Abstract
Although dogs with acute necrotizing pancreatitis (ANP) can develop respiratory complications, there are no data describing lung injury in clinical cases of ANP in dogs. Therefore, we conducted a study to characterize lung injury and determine if pulmonary intravascular macrophages (PIMs) are induced in dogs with ANP (n = 21) compared with control dogs (n = 6). Two pathologists independently graded histologic sections of pancreas from clinical cases to characterize the severity of ANP (total scores of 3–10) compared with controls showing histologically normal pancreas (total scores of 0). Based on histological grading, lungs from dogs with ANP showed inflammation (median score, 1.5; range, 0–3), but the scores did not differ statistically from the control lungs (median score, 0.5; range, 0–2). A grid intersects-counting method showed an increase in the numbers of MAC387-positive alveolar septal mononuclear phagocyte profiles in lungs of dogs with ANP (ratio median, 0.0243; range, 0.0093–0.0734, with 2 outliers at 0.1523 and 0.1978) compared with controls (ratio median, 0.0019; range, 0.0017–0.0031; P < .0001). Only dogs with ANP showed labeling for von Willebrand factor in alveolar septal capillary endothelial cells, septal inflammatory cells, and alveolar macrophages. Toll-like receptor 4 and interleukin 6 were variably expressed in alveolar macrophages and septal inflammatory cells in lungs from both ANP and control dogs. Inducible nitric oxide synthase was detected in alveolar macrophages of dogs with ANP only. These data show that dogs with ANP have lung inflammation, including the recruitment of PIMs and expression of inflammatory mediators.
Keywords
Dogs with acute pancreatitis (AP) represent a common gastrointestinal cause of emergency admissions in small animal veterinary clinics. 24,25 AP can occur in a mild edematous form or in a severe form, known as acute necrotizing pancreatitis (ANP), 41 which is associated with the development of systemic inflammatory response syndrome and, in some cases, multiple-organ dysfunction syndrome with mortality rates as high as 27% to 58%. 24 Among all potential systemic complications, acute lung injury or its more severe form, acute respiratory distress syndrome, occurs in 18% to 46% of people with severe AP and is the main cause of death in human patients with this condition. 1,8,32,39 Although information on the occurrence of respiratory complications in clinical cases of ANP in dogs is scarce, the data from 2 retrospective studies report respiratory clinical signs in 4 of 61 (7%) 25 and 18 of 80 dogs (23%) with AP. 29 Nevertheless, there has not been any histopathological assessment of lung injury or lung inflammation associated with clinical ANP in dogs.
The pathophysiology of acute lung injury is complex and involves a vascular and a cellular response. Mechanisms include the initiation of local inflammatory cascades, including the expression of adhesion molecules, the release of systemic inflammatory mediators, and the recruitment of leukocytes into the lung microvasculature. Neutrophils are normally the first inflammatory cells to be recruited and sequestrated into the lungs. 50 Ultimately, acute lung injury (ALI) is characterized by the failure of alveolar capillary membranes increasing the lung vascular permeability. Furthermore, the lung inflammatory response is strongly modulated by pulmonary resident macrophages, including alveolar macrophages, interstitial macrophages, and, in some species, pulmonary intravascular macrophages (PIMs).
PIMs are highly phagocytic large cells (20–80 μm in diameter) characterized by phenotypic features of mature macrophages and are found strongly adhered to the endothelium of alveolar septal capillaries. 42 Some species in the orders Artiodactyla (cattle, sheep, goats, and pigs), Perissodactyla (horses), and Cetacea (suborder Odontoceti—toothed whales) and some strains of domestic cat have been shown to have constitutive PIMs. 3,6,42,49 Species in the orders Rodentia (rats, mice, guinea pigs, and hamsters) and Lagomorpha (rabbits) do not normally have PIMs, but studies have shown that they can be induced in those species during the course of pathological conditions, such as endotoxemia and liver dysfunction. 9,20 The rare investigations on dogs and humans suggest a lack of constitutive PIMs in these species. 13 –15
It has been demonstrated in many species that the constitutive or induced PIMs enhance the host susceptibility for lung injury in response to microbial challenges. Overall, PIMs are considered proinflammatory cells, and their depletion results in reduced lung levels of proinflammatory cytokines, improved lung histology, and reduced migration of inflammatory cells. 20,30,46 It is still unknown whether PIMs can be induced in dogs with systemic pathological conditions.
Considerable research has been conducted using rodent models to attempt elucidating the pathophysiology of lung injury associated with AP. Briefly, acute lung injury linked to AP is believed to be multifactorial, resulting from the complex combination of the release into the blood circulation of pancreatic-specific proteases causing vascular injury, activation of Kupffer cells and peritoneal macrophages, and gut barrier dysfunction leading to endotoxemia. 1,8,58 Most studies have focused on the role of alveolar macrophages, which are activated by various mechanisms leading to the acquirement of a proinflammatory phenotype. Following their activation, alveolar macrophages cause lung injury by producing proinflammatory mediators, such as tumor necrosis factor α (TNFα), interleukin (IL)–1β, and Macrophage Inflammatory Protein-2 (MIP-2), resulting in Intercellular Adhesion Molecule-1 (ICAM-1)–mediated recruitment of neutrophils, which promotes further lung injury. 18,45,52 Although significant progress has been made to identify the pathophysiological mechanisms leading to the development of lung injury in rodent models of AP, especially the role of alveolar macrophages, no studies have assessed the recruitment and the role of PIMs during this disease.
The majority of our knowledge on ALI associated with AP comes from studies using rodent models of AP, and very little is known about the specific mechanisms occurring in dogs with ANP. Therefore, the objective of this study was to describe lung inflammation in clinical cases of dogs that died with ANP, with a special focus on the recruitment and localization of PIMs.
Materials and Methods
Selection of Cases and Controls
All the studies included in this article were approved by the University of Saskatchewan Committee on Assurance of Animal Care. The cases of dogs that died or were euthanized with a diagnosis of ANP were selected by searching the electronic medical records of the Veterinary Medical Center of the Western College of Veterinary Medicine (WCVM) between the years of 2000 to 2013 using the keywords necrotizing pancreatitis and pancreas necrosis. Two American College of Veterinary Pathologists (ACVP) board-certified pathologists (A.N.A. and B.K.W.) reviewed the hematoxylin and eosin (HE)–stained slides of the pancreas from all selected cases to confirm the diagnosis of ANP and grade the pancreatic lesions (grading system described below). After this selection process, 21 ANP cases remained.
Six dogs euthanized at the Saskatoon Society for the Prevention of Cruelty to Animals (SPCA) for behavioral reasons (aggressive behavior) were used as control dogs. Each dog was visually evaluated before being euthanized for general health status and for signs of respiratory disease, such as coughing, nasal discharge, and abnormal respiratory patterns. All SPCA dogs appeared clinically healthy. Further clinical evaluation could not be performed under agreements made with the SPCA. To ensure that the SPCA dogs had a histologically normal pancreas and therefore were suitable to be controls in this study, the same 2 ACVP board-certified pathologists reviewed the HE-stained slides of the pancreas from each dog (grading system described below).
