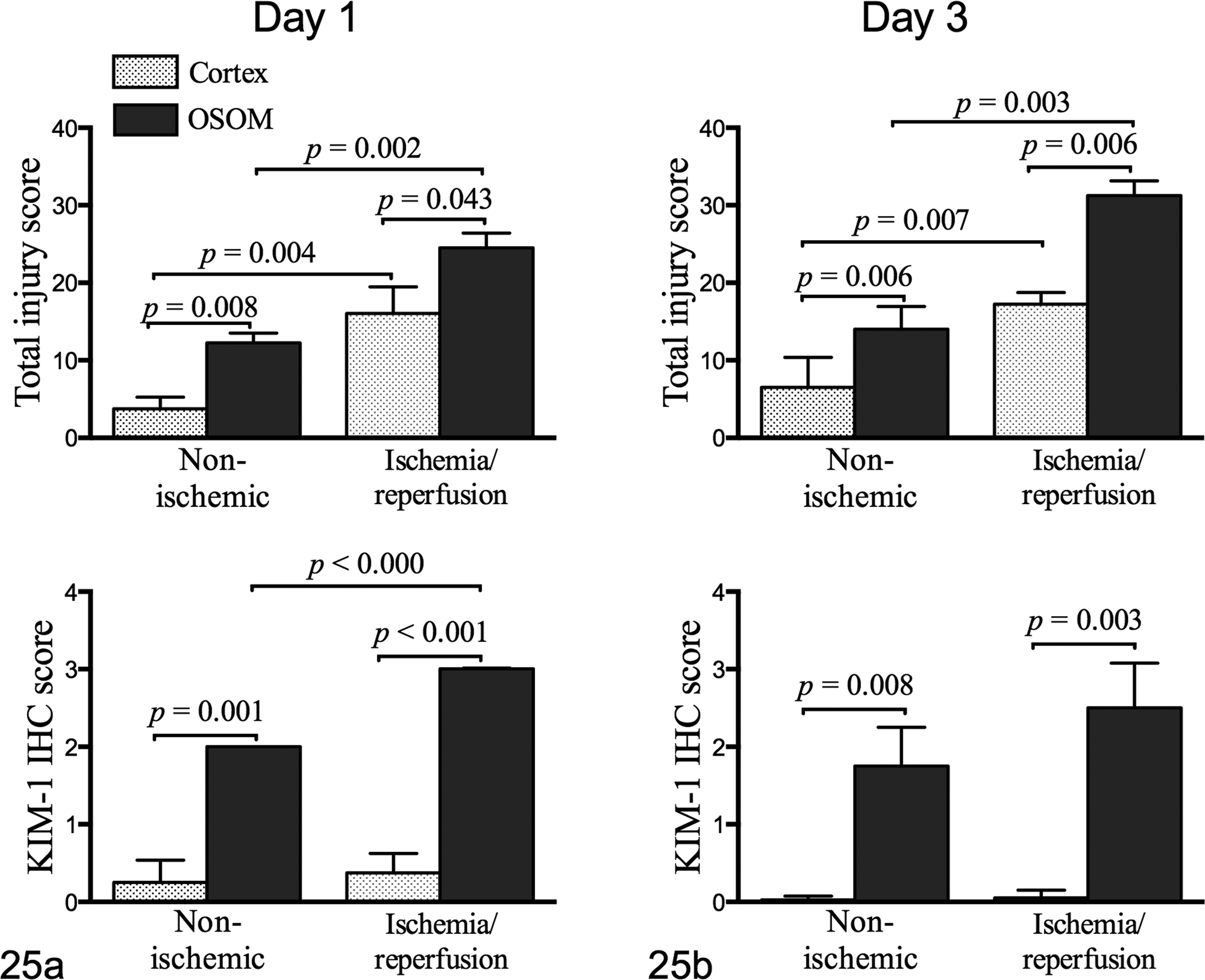
(a) Experimental acute kidney injury. On day 1, injury scores and kidney injury molecule-1 (KIM-1) staining intensity are consistently higher in outer stripe of the outer medulla (OSOM) than in cortex, and in kidneys with ischemia/reperfusion (I/R) (45 minutes) injury relative to contralateral nonischemic kidneys. (b) On day 3, significant differences between OSOM and cortex persist, and injury scores are significantly higher in kidneys with I/R (45 minutes) than in contralateral nonischemic kidneys. IHC, immunohistochemistry.
