Abstract
Multinucleated hepatocytes (MNHs) have been occasionally reported in macaques, as well as chimpanzees and gorillas, as an incidental finding. However, information is sparse on variations in incidence in the cynomolgus macaque (Macaca fascicularis). A survey was conducted to assess the occurrence of MNHs in the liver of stock (nonstudy) animals from SNBL SRC (Alice, TX) and SNBL USA (Everett, WA) submitted for diagnostic purposes. A total of 215 cynomolgus monkeys originally from Cambodia (61), China (5), Indonesia (125), and Mauritius (24) were used for this investigation. From each animal, usually 2 liver samples were processed for histopathology with 2 sections in each slide. An MNH was defined as a hepatocyte with 3 or more nuclei. A threshold of 3 MNHs was selected for the Multinucleated Hepatocyte Grading System: 0 = not remarkable (≤3 MNHs counted from 2–4 liver sections), minimal = 4 to 15 MNHs, mild = 16 to 30 MNHs, moderate = 31 to 59 MNHs, and severe ≥60 MNHs. The incidence of MNHs was 60 of 86 (70%) in males and 72 of 129 (56%) in females for a total overall incidence of 132 of 215 animals (61%). Affected hepatocytes were frequently observed close to the capsule and generally had 3 to 8 nuclei per hepatocyte but as many as 15 occurred in a single cell. Awareness of the incidence of MNHs in cynomolgus monkeys is important for potential use as background data in preclinical safety and toxicity evaluation studies.
In the preclinical safety and toxicity testing of drugs for human medicine, the use of nonhuman primates (NHPs) has increased because of their close phylogenic relationship to humans and increased research and development of drugs and biopharmaceuticals. 1 The cynomolgus macaque, now available purpose-bred for laboratory use, has become the most widely used NHP species. However, there is a dearth of information on multinucleated hepatocytes (MNHs), a spontaneous background pathology finding of laboratory-raised macaques. MNHs have been occasionally reported as an incidental finding in macaques, chimpanzees, and gorillas. 6 Binucleated hepatocytes, pigment in the gallbladder submucosa, and tension lipidosis have also been seen sporadically in the liver of macaques. 2 To our knowledge, no information on incidence variation or pathologic description of MNHs in a large group of stock (nonstudy) cynomolgus monkeys has been published, hence this report. Here we present evidence of the high occurrence and variable severity of MNHs in the liver of cynomolgus macaques that will serve as background data for tissue evaluation in preclinical safety and toxicity studies.
Materials and Methods
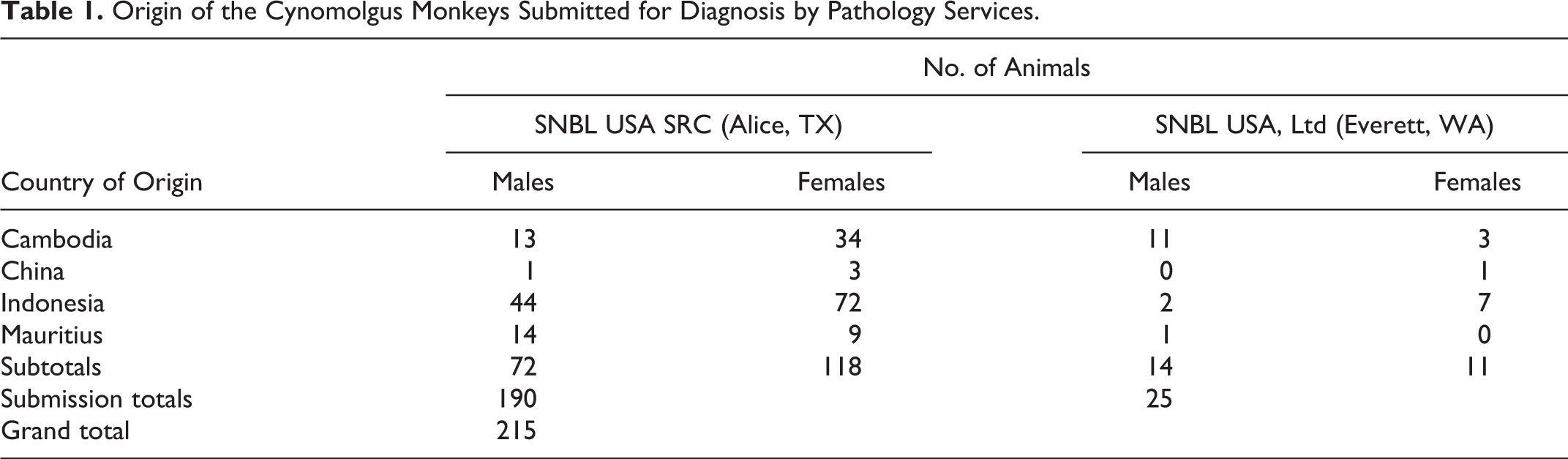