Review of Medical Records of Dogs With ANP
Medical records from the ANP cases were reviewed and data regarding the signalment, presence of respiratory clinical signs, blood analysis including leukograms and serum amylase values, presence of Disseminated Intravascular Coagulation (DIC), and concomitant conditions were recorded. If multiple blood analyses were available, leukograms performed within 48 hours prior to death were used. The presence or absence of concomitant peritonitis and endocrine diseases was based on cytologic examination of abdominal fluid and blood analyses, respectively. The presence of peritonitis or concomitant hepatic, renal, and gastrointestinal diseases was recorded based on the postmortem findings. Leukograms and serum amylase were not available for 7 and 10 ANP cases, respectively.
Histopathology and Grading of Lesions
Formalin-fixed, paraffin-embedded lung and pancreas from 21 ANP cases were provided by the Prairie Diagnostic Services of the WCVM. One paraffin block per animal per organ was used. Lung and pancreas from control dogs were sampled for fixation in 4% paraformaldehyde overnight at 4°C (between 16 and 20 hours) followed by paraffin embedding.
Two ACVP board-certified pathologists graded independently and in a blinded manner pancreas histologic sections from all ANP and control dogs. Grading was based on the following criteria: Necrosis: 0 = absence; 1 = <10% of the pancreas parenchyma; 2 = 10% to 25% of the pancreas parenchyma; 3 = 25% to 50% of the pancreas parenchyma; 4 = >50% of the pancreas parenchyma Edema: 0 = absence; 1 = focally increased space between lobules; 2 = diffusely increased space between lobules; 3 = pancreatic acini disrupted and separated Inflammatory cell infiltrate: 0 = absence; 1 = rare or around ductal margins; 2 = in the pancreas parenchyma (<50% of the lobules); 3 = in the pancreas parenchyma (>50% of the lobules). The primary type of inflammatory cells (neutrophils, monocytes, lymphocytes, or mixed) infiltrating the pancreas was also recorded. Pancreas histology score: sum of necrosis, edema, and inflammatory cell infiltrate scores. The scores generated by both pathologists were averaged for each of the categories.
The same 2 board-certified pathologists graded independently and in a blinded manner lung histologic sections from all ANP and control dogs for signs of lung inflammation. The primary type of inflammatory cells (neutrophils, mononuclear phagocytes, or mixed) infiltrating the pulmonary alveolar septa was recorded for each dog. Grading was based on the following criteria: Edema: 0 = absence; 1 = mild; 2 = moderate; 3 = severe Inflammatory cell infiltrate: 0 = absence; 1 = mild; 2 = moderate; 3 = severe Lung histology score: sum of edema and inflammatory cell infiltrate scores
The scores generated by both pathologists were averaged for each of the categories.
Immunohistochemistry
Immunohistochemical labeling was performed on lung sections from all dogs with ANP and controls, as previously described. 47 Briefly, sections were deparaffinized, rehydrated in descending concentrations of ethanol, and incubated with 0.5% H2O2 in methanol for 20 minutes to quench the endogenous peroxidase activity. Antigen retrieval was performed using pepsin (2 mg/ml in 0.01 N HCl) for 1 hour (for macrophage, IL-6, inducible nitric oxide synthase [iNOS], and von Willebrand factor [vWF] labeling) or by heat-induced epitope retrieval using boiling citrate buffer (pH 6.0) for 20 minutes (for Toll-like receptor 4 [TLR4] labeling), after which blocking was done with bovine serum albumin (BSA) (1% in phosphate-buffered saline [PBS]) for 30 minutes at room temperature. Slides were incubated overnight (16 hours) at 4°C with the following primary antibodies: MAC387 (1:75; AbD Serotec), IL-6 antibody sc-1265 (1:50), TLR4 antibody sc-12511 (1:50), and iNOS antibody (1:25) (all 3 from Santa Cruz Biotechnology, Dallas, Texas, USA) and vWF antibody P0448 (1:500; DAKO, Ottawa, Canada). Then, sections were incubated for 30 minutes at room temperature with the appropriate horseradish peroxidase (HRP)–conjugated secondary antibody (1:100, except against vWF: 1:300, all from DAKO). Color development was performed using a commercial kit (Vector Laboratories, Ontario, CA) followed by counterstaining with methyl green to delineate the nuclei.
Immunohistochemical controls consisted of omission of the primary antibody for all different secondary antibodies used and incubation with the appropriate isotype-matched antibodies. In addition, labeling with vWF was used as a positive control. Both negative controls resulted in an absence of labeling, whereas vWF labeled the vascular endothelium but not the airway epithelium.
Quantification of Cells Labeled With MAC387 Antibody
The MAC387 antibody binds to calprotectin, 7 which is mainly present in macrophages but can also be present in monocytes and neutrophils. Therefore, based on the nuclear morphology, only positively labeled mononuclear phagocyte profiles present in alveolar septa were counted, while discounting neutrophil nuclear profiles. A 2-dimensional (2D)–modified morphological analysis was used to count the number of positively labeled cells; photographs of 15 random fields of view at 600× magnification were captured using a light microscope (Olympus BX53; Olympus, Richmond Hill, Ontario, Canada) and a digital camera (Olympus DP21) for each control and dogs with ANP. Fields of view including large blood vessels or airways were ignored, continuing to the following field of view in a constant predetermined pattern. Then, using ImageJ software (1.49v Rasband, W.S.; ImageJ, National Institutes of Health, Bethesda, Maryland), a 12 × 12 intersects-counting grid covering 15 800 μm2 was superimposed onto each microscopic captured field of view. For every field of view, the number of grid intersects overlaying MAC387-positive mononuclear phagocyte profiles present in alveolar septa was recorded. This was performed for every control and ANP dog. To normalize for the potential changes in lung morphology between cases due to compression or expansion artifacts during sampling and fixing of lung tissues, the number of grid intersects overlaying alveolar septa in each captured image was simultaneously recorded. Totals of grid intersects overlaying positive MAC387 mononuclear phagocyte profiles and alveolar septa were calculated using all 15 fields of view per animal. Then, for each control and ANP dog, a ratio was calculated dividing the total number of grid intersects overlaying positive MAC387 mononuclear phagocyte profiles by the total number of grid intersects overlaying alveolar septa, thus controlling for the variability of lung morphology alterations between cases.
Lung Electron Microscopy
As only paraffin blocks of lung tissues were available for ANP cases, 20-μm sections were prepared from the blocks from 4 dogs with ANP. Sections were deparaffinized, dehydrated, and embedded in Epon while still on the glass slides. Following this, the sections were scraped and transferred to plastic capsules for further polymerization in Epon and sectioning, as described previously. 5 The sections were examined in a Hitachi transmission electron microscope (Hitachi, Illinois, USA).
Statistical Analysis
Data were analyzed using GraphPad Prism 6 (GraphPad Software, La Jolla, California). Data from the histological grading and MAC387 labeling grid-intersects ratios are expressed as median with the range. Comparisons between medians were done with the nonparametric Mann-Whitney U test.
Results
Clinical Description of Dogs With ANP
Various breeds were represented in the ANP group with no sex predisposition. Dogs with ANP were generally older with a median age of 8 years. The signalment for ANP and control dogs is included in Supplemental Table S1. All dogs with ANP except for 3 were presented to the emergency clinic and died or were euthanized within 48 hours. The other 3 dogs were admitted 5 to 10 days prior to death. Clinical evidence suggesting pulmonary complications was recorded in 4 of 21(19%) dogs and included pleural effusion in 3 dogs (1 dog with dyspnea) and increased lung sounds in the fourth dog. Concomitant conditions/complications included liver diseases, kidney diseases, peritonitis, DIC, diabetes mellitus, diabetic ketoacidosis, and enteritis. The occurrence of each concomitant condition/complication and the results from blood analysis are described in Supplemental Table S2.