Injury and KIM-1 scores on day 3 also differed significantly between cortex and OSOM. For nonischemic sections, total injury scores were 6.5 and 14.0 in cortex and OSOM, respectively (P = .006), and for I/R sections, total injury scores were 17.3 and 31.3, respectively (P = .006). Injury scores were significantly higher in I/R than in nonischemic sections (P = .003). Mean KIM-1 scores for nonischemic cortex and OSOM were 0 and 1.75 (P = .008), respectively, and for kidneys with I/R injury, they were 0 and 2.5 (P = .003), respectively (Fig. 25b). Analysis of the change in SCC over days 0, 1, and 3 by repeated-measures 2-way ANOVA indicated a significant effect of time within individuals (P = .02), whereas USG was not significantly different. In cats that underwent 60 minutes of I/R, KIM-1 scores were significantly higher in OSOM than in cortex on days 3 and 6 (Fig. 26).
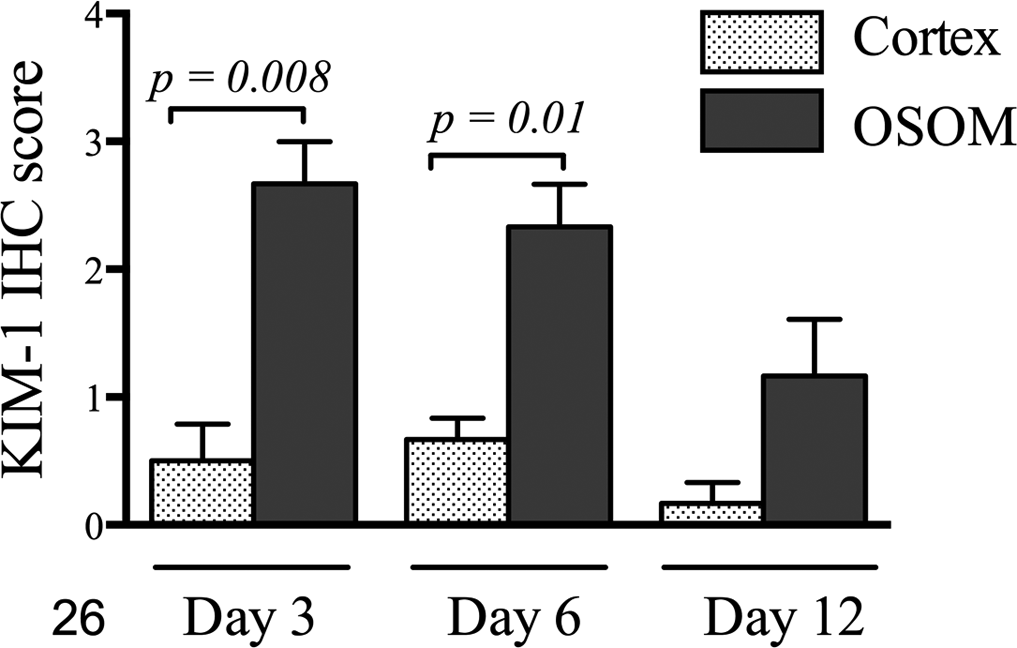
Experimental acute kidney injury, ischemia/reperfusion (60 minutes). Kidney injury molecule-1 (KIM-1) immunohistochemistry (IHC) scores were significantly higher in outer stripe of the outer medulla than cortex on days 3 and 6, and overall KIM-1 staining intensity decreased over time. OSOM, outer stripe of the outer medulla.
In cats with experimental AKI (45 minutes I/R), SCC concentration sharply increased by day 1 and then decreased to values at or above baseline by day 3 (Fig. 27). The change within individuals was significant with repeated-measures 1-way ANOVA for nonparametric data (P = .047) over 3 days. USG decreased from day 0 to day 1, and by day 3 had increased back to baseline in 3 of 4 cats (P = .273).
