Abstract
Seven emu chicks (Dromaius novaehollandiae) from a farm with poor hatchability (16–18%) and increased neonatal mortality were presented for necropsy with a history of death at or within a few days after hatching. Macroscopic examination revealed subcutaneous edema and hemorrhages and swelling of the pipping muscles in the proximal neck (71%), pale liver with hemorrhages (71%), noninternalized residual yolk sac (86%) and anasarca (14%). Histologically, the most remarkable findings were necrosis of the musculus complexus (100%) of the pipping muscles, as well as myocardial necrosis and mineralization (29%). Liver contained severe multifocal hepatocellular necrosis and hemorrhages (57%), and both eyes exhibited swollen and vacuolated lenticular fibers in 5 chicks (100%) in which the eyes were examined. The lesions observed here are suggestive of a nutritional deficiency. The deficiency was confirmed by finding low levels of vitamin E in the liver, and vitamin E and vitamin A levels in the feed.
Emus’s natural reproductive success rate is not fully reproducible in artificial rearing conditions, where hatchability is highly variable and relatively low (36%-73%) and an increased posthatching loss occurs. 10 Several factors affect hatching success rate, including breeders’ fertility and age, genetics, incubation management and nutrition.
Vitamin E is considered one of the key factors in ratite nutrition and management, since its absorption is limited in newly hatched birds and thus the embryonic stores may be depleted before the onset of an efficient dietary assimilation. 10
Few cases of encephalomalacia and nutritional myopathy caused by vitamin E deficiency have been described in adult emus, 1,7 while reports on similar lesions in embryos or newly hatched chicks are lacking. The aim of this case report was to describe and characterize the gross and histological lesions in skeletal muscles, liver and eye of vitamin E-deficient emu chicks. Other deficiencies (vitamin A, zinc) are considered as contributing factors to the severity of the lesions.
One 1-day-old, female, dead emu chick and 6 emu eggs, containing fully developed chicks but dead in shell, were presented to the California Animal Health and Food Safety Laboratory System (Tulare Brach) of the University of California, Davis for diagnostic investigation. In addition, feed samples were submitted for biochemical analyses. The 7 chicks originated from a farm of 130 emus with a history of poor hatchability (16–18%) and increased neonatal mortality.
Complete necropsy and histopathological examination were performed on all birds.
The weight of the dead chick was 330 g (reference weight: 400–500 g). The most striking gross lesions were subcutaneous edema, hemorrhages and swelling of the pipping muscles in the proximal neck (Fig. 1) and a yellow discoloration of the liver, with few petechial hemorrhages (Fig. 2). All 6 eggs contained fully developed chicks within a broken shell. The shell was broken by the owner to facilitate hatching. The mean egg weight of 6 eggs was 668 g (range: 550 – 1040 g; reference weight: 500–700 g). Grossly, a noninternalized residual yolk sac, containing abundant mucoid gray material, was observed in 5 of 6 chicks (83%). Four of 6 chicks (67%) had mild to severe subcutaneous edema of the neck and in all of them the underlying musculus complexus (pipping muscles) appeared pale, enlarged and edematous, with multifocal hemorrhages. Anasarca was observed in 1 of 6 chicks (17%) and fatty and mottled red livers in 4 of 6 chicks (67%).
