Abstract
A previously unrecognized condition is described in wild free-ranging Pribilof arctic foxes (Alopex lagopus pribilofensis) from the Pribilof Islands, Alaska, USA. This condition is called shaggy lame fox syndrome (SLFS) denoting the primary clinical signs first observed. Criteria used to suspect SLFS on gross examination included emaciation, failure to shed winter pelage and moderate to severe polyarthritis. Criteria used to confirm SLFS histologically included polyarthritis (characterized by lymphoplasmacytic synovitis, tenosynovitis, bursitis, periosteal bony proliferation, and periarticular lymphoplasmacytic vasculitis) and systemic leukocytoclastic vasculitis. Other histological lesions often found included renal cortical infarcts, myocarditis with myocardial infarcts, lymphoplasmacytic meningitis, lymphoplasmacytic cuffing of meningeal and a few cerebral vessels, and cavitating infarcts of the brainstem and thalamus. The cause of SLFS is not known at this time; however, the gross and histological lesions suggest that the cause of SLFS may be a bacterial polyarthritis with a secondary immune-mediated vasculitis. These lesions are consistent with changes described with Erysipelothrix rhusiopathiae in domestic dogs; E. rhusiopathiae was identified from the synovial membrane of a swollen stifle joint and the kidney from one fox using real-time polymerase chain reaction and with culture from a fox that had gross and histological lesions of SLFS. Therefore, E. rhusiopathiae is a possible etiological agent for SLFS.
Keywords
The Pribilof fox (Alopex lagopus pribilofensis) is a subspecies of arctic fox endemic to the Pribilof Islands, Alaska. 3,14,21 We have observed a novel syndrome occurring in Pribilof arctic foxes characterized by severe lameness and failure to shed the winter coat. Most Pribilof arctic foxes are dark “blue” color phase animals, however a small percentage (<3%) of Pribilof arctic foxes occur as the white color phase. 21 Unaffected Pribilof arctic foxes have a winter hair coat that is characterized by long, dark blue or brown fur (Fig. 1) and a summer coat that is short, dark, gunmetal blue/brown (Fig. 2). In contrast, Pribilof arctic foxes affected with SLFS have an unshed winter coat and severe lameness (Figs. 3 and 4). Ultimately this condition is fatal in most but not all cases with death resulting from complications of starvation. Here we describe the clinical features, gross and histological lesions, and associated microbiological findings of shaggy lame fox syndrome (SLFS), named for the initial clinical signs observed.

Material and Methods
Study Population
During population surveys and behavioral studies of Pribilof arctic foxes 21 and mortality studies of northern fur seals (Callorhinus ursinus) 17 on St. Paul Island, Pribilof Islands, Alaska, between 1986 to 2014, we encountered arctic foxes with shaggy, unshed winter coats late into the spring and summer months. These foxes also were severely lame and emaciated. Necropsies were carried out on 24 foxes found dead or euthanized in the months of July and August between 1986 and 2014 that had little to no autolysis. Selected tissues were frozen for microbiological studies or preserved in 10% neutral buffered formalin. Fixed tissues were trimmed, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin.
In addition to the foxes sampled in this study, several more Pribilof arctic foxes exhibiting similar signs were observed alive but were not captured or sampled. Serum samples used in this study were collected during a behavioral study by one of the authors (PAW) or from moribund foxes.
Diagnostic Tests
Tissues from Pribilof arctic foxes with and without gross and histologic lesions of SLFS were tested for E. rhusiopathiae using real-time polymerase chain reaction (PCR) and culture using 2 types of enriched media, by the Iowa State Diagnostic Laboratory (Ames, IA). 4,13 Tissues cultured on these 2 enriched media included synovial membrane of the stifle joint (from 7 foxes with SLFS and 3 unaffected foxes), stifle joint fluid (2 with SLFS), kidney (2 with SLFS and 2 unaffected), lung (2 with SLFS), and liver (1 with SLFS). Tissues tested by PCR included synovial membrane of the stifle joint (from 7 foxes with SLFS and 5 unaffected foxes), kidney (4 with SLFS and 4 unaffected), lung (4 with SLFS and 2 unaffected), medial iliac lymph node (3 with SLFS), synovial fluid of the stifle joint (2 with SLFS), liver (2 with SLFS), urine (2 with SLFS), superficial cervical lymph node (1 with SLFS), and spleen (1 with SLFS).
A further 23 microbiological cultures were done from 8 foxes with SLFS and 6 foxes that had no gross lesions of SLFS at the Colorado State University Diagnostic Laboratory (Fort Collins, CO). Tissues tested included the stifle joint (8 with SLFS and 6 unaffected), lung (4 with SLFS), superficial cervical lymph node (2 with SLFS and 1 unaffected), bursa on the palmar surface of the right carpal joint (1 with SLFS), and liver (1 with SLFS). Bacterial swabs were cultured aerobically on blood agar, MacConkey, and xylose-lysine 4 plates.
PCR and routine cultures for Mycoplasma sp. were done on the synovial membrane of the stifle joint from 3 foxes with SLFS. PCR tests for Chlamydophila sp., feline calicivirus, and felid herpesvirus 1 were done at the Colorado State University Diagnostic Laboratory (Fort Collins, CO) on synovial membrane of the stifle joint from 3 foxes with SLFS.
PCR testing for detection of Encephalitozoon cuniculi was done on 39 tissue samples from 11 foxes with SLFS and 5 unaffected foxes by the Reach Animal Diagnostic Laboratory (University of Missouri, Columbia, MO), including synovial membrane of the stifle joint (8 foxes with SLFS and 10 unaffected foxes), kidney (7 with SLFS and 4 unaffected), lung (1 with SLFS and 2 unaffected), medial iliac lymph node (1 with SLFS), synovial fluid of the stifle joint (2 with SLFS), liver (1 with SLFS), and urine (3 with SLFS) foxes.
Complement fixation tests for Coxiella sp. were done on sera from 16 foxes with SLFS and 10 foxes that had no gross evidence of SLFS and appeared to be healthy at the time of capture. Tests were done at the College of Veterinary Medicine, Texas A&M University, College Station, TX and the National Disease Laboratory, Ames, IA.