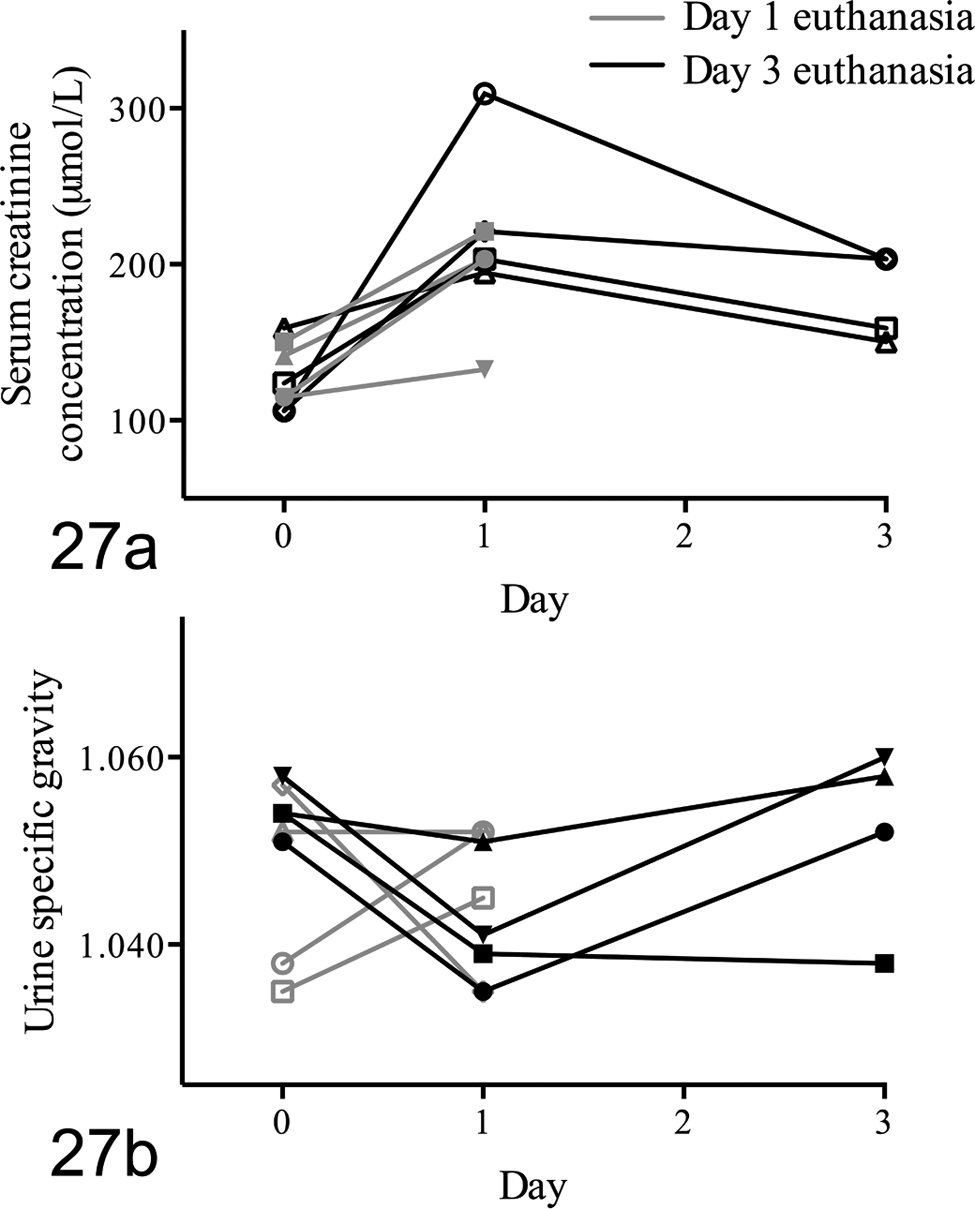
Experimental acute kidney injury, ischemia/reperfusion (I/R) (45 minutes). (a) Analyzed by repeated-measures analysis of variance, serum creatinine concentration (SCC) was increased significantly in cats euthanized 3 days after I/R (P = .047). Only results from cats with 3 SCC values were analyzed. (b) Urine specific gravity was decreased in some cats and increased in others by day 1, and analysis over 3 days did not indicate significant change.
