Abstract
Posttransplant lymphoproliferative disorders (PTLDs) are a heterogeneous group of lymphoid proliferations that occur in the setting of depressed T-cell function due to immunosuppressive therapy used following solid organ transplantation, hematopoietic stem cell transplantation, and also xenotransplantation. In the present study, 28 immunosuppressed parkinsonian
Keywords
Posttransplant lymphoproliferative disorders (PTLDs) are a heterogeneous group of lymphoproliferative diseases that arise after transplantation of solid organ and other tissues. 9 They comprise a wide spectrum of clinical and pathological manifestations ranging from a benign self-limited form of lymphoproliferation to an aggressive and disseminated lymphoma. 2 PTLD incidence is as high as 10% in solid organ transplantation 4 and may occur at any time after transplantation, but the risk appears to be greatest within the first year posttransplantation. 19 PTLD World Health Organization (WHO) classification recognizes 3 main pathologic subsets of the lymphoproliferative lesions: early, polymorphic, and monomorphic lesions. 21 Early lesions, more frequently involving tonsils, adenoids, or lymph nodes than extranodal sites, include 2 morphological types: plasma cell hyperplasia and infectious mononucleosis-like lesions. Polymorphic PTLDs affect nodal and extranodal tissues and show loss of tissue architecture and necrosis. Polymorphic PTLDs are composed of a mixed population of immunoblasts, plasma cells, intermediate-sized lymphoid cells (incorporating a full range of B-cell morphology and differentiation), and occasional Hodgkin Reed Sternberg–like cells. Monomorphic PTLDs can be of B-cell or T-cell lineage, disturb the tissue architecture, and spread to other organs. They are classified according to the WHO classification of lymphomas in immunocompetent patients, showing features of different morphologic variants of diffuse large B-cell lymphoma (DLBCL), Burkitt lymphoma (BL), or plasmablastic lymphoma (PL). 21
Approximately 50% to 80% of PTLD biopsy specimens are positive for human herpesvirus 4 or Epstein-Barr virus (EBV) within the tumor cells, reflecting the critical role of reduced cellular immunosurveillance against EBV in its pathogenesis. 18
EBV-related herpesvirus, or lymphocryptovirus (LCV), is similarly associated with lymphomas in immunosuppressed nonhuman primates (NHPs). 22 In common with human EBV infection, primate LCV is endemic in Old World macaques, both those born in the wild and those raised in captivity, and resides latently in B cells in most adult animals. 16 Likewise, porcine lymphotropic herpesviruses (PLHVs) are widespread among domestic pig populations 5 and have been associated with PTLD in pigs receiving allogeneic hematopoietic cell transplants. 11 Although the zoonotic potential of PLHVs is unknown, PLHV-related PTLD should be considered a risk in recipients of porcine-derived xenografts. This small risk should be diminished by the use of specific pathogen-free housed donor animals. 10
Several reports have suggested that the use of cyclosporine in NHPs, either alone or in combination with conventional immunosuppressive therapy, might be associated with an increased risk of lymphoma. 16 Malignant lymphomas were detected in 2 of 5 nontransplanted cynomolgus monkeys receiving cyclosporine and in simian immunodeficiency virus (SIV)–immunosuppressed cynomolgus and rhesus monkeys. 13 These lymphomas were mostly extranodal, aggressive and in all cases associated with LCV. 16 Endogenous LCV is capable of producing PTLD in immunosuppressed macaques that are recipients of kidney allografts with a frequency of 9 of 160 (5.6%). 20 Similarly, in a kidney xenotransplantation from pig to cynomolgus monkey and in kidney allotransplantation, LCV-related PTLD developed in 10 of 245 xenotransplanted (pig to cynomolgus monkey) (4%) and 9 of 231 (3.8%) allotransplanted primates. 16 The prevalence of PTLD is probably determined by the intensity of immunosuppression and not by the regimen used. The highest frequency of PTLD occurred in those animals that were on an aggressive immunosuppressive protocol, 16 and others have reported that there was no clear relationship between the use of a particular drug or drug combination in maintenance of immunosuppression and the occurrence of PTLD. 16 Furthermore, a high-dose regimen of cyclophosphamide and rapamycin led to a high incidence of PTLD in a life-supporting functional kidney and liver baboon-to-monkey grafts. 23 In a nonhuman primate model of facial composite tissue allotransplantation, a continuous infusion of tacrolimus provided prolonged rejection-free survival of the grafts. This was associated with a high frequency of donor-derived PTLD, possibly due to a large volume of transplanted vascularized bone marrow. 3 In summary, in humans, the pathogenesis of PTLDs seems to be multifactorial: aside from the accumulation of genetic alterations of oncogenes and tumor suppressor genes, the role of EBV, among the possible infective causes, seems to be central in driving the proliferation of EBV-infected B cells and subsequent development of the lymphoproliferative disorder in the setting of a prolonged and profound immunosuppression that decreases the pool of specific cytotoxic T lymphocytes. 7 –21
In this study, we characterize PTLD occurring in the setting of neural precursors xenotransplantation to treat a pharmacologically induced form of Parkinson disease (PD), 1 with respect to tissue localization, histopathology, immunophenotype, and molecular viral transcript expression of primate LCV.