Pancreas Histologic Grading
The pancreas histology scores for SPCA dogs were zero in every grading category. The scores from each pancreas histology grading category were added to give the total pancreas histology scores for each dog with ANP; the median was 4.5 and the range was 2.5 to 9.5 (Fig. 1), indicating the presence of some variation in the severity of pancreatitis among cases. The presence of pancreatic necrosis confirmed the diagnosis of ANP. The type of pancreatic inflammation when present was mainly neutrophilic (n = 12/17) but also monocytic (n = 3/17), mixed (n = 1/17), and lymphocytic (n = 1/17).
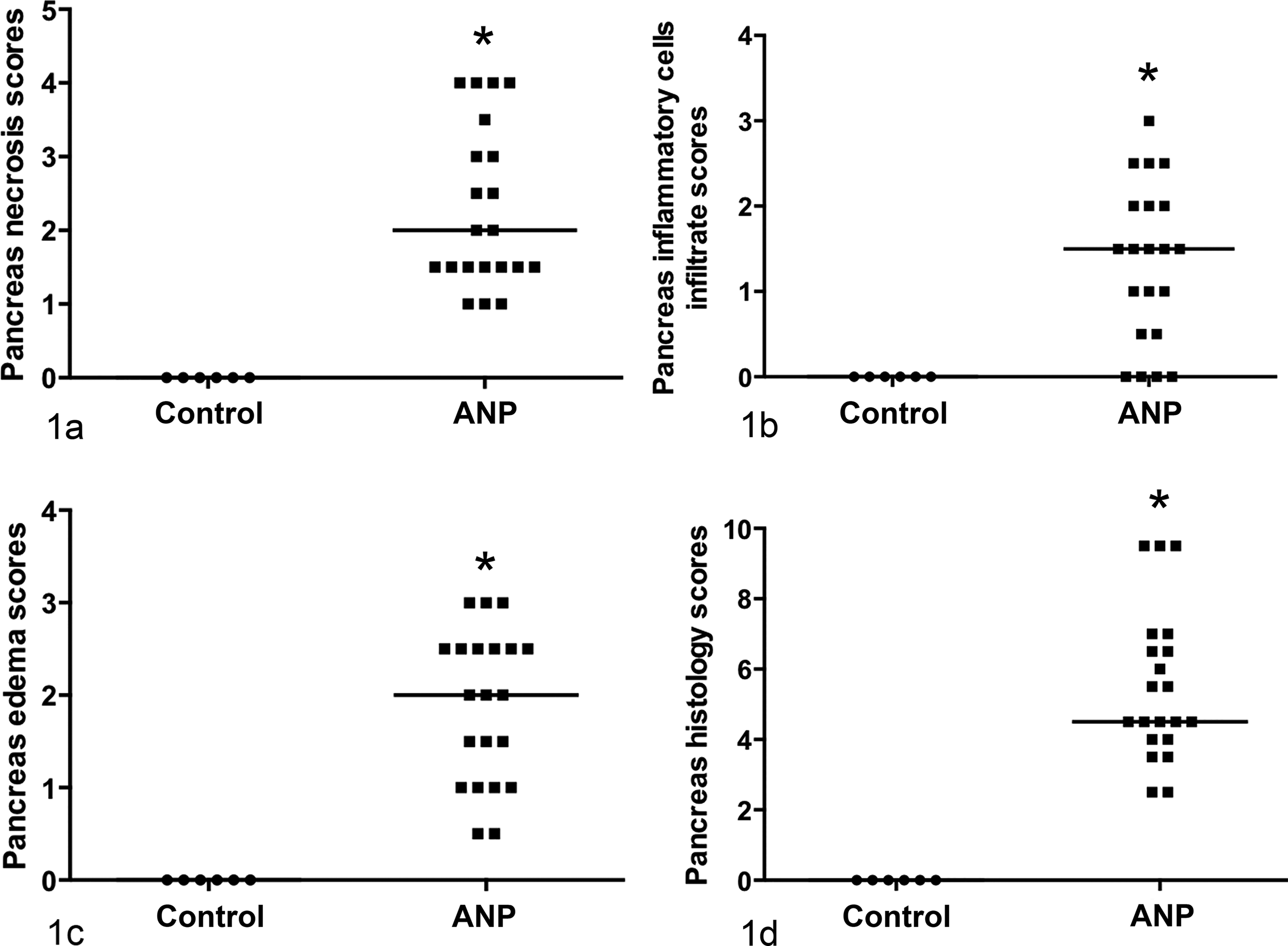
Pancreatic histology grading scores of dogs with acute necrotizing pancreatitis (ANP) (n = 21) and control (n = 6) dogs. (a) Pancreatic necrosis scores. (b) Inflammatory cell infiltrate scores. (c) Pancreas edema scores. (d) Total histology scores (representing the sum of the 3 different scoring categories). Median values are represented by horizontal lines. Significant differences from control dogs at P < .05 are denoted with asterisks (*), Mann-Whitney U.
Lung Histologic Grading
Histologically, most dogs with ANP had accumulation of inflammatory cells, mostly mononuclear cells, in alveolar septa, and an increase in alveolar macrophages compared with control dogs (Figs. 2, 3). In addition, 12 of 21 (60%) dogs with ANP presented alveolar edema. However, lung histology scores for dogs with ANP were not statistically different from those of the control dogs (Fig. 4).