Mean total injury scores for cats with natural AKI were 12.8 and 19.1 in the cortex and OSOM, respectively (Table 3). Total injury scores in the OSOM were significantly correlated with those in cortex (P < .01) and with SCC (P = .022, Fig. 28) but not with KIM-1 IHC scores.
Lesion and Immunohistochemical Scores in Sections From Cats With Natural Acute Kidney Injury.
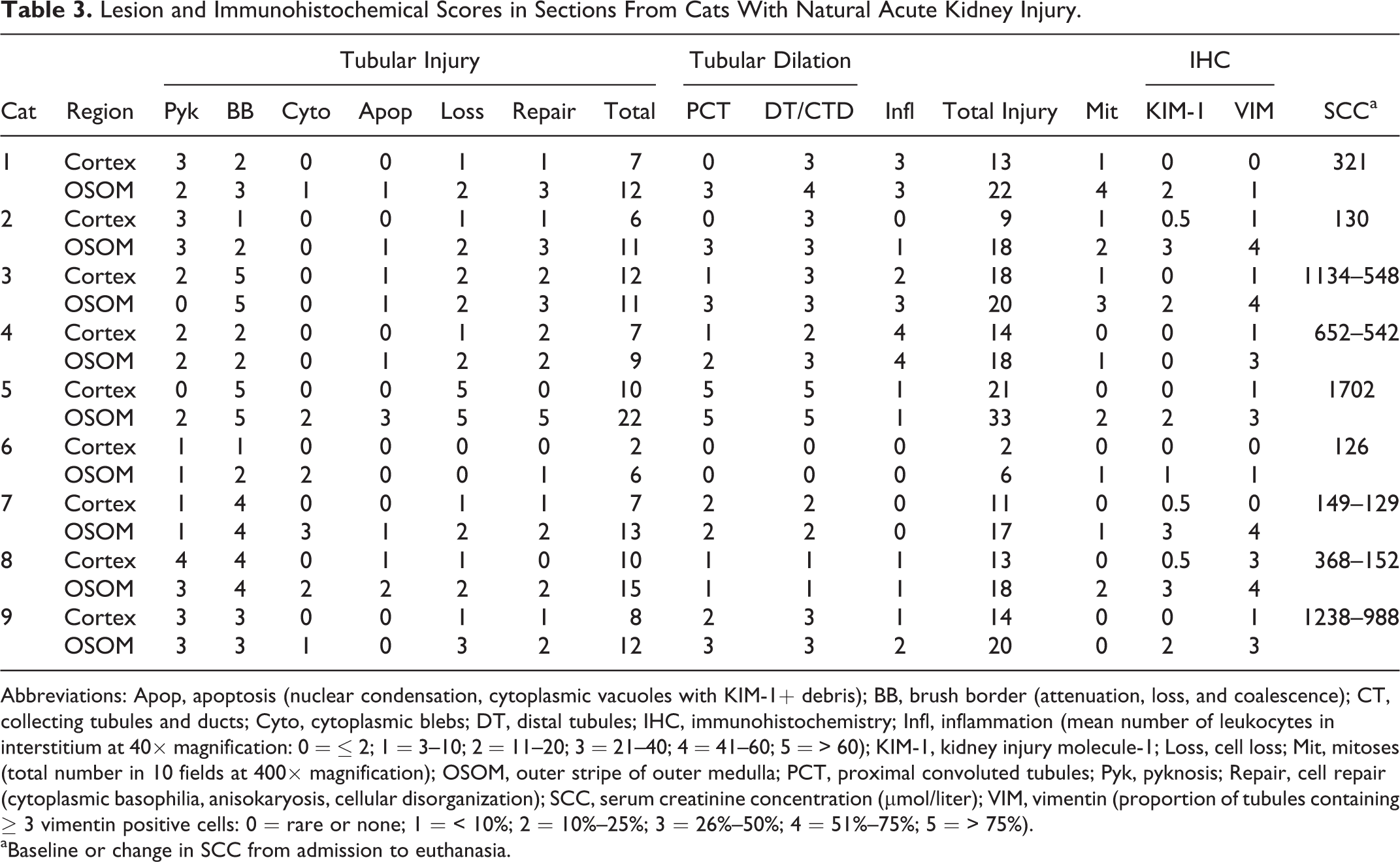
Abbreviations: Apop, apoptosis (nuclear condensation, cytoplasmic vacuoles with KIM-1+ debris); BB, brush border (attenuation, loss, and coalescence); CT, collecting tubules and ducts; Cyto, cytoplasmic blebs; DT, distal tubules; IHC, immunohistochemistry; Infl, inflammation (mean number of leukocytes in interstitium at 40× magnification: 0 = ≤ 2; 1 = 3–10; 2 = 11–20; 3 = 21–40; 