The results of histopathologic examination of livers of 215 stock (nonstudy) cynomolgus monkeys from SNBL SRC (Alice, TX; 190 cases) and SNBL USA (Everett, WA; 25 cases) submitted for diagnostic purposes from fall 2009 to summer 2012 were evaluated (Table 1). These monkeys were purpose-bred, originally from breeding and/or holding facilities in Cambodia, China, Indonesia, and Mauritius. Animals were brought first to SNBL SRC (Alice, TX) for quarantine and then shipped to Everett, Washington, for use in contract research studies. All 190 animals from Texas and 6 of 25 from Everett were naive to treatment with investigational drugs or surgical procedures. Of the remaining 19 Everett stock animals, 15 were necropsied 2 weeks or more following their recent washout period of test and/or control articles. The remaining 4 animals necropsied did not complete their washout period.
Origin of the Cynomolgus Monkeys Submitted for Diagnosis by Pathology Services.
The monkeys, which ranged in age from 0.25 to 9.9 years, were either found dead or euthanized and necropsied due to debilitating clinical condition or terminated for other reasons. At necropsy, samples of organs and tissues deemed important for diagnosis were collected into 10% neutral buffered formalin fixative and submitted to Pathology Services (SNBL USA, Everett, WA). Following adequate fixation, tissue samples, including the liver, were trimmed, processed to paraffin block, and sectioned. Slides were stained with hematoxylin and eosin (HE) and examined by light microscopy by American College of Veterinary Pathologists (ACVP) board-certified pathologists. For transmission electron microscopy, selected wet liver samples corresponding to the paraffin-embedded tissue blocks of HE sections with MNHs were processed in the usual manner and examined with the Philips CM 200 electron microscope (FEI, Hillsboro, OR) at the Franchesi Microscopy and Imaging Center, Washington State University, Pullman, Washington.
At SNBL USA, usually 2 liver samples (1 from the median lobe and 1 from the left lateral lobe) are processed for histopathology with 2 sections in each slide (1 median lobe section included gallbladder). The MNH was defined as a hepatocyte with 3 or more nuclei. In this survey, a threshold of greater than 3 MNHs was used for the Multinucleated Hepatocyte Grading System as follows: 0 = not remarkable (≤3 MNHs), minimal = 4 to 15 MNHs, mild = 16 to 30 MNHs, moderate = 31 to 59 MNHs, and severe ≥60 MNHs.
Results
The incidence, severity, and distribution of MNHs in cynomolgus monkeys are presented in Table 2. The incidence of multinucleated hepatocytes (61% overall) was not related to geographical origin (Table 3). Average age was 5.7 years for affected animals and 6.2 years for unaffected animals. None of the 4 infants examined (0.25–0.7 years old) were affected. More males had MNHs (60 of 86 or 70%) compared with females (72 of 129 or 56%) with more minimal to mildly affected animals than moderate to severely affected animals. Our survey indicated that slightly more than half of animals with a 0 grade had no detectable MNHs.
Incidence and Severity of Multinucleated Hepatocytes (MNHs) in Cynomolgus Macaques From Different Countries of Origin.