Routine serological tests were performed for evidence of exposure to canine distemper virus (serum neutralization titer >1:16 was considered positive), canine parvovirus (hemo-agglutination inhibition titer >1:16 was considered positive), leptospirosis (micro-agglutination titer >1:100 was considered positive), brucellosis (card agglutination), and Feline calicivirus (serum neutralization titer >1:2 was considered positive) in 15 foxes with and 15 foxes without SLFS. A modified agglutination test (titer >1:40 was considered positive) was done on sera for detection of Toxoplasma gondii from 10 foxes with and 5 foxes without SLFS. Serological tests were done at the Colorado State University Diagnostic Laboratory, Fort Collins, CO.
Osteological Preparations
Forty complete Pribilof arctic fox skeletal preparations were available for examination at the Museum of Vertebrate Zoology, University of California, Berkeley, CA. These skeletons were opportunistically salvaged from 1990 through 2008 from St. Paul Island and cleaned and deposited in the museum by one of the authors (PAW). Forty-one anatomical sites, as defined in domestic dogs, 5 were examined from each skeletal preparation for the presence of periarticular bony proliferation.
Results
Demographics
This study describes gross and histological lesions and microbiological findings in 24 Pribilof arctic foxes from St. Paul Island, Pribilof Islands, Alaska of which 18 foxes (12 male, 5 female, 1 not recorded) had gross evidence of SLFS and 6 foxes (3 male, 3 female) had no gross evidence of SLFS. The 6 unaffected foxes had been killed by the local people or been hit by a car. Ages of the foxes (affected and unaffected) ranged from 1 to 5 years (2.25 ± 1.1 years, mean ± SD) as determined by tooth cementum annular rings. 7
Clinical Signs
Most foxes with SLFS were readily distinguishable as still maintaining a shaggy, dull, light brown winter coat into the summer months (July–August), past the time when other foxes had shed most of their winter pelage (Fig. 3). This ragged, faded winter coat typically remained over the head, shoulders, chest, abdomen, and tail. Over the lower back, flank, and head, the winter coat was occasionally shed and the dark summer coat was visible. Occasionally, extremely lame foxes that had shed most of their winter hair coat were observed in mid-August (Fig. 4).
When observed at a distance and not disturbed, foxes with SLFS walked with a short choppy gait. They usually did not run on their own volition, but rather would walk short distances before sitting. Occasionally, when they sat down, they seem to “fall” the last few centimeters onto their rump. This was believed to be due to joint pain and loss of range of motion of the stifle joint. Affected foxes were often encountered unexpectedly at close range due to their penchant for lying down behind rocks or other windbreaks. Upon discovery, some foxes struggled to their feet and could “warm out” of their lameness to some degree and run for a short distance, although they appeared to tire quickly. Others literally toppled over when they tried to rise, or did not attempt to stand and were considered to be moribund.
Although physically incapacitated, foxes appeared to have normal mentation. Their eyes appeared normal and evidence of blindness or other neurological dysfunctions were not observed except in 1 affected fox that had bilateral phthisis bulbi.
Gross Lesions
The gross criteria for the diagnosis of SLFS were emaciation, failure to shed winter pelage, and moderate to severe polyarthritis (Table 1). Other gross lesions that were present in foxes with SLFS included renal cortical infarcts and, occasionally, pale foci in the myocardium that were considered to be infarcts.
Summary of Gross Lesions in 24 Pribilof Arctic Foxes (Alopex lagopus pribilofensis) With and Without Gross Lesions of Shaggy Lame Fox Syndrome (SLFS).
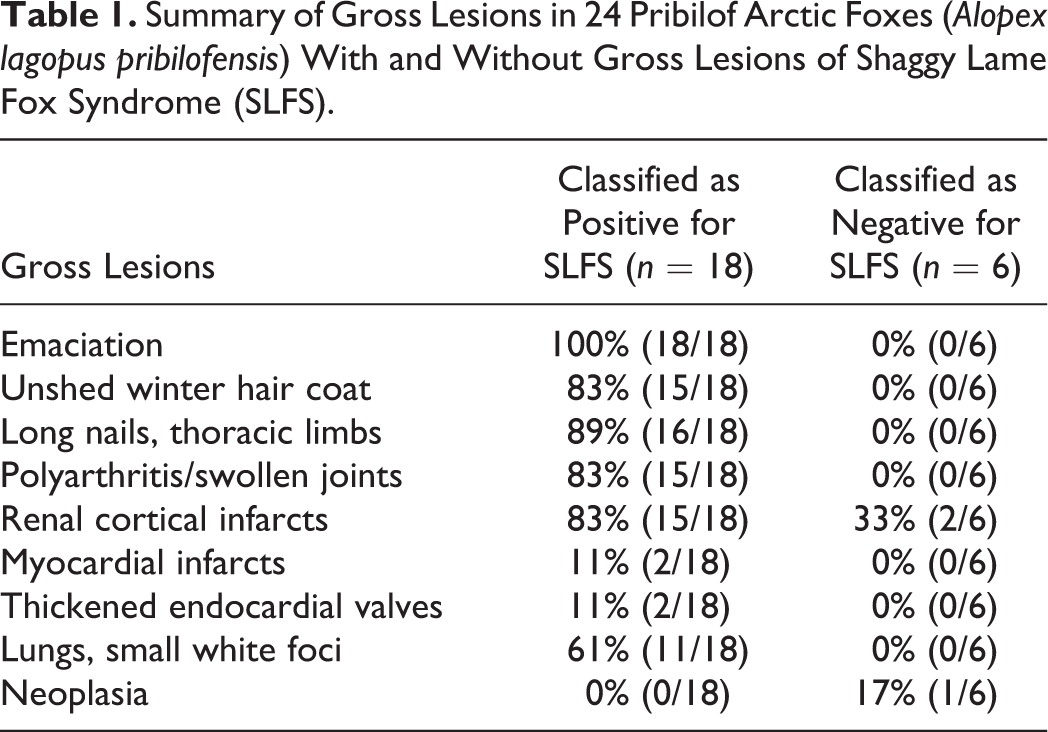
Emaciation with severe generalized muscle atrophy was present in all 18 foxes with SLFS but none of the 6 unaffected foxes. Body weights of foxes with SLFS were 3.0 ± 0.2 kg for males and 2.6 ± 0.5 kg for females (mean ± SD). This was significantly less than weights of apparently healthy male (4.6 ± 0.5 kg; Student’s t = 8.96, df = 58, P < .0001) and female (3.7 ± 0.5 kg; Student’s t = 3.66, df = 48, P < .001) Pribilof arctic foxes of similar age weighed as part of a behavioral study by one of the authors (PAW). 21 Subcutaneous and abdominal adipose tissues were depleted of lipid. Bone marrow was yellow and gelatinous. Sixteen of the emaciated foxes also had overgrown nails of the thoracic limbs and, to a lesser degree, of the pelvic limbs whereas the 6 unaffected foxes had nails of normal length.