4 = 41–60; 5 = > 60); KIM-1, kidney injury molecule-1; Loss, cell loss; Mit, mitoses (total number in 10 fields at 400× magnification); OSOM, outer stripe of outer medulla; PCT, proximal convoluted tubules; Pyk, pyknosis; Repair, cell repair (cytoplasmic basophilia, anisokaryosis, cellular disorganization); SCC, serum creatinine concentration (μmol/liter); VIM, vimentin (proportion of tubules containing ≥ 3 vimentin positive cells: 0 = rare or none; 1 = < 10%; 2 = 10%–25%; 3 = 26%–50%; 4 = 51%–75%; 5 = > 75%).
aBaseline or change in SCC from admission to euthanasia.
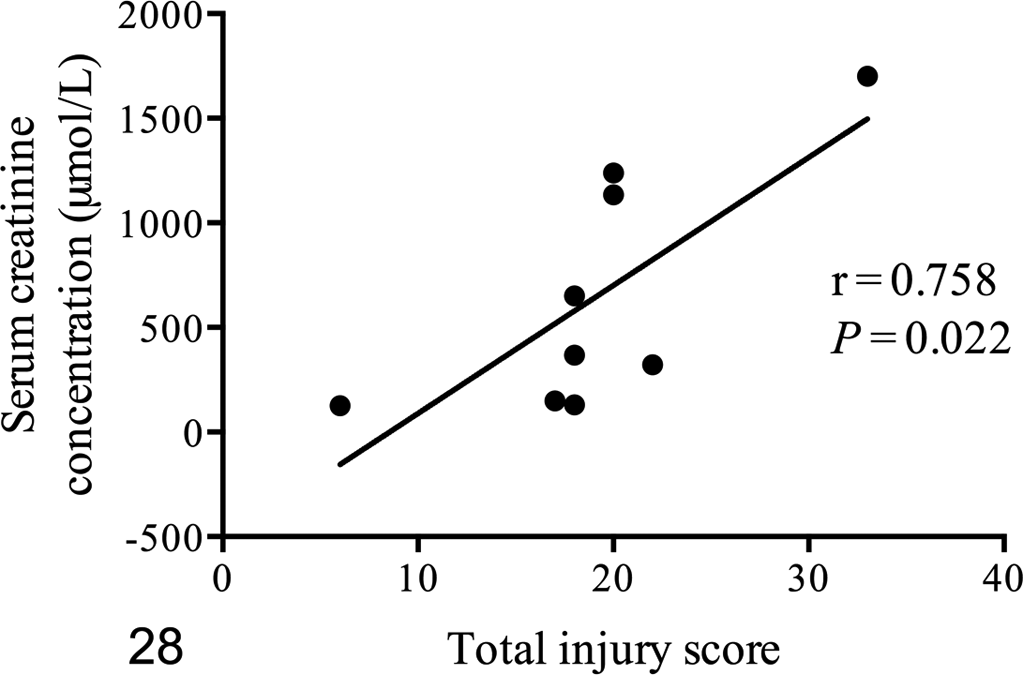
Naturally occurring acute kidney injury. Total injury scores in the outer stripe of the outer medulla of 9 cats were significantly correlated with serum creatinine concentration.