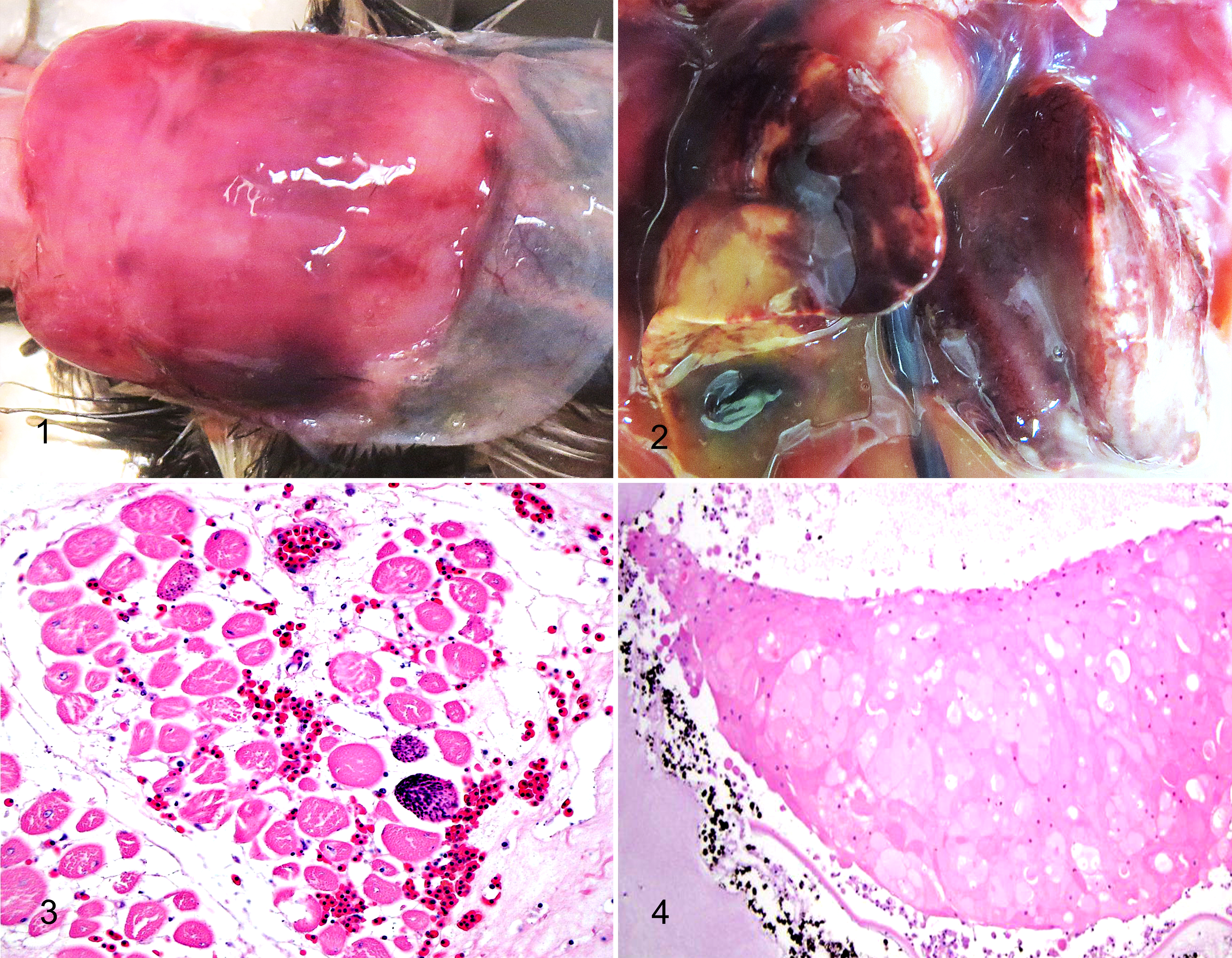
Microscopically, the musculus complexus of all 7 chicks presented various degrees of degeneration with increased eosinophilia, loss of cross striations, variation in fiber size and loss of myocyte nuclei. In addition, 5 chicks (71%) had floccular degeneration and necrosis of the muscle fibers, as well as interstitial edema and hemorrhages. A mild mononuclear cell infiltration was present in 6 chicks (86%) and mild multifocal mineralization in 1 of 7 (14%) (Fig. 3). In 4 of 7 chicks (57%), skeletal muscles in the legs and thighs and pectoral muscles had mild degeneration. In 2 chicks, there was multifocal degeneration of cardiomyocytes with focal mineralization (Supplemental Fig. 1). The liver was congested in 5 of 7 chicks (71%), and contained multifocal severe hepatocellular necrosis and hemorrhages in 4 of 7 chicks (57%) (Supplemental Fig. 2). The same number of birds (57%) showed renal tubular necrosis and mineralization. In 5 of 7 chicks (71%), both eyes were swollen with vacuolation of the lenticular fibers and multifocal formation of eosinophilic globules (Fig. 4). Wrinkling of the lens capsule was observed in 2 of 5 chicks and mild inflammation of the anterior lens capsule in 1 chick.
Routine aerobic bacterial culture was performed on skin, liver and yolk sac, as well as Salmonella enrichment for cultures of intestines and yolk sac. Escherichia coli was isolated from the liver of 5 of 7 chicks, which were probably postmortem contaminants as the owner had broken the shells to facilitate hatching. TEM analysis of the intestinal contents was negative for viruses.
Liver samples were analyzed for heavy metals and selenium by inductively coupled plasma mass spectrometry (ICP-MS) and for vitamin E by high pressure liquid chromatography (HPLC). The limited amount of hepatic tissue collected during necropsy hindered the examination of vitamin A level in the liver. Feed levels of vitamin E and vitamin A were also examined by HPLC. In the liver of all 7 chicks, vitamin E were low, ranging from 0 IU/kg (chick 6) to 11 IU/kg (chick 4) (normal reference range = 15–40 IU/kg). 6 Selenium was slightly increased in 3 (chick 3 and 6 = 1.7 IU/kg; chick 7: 2.1 IU/kg; normal reference range = 0.35–1 IU/kg) of 7 chicks. Regarding heavy metals, the concentrations of arsenic and lead were within the expected ranges for poultry in all chicks, while cadmium, copper, mercury and molybdenum were slightly increased in 1, 2, 5 and 2 chicks, respectively. An increased level of iron was observed in 2 of 7 chicks . Manganese concentration was slightly decreased in 4 chicks, while zinc was low in 5 (Supplemental Table 1).