Varying degrees of shedding of the winter pelage were present in 15 of the 18 foxes with SLFS, whereas the 6 unaffected foxes had normal summer pelage.
Multiple swollen joints (polyarthritis) were present in 15 of the 18 foxes with SLFS but none of the 6 unaffected foxes. Joints most commonly affected were the shoulder (15/18), stifle (15/18), carpal/metacarpal (12/18), tarsal/metatarsal (12/18), metacarpal/phalangeal (10/18), metatarsal/phalangeal (12/18), and interphalangeal joints (7/18). Affected joints were mildly to severely swollen and surrounded by a thickened joint capsule (Fig. 5). The joint cavity contained a slightly excessive amount of dark, clear, yellow synovial fluid. Occasionally, there was swelling of the bursas under flexor tendons on the palmar surface of the carpal/metacarpal joints, tarsal/metatarsal joints, and metacarpal/phalanges and metatarsal/phalanges (Fig. 6). Many affected joints had mild to severe periarticular bony proliferation with a decreased range of motion.
Kidneys were slightly small and often had an irregular surface with pale triangular foci within the cortex (infarcts). These lesions were present in 15 of 18 affected foxes and in 2 of the 6 foxes that did not have gross evidence of SLFS. Small 0.5–1 mm white foci were present scattered throughout the lungs in 11 of 18 affected foxes and in none of the unaffected foxes, and were most common in the diaphragmatic lobes. In 2 of 18 affected foxes, the left atrioventricular valves of the heart were thickened and had a slightly yellow granular surface. These 2 foxes also had several small pale foci within the left ventricular myocardium.
Gross lesions were not present in the eyes (except for 1 fox with bilateral phthisis bulbi), mouth (other than a few broken teeth), trachea, thyroid glands, parathyroid glands, liver, gall bladder, stomach, small and large intestine (except for a few cestodes in the small intestine), adrenal glands, pancreas, urinary bladder, or gonads in the 18 foxes with SLFS or in the 6 foxes that had no gross evidence of SLFS. However, 1 of the foxes that did not have SLFS had an adrenal cortical carcinoma with widespread metastasis.
Histopathology
The histologic criteria for diagnosis of SLFS were systemic leukocytoclastic vasculitis and polyarthritis characterized by lymphoplasmacytic synovitis, tenosynovitis, bursitis, periosteal proliferation and periarticular lymphoplasmacytic vasculitis (Table 2).
Summary of Histologic Lesions in 24 Pribilof Arctic Foxes (Alopex lagopus pribilofensis) With and Without Gross Lesions of Shaggy Lame Fox Syndrome (SLFS).
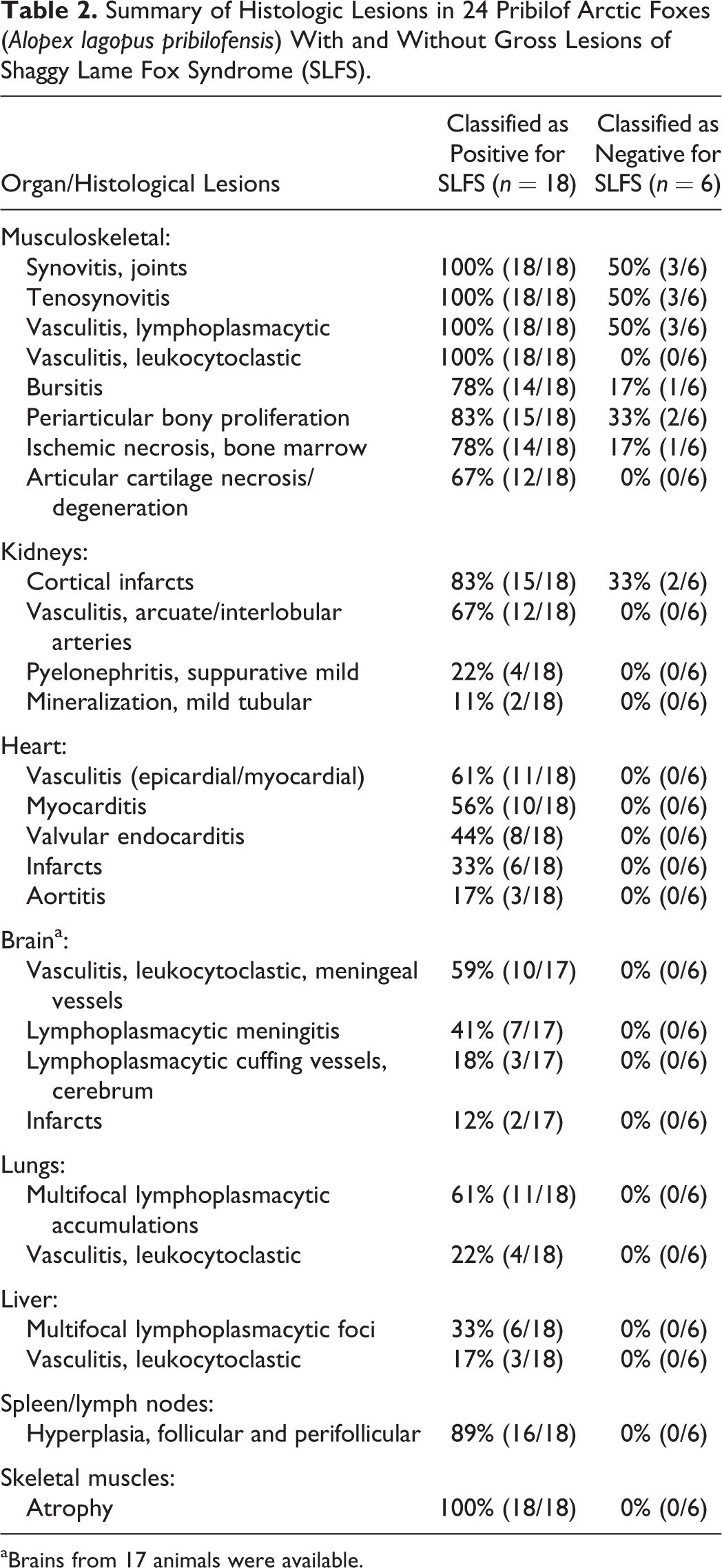
aBrains from 17 animals were available.