IRIS Grade for AKI
Applying IRIS criteria for grading AKI at 24 hours after I/R indicated that 7 of 8 cats met criteria for AKI (increase in SCC of ≥ 26.4 μmol/liter). 24 Four of 8 cats met criteria for AKI grade 2 (increase in SCC of ≥ 26.4 μmol/liter with SCC between 141 and 220 μmol/liter) and 3 cats met criteria for AKI grade 3 (increase in SCC of ≥ 26.4 μmol/liter with SCC between 221 and 439 μmol/liter). In cats with natural AKI, IRIS grade of AKI could not be assessed since appropriately timed SCC results were unavailable.
Discussion
In this study, kidney sections from cats without KD, with experimentally induced AKI and with AKI from naturally occurring causes, were assessed for presence and severity of injury in relation to location and expression of KIM-1.
Based on anatomic location, cellular arrangement, characteristic PAS–brush border staining, pattern of cytoplasmic vacuolation, and expression of AQP-1, 3 distinct PT segments in the feline nephron were identified. Morphologically similar PT segments exist in rats, where S3 corresponds to the straight PT portion in the OSOM, and S1 and S2 to the convoluted portion of the PT. 21,40,49,50 There are well-defined functional differences among specific segments of the PT in rats, 25 and results from this study suggest that different PT segments in cats also have unique functions and susceptibility to injury. The feline nephron was previously differentiated into convoluted (pars convoluta) and straight (pars recta) portions based on differences in intracellular lipid, alkaline phosphatase reactivity, and PAS staining. 4,41,44,45 Using expression of AQP-1 and detailed morphologic assessment, S1 and S2 were localized to the convoluted PT in the cortex, and S3 to the straight PT extending from the inner cortex to the OSOM within the medullary ray. Expression of VIM was rare in PT of nondiseased kidneys and prominent in cortical collecting ducts, consistent with findings in humans and rats. 2,64,69
Mild injury such as low-grade brush border attenuation, loss, and coalescence was noted in sections from some cats without KD and might result from reduced renal perfusion associated with sedation and anesthesia or from degeneration after euthanasia. 31 Although the time frame between sedation and fixation of kidneys was only approximately 30 minutes in cats without KD, highly metabolically active S3 cells may be as prone to rapid degeneration in cats as in rats. 51 Even mild S3 injury was associated with focal KIM-1 expression, which suggests that KIM-1 may be a very sensitive marker of injury or functional alteration.
All sections from cats with 45 minutes of I/R had marked PT injury. Injury was more readily identified with PAS than HE staining and was consistently more severe in OSOM containing most S3 than in cortex with mostly S1 and S2. Injury of S3 involved loss of brush border, membrane blebs, and single cell and whole tubule necrosis and sloughing on both days 1 and 3 after injury. On day 3, there was OSOM infiltration by inflammatory cells and mitoses among S3 cells. Persistent OSOM injury, as seen on day 3, has been attributed to inadequate blood reflow after I/R to metabolically highly active but relatively hypoxic S3 located remote to renal blood vessels. 29,70,78 Ischemia results in initiation of AKI characterized by cellular depletion of adenosine triphosphate (ATP) and activation of epithelial and endothelial production of proinflammatory chemokines and cytokines such as monocyte chemoattractant protein-1, interleukin (IL)-1, -6, -8, and -18, tumor necrosis factor-α, and transforming growth factor-β, in addition to failure of up-regulation of anti-inflammatory cytokines. 5,56,70,82,83 Damaged endothelial cells also up-regulate intercellular adhesion molecule-1, and P- and E-selectin, leading to enhanced leukocyte adhesion and activation, which in turn perpetuates inflammation and worsens hypoxia. 55 The subsequent extension phase of AKI consists of continued hypoxia in the OSOM and inflammation with accumulation of capillary and interstitial leukocytes, as seen on day 3. 12,35,56,78 In cats, S3 has a unique complement of mitochondria, peroxisomes, and endoplasmic reticulum, which may impart greater susceptibility to injury from I/R and metabolic activation, whereas S1 and S2 may be more susceptible to injury from lysosomal overload. 4
Acutely injured but intact S3 and luminal sloughed cells were strongly KIM-1 positive on days 1 and 3, suggesting KIM-1 expression prior to necrosis or apoptosis. KIM-1 positive anucleate fragments (cytoplasmic blebs) in lumens likely represent dislodged membranous/cytoplasmic fragments of S3 cells that arose from sublethal injury. In other models of AKI, sublethal I/R initially causes S3 cellular depletion of ATP, leading to alteration in the apical actin cytoskeleton and microvillar rearrangement, which then leads to the formation of membrane-bound extracellular vesicles or cytoplasmic blebs. 26,78,80,83 These blebs are shed into the lumen and subsequently lost into the urine, or phagocytosed by neighboring intact S3 cells that acquire a phagocytic phenotype with induction of KIM-1 expression. 36 Phagocytic KIM-1 positive S3 cells are believed to be important to eliminate intraluminal debris and apoptotic cells and to thereby prevent tubular obstruction. 34,82 KIM-1 expression readily identified cytoplasmic blebs adjacent to the surface of S3 cells and within membrane-bound intracellular vesicles, but these were not readily apparent with HE or PAS stain.
