Abstract
The histopathologic characteristics of colorectal inflammatory polyps that formed in Miniature Dachshunds were compared with those of other colorectal proliferative lesions, including adenomas and adenocarcinomas. Fifty-three colorectal polypoid lesions were histopathologically classified into inflammatory polyps (26 cases), adenoma (18 cases), and adenocarcinoma (9 cases). All 26 dogs that were diagnosed with inflammatory polyps were Miniature Dachshunds, indicating that colorectal inflammatory polyps exhibit a marked predilection for this breed. The inflammatory polyps had complex histopathologic features and were classified into 3 stages based on their epithelial composition. In early stage (stage 1), the polyps tended to exhibit a thickened mucosa containing hyperplastic goblet cells, dilated crypts filled with a large amount of mucus, and mild lymphocyte and macrophage infiltration. In later stages (stages 2 and 3), more severe neutrophil infiltration, interstitial mucus accumulation, granulation tissue, and occasional osteoid tissue were seen. Also, a few small foci of dysplastic epithelial cells were detected. The hyperplastic goblet cells, which were a major component of the epithelium of the inflammatory polyps, were positive for cytokeratin 20 (CK20), while the dysplastic epithelial cells found in inflammatory polyps (stage 3) and the tumor cells of the adenomas and adenocarcinomas were negative for CK20. These CK20-negative epithelial cells exhibited cytoplasmic and nuclear immunoreactivity for beta-catenin. In addition, the epithelial cells in the inflammatory polyps demonstrated significantly higher cyclooxygenase 2 and fibroblast growth factor 2 expression than did those of the adenomas and adenocarcinomas, suggesting that the arachidonate cascade is involved in the development of colorectal inflammatory polyps in miniature dachshunds.
Due to recent advances in endoscopic veterinary examination techniques, colorectal polyps are being detected with increasing frequency in domestic animals. Intestinal polyps are histopathologically defined as nonneoplastic pedunculated intraluminal growths. However, this term is generally used to refer to pedunculated intraluminal growths that are detected at clinical sites during endoscopic and/or gross examinations. Among the polypoid growths found in the intestines of dogs, the major histopathologic subtypes include nonneoplastic (hyperplastic) polyps, papillary adenoma, and papillary adenocarcinoma. Although there have been some reports about canine colorectal tumors, the pathology of inflammatory polyps has not been described in detail, presumably due to the small number of cases. 14,19,22,26,27
In Japan, colorectal inflammatory polyps are often encountered in Miniature Dachshunds with tenesmus, hematochezia, and/or periodic intermittent diarrhea. Endoscopically, these polyps appear as single or multiple outgrowths localized between the descending colon and rectum. 17 Colorectal inflammatory polyps are usually treated with a combination of prednisolone, cyclosporine, and endoscopic polypectomy. 17 However, recurrence often occurs after endoscopic polypectomy 25 ; furthermore, the lesions can progress into adenomas in some cases. 11 It remains unclear why these polyps arise more commonly in Miniature Dachshunds than in other dog breeds and how they are related to adenoma and adenocarcinoma. Here, we describe the histopathologic characteristics and pathogenesis of colorectal inflammatory polyps in Miniature Dachshunds by comparing them with other colorectal polypoid lesions, such as adenoma and adenocarcinoma. Furthermore, findings indicative of neoplastic transformation of these polyps are discussed.