Joints
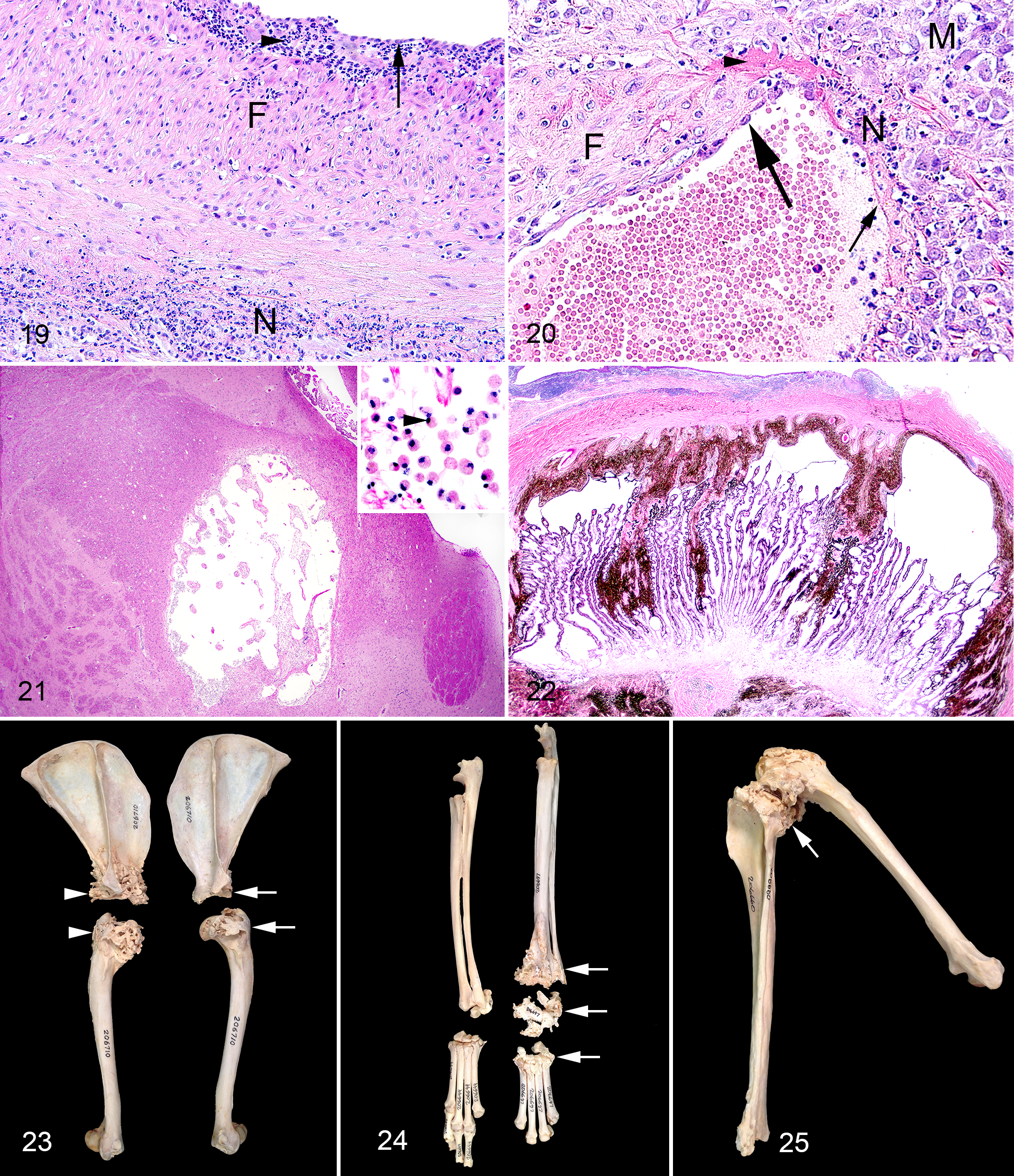
The most consistent lesions in foxes with SLFS involved the joints, periarticular tissues and blood vessels. Mild to severe, acute to chronic-active, proliferative lymphoplasmacytic synovitis, tenosynovitis, and bursitis were the most consistent periarticular lesions. Synovitis and tenosynovitis were present in 18 of 18 foxes with SLFS, and mild lesions were present in 3 of the 6 foxes with no gross lesions of SLFS. Bursitis was present in 14 of 18 foxes with SLFS and mild bursitis was present in 1 of 6 foxes without gross evidence of SLFS. Changes in the synovial membranes of the joints (Figs. 7 and 8), tendons (Figs. 9 and 10), and bursae (Fig. 11) were characterized by a mild to moderate infiltration of lymphocytes, plasma cells, a few macrophages and neutrophils with mild to moderate edema and varying degrees of fibrosis. Erosion of synoviocytes were present and were often covered with a thin layer of fibrin with a few neutrophils. Mild to severe, subacute to chronic, periarticular bony proliferation with osteophyte formation was present in 15 of 18 foxes with SLFS and in 2 of 6 foxes without gross evidence of SLFS (Fig. 12). Degeneration and loss of articular cartilage were present in 12 of 18 affected foxes and not present in the 6 foxes without gross evidence of SLFS. Focal areas of ischemic necrosis were present within the bone marrow in 14 of 18 foxes with SLFS and in 1 of 6 foxes without gross evidence of SLFS (Fig. 13).