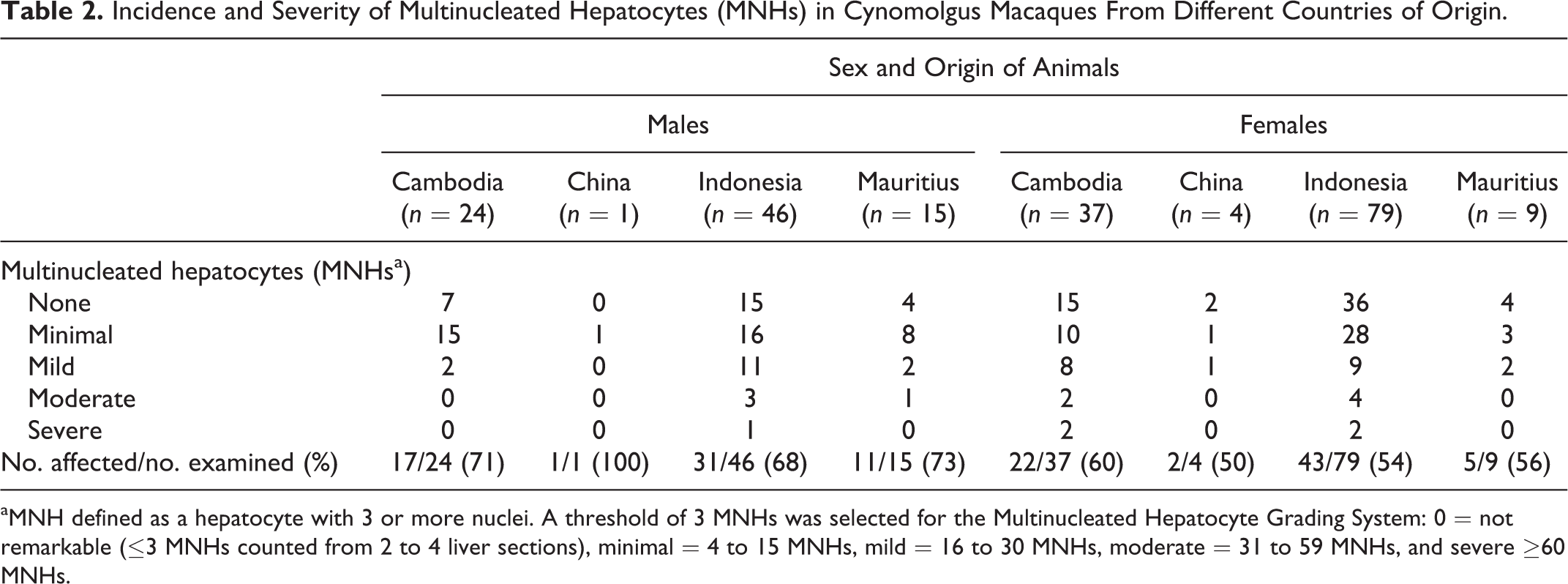
aMNH defined as a hepatocyte with 3 or more nuclei. A threshold of 3 MNHs was selected for the Multinucleated Hepatocyte Grading System: 0 = not remarkable (≤3 MNHs counted from 2 to 4 liver sections), minimal = 4 to 15 MNHs, mild = 16 to 30 MNHs, moderate = 31 to 59 MNHs, and severe ≥60 MNHs.
Distribution of Multinucleated Hepatocytes (MNHs) in Cynomolgus Macaques.

Affected hepatocytes were frequently observed close to the liver capsule but sometimes appeared in the periportal, midzonal, and centrilobular zones. Generally, MNHs had 3 to 8 nuclei and the affected hepatocyte apparently enlarges to accommodate more nuclei. As many as 15 nuclei occurred in a single hepatocyte (Fig. 1a,b). The multiple nuclei had staining similar to those in the adjacent normal (uni- and binucleated) hepatocytes. No mitotic figures were observed in the MNHs or the normal hepatocytes.