Materials and Methods
Tissue Samples and Histopathology
At total of 111 colorectal mass lesions were removed from dogs at the Veterinary Medical Center of the University of Tokyo or the Japan Small Animal Medical Center between 2008 and 2013. The lesions were diagnosed at the Department of Veterinary Pathology of the University of Tokyo. Among them, 53 cases of colorectal polypoid lesions with precise clinical histories were examined in this study (Suppl. Table 1). All of the tissue samples were readily fixed in 10% neutral buffered formalin, routinely embedded in paraffin wax, and cut into 4-µm-thick serial sections, and deparaffinized sections were stained with hematoxylin and eosin. Selected sections were also stained with periodic acid–Schiff, alcian blue (pH 2.5), or Warthin-Starry silver stain. The lesions were classified into 3 types—inflammatory polyps, adenoma, and adenocarcinoma—by 2 experienced Japanese College of Veterinary Pathologists–certified veterinary pathologists (J.K.C. and K.U.) based on the histologic and cellular atypia, invasive ability, and proliferative activity of their epithelial cells. Image analyses were performed with Adobe Photoshop CS6 Extended (Adobe Systems, San Jose, CA, USA). The area of the epithelial component of each lesion was measured in a randomly selected field under ×40 magnification. In each field, the epithelial area was marked manually on the computer screen; then, the percentile area was measured with the software tool. Lesions in which >15%, 5%–15%, and <5% of the tissue consisted of epithelial cells were defined as stage 1, stage 2, and stage 3 lesions, respectively.
Immunohistochemistry
After being subjected to deparaffinization and antigen retrieval (Suppl. Table 2), the tissue sections were treated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes and then incubated with 8% skimmed milk in Tris-buffered saline at 37°C for 40 minutes to block nonspecific reactions. Subsequently, the sections were incubated overnight with primary antibodies (Suppl. Table 2) at 4°C. After being washed in Tris-buffered saline, the sections were incubated with horseradish peroxidase–labeled polymer (Dako Envision+ System; Dako, Carpinteria, CA, USA) or biotinylated anti-goat immunoglobulin G (KPL, Guildford, UK) before being incubated with horseradish peroxidase–labeled streptavidin (Dako). Finally, the reaction products were visualized with 0.05% 3-3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris/HCl buffer and then counterstained with Mayer’s hematoxylin. Colorectal tissue samples from dogs without lesions were used as positive controls. The primary antibodies were omitted to produce negative controls of normal colorectum, inflammatory polyp, adenoma, and adenocarcinoma.