Materials and Methods
Animals
Twenty-eight (20 females and 8 males), purpose-bred, 6- to 12-year-old cynomolgus monkeys (
Experimental Xenotransplant Model
To obtain a PD-like syndrome due to a progressive degeneration of the nigrostriatal dopaminergic system, all monkeys were treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) (Sigma, St Louis, MO) by systemic administration, as described.
1
Once stable PD lesions were obtained, 24 monkeys were unilaterally injected in the left putamen with wild-type (
Pathological Study
At the end of each experimental study or on the basis of their clinical condition, monkeys were euthanized and perfused transcardially with normal saline followed by 4% paraformaldehyde (Sigma). The brains were removed and postfixed for approximately 18 hours in 4% paraformaldehyde and then cryopreserved by immersion in a graded (10%, 15%, 20%) sucrose/0.1 M phosphate-buffered saline (pH 7.2) solution. Soon after euthanasia, collection of blood by aorta cannulation, cerebrospinal fluid by lumbar puncture, and bone marrow from femur was also performed. The primates underwent a complete postmortem examination by a veterinary pathologist (L.C.) familiar with necropsies of transplanted primates, with pictures of every organ and lesion, and samples of all tissues were formalin-fixed as well as cryopreserved at –80°C. Paraffin-embedded, 3- to 4-μm-thick sections were stained with hematoxylin and eosin and observed with a light microscope in a blinded manner by 2 independent pathologists (L.C. and S.W.).
Additional investigations on neoplastic tissues from 9 of 28 primates included immunohistochemistry (IHC) using monoclonal antibodies with the following specificities: CD3 (dilution 1:100, clone PC3/188A; DAKO, Milan Italy), CD5 (dilution 1:50, clone 54/F6; DAKO), CD20 (dilution 1:50, clone L-26; BioGenex, Menarini, Florence, Italy), CD79αcy (dilution 1:50, clone HM57; DAKO), a noncommercial antibody specific for IE1 protein of rhesus cytomegalovirus (CMV) (courtesy of Prof. Peter Barry, California Medical Research Center, dilution 1:3000 and antigen retrieval at 95°C for 60 minutes), pancytokeratin (dilution 1:100, clone AE1/AE3 M3515; DAKO), and vimentin (dilution 1:100, clone V9; DAKO). Sections (4 μm) were mounted on Superfrost Plus microscope slides (Menzel GmbH, Braunschweig, Germany) and dried at 37°C for 30 minutes. All samples were tested with an automated immunostainer (Benchmark; Ventana Medical Systems, Tucson, AZ), which included dewaxing and rehydration, antigen retrieval, primary antibody incubation, antigen detection with an ultraView Universal diaminobenzidine (DAB) kit (Ventana Medical Systems), and counterstaining with Mayer’s hematoxylin. Finally, slides were manually dehydrated through a graded series of alcohols and mounted (Eukitt mounting medium; Eukitt, Fort Washington, PA). Primary antibody dilutions were performed using a commercial antibody diluent (Ventana Medical Systems).
For double-labeling IHC, formalin-fixed tissues were sectioned at 5 μm. Slides were deparaffinized, rehydrated, and then blocked for peroxidase. Antigen retrieval was done in a microwave by boiling in citrate buffer (pH 6.0) and cooling for 20 minutes at room temperature. Slides were then blocked sequentially for biotin and DAKO protein block. The slides were incubated with either CD20 (M0755; Dako, Carpinteria, CA) or CD3 (Pan T cell Pc #A0452; Dako) for 60 or 30 minutes at room temperature, respectively, followed by biotinylated secondary for 30 minutes and the Vector ABC Alkaline Phosphatase (Vector Labs, Burlingame, CA). Slides were incubated with the liquid permanent red for the cellular marker detection. Slides were then blocked sequentially for biotin, Mouse IgG Blocking Reagent (Vector Labs), Universal Block (KPL, Gaithersburg, MD), and DAKO protein block. Slides were then incubated overnight at 4°C with a monoclonal Epstein-Barr nuclear antigen 2 (EBNA-2) antibody (NCL-EBV-PE2,;Novocastra, Leica, Buffalo Grove, IL) followed by the biotinylated secondary antibody and Vector ABC peroxidase (Vector Labs). EBNA-2 detection was performed with chromogen DAB (Dako). Slides were finally counterstained with Mayer’s hematoxylin and coverslipped.
