Abstract
Oral squamous cell carcinomas (OSCCs) are common cancers of cats. While papillomaviruses (PVs) are an important cause of human OSCCs, there is currently little evidence that PVs cause squamous cell carcinomas (SCCs) of the mouth or other mucosal surfaces in cats. In the present cat, in situ carcinomas developed on the gingiva and nictitating membrane. Neoplastic cells within both cancers contained prominent PV-induced cellular changes consistent with those caused by Felis catus PV3 (FcaPV3), and FcaPV3 DNA was amplified from both cancers. Neoplasms also contained intense nuclear and cytoplasmic p16CDKN2A protein (p16) immunolabeling, suggesting PV-induced degradation of retinoblastoma protein. The molecular and histological features strongly suggested the cancers were caused by FcaPV3 infection. This is the first report of an association between PV infection and the development of an in situ carcinoma of the mucosa of cats. The identification of these lesions suggests that PVs might cause a proportion of OSCCs, and SCCs from other mucosal surfaces, in cats.
Keywords
Papillomaviruses (PVs) are small circular double-stranded DNA viruses that generally infect epithelium. 3 While most PV infections are asymptomatic, these viruses can influence cell growth and differentiation, and PVs can cause hyperplastic lesions (warts) as well as some types of cancer. 8 In humans, PVs are well recognized to cause cancers of the squamous epithelial mucosal surfaces, especially squamous cell carcinomas (SCCs) of the cervix and of the mouth. These cancers develop due to a well-defined progression from a virally induced in situ carcinoma to an invasive SCC. 6
In cats, PVs are well established to cause bowenoid in situ carcinomas (BISCs) of the haired skin. 16 BISCs can progress to SCCs 10 and PVs are thought to cause a proportion of cutaneous SCCs in cats.2,20 Oral SCCs (OSCCs) are very common in cats. As some human OSCCs are caused by PV infection and PVs cause skin SCCs in cats, previous studies have investigated whether PVs cause feline OSCCs. However, evidence from molecular studies is inconclusive. While Felis catus papillomavirus (FcaPV)2 DNA was detected in 10 of 32 (31%) feline OSCCs in one study 1 and in 12 of 28 (43%) in another, 22 feline PV DNA was detected in none, or a small proportion, of feline OSCCs in six other studies.4,11,13,14,18,23 Evidence from human OSCCs and feline cutaneous SCCs suggests that, if feline OSCCs are caused by PV infection, these cancers are likely to develop as the result of progression from a PV-induced in situ carcinoma. However, no such PV-induced precursor has been reported in the mouth of cats.
In human pathology, p16CDKN2A protein (p16) immunolabeling is used to determine whether an OSCC was caused by PV infection. 5 This is because human PVs consistently cause cancer by degrading the retinoblastoma protein resulting in an accumulation of p16 within the neoplastic cells. 19 Similarly in cats, PVs have been shown to degrade the retinoblastoma protein, increasing cell p16. Therefore, both human and feline SCCs that have been caused by PV infection are characterized by intense nuclear and cytoplasmic p16 immunolabeling. 9 While some variability in p16 immunolabeling has been reported in feline OSCCs,1,14 no feline OSCC has previously been reported to contain the intense nuclear and cytoplasmic immunolabeling that would indicate a PV etiology in a human OSCC or a feline BISC.
If PVs cause OSCCs in cats, these are likely to develop as a result of progression from an in situ carcinoma that contains PV DNA sequences, PV-induced cell changes, and p16 immunolabeling. Herein is reported a cat that developed in situ carcinomas of the mucosal epithelium of the gingiva and nictitating membrane. DNA sequences from FcaPV3 were amplified from both carcinomas, and neoplastic cells contained prominent PV-induced cell changes and intense p16 protein immunolabeling. This is the first time that histological, immunohistochemical, and molecular evidence of a PV etiology have all been identified within cancer of either the mouth or the nictitating membrane of cats.
An 11-year-old male domestic short-haired cat was presented to a veterinary clinic due to the presence of small lesions covered by a serocellular crust on the lower right eyelid and nasal planum. The eyelid mass enlarged over the following 4 months and, as an SCC was considered most likely, enucleation was performed. The globe and surrounding tissues were submitted for histopathology. The nose was treated with cryotherapy as an early SCC was suspected.
Examination of the nictitating membrane revealed irregular hyperplasia of the epithelium with the presence of thick trabeculae of epithelial cells that extended into the underlying submucosa. The cells appeared to push into the underlying tissue and the cells remained confined by the basement membrane with no evidence of invasion into the submucosa (Fig. 1). The cells within the hyperplastic epithelium showed significant dysplasia with crowding of cells within the deeper layers of the epithelium and loss of nuclear polarity. Many of the cells had an enlarged nucleus that was surrounded by a clear cytoplasmic halo (koilocytosis). In addition, linear or brick-like basophilic bodies were visible within many cells (Fig. 2). These bodies were typically arranged so that they were parallel to the cell nucleus and were consistent with those previously described to be due to FcaPV3.16,17 Examination of the haired skin of the eyelid revealed features of an SCC with invasion of the underlying dermis by nests and trabeculae of neoplastic epithelial cells. Clusters of enlarged cells with perinuclear clearing and basophilic bodies were also visible within the mass from the haired skin of the eyelid.