Quantitative Data Analysis
The numbers of inflammatory cells that were positive for CD3, CD20, Iba-1, or myeloperoxidase were counted under ×400 magnification in 5 randomly selected fields. In addition, the number of von Willebrand factor–positive vessels was counted under ×200 magnification in 3 randomly selected fields in each section. Finally, the percentages of Ki67-, cyclooxygenase 2 (COX-2)–, and fibroblast growth factor 2 (FGF-2)–positive epithelial cells in total epithelial cells were measured under ×400 magnification in 5 randomly selected fields. Comparisons of the means of different groups were performed with 1-way analysis of variance, followed by Tukey honestly significant difference test with the software SPSS version 22 (IBM, Tokyo, Japan). Differences associated with P values <.01 were considered to be significant.
Results
Clinical Features of the Colorectal Polyps
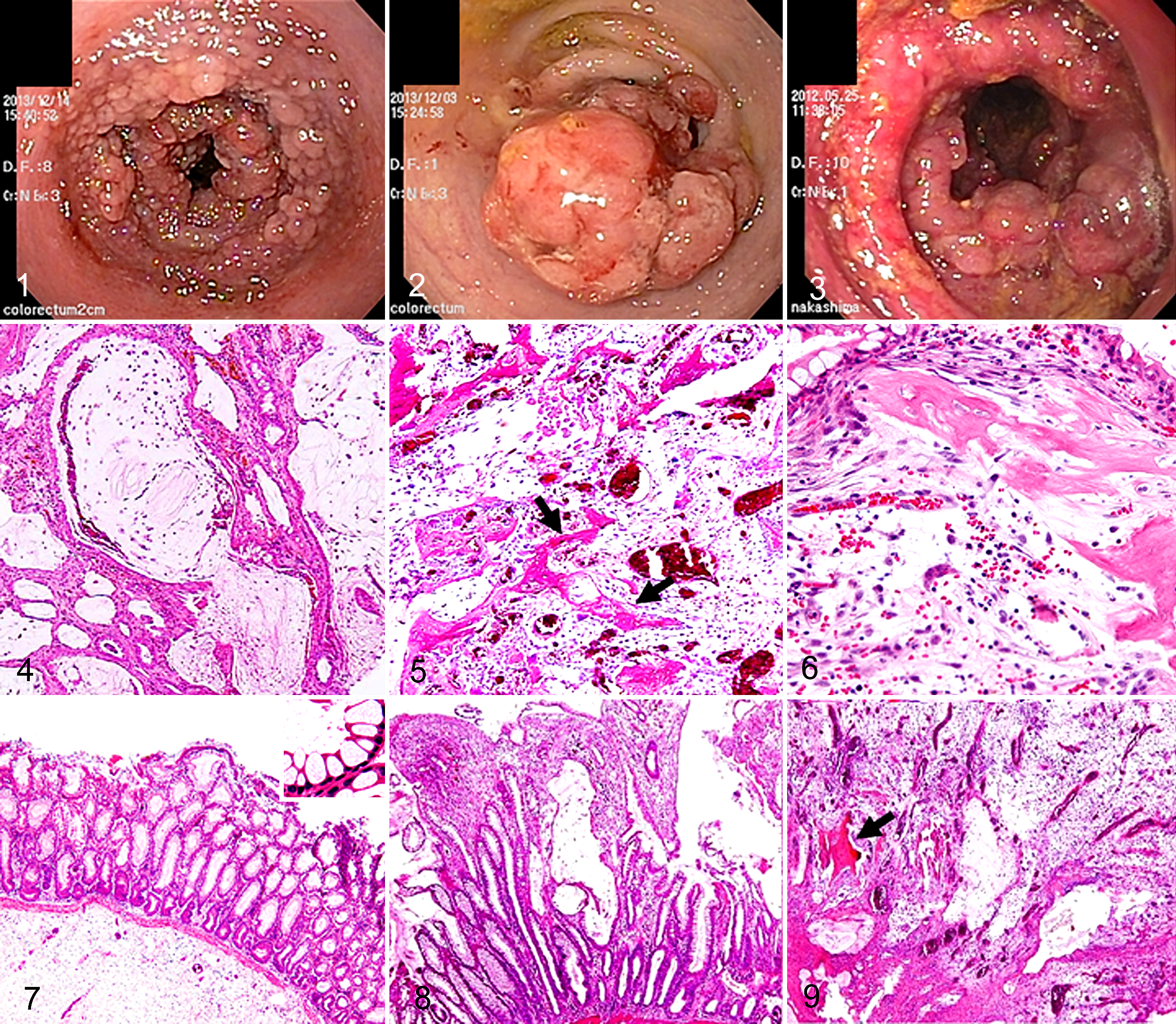
The tissue samples from the 53 cases of colorectal polyps were classified into inflammatory polyps (26 cases, 49%), adenoma (18 cases, 34%), and adenocarcinoma (9 cases, 17%) according to their histopathologic features (Suppl. Table 1). During endoscopic examinations, the above-mentioned polyps were detected as single or multiple lesions. As for their morphology, they appeared as pendulous, protruding, or irregularly thickened masses in the colorectal mucosa (Figs. 1–3). All 26 lesions that were diagnosed as inflammatory polyps were found in Miniature Dachshunds, and the mean age of the affected dogs was 9 years 3 months. The condition did not exhibit a predilection for either sex (males: 14 cases, females: 12 cases). Most of the dogs with inflammatory polyps displayed hematochezia or tenesmus at 2 to 78 months before they underwent an endoscopic biopsy or polypectomy. Among the 18 cases of adenoma, 7 involved Miniature Dachshunds (39%; mean age: 9 years 11 months). The remaining 11 involved Jack Russell Terriers (2 cases), French Bulldogs (2 cases), Toy Poodles (2 cases), or other breeds (5 cases). The dogs that were diagnosed with adenoma exhibited hematochezia or tenesmus at 5 to 29 months before they underwent an endoscopic biopsy or polypectomy. Of the 9 cases of adenocarcinoma, 2 involved Miniature Dachshunds (22%; mean age: 11 years 1 month). Adenocarcinoma did not exhibit a marked predilection for any particular breed. The dogs that were diagnosed with adenocarcinoma displayed hematochezia or tenesmus at 2 to 20 months before they underwent an endoscopic biopsy or polypectomy.