Scoring for Ki-67 IHC
Immunohistochemistry slides for Ki-67 were evaluated by a pathologist and scored on a 1+ to 4+ scale based on percent positivity assessed in 5 high-power fields (1 = <25%; 2 = 25%–50%; 3 = 51%–75%; 4 = >75%).
Molecular Study
RNA extraction
Total RNA was extracted from PTLD and lymph node specimens using an RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s specifications. The quality of the RNA was checked by gel electrophoresis on a 1% agarose gel containing SYBR Safe DNA Gel stain 10,000× (Invitrogen, Carlsbad, CA). RNA concentration was determined using a UV-Vis spectrophotometer, NanoDrop ND-1000 (NanoDrop Technologies, Wilmington, DE).
Amplification of EBNA-1, EBNA-2, and latent membrane protein 1 complementary DNAs
A total of 300 ng total RNA was reverse transcribed using a reverse transcriptase (Superscript II; Invitrogen) with random hexamers to obtain first-strand complementary DNA (cDNA). The cDNA was then used as a template for the subsequent polymerase chain reaction (PCR). Cycling conditions were as follows: initial incubation at 94°C for 2 minutes followed by 45 cycles at 94°C for 30 seconds, annealing temperature for 40 seconds, and 72°C for 40 seconds. A final extension step at 72°C for 5 minutes was added at the end of the last cycle. For each primer pair, annealing temperature was set according to the predicted melting temperature. One-fifth of each PCR reaction was electrophoresed on a 1.8% agarose gel, stained with SYBR Safe DNA Gel stain, and visualized under UV light. Primers for amplification of Epstein-Barr nuclear antigen 1 (EBNA-1) and latent membrane protein 1 (LMP-1) transcripts were designed according to cynomolgus LCV sequences already stored on the GenBank database (www.ncbi.nlm.nih.gov) under accession numbers AB037276 and AAD56946, respectively (Suppl. Table S1). A first EBNA-2 fragment was amplified using degenerate primers designed on the basis of the multiple alignment of publicly available nucleotide sequences of 3 primates LCV transcripts: macacine EBNA-2 (
PLHV testing
DNA was prepared from porcine peripheral blood mononuclear cells (PBMCs) using the Qiagen DNeasy kit (Qiagen, Crawley, UK) according to the manufacturer’s instructions. Isolated DNA was then screened for the presence of PLHV by nested PCR using primer sets previously described. 8 All reactions were carried out using Core Taq PCR reagents (Qiagen).
Macaque lymphocryptovirus screening
Viral nucleic acid was isolated from the macaque sera using the PureLink viral RNA/DNA extraction kit (Life Technologies, Paisley, UK). Viral DNA preparations were screened for the presence of macaque lymphocryptovirus (macLCV) DNA by quantitative PCR (PCR), using primers designed to correspond to the DPOL gene of macLCV (macLCVqPCR-F, 5′-GGACGTGCACTACAAGGAGA-3′; macLCVqPCR-R, 5′- GAATCYTGCACGCAGTACAT-3′; and probe macLCVqPCR-P, 5′-6FAM-TCGCCTCTTTGCAGAGGGCC-TAMRA-3′) and Taqman Gene Expression Master Mix reagents (Life Technologies). Amplification was achieved with 40 cycles of 95°C for 15 seconds, 53°C for 15 seconds, and 60°C for 30 seconds and carried out alongside known copy number controls to allow quantification of each sample and to test reaction efficiency. All samples were spiked with an Internal Positive Control (Life Technologies) to test for the presence of inhibitory factors. The assay’s limit of detection was 10 virus particles per milliliter of serum.