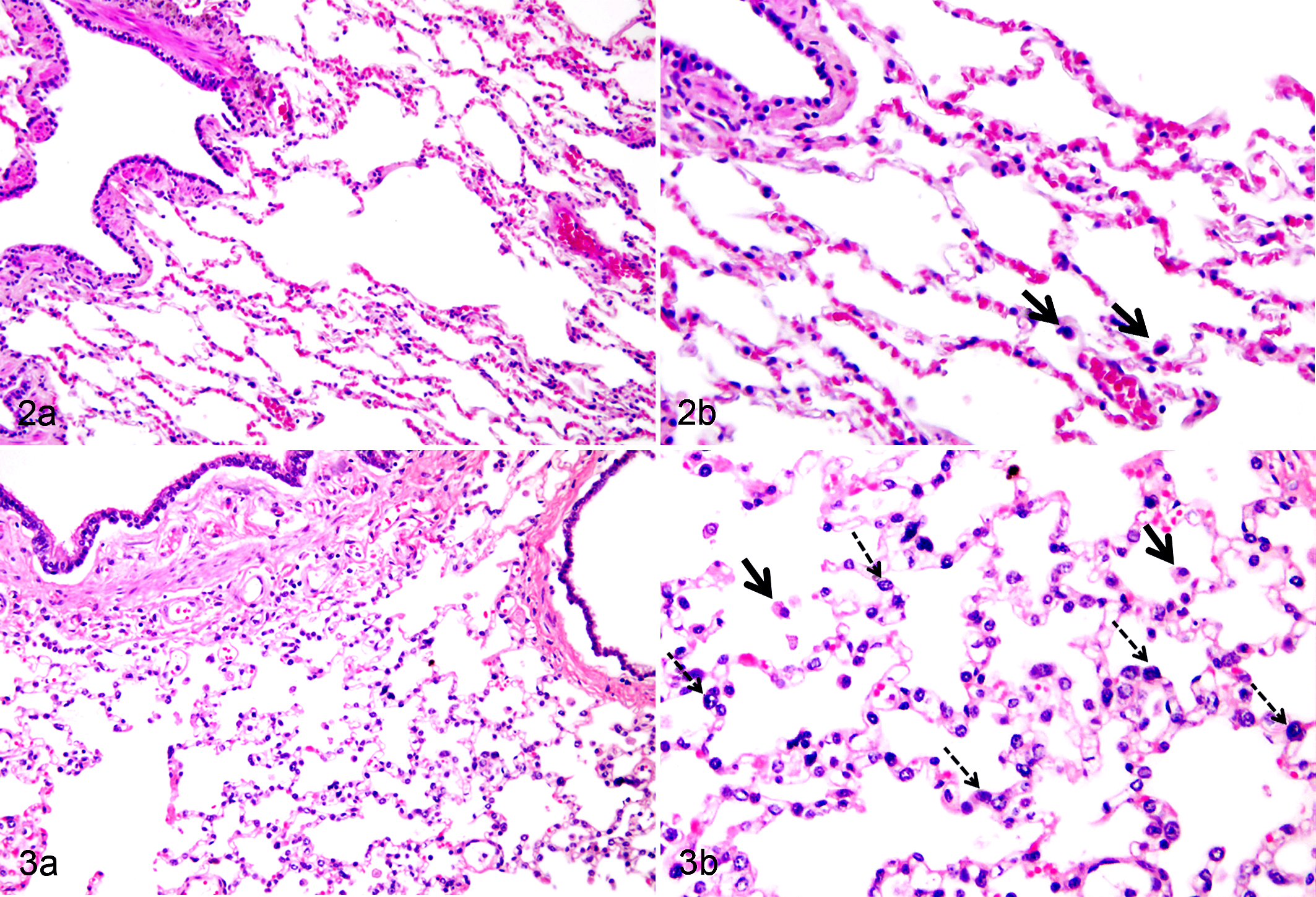
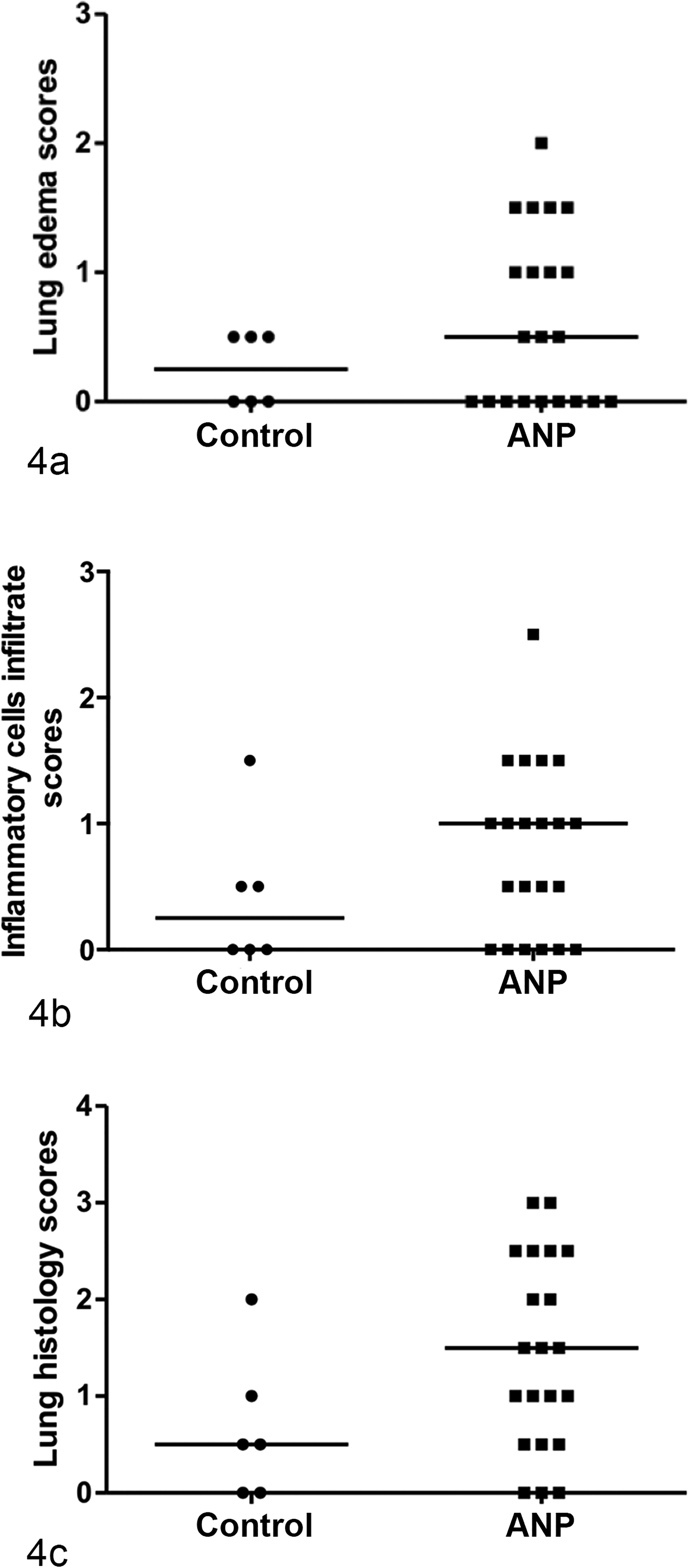
Lung histologic grading scores for dogs with acute necrotizing pancreatitis (ANP) (n = 21) and control dogs (n = 6). (a) Lung edema scores. (b) Inflammatory cell infiltrate scores. (c) Lung histologic scores (representing the sum of the 2 different scoring categories). Median values are represented by horizontal lines.