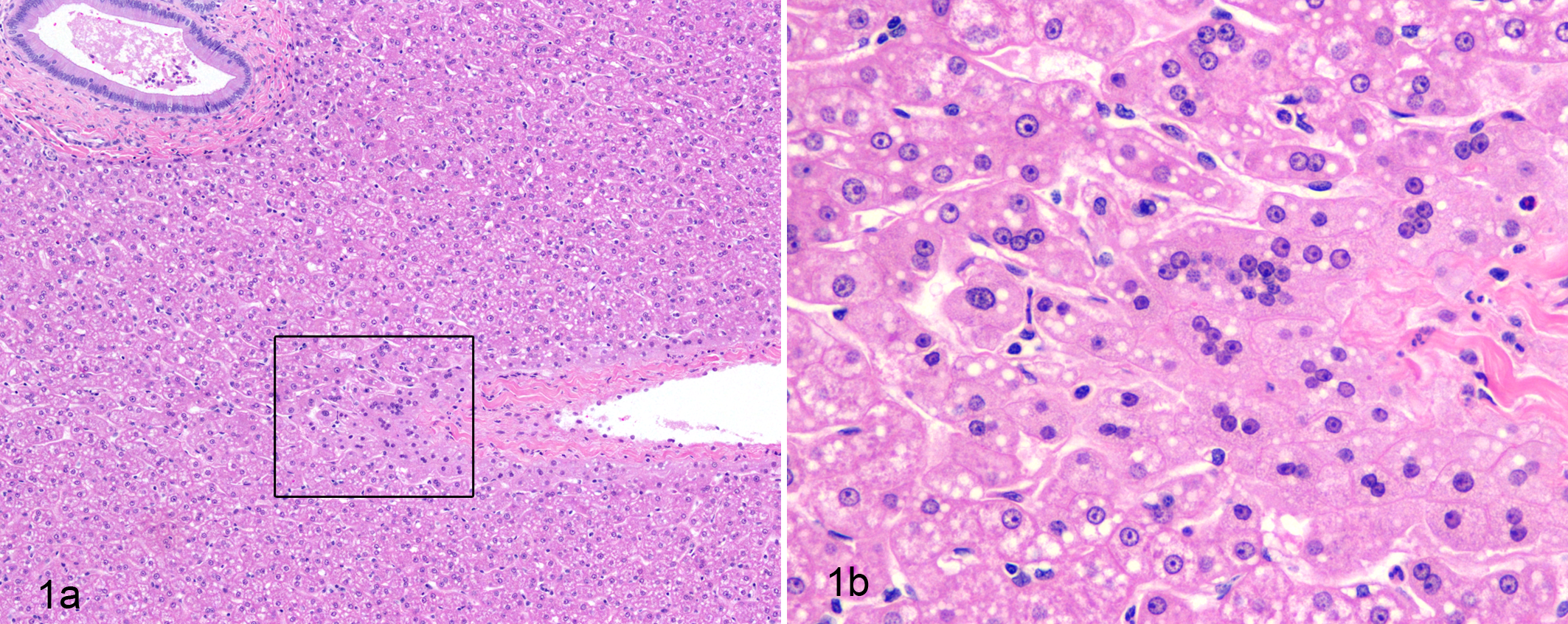
(a) Liver; cynomolgus macaque. Low-power photomicrograph shows multinucleated hepatocytes adjacent to normal uni- and binucleated hepatocytes. (b) Higher magnification of the boxed area in (a) shows 3 to as many as 15 nuclei in a single hepatocyte. Hematoxylin and eosin.
Electron microscopy confirmed the normal features of the nuclei and organelles. No cell membranes were observed within the cytoplasm of MNHs. Multinucleated hepatocytes had nuclei similar to adjacent normal hepatocytes (Fig. 2).