Nonischemic kidneys from cats with experimental AKI had injury scores slightly higher than those of cats without KD. Prolonged anesthesia during I/R and clamping of the contralateral renal artery and vein may have altered perfusion to the nonischemic kidney. In addition, inflammatory cytokine release from the I/R kidney may have caused cellular injury in contralateral kidneys. Overall higher KIM-1 expression, and presence of KIM-1 positive cytoplasmic blebs, intracellular vacuoles, and luminal debris, indicated relatively greater injury in nonischemic kidneys than in those from cats without KD. Nonischemic kidneys from cats with experimental AKI had fine VIM-positive filaments, whereas those from cats without KD did not. VIM, a cytoskeleton filament expressed in mesenchymal cells, is re-expressed in injured epithelial cells. 54 Fine VIM-positive filaments suggest early dedifferentiation or repair of cells from nonlethal injury and, more specific, may reflect repair of sodium-glucose channels and maintenance of the cytoskeleton with osmotic changes in cells sublethally injured. 14,64 On the other hand, thick VIM-positive filaments such as those seen on day 3 post I/R are thought to indicate dedifferentiation rather than repair. 15,64
Forty-five minutes of I/R resulted in moderate though individually variable increases in SCC. This finding highlights 2 aspects concerning routine interpretation of SCC: First, increase in SCC beyond the RI is insensitive of AKI. Increases in SCC beyond the RI are most often associated with irreversible KD, whereas transient increases in SCC are typically classified as prerenal azotemia and considered insignificant. However, as shown here, mild to moderate transient increases in SCC were associated with profound tissue injury. Second, SCC should be assessed over time within each individual regardless of population-based reference intervals. 3,61,67,80,81 Recently updated IRIS guidelines emphasize the importance of interpreting SCC in relation to muscle mass and that even small increases in SCC may indicate AKI. 24 Furthermore, the often-held contention that AKI is associated with reduced ability to concentrate urine was unsupported, since there was no significant decrease in USG.