MAC387 Immunohistochemistry
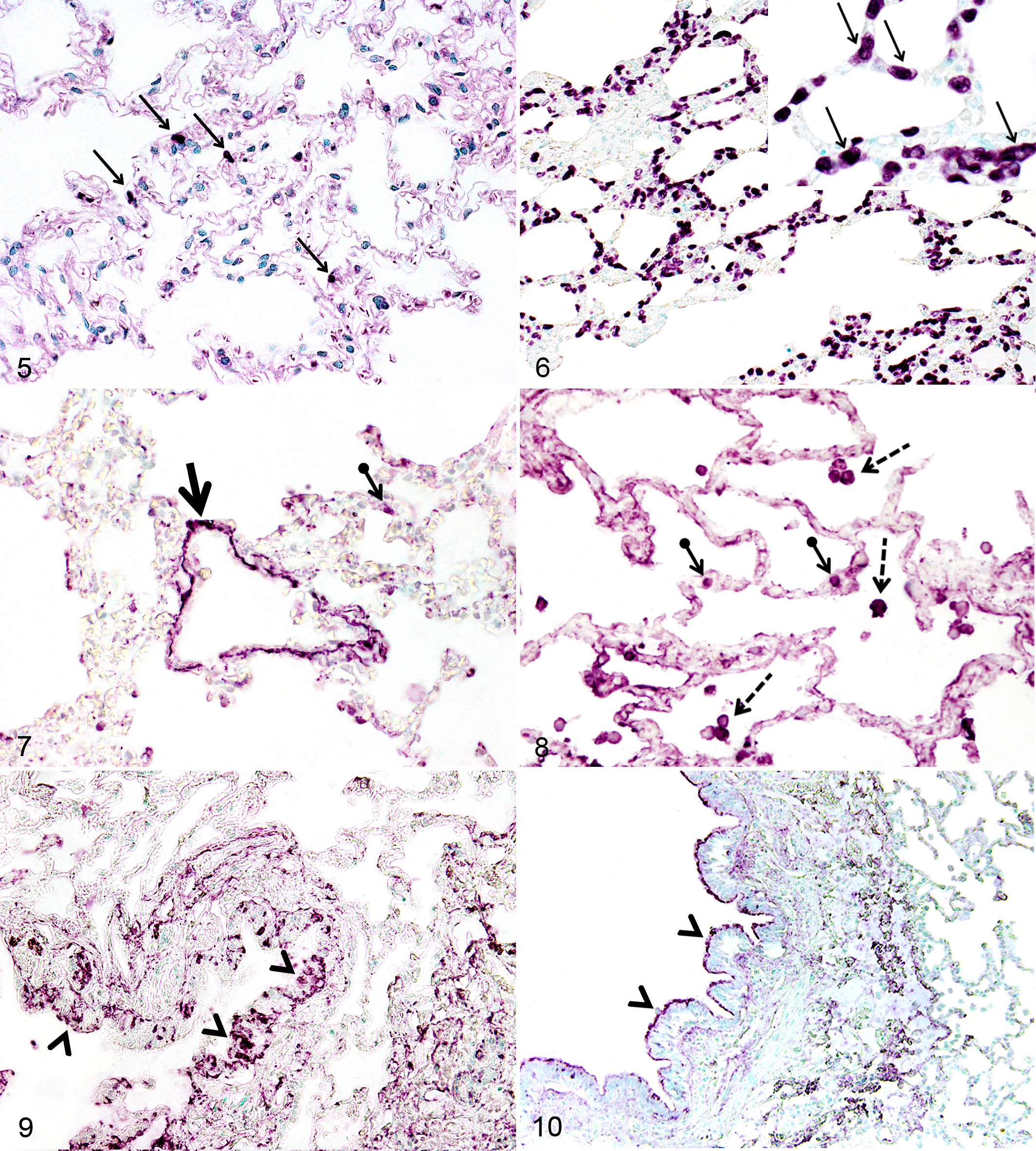
Immunohistochemical labeling with MAC387 revealed an increased infiltration of mononuclear phagocyte profiles in the pulmonary alveolar septa of dogs with ANP compared with controls (Fig. 5, 6). The potential impact of unequal conservation of morphology between cases on the interpretation of MAC387-positive cell profile counts was eliminated through the ratio of the total number of grid intersects overlaying MAC387-positive mononuclear phagocyte profiles to the total number of grid intersects overlaying alveolar septa. The results showed a significant increase in such ratios in ANP dogs (ratio median, 0.0243; range, 0.0093–0.0734, with 2 outliers at 0.1523 and 0.1978) compared with control dogs (ratio median, 0.0019; range, 0.0017–0.0031; P < .0001, Mann-Whitney U, dogs with ANP; n = 21, controls; n = 6) (Suppl. Fig. S1).
Lung Electron Microscopy
We confirmed the intravascular location of macrophages to denote them as PIMs by performing electron microscopy (EM) on tissues rescued from paraffin blocks. The data show many PIMs containing lysosomes in addition to being in apposition with the endothelium of alveolar septal capillaries (Suppl. Fig. S2).
TLR4 Immunohistochemistry
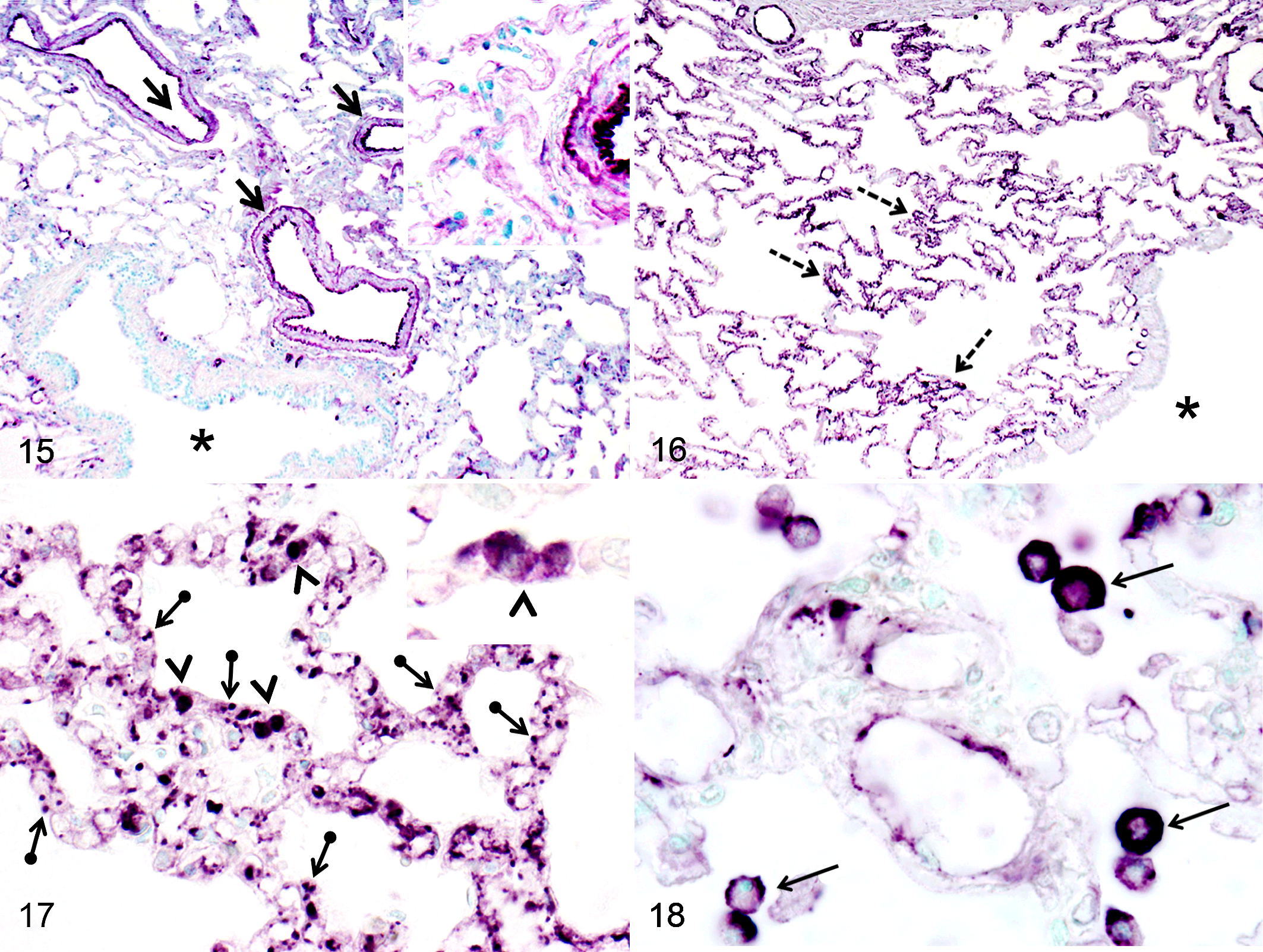
Every control dog and 18 of 21 (86%) dogs with ANP showed TLR4 labeling in the vascular endothelium of medium-sized blood vessels (Fig. 7). Only faint occasional labeling was seen in alveolar septa of lungs from control dogs (Fig. 7). Control dogs lacked TLR4 labeling in alveolar macrophages, whereas half of dogs with ANP showed strong labeling in the cytoplasm and nucleus of alveolar macrophages (Fig. 8). In addition, 18 of 21(86%) dogs with ANP showed labeling for TLR4 in mononuclear phagocytes in alveolar septa, and 4 of 21 (19%) dogs with ANP showed labeling of the alveolar septal capillary itself (Fig. 8). Some basal labeling for TLR4 was present in the cytoplasm or in the intercellular space of airway epithelial cells in every control dog (Fig. 9). Interestingly, the labeling pattern for TLR4 in airway epithelial cells in dogs with ANP varied from cytoplasmic labeling in 4 of 21 dogs (19%), to apical labeling in 3 of 21 dogs (14%) (Fig. 10), to basal labeling in 9 of 21 dogs (43%), to granular and irregular labeling of the cytoplasm as in control dogs in 8 of 21 dogs (38%), and to a lack of labeling of some airway epithelium in 8 of 21 dogs with ANP (38%) (Table 1).