Histopathologic Features of the Inflammatory Polyps
The inflammatory polyps displayed a thickened mucosa composed of hyperplastic goblet cells, increased mucus production, dilated crypts filled with abundant periodic acid–Schiff–positive mucinous materials, and mild infiltration (mainly by lymphocytes and macrophages; Fig. 4). The larger polyps had severe leukocyte infiltration (mostly by neutrophils and macrophages), interstitial accumulation of mucus or cystic spaces filled with mucinous materials, proliferation of granulation tissue, and occasional osteoid formation (Figs. 5, 6). In these lesions, no specific infectious organisms were detected by Warthin-Starry silver staining. Changes apparently associated with the more severe inflammatory changes observed in larger polyps included decreased size of the epithelial component, while small foci of dysplastic epithelial cells appeared within the inflammatory and granulation tissue. The epithelial cells in these foci exhibited anisocytosis and a loss of cellular polarity.
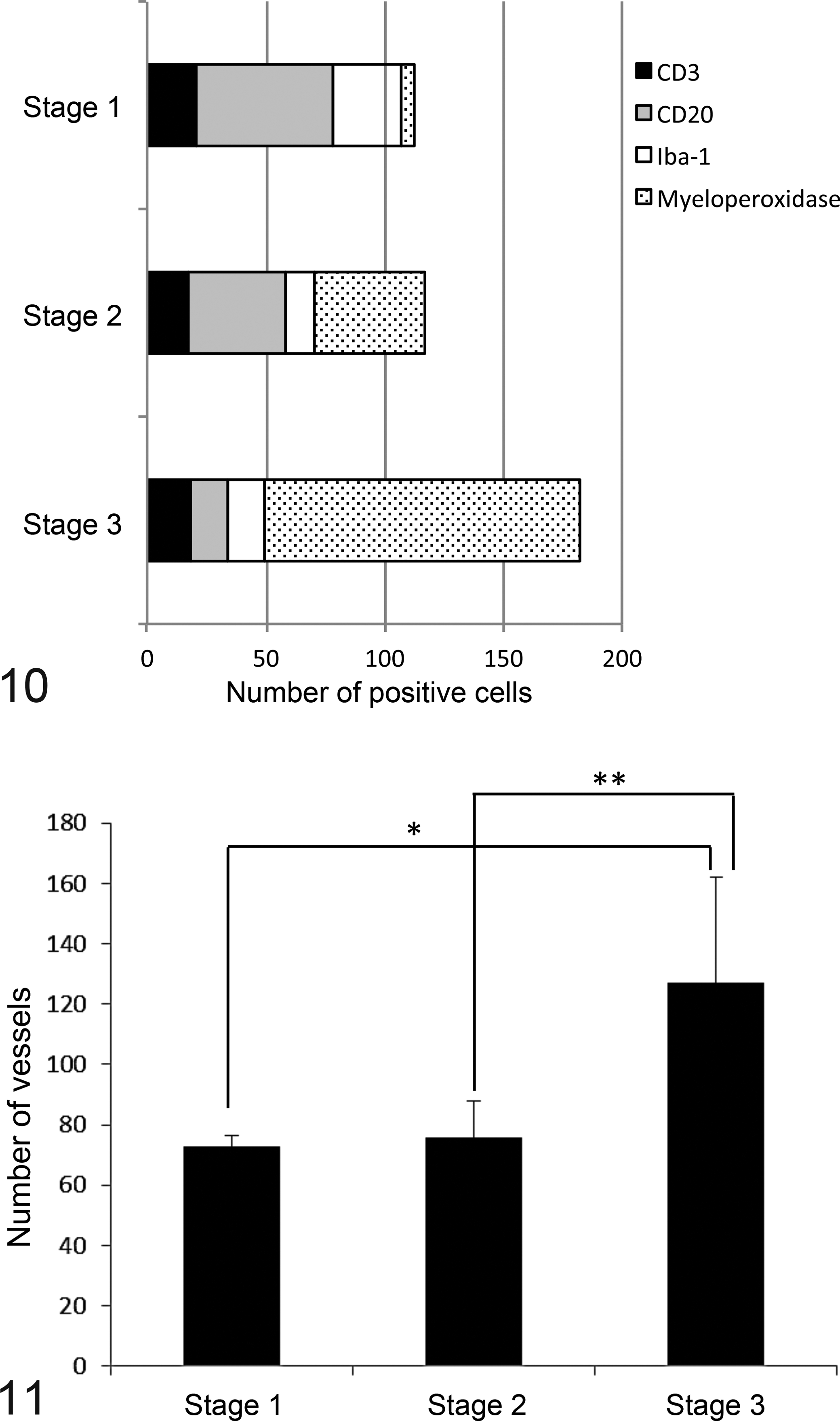
As assessed by image analysis, lesions in which >15%, 5%–15%, and <5% of the tissue consisted of epithelial cells were defined as stage 1 (n = 3, Fig. 7), stage 2 (n = 8, Fig. 8), and stage 3 (n = 23, Fig. 9) lesions, respectively. The inflammatory cells found in stage 1 lesions mainly consisted of mononuclear cells—namely, CD20-positive B cells, CD3-positive T cells, and Iba-1-positive macrophages (Fig. 10), with B cells being predominant. The total number of inflammatory cells increased with the stage of the condition. Specifically, increased numbers of myeloperoxidase-positive neutrophils were seen in the stage 2 lesions, and these cells accounted for >70% of the inflammatory cells in the stage 3 lesions. Conversely, the number of B cells decreased as the stage increased. To assess the proliferation of the lesions’ stromal components, the number of small vessels was counted in each lesion. The number of small vessels was significantly higher in the stage 3 lesions than in the stage 1 and 2 lesions, which was indicative of excessive granulation tissue formation (Fig. 11).
Epithelial Cell Characterization in the Colorectal Inflammatory Polyps, Adenomas, and Adenocarcinomas
A considerable number of the epithelial cells in the stage 1 and 2 inflammatory polyps exhibited goblet cell differentiation (Fig. 12) and were positive for cytokeratin 20 (CK20; Fig. 13). Beta-catenin was expressed in the cytoplasm of the epithelial cells in stage 1 and 2 lesions (Fig. 14). However, the dysplastic epithelial cells observed in stage 3 lesions (Fig. 15) were negative for CK20 but expressed beta-catenin in both their cytoplasm and nuclei (Fig. 16).
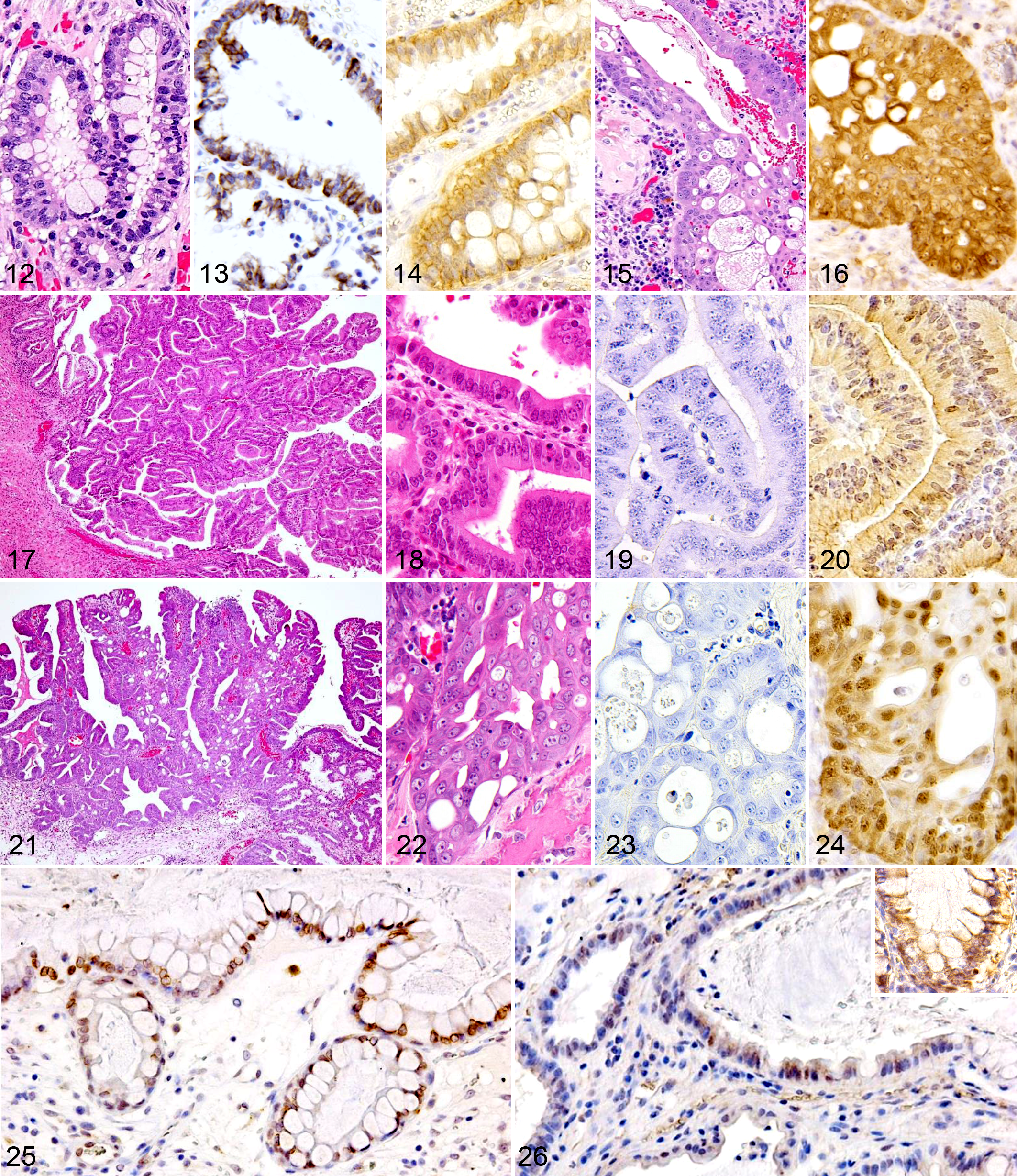