Results
Pathological Study
Nine of 28 immunosuppressed macaques developed masses compatible with PTLD. Seven were recipients of neuronal precursors from CTLA4-Ig transgenic pig embryos, 1 was from wild-type pig embryos, and 1 received only MPTP treatment and immunosuppression. Clinical signs that led to euthanasia were related to the presence of a nasal mass (
Macroscopically, nasal masses had different levels of severity, tissue infiltration, and size. The lesions included diffuse bilateral thickening of nasal mucosa of both sides (
Microscopically, the masses were consistent with large-cell lymphomas composed of a diffuse atypical population of round cells, measuring up to 20 μm, with scant to moderate eosinophilic cytoplasm, lacking distinct cell boundaries, located within the subepithelial connective tissue and occasionally in the epithelium of the nasal cavity, intestinal tract, and many visceral lymph nodes. Occasionally, the thoracic and visceral masses were severely effaced by the atypical population, and a possible lymph nodal origin was considered. The cells were organized in broad sheets intermingled with a moderate fibrovascular stroma. The nuclei, 10 to 15 μm in diameter, were central, round to ovoid, and frequently cleaved with finely granular chromatin and 1 prominent nucleolus. There were moderate anisokaryosis and anisocytosis. The mitoses were 5 to 20 in 10 high-power fields (Figs. 1, 2), and scattered apoptotic cells were present. In the nasal masses (3 of 3 cases), the atypical population was associated with massive necrosis and destruction of the nasal bone. Scattered around the periphery of the masses were moderate, multifocal lymphoplasmacytic aggregates. As for the intestinal masses, in association with the atypical cells, focally extensive areas of necrosis and a peripheral rim of lymphocytes were present. A diffuse atypical population with the same features of other locations characterized the nodal masses. Based on cellular morphology and the rather homogeneous characteristics of all the nodal masses and the nasal and intestinal lesions, the diagnosis in the 9 affected primates was of monomorphic PTLD according to the patterns described in the WHO classification. 21
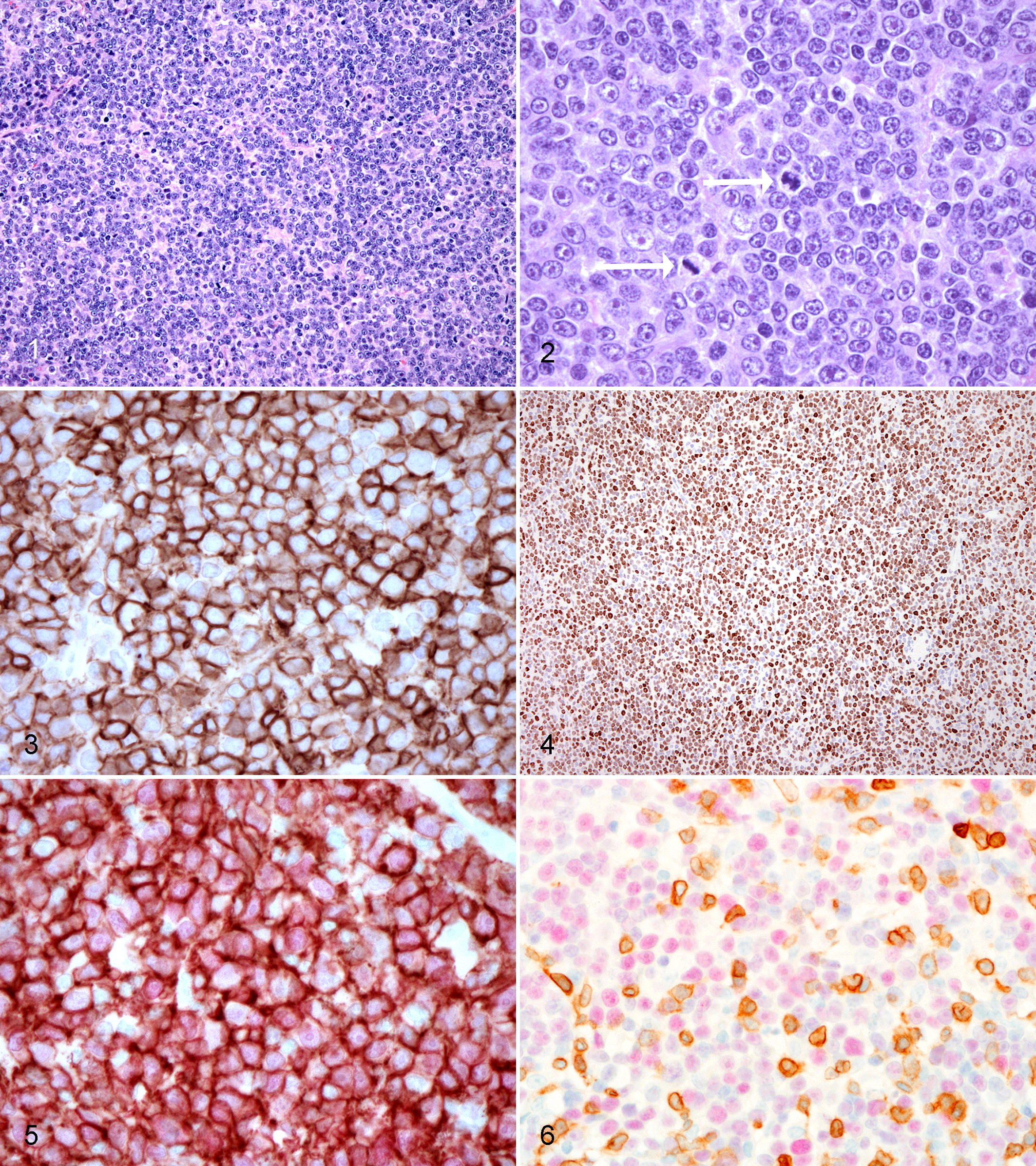
IHC revealed a uniform pattern of marker expression in all PTLD tumors examined. Neoplastic cells were consistent with B cells, diffusely labeling CD20 (Fig. 3) and CD79, and for vimentin intermediate filaments (data not shown for CD79 and vimentin). Neoplastic cells were negative for cytokeratins, CMV, CD3, and CD5 markers; however, CD3+ and CD5+ lymphocytes were scattered at the periphery of most tumors, occasionally infiltrating the neoplastic cells (data not shown for CD3 and CD5). Cellular proliferation, evidenced by