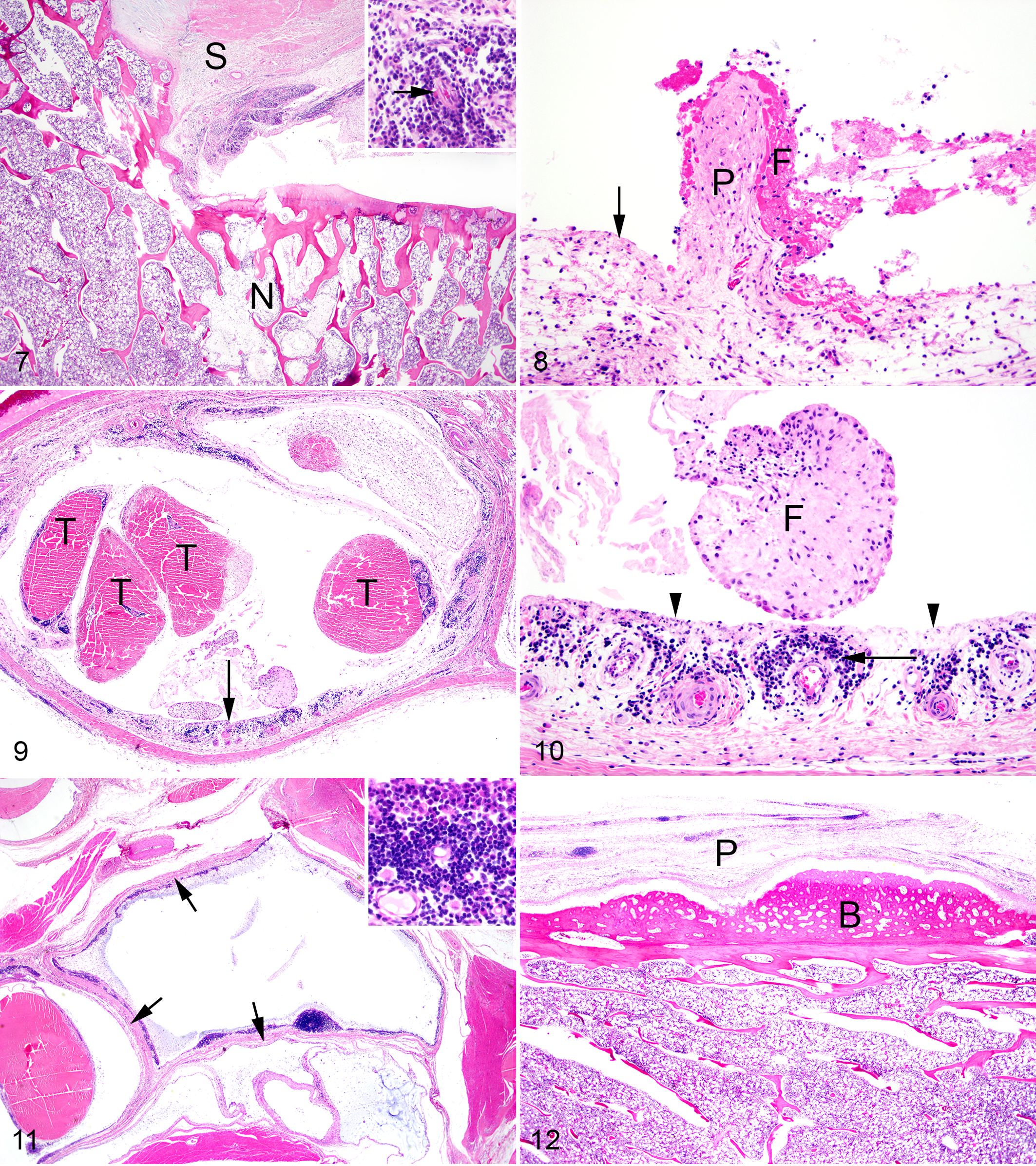
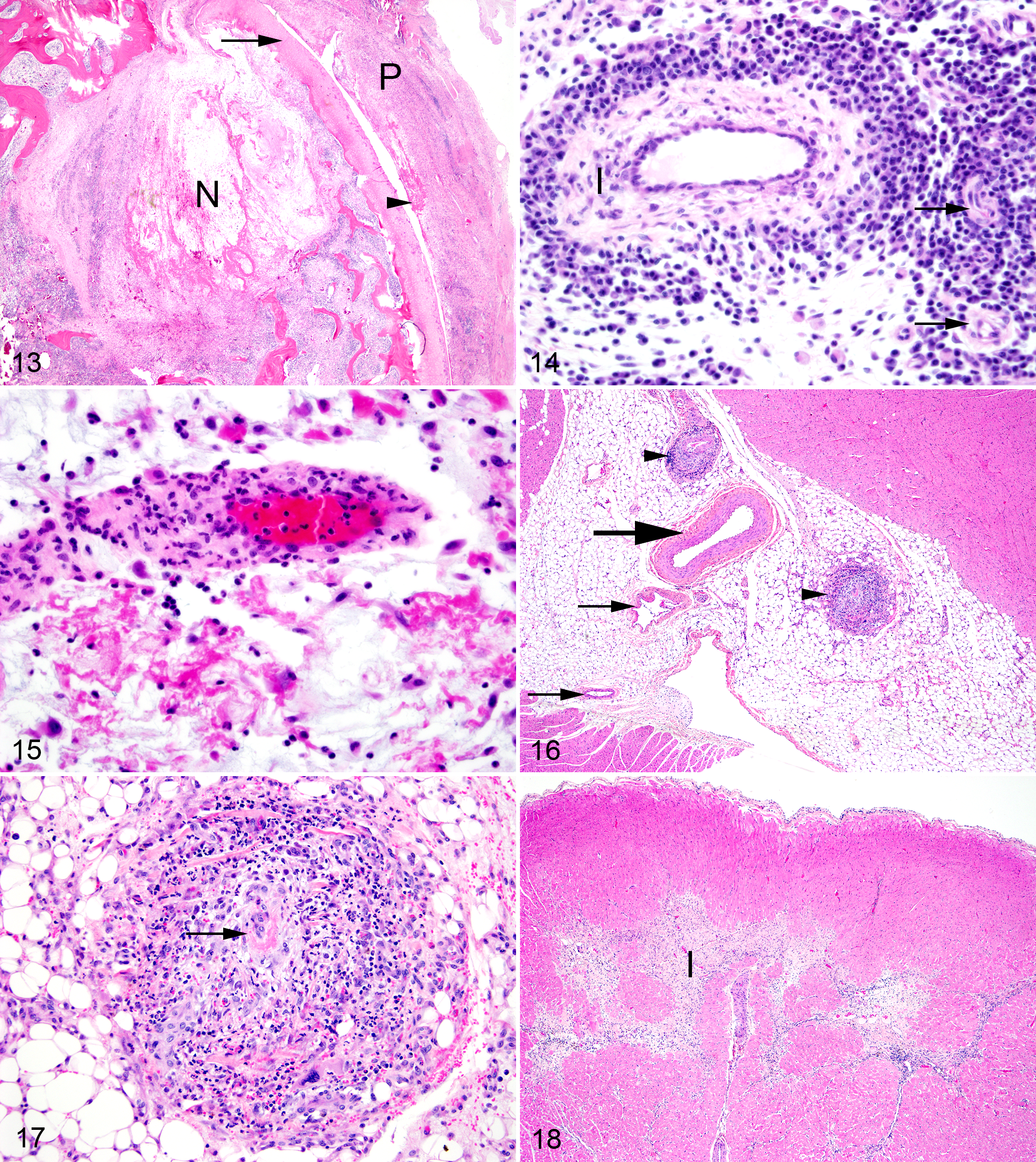
Vasculature
The most common vascular change was lymphoplasmacytic cuffing of small periarticular vessels (Fig. 14). This was present in 18 of 18 foxes with SLFS and in 3 of 6 foxes with no gross evidence of SLFS; these 3 foxes also had mild arthritis and tenosynovitis. The second, more severe, vascular lesion affected small to medium-sized arteries and was characterized by fibrinoid necrosis of vessel walls with a moderate to severe infiltration of lymphocytes, plasma cells, macrophages, and neutrophils (leukocytoclastic vasculitis) 19 (Fig. 15). Fibromuscular intimal proliferation within medium sized arteries also was observed in many affected vessels. This leukocytoclastic vasculitis was present in 18 of 18 foxes with SLFS, but not in the 6 foxes without gross evidence of SLFS. Leukocytoclastic vasculitis was present in a patchy distribution in the kidney, heart, and meninges of the brain, and less commonly in the periarticular tissues, lung, and liver.
Kidneys
Multifocal, mild to moderate, acute to chronic areas of infarction were present in the renal cortex of 15 of 18 foxes with SLFS and in 2 of 6 foxes that did not have evidence of SLFS on gross examination. Glomeruli were considered mildly sclerotic in the infarcted areas and normal in the noninfarcted areas. Other lesions included mild to moderate, subacute suppurative pyelonephritis, mild tubular mineralization, and leukocytoclastic vasculitis. Vasculitis was most common in the arcuate and interlobular arteries of the kidneys and within the capsule. Leukocytoclastic vasculitis, pyelonephritis, and mineralization were present in 18, 2, and 12 of the 18 foxes with SLFS, respectively, but were not present in the unaffected foxes.
Heart
Mild to severe leukocytoclastic vasculitis was present within the myocardium and/or epicardial connective tissues in 11 of 18 foxes with SLFS (Figs. 16 and 17). Myocarditis characterized by an infiltration of inflammatory cells between degenerate myocardial cells was present in 10 of 18 affected foxes. Myocardial infarcts were present in 6 of 18 affected foxes (Fig. 18). Valvular endocarditis