The injury associated with 60 minutes of I/R was previously reported and was similar to but more severe than that of 45 minutes of I/R. 65 Evaluation of KIM-1 expression in day 3, 6, and 12 samples from that study indicated differences in intensity and cell distribution over time. Expression on days 6 and 12 was less intense than on day 3 despite ongoing injury, which may be because injured cells were sloughed and lost in urine, there was epithelial repair with KIM-1 negative cells, or maximal expression of KIM-1 may occur only for a few days after injury. Viable cells may continue to express KIM-1 at low levels if injury is ongoing or there is maladaptive repair, which was suggested by worsening inflammation and increasing VIM expression between days 3 and 12. It is most likely that KIM-1 expression is maximal between days 1 and 3 after a single injury. In rodents, KIM-1 is most highly excreted in urine at the time of AKI prior to increase in SCC, suggesting that KIM-1 tubular expression is induced immediately during injury and decreases thereafter. 10,38,73 KIM-1 and VIM were expressed in the same injured and regenerating S3 of all cats with AKI. VIM persisted in injured S3, although there was only faint KIM-1 expression, indicating either that tubular injury persisted or that tubular repair was incomplete by day 12. Heterogeneous VIM staining of individual cells that also expressed KIM-1 was interpreted to reflect a variable degree of injury perpetuated from initial ischemia through reperfusion, influx of inflammatory cells, and repair processes. These findings are consistent with observations in rats where VIM and KIM-1 were coexpressed in the dedifferentiated cells undergoing regeneration after ischemia. 37
Despite lack of KIM-1 expression in cortex on days 1 and 3 after I/R, KIM-1 was faintly expressed in S1 and S2 cortical biopsies immediately after onset of ischemia, intensely after 1 hour of ischemia, and less intensely 1 hour after reperfusion. Rats with unilateral I/R expressed KIM-1 immunoreactivity only in the regenerating S3 segments at 48 hours postinjury. 37 Temporal KIM-1 expression in different PT segments likely relates to differences in the susceptibility to injury and the rapidity of repair. In S1/S2, I/R causes rapid brush border loss, progressive chromatin clumping, cell swelling, and cell loss, but within 4 hours of reperfusion, there is brush border recycling, cell restoration, and repair. 33,77,79 In S3, I/R induces progressive cell injury and death followed by cell sloughing and eventual repair. 29,78 These functional and morphologic differences between PT segments underscore the need to focus histopathologic evaluation of ischemic kidney injury on S3.
Cats with natural AKI also had more extensive injury in OSOM than cortex, but pre-existing CKD at times rendered segment identification challenging with HE. Fidelity and pattern of AQP-1 expression and PAS staining were helpful to identify S3 and then allowed assessment of KIM-1 expression. Injuries in cortex and OSOM were significantly correlated, suggesting that the type and duration of insults leading to AKI were heterogeneous. In this group, injury score correlated significantly with SCC, likely because most cats had conditions resulting in kidney injury over several weeks prior to death, and some were deemed to have irreversible disease due to lack of treatment response. Expression of KIM-1 did not significantly correlate with total injury, likely because injury was less acute and more variable than in cats with I/R. Persistent low-grade regeneration and repair in cats with CKD may also affect KIM-1 expression. This I/R model of AKI results in what is considered acute tubular necrosis in other models, whereas AKI associated with conditions such as sepsis causes relatively little necrosis. 26
There are limitations to this study. First, the number of animals per group was small. However, findings were very similar within groups of I/R, suggesting that additional animals may not have revealed different outcomes. Second, the experimental study was time limited. This limitation did not allow assessment of KIM-1 expression over the time frame when AKI might progress to CKD. Longer term studies are needed to assess the risk for development of CKD, but there is evidence of progression of AKI to CKD in people. The degree of tubular injury and inflammation in cats with experimental AKI characterized here, and reported previously, 65 suggests that AKI may also progress to CKD in cats. Third, only PT were examined systematically due to the focus of this study on KIM-1. The TAL also had features of injury, albeit not to the same extent as S3. Fourth, this model of I/R induces extensive necrosis, whereas less severe injury due to sepsis or toxins with components of necrosis, apoptosis, necroptosis, and functional tubular changes may result in different KIM-1 expression. Finally, natural AKI results from various causes, is of variable duration, and is affected by pre-existing CKD and different therapies, which limits the applicability of experimental results.
Kidney disease is extremely common in aged cats and is likely a consequence of single or repeated AKI. Available assays to detect AKI are of limited sensitivity. KIM-1 is highly yet transiently expressed in the tubular segments most susceptible to injury. Further validation of expression with clinically relevant causes of injury and development of assays for detection may be warranted.
Footnotes
Authors’ Note
This article was presented in part as abstracts at the annual meeting of the American College of Veterinary Pathologists in Montreal (2013) and Atlanta (2014).
Acknowledgements
We sincerely appreciate the technical expertise and subject matter enthusiasm of Susan Lapos and Maureen Harron, Histopathology, Animal Health Laboratory, University of Guelph.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Pet Trust Foundation at the University of Guelph and the Natural Sciences and Engineering Research Council of Canada.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