expression of Ki-67, was high with greater than 50% positivity of the cells (based on at least 5 high-power fields) for most PTLD tumors. Tumors from multiple sites in the same animal exhibited similar proliferation rates. A nasal cavity lymphoma in 1 primate had a low proliferation rate with <25% of cells Ki-67+ (based on at least 5-high power fields). EBNA-2 labeling revealed a diffuse nuclear positivity of the tumors (Fig. 4).
Double-labeled IHC for CD20 and EBNA-2 confirmed LCV infection of CD20+ neoplastic cells of B-cell origin (Fig. 5). In 1 case, roughly 90% of EBNA-2+ neoplastic cells were CD20+ B cells, while the remaining 10% were either B cells with downregulated CD20 or were not B cells. In the same case, in another section, only 50% of the EBNA-2+ cells were positive for CD20, indicating a population of CD20+ cells that were not LCV infected. In another case, only 50% of the EBNA-2+ cells were positive for CD20, while 80% of CD20+ B cells were positive for EBNA-2. Double-labeling with CD3 and EBNA-2 in this case failed to demonstrate that additional EBNA-2+ cells were CD3 lymphocytes (Fig. 6).
Molecular Study
A preliminary survey on EBNA-1, EBNA-2, and LMP-1 gene expression was carried out using end-point real time (RT)-PCR on neoplastic cells from 5 different PTLDs and 2 hyperplastic lymph nodes, confirmed at histology and immunohistochemistry and taken from PTLD-affected primates. Gene-specific primers were used for both EBNA-1 and LMP-1 amplification and 1 primer pair for EBNA-1, while 4 different primer combinations were employed for LMP-1. EBNA-1 expression was demonstrated in all PTLD specimens by the amplification of a region of 453 bp showing a 98.6% identity with reference cynomolgus sequences in GenBank, while no expression was found in controls. Noticeably, 4 different RT-PCR assays failed to detect LMP-1 transcript in any of the specimens (Fig. 7). Degenerate primers designed on the multiple alignments of 3 primate EBNA-2 sequences allowed isolation of the cynomolgus LCV EBNA-2 transcript. The sequencing of two overlapping regions led to the assembly of an 832-bp sequence (GenBank accession number JQ845949). Sequence analysis revealed a 76% identity at the protein level with macacine herpesvirus 4 EBNA-2 (AAK95414) but considerably lower homology with human and papiine counterparts, showing 49% and 33% identity, respectively. The cynomolgus LCV EBNA-2 transcript in the present study showed a 99.3% identity at the nucleotide level with the complete genome of a lymphocryptovirus isolated from a cynomolgus monkey (NCBI accession number KP676001.1). EBNA-1 and EBNA-2 transcripts were detected in all PTLD specimens, revealing viable virus in neoplastic cells, while no expression was observed in hyperplastic lymph nodes (Fig. 7).