Number of Lung Samples From Control Dogs and Dogs With ANP Positive by Immunohistochemistry for Various Antigens.
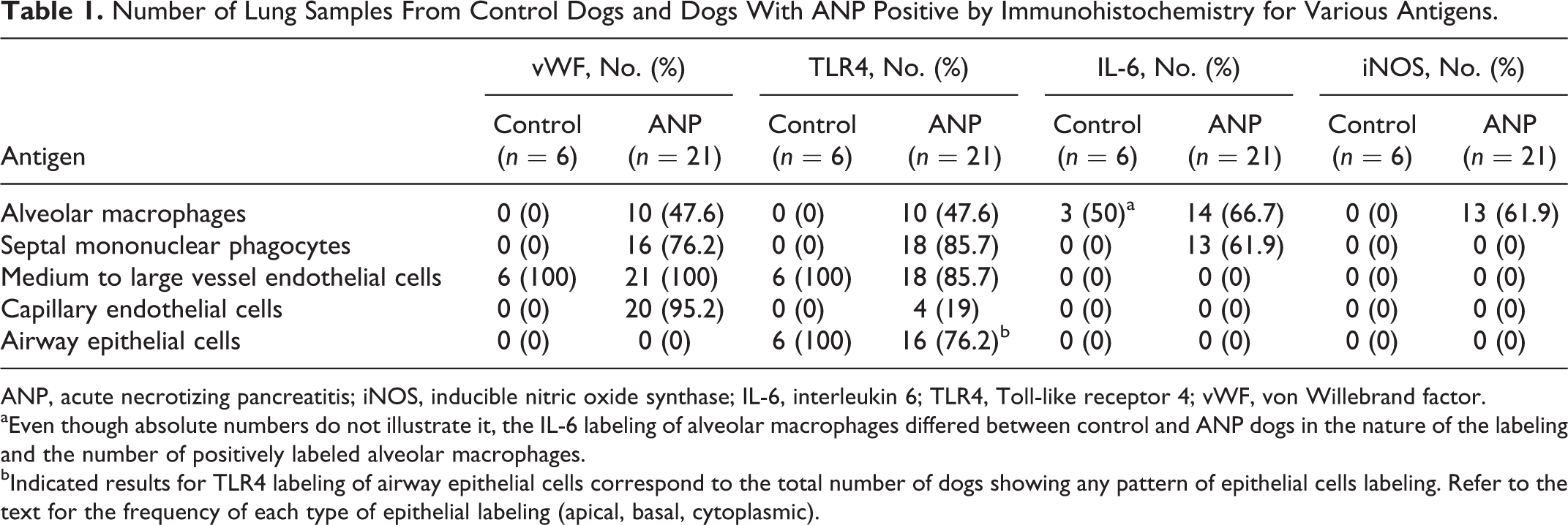
ANP, acute necrotizing pancreatitis; iNOS, inducible nitric oxide synthase; IL-6, interleukin 6; TLR4, Toll-like receptor 4; vWF, von Willebrand factor.
aEven though absolute numbers do not illustrate it, the IL-6 labeling of alveolar macrophages differed between control and ANP dogs in the nature of the labeling and the number of positively labeled alveolar macrophages.
bIndicated results for TLR4 labeling of airway epithelial cells correspond to the total number of dogs showing any pattern of epithelial cells labeling. Refer to the text for the frequency of each type of epithelial labeling (apical, basal, cytoplasmic).
IL-6 Immunohistochemistry
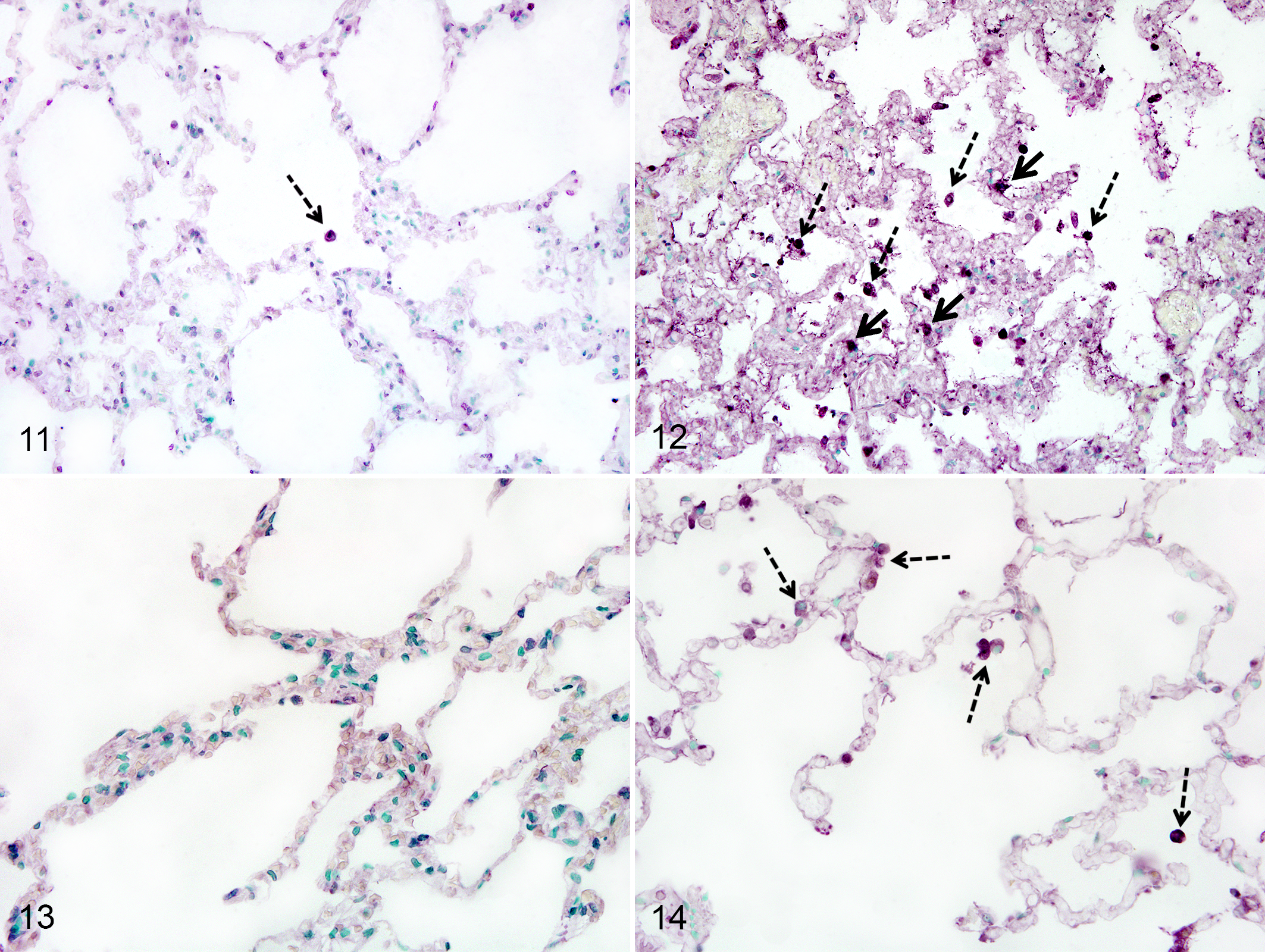
The IL-6 labeling was mainly observed in alveolar macrophages and mononuclear phagocytes present in alveolar septa. While nearly half of the control dogs rarely showed an alveolar macrophage positive for IL-6 (Fig. 11), 14 of 21 (66%) dogs with ANP showed robust labeling for IL-6 in alveolar macrophages and in some mononuclear phagocytes present in alveolar septa (Fig. 12, Table 1). There was no labeling of other structures of the lungs of dogs with ANP or controls.