characterized by mild thickening valves covered by a thin layer of fibrin mixed with a few neutrophils was present in 8 of 18 foxes with SLFS and included the atrioventricular and semilunar valves (Table 2). Mild to moderate, subacute to chronic aortitis characterized by an infiltration of mononuclear inflammatory cells within the vessel wall and fibromuscular intimal proliferation was present in 3 of 18 foxes with SLFS (Fig. 19). Lesions were not present in the heart or aorta of the 6 foxes without gross evidence of SLFS.
Brain
The primary lesion of the brain was a leukocytoclastic vasculitis within the meninges that was present in 10 of 17 foxes with SLFS (Fig. 20) (1 fox with SLFS was hit by a car and the brain was not suitable for examination). The vasculitis was especially prominent within basilar and middle cerebral arteries. A mild infiltration of lymphocytes and plasma cells was present within the meninges in 7 of 17 affected foxes. Three of these foxes also had mild cuffing of a few vessels primarily within the cerebral cortex. Cystic areas were present in the brain stem and thalamus in 2 of 17 foxes; these cystic regions were infarcts with infiltration by gemistocytic astrocytes (Fig. 21). Lesions were not present in the brains of the 6 foxes without gross evidence of SLFS.
Lungs
Small, multifocal accumulations of plasma cells, lymphocytes, and macrophages were present in the pulmonary parenchyma in 11 of 18 affected foxes. Mild leukocytoclastic vasculitis was present in pulmonary arteries in 4 of the 18 affected foxes. Mild suppurative bronchitis was present in 1 of 18 affected foxes. Interstitial pneumonia was not a feature in any of the foxes. Lesions were not present in the lungs of the 6 foxes without gross evidence of SLFS.
Liver
Several small focal areas of hepatocellular necrosis with infiltration of macrophages, plasma cells and lymphocytes were present in 6 of 18 affected foxes. These lesions were suggestive of parasitic migration tracts and/or previous bacteremia. Mild leukocytoclastic vasculitis was present in hepatic arteries in 3 of 18 affected foxes. Lesions were not present in the liver of the 6 foxes that were considered to be free of SLFS.