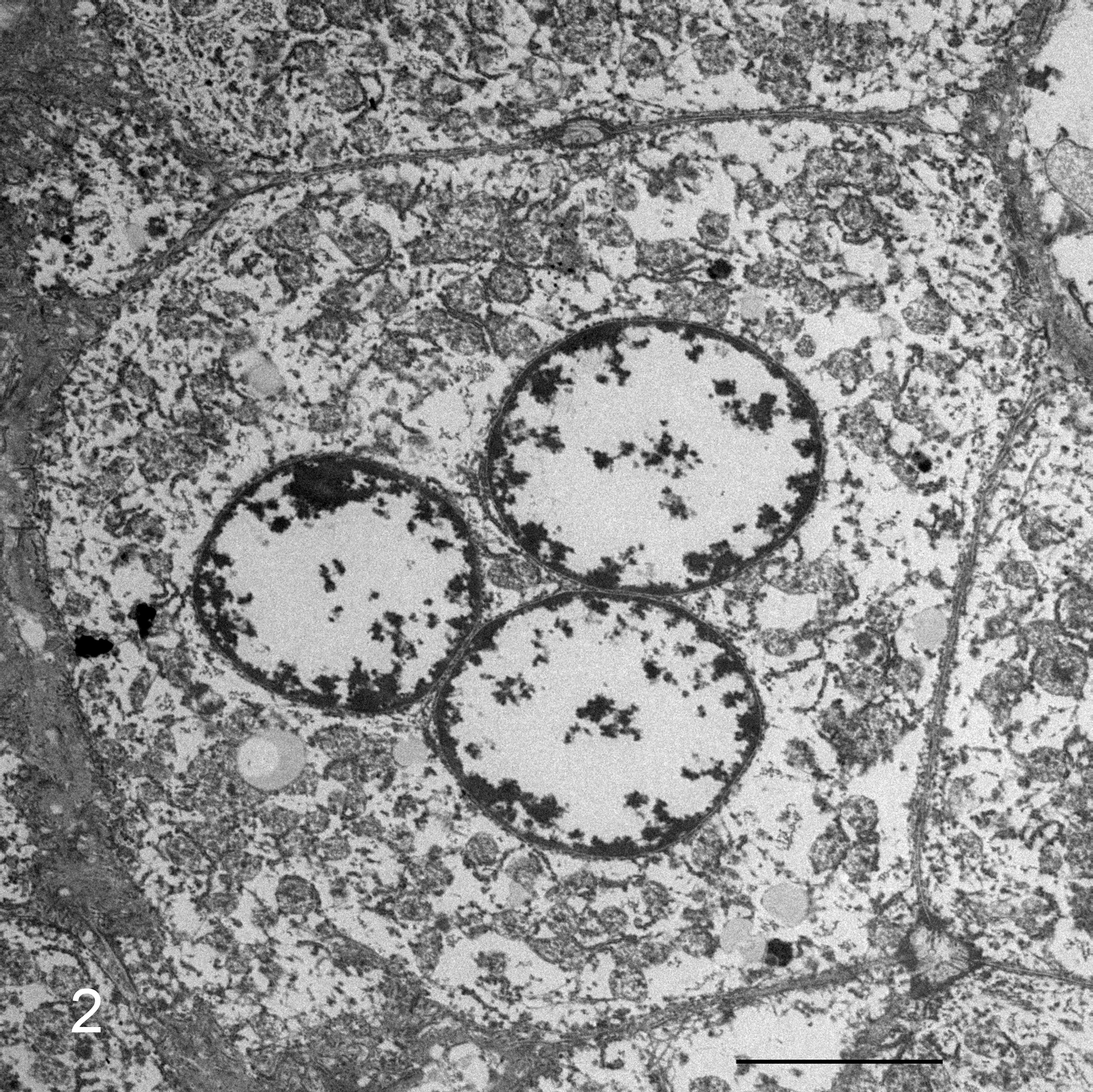
Liver; cynomolgus macaque. Electron micrograph showing a single hepatocyte with 3 nuclei. Stained with 4% uranyl acetate, trace of 10% potassium permanganate followed by Reynolds lead. Bar = 5 μM.
Diagnostic findings in the animals were unrelated to the incidence of multinucleated hepatocytes. Eighty-five animals, from a cohort suspected to have been exposed to tuberculosis (TB) in the country of origin, were euthanized and necropsied during quarantine but did not have TB lesions.