iNOS Immunohistochemistry
iNOS labeling was identified in alveolar macrophages of 13 of 21 (62%) dogs with ANP (Fig. 14). iNOS labeling was absent from the alveolar macrophages of control dogs (Fig. 13). There was no labeling of other structures of the lungs of dogs with ANP or controls (Table 1).
vWF Immunohistochemistry
All dogs with ANP and controls showed vWF labeling of endothelial cells of larger blood vessels, including arteries, arterioles, venules, and veins (Fig. 15). However, in dogs with ANP, there was also strong labeling in alveolar septal capillaries, seen as granular labeling, which was not observed in control dogs (Figs. 16, 17). Also, dogs with ANP showed intracytoplasmic labeling for vWF in mononuclear phagocytes infiltrating alveolar septal capillaries (Fig. 17) and, in some cases, in alveolar macrophages (Fig. 18). These 2 findings were not observed in control dogs (Table 1).
Discussion
Until now, there was only a single case report describing histologic changes in the lung of a dog with ANP, involving a 3-year-old dyspneic miniature poodle dog. 23 To our knowledge, this is the first systematic evaluation of the lung histopathology in clinical cases of ANP in dogs. Although only 4 of 21(19%) dogs with ANP had respiratory clinical signs based on the review of medical records, most dogs with ANP showed histological lesions of lung inflammation and increased levels of inflammatory markers such as IL-6, TLR4, and iNOS. In addition, immunohistochemical analysis and EM revealed a significant increase in numbers of mononuclear phagocytes in alveolar septal capillaries of dogs with ANP, which suggests that PIMs are induced in this condition. Interestingly, intense vWF labeling was noticed in the endothelium of alveolar septal capillaries in dogs with ANP, suggesting that the lung microvasculature is inflamed in this disease. The histology, immunocytochemistry, and EM results show hitherto unknown recruitment of PIMs in dogs with ANP.
Using labeling with the MAC387 antibody and EM, this study reports evidence that mononuclear phagocytes are markedly increased in alveolar septa of the lungs of dogs with ANP compared with control dogs. We used a well-established system that controls, to a degree, for the lung volume changes caused by different fixation methods and provides reliable cell profile counts based on a ratio of line intersects. We selected the MAC387 antibody based on publications that described its use for the identification of canine macrophages 19 and its use to identify PIMs with immunofluorescence, immune-electron microscopy, and immunohistochemistry in horses, rats, and humans. 30,31,42 Furthermore, calprotectin or L1 antigen, which the MAC387 antibody labels, has been shown to be formalin resistant and is considered a useful marker for macrophages in routinely processed clinical material. 7 Although there is always a challenge with light microscopy to distinguish cells residing in the alveolar septal capillaries versus the interstitium, it is well accepted that the basement membranes of pulmonary capillary endothelial cells and alveolar epithelial cells are mostly fused except at the thick part of the alveolar septum. Therefore, the interstitium may not provide the space required to contain large numbers of cells as large as monocytes or macrophages, which supports that MAC387-positive cells are located in the capillaries of alveolar septa. The electron microscopic data conclusively prove the intravascular and not interstitial location of macrophages in lungs of dogs with ANP. The intravascular cells showed ultrastructural characteristics of macrophages, including adhesion to endothelial cells and the presence of lysosomes. To our knowledge, this is the first evidence of the induction of PIMs in the lungs of dogs with a clinical condition. Only 1 other study has showed the absence of constitutive or induced PIMs in 2 normal dogs and 2 dogs receiving a transplant of dead Dirofilaria immitis adult nematodes. 15 It is interesting to note that unlike the data from most studies using rodent models of AP, the lungs from dogs with ANP did not show recruitment of neutrophils in alveolar septa or alveoli. 21
PIMs are now established as proinflammatory regulators of lung inflammation in species where they are constitutively present, as well as those in which they are induced during pathological conditions. Indeed, protective effects of PIMs’ depletion were noted in models of ALI in species with constitutive PIMs, such as in endotoxin-mediated lung injury in sheep 48 and horses 31 and in calves infected with Mannheimia hemolytica. 46 Proinflammatory signaling pathways in PIMs are suggested to occur through TLRs, such as TLR4, TLR9, and TLR2, during sepsis and endotoxemia. 43,47 Following activation of TLRs, PIMs potentially produce large amounts of proinflammatory mediators such as TNF-α and IL-1β, which can stimulate local endothelial cells or cross alveolar septa to stimulate alveolar macrophages. 10,12,20 Histologically, PIMs’ stimulation is associated with vascular congestion and the recruitment of inflammatory cells. 10 Also, PIMs’ activation was linked to the aggregation of IL-8–rich platelets in the lungs of calves challenged with intratracheal M. hemolytica. 46 Based on our understanding that PIMs exacerbate lung inflammation following microbial challenges, their presence in the lungs of dogs with ANP could have significant detrimental consequences, especially in patients with blood-borne microbes or microbial molecules such as endotoxins. 2,36,37
Since endotoxins or bacterial translocation to the blood circulation is a possible occurrence during ANP, the expression of TLR4 in the lungs of dogs with ANP was assessed with immunohistochemical labeling. 58 The labeling for TLR4 observed in control dogs corresponds to what was previously reported in normal lungs from cattle, pigs, and horses. 27,47,53 In lungs of dogs with ANP, TLR4 labeling was localized in the numerous newly recruited mononuclear phagocytes, mostly PIMs, and in alveolar macrophages of some cases. TLR4 labeling in PIMs has been shown in pigs, horses, and cattle, 47,53 and the role of PIMs in adding to the total amount of TLR4 messenger RNA (mRNA) in the lung was demonstrated through significant reduction in TLR4 mRNA amount following depletion of PIMs. 43,47 The recruited PIMs and alveolar macrophages in dogs with ANP may increase the total amount of TLR4 in the lungs, which would increase the susceptibility to endotoxin-induced lung inflammation as well as contribute to the release of cytokines into circulation. Interestingly, in contrast to the control dogs, some medium-sized blood vessels and airway epithelium in dogs with ANP did not show TLR4 labeling. These observations were also reported in inflamed lungs from M. hemolytica–infected calves and Pasteurella multocida–infected water buffalo. 44,53 The reason for this loss of TLR4 expression in blood vessels and airway epithelium is unclear but could be a protective mechanism to reduce endotoxin-induced inflammation of the infected lungs. Furthermore, it has been shown in rats that acute hemorrhagic necrotizing pancreatitis was associated with lung injury and led to an increased expression of TLR4 in the lungs assessed by mRNA and protein expression. 56
The proinflammatory response in lungs of dogs with ANP was further assessed using immunohistochemistry for IL-6 and iNOS, which are important mediators of inflammation. Similarly to the expression of TLR4, the labeling for IL-6 in lungs of dogs with ANP was most likely due to the recruitment of PIMs and activation of alveolar macrophages, suggesting a proinflammatory role for these cells in ANP. Some ANP cases presented iNOS labeling in alveolar macrophages, which correlates with various experimental AP studies reporting high expression of iNOS in alveolar macrophages. The resulting increased levels of nitric oxide in the lungs have been associated with controversial roles depending on the studies; in some studies, nitric oxide is associated with further lung injury, 11,52 whereas in others, nitric oxide was protective and reduced the severity of acute lung injury. 56 Although this is only speculation, the induction of iNOS in lung macrophages may constitute a protective mechanism against lung injury during ANP.