Lymphoid tissues
Hyperplasia of both follicular and perifollicular lymphocytes was a common finding in lymph nodes (superficial cervical, axillary, medial iliac, prefemoral, and popliteal nodes) and spleen of 16 of 18 affected foxes. Lesions were not present in lymphoid tissues of the 6 foxes that were considered to be free of SLFS.
Skin and skeletal muscle
The epidermis was thin in all animals, and atrophy of skeletal myocytes was present in all emaciated foxes.
Lungworms compatible in size and shape with Crenosoma vulpis were present in the bronchi and pulmonary parenchyma in 3 foxes with little or no inflammatory reaction. Sarcocysts were present in myocytes of skeletal muscle in 2 foxes and a larval nematode compatible in size and shape with Trichinella nativa was present in the skeletal muscle of 1 fox. Ear mites were present in the external ear canal in 3 foxes.
Microscopic lesions were not present in hair follicles or adnexa, eye, urinary bladder, ureter, testis, epididymis, pampiniform plexus, ovary, uterus, gall bladder, thyroid gland, parathyroid gland, adrenal gland, celiac ganglia, esophagus, stomach, small and large intestine, pancreas, peripheral nerve, spinal cord, or trachea of the 24 foxes, except the aforementioned phthisis bulbi (Fig. 22) and adrenal cortical carcinoma.
Microbiology
Real-time PCR (n = 37 tissues) and culture (n = 19) were done to search for E. rhusiopathiae in 7 foxes with SLFS and 5 foxes without SLFS. In 1 fox with gross and histological lesions of SLFS, culture and PCR testing of the stifle joint and kidney were positive for E. rhusiopathiae serotype 2 (Supplemental Table 1).
Routine bacteria cultures were done from tissues from 8 foxes with SLFS and 6 foxes without SLFS. Streptococcus bovis was isolated from the joint fluid and synovial membrane from the stifle joint of 1 fox, and Salmonella group D was isolated from the lung of another fox. These 2 foxes had gross and histological lesions of SLFS (Supplemental Table 1). The following bacteria were isolated in low numbers from tissues of foxes with and without gross and histological lesions of SLFS and were considered to be contaminants or of little significance: Moraxella phenylpyruvica, Staphylococcus intermedius, coagulase negative Staphylococcus, Enterococcus faecalis, Pseudomonas fluorescens, Comomonas testosterone, Escherichia coli, Enterobacter cloacae, Citrobacter freundii, and Clostridium perfringens.
Culture and PCR for Mycoplasma sp. were negative in the 3 SLFS foxes tested. PCR for Chlamydia sp, feline calicivirus, and felid herpesvirus 1 were negative using synovial tissues from 3 foxes with SLFS. Encephalitozoon cuniculi was not detected by PCR in any of the 39 tissues tested from affected and unaffected foxes (Supplemental Table 1).
Complement fixation tests for Coxiella sp. were all negative from both laboratories. Serological tests for evidence of exposure to canine distemper virus, canine parvovirus, leptospirosis and brucellosis were all negative. Two foxes that had no gross or histological lesions of SLFS had a titer of 1:2 to feline calicivirus. Of the 15 sera tested for antibodies to Toxoplasma gondii, 10 had a titer of <1:40, 3 of 1:40, 1 of 1:8,100, and 1 of 1:24,300. The 3 foxes with lower titers had SLFS and the 2 foxes with higher titers did not have SLFS (Supplemental Table 2).
Bone Lesions
Examination of opportunistically collected skeletal preparations archived in the Museum of Vertebrate Zoology, University of California, Berkeley verified the importance and relative significance of chronic active polyarthritis in the general Pribilof arctic fox population; 15 of 40 (37%) skeletons had evidence of periarticular bony proliferation. Joints most commonly affected were the shoulder (Fig. 23), carpal/metacarpal (Fig. 24), stifle (Fig. 25), tarsal/metatarsal, metacarpal/phalangeal, and interphalangeal joints. Joints occasionally affected included the antebrachial/carpal, tibial/metatarsal, atlantooccipital, temporal/mandibular, hip, and vertebral joints. The facets of the vertebrae and the costal/vertebral joints appeared to be rarely affected. This pattern also was observed in the gross examinations of the foxes with SLFS. While these museum skeletons were not confirmed cases of SLFS, they did share one of the most prominent lesions associated with this condition.
Discussion
Shaggy lame fox syndrome appears to be a previously undescribed condition in wild Pribilof arctic foxes. The syndrome was first observed on St. Paul Island by one of the authors (TRS) in July 1986. Thus far, foxes with SLFS have only been documented on St. Paul Island, but the condition is suspected to be present on other islands in the Pribilof Archipelago based on observations by one of the authors (TRS).
The primary clinical signs in affected foxes were mild to severe lameness and failure to shed the winter hair coat. The lameness can easily be explained by the gross and histological lesions in affected foxes. Nearly 50% of the foxes lacking typical gross changes of SLFS did have histological lesions compatible with this condition, suggesting that these foxes were in an early stage of the disease or were recovering. In a previous behavioral and tagging study by one of the authors (PAW), it was documented that even severely affected animals could survive for several months and perhaps longer. This prolonged survival may be related to the behavior of healthy foxes that have been observed bringing food to sick family members (observations by one of the authors, PAW).
The failure of affected foxes to shed the winter coat may be secondary to the arthritic lesions. Joint swelling can lead to pain which could result in a decreased tendency to scratch thereby inhibiting shedding of the winter coat. In particular, lesions in the stifle joints probably caused a reduced range of motion in the pelvic limbs and could be partially responsible for the unshed winter pelage on the neck, shoulders, chest, and abdomen. In contrast, shedding over the pelvic limbs, even in severely affected individuals, was facilitated by foxes pulling out the winter fur with their teeth. In foxes that started to show clinical signs in summer after the winter coat had already been shed, or with only mild clinical signs, the winter coat was partially or totally shed and the animals had the dark, gunmetal blue summer hair coat. There was no gross or histological evidence that any of these foxes had any type of lesions of the skin, dermal ectoparasites or endocrinopathy to account for the lack of shedding.