Vitamin E (89 IU/kg) and vitamin A (3499 IU/kg) levels in the feed material were considered insufficient to meet the requirements suggested for both emu starters (vitamin E = 99 IU/kg; vitamin A = 15432 IU/kg) and emu breeders from 4 or 5 weeks before onset of egg production (vitamin E = 99 IU/kg; vitamin A = 8818 IU/kg). 9
The gross and microscopic lesions observed in the skeletal muscles of the 7 chicks and in the myocardium of 2 chicks that died before hatching are suggestive of a nutritional myopathy, most likely secondary to vitamin E deficiency, as confirmed by the biochemical analyses. Management factors, such as a high relative humidity during incubation, which has been associated with degeneration of the musculus complexus and the pelvic limb muscles in ostrich chicks, 5 may be potentially considered additional contributing factors, although information on rearing conditions were not available.
In our cases, the involvement of the muscles in the proximal neck region, and specifically the musculus complexus (also known as “pipping muscle”), could have compromised the hatching process. The musculus complexus is a paired muscle, located superficially in the anterodorsal cervical region, and running from the cervical vertebrae to the posterior end of the parietal bones. 3 Its contraction, at the end of the incubation, elevates the head of the chick and therewith the beak, thus providing the driving force to the egg tooth, responsible for the internal and external pipping. 3
A common lesion described in ruminants and pigs affected by vitamin E and/or selenium deficiency is hepatosis dietetica, characterized by coagulative necrosis of individual or irregular small groups of hepatic lobules. Similar hepatic changes were observed in all chicks in this study, although the typical lobular distribution observed in other species was not obvious in our cases due to the normal structure of the avian liver lacking distinctive interlobular septa. The histological lesions described in the eye are commonly associated with impaired levels of vitamin E but also vitamin A, 2 further confirming the pathogenesis of the condition in our cases.
The biochemical analyses performed by HPLC and ICP-MS on liver and feed samples support the nutritional etiology of the pathological lesions observed. Unfortunately, no data were available on the normal levels of nutrients and minerals in tissues of ratites. Thus, extrapolation from what is known for other avian species, specifically poultry, appeared to be the only available option.
The deficiency observed in our chicks is most likely due to improper supplementation. Since the absorption of lipid-soluble vitamins is strictly related to fat digestion, their bioavailability may have been compromised by an inadequate fat content of the diet or impaired hepatic and pancreatic function.
Another factor to be considered in the pathogenesis of this condition is the amount and quality of feed administered to the emu breeders. Both parents need to build ample energy reserves (fat pad) at the onset of breeding, since food intake progressively decreases and becomes erratic during reproduction. 9 Improper nutrition may result in low fertility and production of nutritionally deficient eggs.
A few months later, during the following reproductive season, the farm experienced the same problem of poor hatchability (22–25%). Therefore, 3 more emu eggs containing fully developed chicks but dead in shell were submitted to the CAHFS Laboratory System (Tulare branch) for evaluation. Myopathy of the pipping muscles and cataract were observed in 3 chicks and hepatosis dietetica in 2 of 3. Unfortunately feed was not submitted for evaluation. However, vitamin E analyzed in the liver from 2 chicks was low (chick 1= 9.6 ppm, chick 2 = 0.29 ppm). In addition, the hepatic level of vitamin A in 1 animal appeared low (7.8 ppm). These results add further evidence that deficiency of vitamin E and vitamin A were involved in causing lesions in the pipping muscles, liver and lens, leading to low hatchability in the emu flock. After this experience, the owner changed the feed to 1 containing adequate levels of vitamin A and E, which resulted in significant improvement in hatchability (from 3–5% to 35% after the change of feed) and chick survival (from 12% to 75% after the change of feed).
The anasarca and noninternalized residual yolk sacs observed in our cases are suggestive of insufficient water loss during the incubation process, which could be related to poor management or an abnormal structure of the eggshell. One of the most important contributing factors is the high humidity during incubation. This results in poorly developed air cells and poor gas exchange, which lead to weak and “water logged” (edematous) chicks. These chicks frequently die at/near point or soon after hatching, secondary to hypoxia. 4 Vitamin A may also have a role in this process. Vitamin A deficiency causes reduced translocation of zinc through the intestinal wall and reduced transport to the liver, and low levels of hepatic zinc were demonstrated in our chicks. Zinc is incorporated in the molecular structure of a large number of metalloenzymes and is a cofactor of carbonic anhydrase, involved in the formation of the eggshell. 8
In conclusion, we have described necrosis of the pipping muscles, heart and liver, as well as cataract in emu chicks associated with a primary vitamin E deficiency and reduced vitamin A and Zinc as minor contributing factors. Therefore, nutritional deficiencies and poor incubation management may play a critical role in the perinatal and neonatal death of emus. This case report also emphasizes the importance to provide standardized parameters during incubation and optimal dietary requirements for this species at different ages.
Footnotes
Acknowledgments
The authors wish to acknowledge and thank the California Animal Health and Food Safety laboratory staff of Tulare (histology and bacteriology section) and Davis (toxicology and electron microscopy section) for their excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