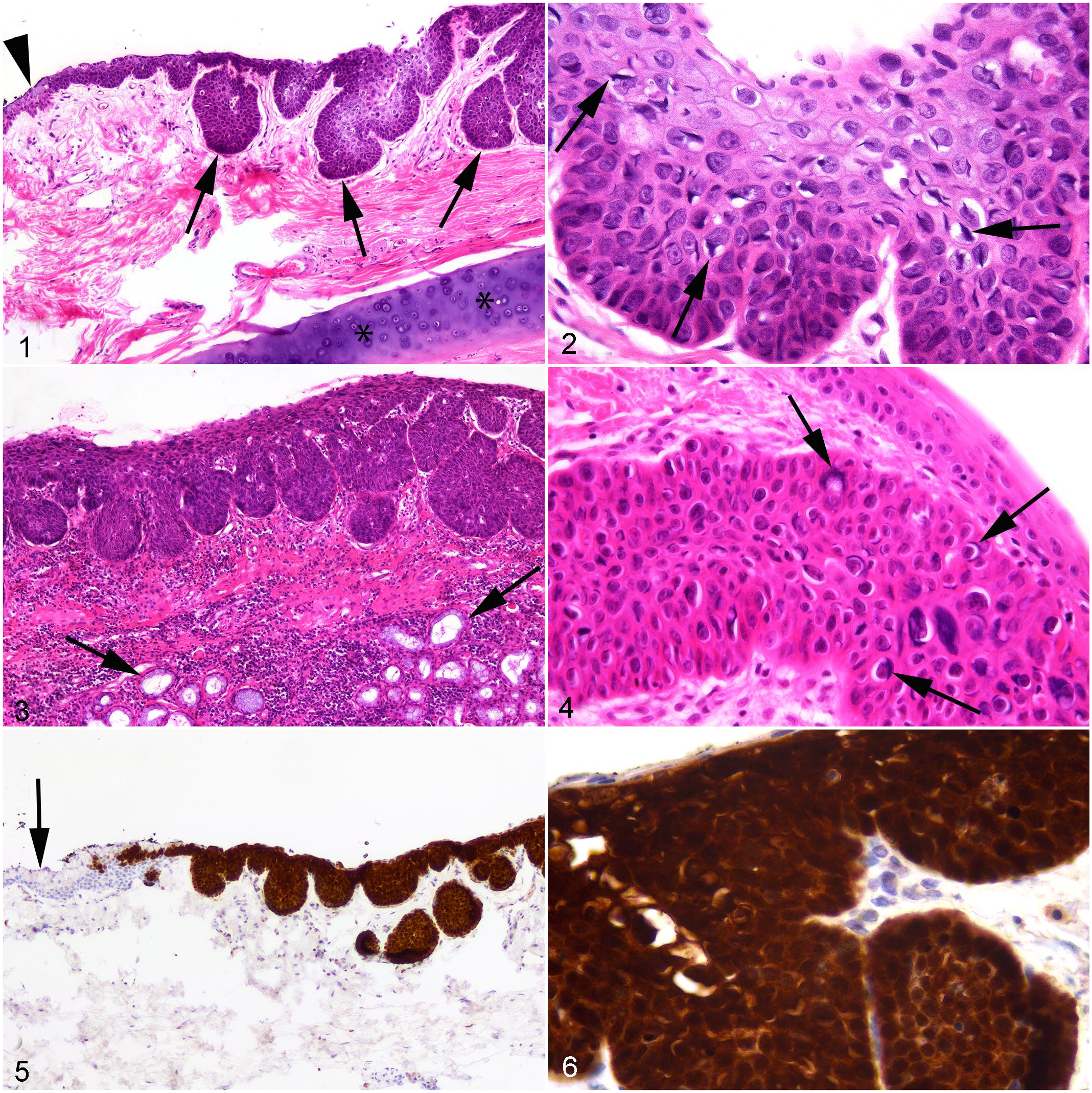
In situ carcinoma, cat.
Two months later, the cat was represented due to oral pain. Examination of the mouth revealed a reddened, slightly raised area of gingiva within the right side of the oropharynx caudal to the dental arcade. An OSCC was suspected and the cat was treated symptomatically for 6 months. He was then anesthetized for repeat cryotherapy of the nasal lesion and two biopsies of the affected gingiva were taken and submitted for histopathology.
Examination of samples from the mouth revealed irregular hyperplasia of the gingival epithelium with the formation of irregular broad trabeculae that bulged into the underlying lamina propria. Epithelial cells in the trabeculae remained confined by the basement membrane (Fig. 3). Normal maturation of the epithelium was lost within the affected epithelium with cells within the basal layers crowded, elongated, and having loss of nuclear polarity. Cells within the middle to more superficial layers of the epithelium had nuclei that were expanded by darkly basophilic material. Some cells also had a clear cytoplasmic halo containing a darkly basophilic body (Fig. 4). While metastasis from the eyelid SCC was considered, it would not be possible for a neoplasm confined to the mucosa to develop by this mechanism.