The nails of the thoracic limbs and, to a lesser degree, the pelvic limbs of foxes with SLFS were longer as compared to healthy foxes. This may be the result of decreased wear of nails secondary to changes in behavior patterns of affected foxes that resulted in decreased walking, running and digging.
The cause of SLFS is not known; however, the gross and histological lesions were similar to those described in domestic dogs and pigs infected with E. rhusiopathiae. 6,8,15,18,19 Both culture and PCR were positive for E. rhusiopathiae type 2 using synovial membrane from a swollen stifle joint and kidney from 1 fox with SLFS. E. rhusiopathiae has been isolated from an aborted northern fur seal fetus on St. Paul Island (Spraker, unpublished data). Thus, the organism has been documented to be present (with unknown prevalence) in the primary food items utilized by Pribilof arctic foxes.
The vascular lesions present in cases of SLFS resemble those described in ranch-raised arctic foxes in Sweden that were infected with E. cuniculi. 1,2,9 –12 These authors suggested this condition to be similar to polyarteritis nodosa in domestic dogs, especially in regards to the severe generalized vasculitis. 2 Several major differences were observed between the E. cuniculi infections in ranch-raised arctic foxes as compared to shaggy lame Pribilof arctic foxes. First, E. cuniculi primarily infects young of the year (pups), whereas SLFS primarily affects foxes that are 2 years of age and older (only 1 fox with SLFS was a yearling). Second, E. cuniculi can be observed with histopathology and such organisms were not present in tissues of Pribilof arctic foxes suffering from SLFS. Third, multiple tissues from Pribilof arctic foxes with and without SLFS have been examined using PCR for E. cuniculi and all were negative. Last, polyarthritis was not a prominent feature with E. cuniculi infections in foxes. 1,2,9 –12
Severe vasculitis is the hallmark lesion in polyarteritis nodosa of humans and domestic animals, especially in domestic dogs in which the cause is unknown. 19,20 However, severe chronic active polyarthritis with periarticular bony proliferation is not a feature of polyarteritis nodosa of domestic dogs. 19,20
The importance of the bacteria that were isolated from swollen joints and tissues from Pribilof arctic foxes with SLFS was undetermined. However, histological lesions in these animals were all similar and were hallmarked by subacute to chronic polyarthritis (many times fibrinous), tenosynovitis, bursitis, periarticular bony proliferation and systemic leukocytoclastic vasculitis. E. rhusiopathiae is known to cause similar lesions in dogs and swine in SLFS foxes. 6,8,15,18 –20 Streptococcus sp. are known to cause an immune mediated vasculitis/disease such as rheumatic fever in humans by mimicking host molecules, but articular lesions are usually not associated with this condition. 16 The gross and histological lesions combined with the microbiological results suggest that SLFS may be a primary bacterial infection with a secondary immune mediated vasculitis as is seen in domestic dogs and pigs infected with E. rhusiopathiae.
The mode of transmission of SLFS is unknown; however, if the cause is bacterial then 1 possible source of infection could be ingestion of bacteria-laden food. The majority of Pribilof arctic foxes that are seen with SLFS live adjacent to seal rookeries. The diet of these foxes is mostly northern fur seal placentas and scavenged dead pups and adult seals that foxes find on the rookeries and haul-out areas. E. rhusiopathiae has been isolated from an aborted northern fur seal fetus on St. Paul Island and Streptococcus sp. has been isolated from the mouths of foxes, as well as from wounds/cellulitis of northern fur seals from St. Paul Island (Spraker, unpublished data). The foxes that primarily live adjacent to bird colonies and nesting cliffs rarely show evidence of SLFS; their diet is primarily marine birds and eggs, not seals. 21 Direct transmission of SLFS also could possibly occur via direct contact (bite wounds and fighting injuries). Although there are no known differences in scavenging habits between male and female Pribilof foxes, male foxes are more aggressive in defending their territory. Males are over represented among SLFS cases (ratio 2:1) on St. Paul Island, and fights resulting in wounds were more common among male foxes as compared to female foxes (White, unpublished data).
Since 1995, the Pribilof arctic fox population of St. Paul Island appears to be decreasing (White, unpublished surveys). The importance of SLFS as a population-limiting factor is difficult to determine. However, any disease that can cause mortality has the potential to influence population dynamics. Furthermore, this disease has been documented as causing mortality within the reproductively active age class, meaning that population-level effects could be dramatic. This is especially true in small isolated fox populations such as exist on the Pribilof Islands. More data are needed to identify the cause of SLFS and to determine the potential population-level effects that this disease might have on Pribilof arctic foxes.
Footnotes
Acknowledgements
We thank the City of St. Paul and the Tanadgusix Corporation for permission to conduct this research on St. Paul Island, and the Alaska Department of Fish and Game for permission to live-trap and tag foxes and to salvage dead animals (ADF&G Scientific Permit Numbers: 90-78; 91-04; 92-96; 93-09; 00-62; 01-50; 02-88; 03-77; 06-114; 08-132). We thank Karin Holser for finding and freezing foxes found dead during the winters of 2001–2003. We thank Robert and Debbie Zink for sectioning and staining the histopathology slides. We thank Dr Tanja Opriessnig for examining tissues for Erysipelothrix rhusiopathiae and Dr Robert S. Livingston for examining tissues for Encephalitozoon cuniculi. We thank Hana Van Campen, Anita Schiebel, Gail Chinnock, and Brendan Podell for performing the PCR tests for Mycoplasma sp, Chlamydia sp, Feline calicivirus, and Felid herpesvirus 1 and Gail Thomson for the bacteriology. We thank Dr Tetiana Kuzmina for the gross photographs of the healthy and foxes with SLFS and Jay Oaks for helping with the histological photographs. We thank Dr Chris Conroy for photographing the osteological preparations. Osteological photographs are used with the permission of the Museum of Vertebrate Zoology, University of California, Berkeley. We thank Dr Robert Norrdin and the reviewers for Veterinary Pathology for providing helpful comments and suggestions on earlier versions of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