While many animals were considered apparently normal, 93 animals had morphologic and/or etiologic diagnoses associated with morbidity or death. Diarrhea/enteritis was the most frequent diagnosis, observed in 46 (50%) of these animals.
In this survey, diagnoses consisted of nonproliferative lesions, except for 2 benign neoplasms found in 2 male Mauritian cynomolgus macaques. One male diagnosed with osteochondroma had minimal MNHs in the liver, while no MNHs were found in the other male observed with cystic trichoepithelioma and epidermal inclusion cysts. All the proliferative and nonproliferative lesions had no relationship with the occurrence or severity of MNHs in affected animals and hence were considered naturally occurring incidental background changes in the cynomolgus monkey.
Discussion and Conclusions
MNHs have been reported to occur in infectious conditions, nutritional deficiencies, diseases of abnormal storage, and in response to bacterial toxins and to toxic chemicals (reviewed by Scampini et al 7 ). Some reports considered the giant-cell (multinucleated) transformation as a nonspecific reaction rather than diagnostic of any specific disease or condition. 3 Although the occurrence of MNHs has been considered a nonspecific reaction to injury, the pathogenesis is not certain. Spontaneous formation can result from nuclear division of a single cell with incomplete cytoplasmic separation, but an alternative is fusion, such that parenchymal cells coalesce to form the MNHs. 4 Ultrastructural examination of selected liver samples in the present study did not reveal cell membranes within the cytoplasm of MNHs, and their nuclei and organelles were within normal limits.
MNHs have been reported to occur in the liver of Sprague-Dawley rats gavaged with rifabutin in a dose-related manner. 7 The untreated control rats (males or females) had a 10% incidence rate, but no MNHs were observed in mice and cynomolgus monkeys given rifabutin. MNHs were occasionally seen as an incidental finding in macaques, chimpanzees, and gorillas and have been reported in the rhesus monkey, baboon, and marmoset associated with diethylnitrosamine toxicity, pyridoxine deficiency, and aflatoxin B1 toxicity, respectively. 6,7 In addition, increased numbers of MNHs were observed in liver biopsies from 3 of 8 cynomolgus monkeys experimentally infected with hepatitis E virus (HEV). 5
Previous histopathology experiences evaluating livers from NHPs suggested that hepatocytes with 3 nuclei were uncommon, in contrast to binucleated hepatocytes, which were commonly found and considered normal. In this study, a threshold of greater than 3 MNHs was used in a grading system to denote 0 or not remarkable as equal to or less than 3 MNHs in all sections examined from individual animals. Based on our survey, more than half of the animals that had a 0 grade did not have any MNHs.
The incidence rates of MNHs in cynomolgus monkeys by geographical origin were 39 of 61 (64%) from Cambodia, 3 of 5 (60%) from China, 74 of 125 (59%) from Indonesia, and 16 of 24 (67%) from Mauritius for a total incidence of 132 of 215 animals (61%). Although none of the 4 neonatal animals had MNHs, no firm conclusions could be made about the relationship of age to MNHs. More males had MNHs (60 of 86 or 70%) compared with females (72 of 129 or 56%). The high incidence and severity of MNHs were not related to the naturally occurring incidental changes in affected animals and hence were considered to occur spontaneously. Spontaneous background findings may complicate pathologic interpretation. Since fewer numbers of NHPs are used in preclinical studies, the chance of some spontaneous incidental finding, such as MNHs, occurring in a dose-related manner or only in treated animals can lead to misinterpretation of the finding as a test article–related effect and potential for neoplasia. Unless large numbers of MNHs were present (moderate or severe), they would probably be unnoticed in routine evaluation and would not be expected to be a significant factor in preclinical studies. Large numbers of MNHs may be present on occasion, and awareness of the incidence and severity of MNHs may help mitigate concerns during preclinical safety and toxicity evaluation studies in cynomolgus monkeys.
Footnotes
Acknowledgment
We acknowledge and thank Dr Christine Davitt, Washington State University and Barbara Wright, Miki Haraguchi, and Michelle Taylor at SNBL USA for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