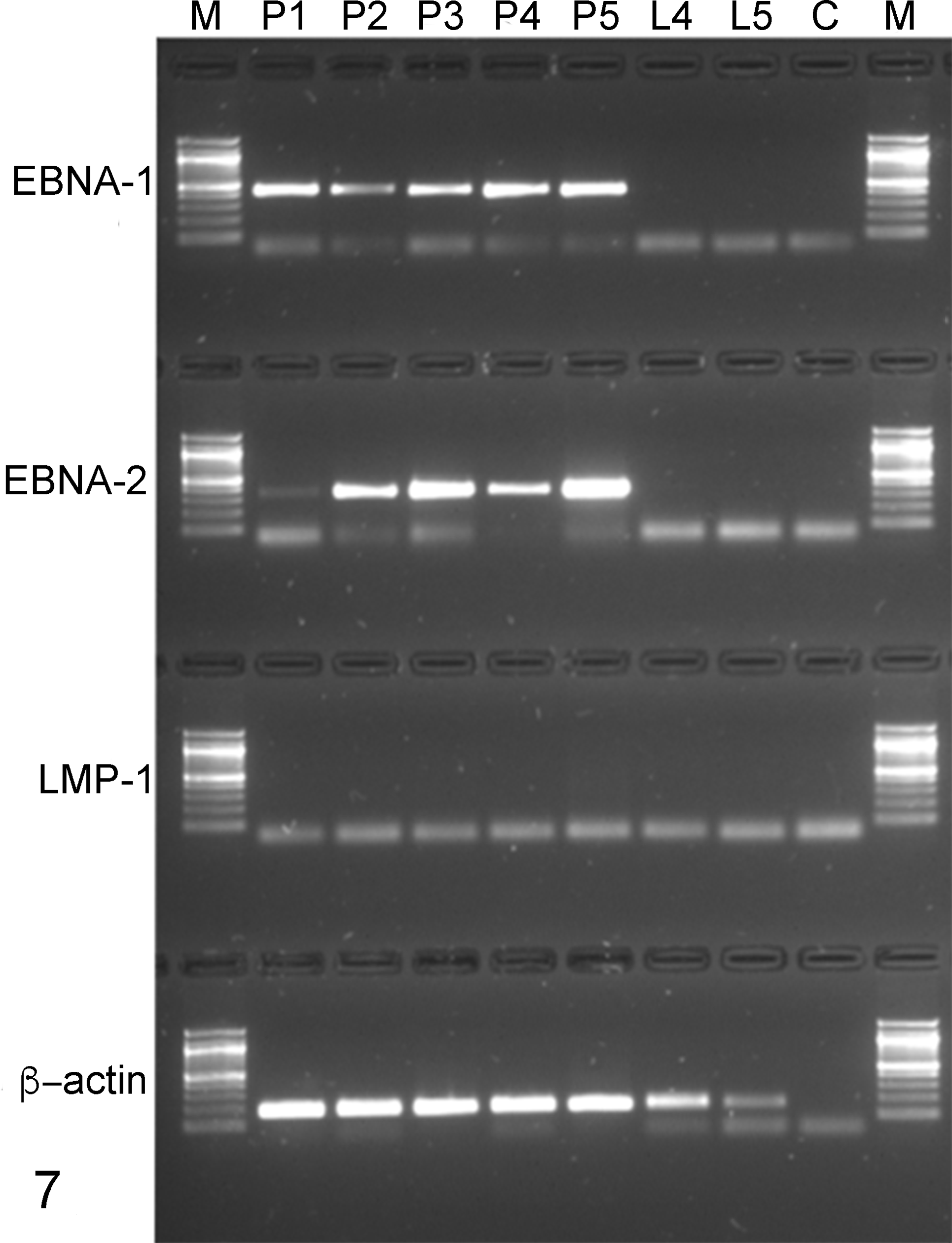
Real time–polymerase chain reaction analysis of cynomolgus Epstein-Barr nuclear antigen 1 (EBNA-1), Epstein-Barr nuclear antigen 2 (EBNA-2), and latent membrane protein 1 (LMP-1) messenger RNA expression. Samples P1, P2, P3, P4, and P5 are posttransplant lymphoproliferative disorder (PTLD)–positive primates; samples L4 and L5 are hyperplastic lymph nodes from P4 and P5 PTLD+ primates. M, molecular marker; C, no template control. EBNA-1 and EBNA-2 transcripts are detected in all PTLD specimens, while no expression is observed in hyperplastic lymph nodes.