The colorectal adenomas exhibited papillary epithelial cell proliferation, but their neoplastic cells did not display invasive activity (Fig. 17). The neoplastic cells demonstrated mild atypia—namely, anisocytosis, basophilic cytoplasm, and dark nuclear staining (Fig. 18). The adenoma cells were negative for CK20 (Fig. 19) but weakly expressed beta-catenin in both their cytoplasm and nuclei (Fig. 20). The colorectal adenocarcinomas were characterized by papillary epithelial cell proliferation, cellular atypia, polymorphism, a loss of cellular polarity, and a greater invasiveness (Figs. 21, 22). The adenocarcinoma cells were negative for CK20 (Fig. 23) but exhibited intense immunoreactivity to beta-catenin in their cytoplasm and nuclei (Fig. 24).
To assess the role of the arachidonate cascade in the progression of inflammatory polyps, the expression levels of COX-2 and FGF-2 were examined in all 53 cases. COX-2 and FGF-2 immunoreactivity was detected in the epithelial cells of the inflammatory polyps (Figs. 25, 26). Semiquantitative analysis revealed that the inflammatory polyps (68.9%) displayed significantly higher epithelial cell COX-2 positivity than did the adenomas (13.9%) and adenocarcinomas (23.9%; Fig. 27). In contrast, no significant difference in epithelial cell COX-2 positivity was detected among the different stages of inflammatory polyps. The inflammatory polyps (43.2%) also demonstrated significantly higher epithelial cell FGF-2 positivity than did the adenomas (3.2%) and adenocarcinomas (12.0%; Fig. 28). However, no significant difference in FGF-2 positivity was observed among the different stages of inflammatory polyps.