Last, another major finding in this study was the robust labeling for vWF in alveolar septal capillaries (ie, the microvasculature, capillaries of ≤10 μm in diameter) in dogs with ANP compared with the lack of labeling in the same compartment in the lungs of control dogs. The paucity of vWF labeling in the microvasculature compared with the macrovasculature (arteries, arterioles, venules, and veins) in normal healthy lungs has been reported previously in humans and in animals with results similar to our findings. 22,35,55 In addition, the marked vWF labeling observed in endothelial cells of the lung microvasculature of dogs with ANP, compared with control dogs, was similarly reported in other studies describing vWF in inflamed lungs or lungs with vascular injury. 38,40
Causes for the differences in the vWF labeling patterns between the microvasculature and the macrovasculature in healthy lungs have not yet been elucidated. Nevertheless, it is well recognized that endothelial cells from the macrovasculature and from the microvasculature circulation present important phenotypic and functional heterogeneity, including variations in the transcription of vWF. 28,35 Furthermore, endothelial cells of alveolar septal capillaries (≤10 μm in diameter), unlike larger blood vessels of the lung, do not possess cytoplasmic Weibel-Palade bodies, 17,57 which store vWF along with IL-8 and P-selectin. 54 Whether vWF is stored or not in quiescent endothelial cells of the lung microvasculature remains unclear. Nevertheless, during vascular injury, endothelial cells from alveolar septal capillaries synthesize and secrete vWF, which does not normally occur in healthy alveolar septal capillary endothelium. 26 This suggests that a stimulus is required to initiate transcription and subsequently for the production of vWF to occur in endothelial cells of the lung microvasculature. During acute pancreatitis, it is known that pancreatic proteases, including trypsin, elastase, and phospholipase A2, are released into the blood circulation and can cause vascular injury to various organs, including the lungs. 32,51 Therefore, it is possible that in dogs with ANP, circulating pancreatic enzymes cause endothelial cells of the lung microvasculature to synthesize and release vWF. After being released, some vWF remains attached to the endothelial cell surface to allow adhesion of platelets during initiation of the coagulation cascade, while a portion is secreted in the blood circulation. 16 The granular vWF labeling pattern lining the capillary endothelial cells may be interpreted as localized accumulation of vWF on the endothelial cell surface or clusters of circulating vWF present in the capillary lumen.
The presence of vWF in alveolar septal capillaries of dogs with ANP could have important roles in promoting or exacerbating lung inflammation. Indeed, in addition to being involved in hemostasis, vWF has a direct role in inflammatory processes such as providing binding sites for leukocyte receptors, including P-selectin glycoprotein ligand 1 (PSGL-1) and integrin-β2, and therefore promotes rolling and adhesion of leukocytes to the endothelium. 34 It was also reported recently that vWF can bind to the cell receptor named Singlec-5 present in various human immune cells, including macrophages, which led to the internalization of vWF in early endosomes. 33 A similar mechanism could be occurring in dogs with ANP, as PIMs and alveolar macrophages of some cases showed intracytoplasmic labeling for vWF. Finally, expression of vWF in the lung microvasculature of dogs with ANP may have indirect proinflammatory effects as it allows platelets to aggregate in the lungs, and various inflammatory roles have now been attributed to platelets, including facilitating leukocyte recruitment to the site of inflammation. 4 The recruitment of platelets by vWF may also be one of the mechanisms for the subsequent recruitment of PIMs in the lungs of dogs with ANP.
This study could have benefited from a larger sample size for both control and dogs with ANP to improve the statistical power. Also, using retrospective clinical cases of ANP in dogs brings the challenge of dealing with varied clinical factors that may influence the findings in the lung. These factors may include the time between the onset of ANP and death; treatments received during hospitalization such as anti-inflammatory drugs, analgesics, diuretics, and antibiotics; and history of lung diseases. In addition, the presence of concomitant conditions/complications in the ANP cases was reported in this study, but their effect on the lungs could not be assessed. Nevertheless, such variations are expected in clinical studies and are a major strength compared with rodent models. In addition, pancreatic enzymes that have been suggested to be involved in the pathogenesis of lung injury associated with AP, including trypsin, elastase, and phospholipase A2, are not routinely assessed in a clinical setting. Assessment of these enzymes would have strengthened the study. This study was also limited to the use of paraffin lung tissues only. Thus, we could not perform stereology analysis or immuno-electron microscopy, and those samples were not optimal for electron microscopy, explaining the reduced quality of the images.
Because there has been evidence of severe respiratory signs in clinical cases of AP in dogs, we sought to determine recruitment of PIMs in this study. The data show significant recruitment of septal mononuclear phagocytes, mostly PIMs and increased expression of vWF and iNOS in the lungs of dogs with ANP. The data suggest that activation of PIMs by circulating inflammatory molecules may incite pulmonary complications in dogs with ANP.
Footnotes
Acknowledgements
We are thankful to Ms. Laura Johnson for her help with the counting of pulmonary intravascular macrophages.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported through grants from the Companion Animal Health Fund of the Western College of Veterinary Medicine and the Natural Sciences and Engineering Research Council of Canada. Dr. Vrolyk was supported through a Fellowship from the Interprovincial Graduate Fund of the Western College of Veterinary Medicine.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