DNA were isolated from PBMCs from the recipients and from the donor pigs. The DNA was screened by PCR for the presence of the porcine homologue to EBV, porcine lymphotropic herpesvirus (PLHV). PLHV was detected in 44% (
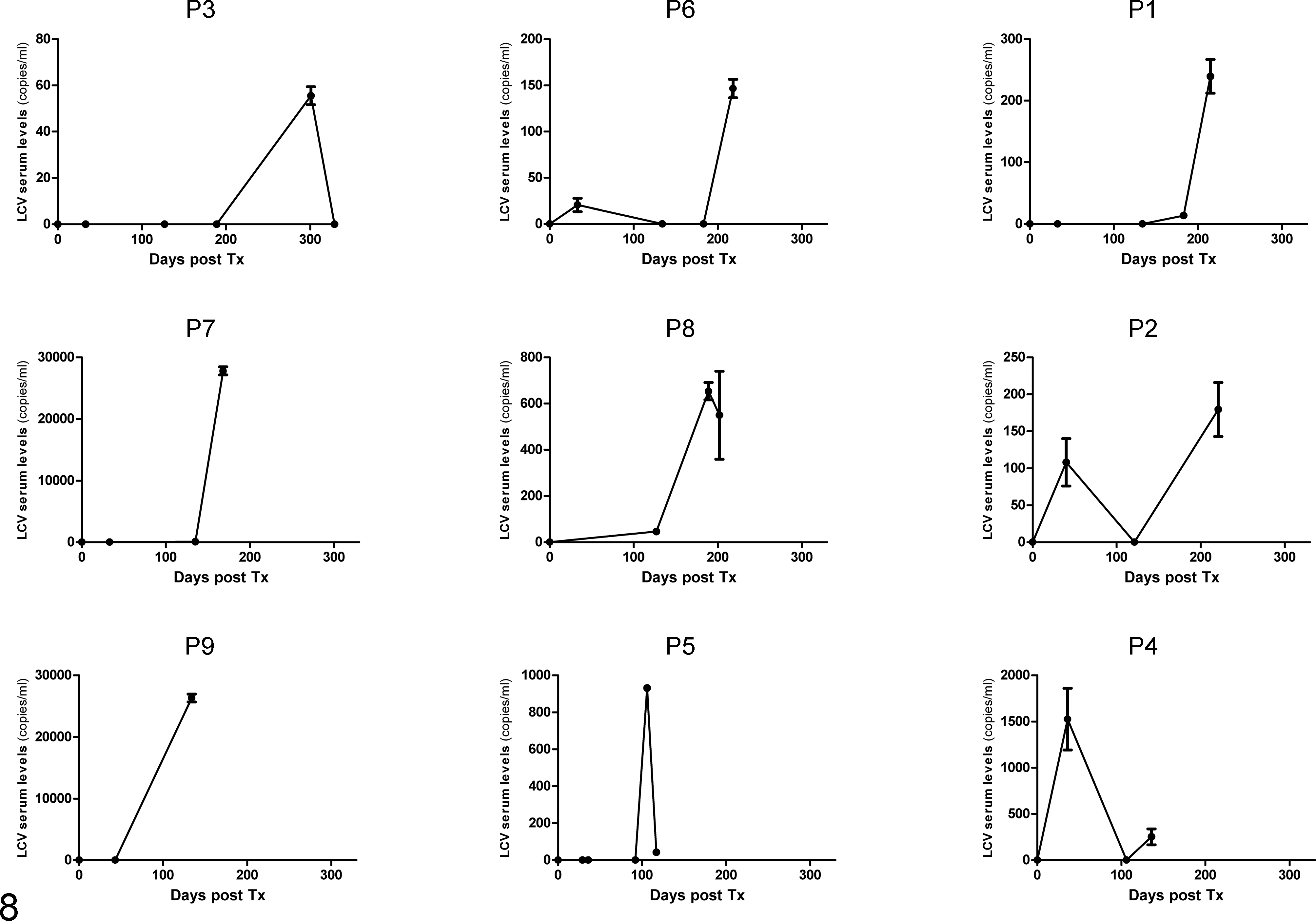
Lymphocryptovirus (LCV) viremia levels in animals affected by posttransplant lymphoproliferative disorder (PTLD). No viremia was detected in PTLD-negative animals.
Discussion
PTLDs are a heterogeneous group of lymphoid proliferations that occur in the setting of depressed T-cell function due to immunosuppressive therapy used with solid organ transplantation or hematopoietic stem cell transplantation. 9 In humans, genetic and epigenetic alterations, microenvironment-resident plasmacytoid dendritic cells, and Treg cells play a critical role, as well as viruses, notably EBV, and contribute to the development of PLTDs. 21
The EBV genome exists in humans as an episome in latently infected B cells. Activated functional T cells play a pivotal role in the control of proliferation and elimination of EBV-infected cells. 15 In immunocompromised subjects, T-cell function is impaired, which may lead to uncontrolled proliferation of EBV-transformed cells, known as EBV-related PTLD. 12 Most Old World NHPs are infected with LCV, a herpesvirus of the same subgroup as EBV, 22 and several of the LCV products are homologues to EBV, such as EBNAs and LMP. 20 A NHP xenotransplantation model may have considerable potential value in elucidating PTLD pathogenesis and diagnosis. In this light, we characterized PTLD occurring in immunosuppressed cynomolgus monkeys undergoing neural precursor xenotransplantation to treat a pharmacologically induced form of PD. 1
In our case series, PTLD occurred in 32% of animals after a long course of immunosuppressive therapy. In particular, we detected clinical signs related to the presence of nasal masses that led to difficulties in breathing, abdominal masses that led to abdominal pain, or both. Clinical signs occurred on average at 172.5 PODs, and the average time between the occurrence of clinical signs and euthanasia was 20 days. In the literature of NHP PTLDs, specifically in the context of solid organ xenotransplantation, different times of occurrence of this disorder are reported: a median of 6.5 and 33.5 PODs in
All above-mentioned publications from other laboratories have reported extranodal masses in xenotransplanted and immunosuppressed primates. In our animals, the principal locations were alimentary and upper respiratory tracts, possibly due to a high density of mucosal associated lymphoid tissue. In the abdominal cavity of primates with larger alimentary tract masses, disseminated enlargement of lymph nodes was observed. In addition, in half of the cases, other masses (possibly lymph nodes) were found close to the trachea, in the mediastinum, or adjacent to the thoracic or abdominal aorta.