Due to the histological evidence of PV infection, samples of the masses from the external eyelid, nictitating membrane, and gingiva were excised from the formalin-fixed paraffin-embedded tissue block using a scalpel. DNA was then extracted from these samples using a NucleoSpin DNA FFPE XS kit (Macherey-Nagel GmbH, Duren, Germany) according to manufacturer’s instructions. The consensus primers MY09/11 and CP4/5 were used as previously described 15 and amplified PV DNA from all three masses. Amplified DNA from the three masses was sequenced and found to be between 99.3% and 99.7% similar to the sequence of FcaPV3 that had been previously deposited in GenBank (accession number U83595).
Immunolabeling for p16CDKN2A protein (p16) was performed as previously described. 12 Examination of the in situ carcinomas from the nictitating membrane and gingiva and the SCC from the external eyelid all revealed intense cytoplasmic and nuclear immunolabeling within the neoplastic cells (Figs. 5, 6). Surrounding epithelium contained no significant p16 immunolabeling and no cells with intermediate amounts of immunolabeling were visible within the sections.
Examination of the cat 3 months later did not reveal progression of the oral lesions, although examination of the mouth was difficult as the cat was conscious during examination.
The detection of PV DNA, the presence of prominent PV-induced cell changes within the neoplastic cells, and the consistent intense nuclear and cytoplasmic p16 immunolabeling of the neoplastic cells suggested that the in situ carcinoma within the mouth of this cat was caused by PV infection. This is the first time that molecular, histological, and immunohistochemical evidence of a PV etiology has all been identified within a feline oral neoplasm. In humans, PV-induced OSCCs develop as the result of progression from a PV-induced in situ carcinoma. 6 Therefore, the detection of an in situ carcinoma containing evidence of a PV etiology in the mouth of a cat suggests that, as in humans, a subset of feline OSCCs might develop as the result of progression from a PV-induced precursor lesion.
DNA sequences from FcaPV3 were amplified from the oral neoplasm. This PV type has been previously detected in feline BISCs that, as in the presently described oral in situ carcinoma, contained prominent PV-induced cell changes and intense p16 immunolabeling. 17 FcaPV3 DNA was detected in 1 of 20 feline OSCCs from American cats. 4 However, this is the only previous report of this PV type in OSCCs, and FcaPV3 was not detected in other series of OSCCs from American, New Zealand, or Japanese cats.13,18,23 While evidence suggests that FcaPV3, and possibly other PV types, could cause a subset of feline OSCCs, most OSCCs in cats appear to develop independently of PV infection.
PV DNA has previously been amplified from feline OSCCs using PCR. However, as PVs can be present as asymptomatic infections it can be difficult to determine the significance of this detection. This uncertainty was illustrated by the detection of FcaPV2 in 32% of OSCCs, but also in 36% of samples of nonneoplastic gingiva. 1 In contrast, the presence of the prominent PV-induced cell changes visible in the present study allowed it to be confirmed that the PV was present in the neoplastic cells. In addition, the p16 immunolabeling strongly suggested that PV proteins were being expressed and PV-induced degradation of retinoblastoma protein had altered normal cell regulation. While it could not be confirmed that FcaPV3 caused the cancer, it could be confirmed that FcaPV3 had infected the neoplastic cells and was influencing their behavior. The presence of these cell features therefore provides the strongest evidence so far that PVs can cause neoplasia within the mouth of cats.
The SCCs from the external eyelid and the in situ carcinoma from the nictitating membrane also contained FcaPV3 DNA sequences, PV-induced cell changes, and intense p16 immunolabeling. This is the first time that a neoplasm from the mucosa of the eyelids can be confirmed to contain PV DNA sequences and the first report of PV-induced cell changes in a neoplasm of the eyelid of a cat. The development of multiple in situ and invasive carcinomas of the eyelid has been previously reported in cats. 7 However, these previous lesions were not evaluated for the presence of PV DNA.
The in situ carcinomas of the gingiva and nictitating membrane contained histological and immunohistochemical features that are also present in feline PV-induced BISCs of haired skin.9,21 This is important as BISCs are known to progress to SCCs suggesting a proportion of OSCCs could develop due to progression from a PV-induced oral in situ carcinoma. However, cutaneous SCCs that develop from BISCs are characterized by the presence of PV DNA and p16 immunolabeling.9,10 As PV DNA and p16 immunolabeling appear to be only rarely present in feline OSCCs, OSCCs in cats may only rarely develop due to progression from a PV-induced in situ carcinoma.
In conclusion, this is the first report of histological and immunohistolabeling evidence of a PV etiology of a feline OSCC. Although PVs appear to contribute to, at most, a small subset of OSCCs in cats, findings from this case broaden the range of lesions associated with PVs in cats, and it may be possible to develop novel ways to prevent some feline OSCCs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