Like most human PTLD- and AIDS-related diseases, the NHP lymphomas are generally of high-grade malignancy. 16 The morphology of the masses of all xenotransplanted cynomolgus monkeys was similar, revealing a centroblastic diffuse B-cell lymphoma with a high proliferation rate assessed with Ki-67 immunolabeling. According to the most recent WHO classification of PTLDs, 21 these lymphomas are classified as monomorphic lymphomas.
To assess whether PTLDs are associated with LCV, 2 different techniques were used: IHC and PCR. The IHC findings revealed that the neoplastic cells were diffusely positive for the EBNA-2 viral protein. Double-labeling for CD20 and EBNA-2 confirmed LCV infection of CD20+ neoplastic B cells.
When EBV infects a B lymphocyte, the virus expresses genes encoding proteins involved in viral DNA replication and viral particle synthesis. These include EBV nuclear antigens, EBNAs, and 3 LMPs associated with transforming activity. 7 In particular, EBNA-1 supports the EBV genome as a circular episome and is an important protein for immortalization and tumorigenesis of infected cells, and EBNA-2 stimulates B-cell proliferation through upregulation of LMP-1 and prevents reactivation from latency by upregulating the expression of LMP-2. In addition, EBV proteins have properties that help avoid host immune responses. During latent infection, EBV-infected B cells are protected from apoptosis by LMP-1. LMP-1 is a CD40-mimic protein, which upregulates cellular Bcl-2, A20, and other proteins that inhibit apoptosis and activates the nuclear factor–κB signaling pathway to stimulate cell growth. In humans, EBV-associated malignant diseases can be divided into 3 patterns of latency, depending on the viral genes expressed. In type III latency, all latency genes are expressed and development of PTLD occurs. 6 In our study, PCR revealed diffuse expression of EBNA-1 and EBNA-2 in neoplastic tissues, in comparison to reactive lymph nodes of the same animals. In contrast to what is observed in humans, we detected only the expression of EBNA-1 and EBNA-2 and not LMP-1, revealing a possible new pattern of viral protein expression during the development of PTLD in nonhuman primates.
To establish whether variation of viral DNA could be an early diagnostic tool for PTLD progression in primates, we designed a quantitative PCR that was used on sera of 3 distinct phases of the experimental life of our primates: pretransplant, posttransplant (an intermediate time between transplantation and euthanasia), and euthanasia. EBV monitoring in humans suggests that high EBV loads are only predictive of PTLD if associated with low cytotoxic T-cell responses. Instead, in the present study, the data suggest that macLCV monitoring may be valuable in the early detection of PTLD in nonhuman primate xenotransplantation trials, allowing preemptive treatment such as a reduction of immunosuppression therapy. In addition, there was no evidence of PLHV zoonotic transmission through xenoneuronal grafts in this study.
In conclusion, even if a systemic immunosuppression was needed to sustain the life of a neuronal cell graft in this PD xenotransplantation model, PTLD occurred in a third of the immunosuppressed
Footnotes
Acknowledgements
We thank people involved in the XENOME project (European Commission’s Sixth Framework Program, under the priority thematic area Life Sciences, Genomics, and Biotechnology for Health, contract LSHB- CT- 2006-037377), Dr Peter Barry (California Medical Research Center) for providing the noncommercial antibody specific for IE1 protein of rhesus CMV, the CORIT (Consortium for Research in Organ Transplantation, Padua, Italy) primates’ keepers (Daniele Ramon, Andrea Barzon) and veterinary staff (Fabio Fante, Massimo Boldrin) for the invaluable support and care, Dr Giuseppe Palmisano and Emanuele Zanetti (BCA Department, Padua University) for postmortem room technical help, and Dr Silvia Ferro for editing support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