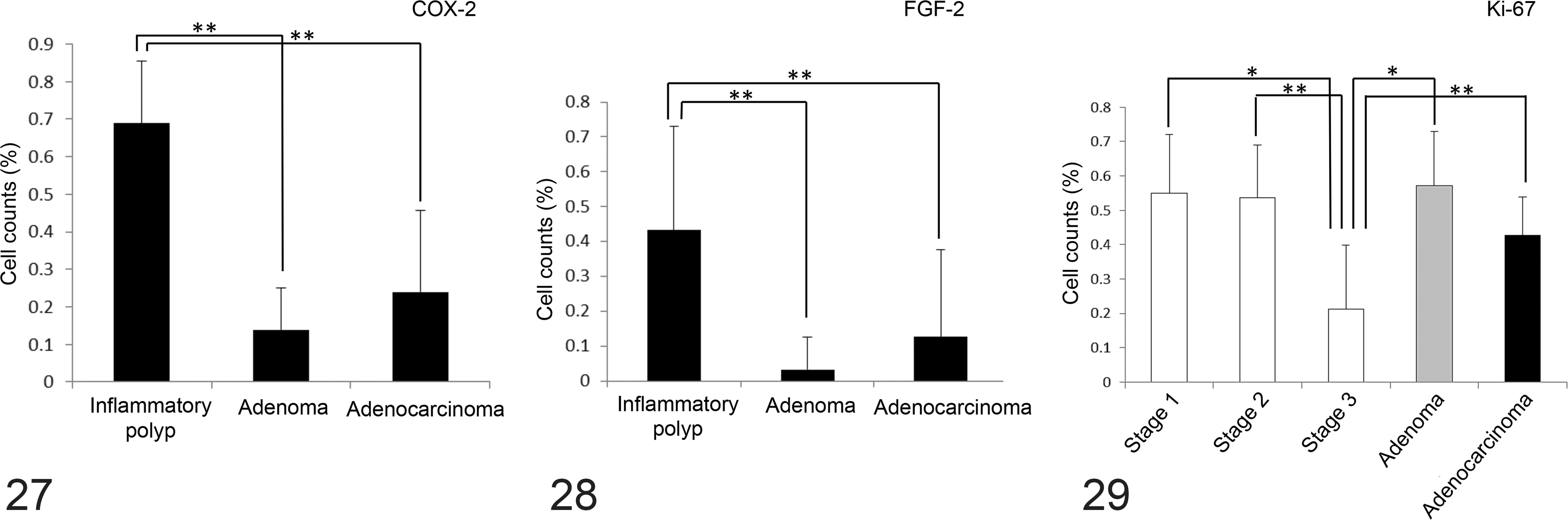
To assess the proliferative activity of the epithelial cells, their Ki67 positivity was examined. Ki67-positive epithelial cells were confined to the deeper crypts of the inflammatory polyps, whereas they were scattered randomly throughout the adenomas and adenocarcinomas. 2 Stage 3 inflammatory polyps displayed significantly lower Ki67-positive epithelial cell percentages than those of stage 1 and 2 inflammatory polyps, adenomas, and adenocarcinomas (Fig. 29).
Discussion
The present study clearly showed that colorectal polyps are common in Miniature Dachshunds in Japan. Interestingly, the nonneoplastic polyps seen in this breed displayed distinct histopathologic characteristics—that is, goblet cell hyperplasia combined with inflammation. We propose to refer to these polyps as inflammatory polyps of Miniature Dachshunds (IPMDs).
In this study, we histopathologically classified IPMDs into 3 stages based on the relative content of epithelial components and then compared the inflammatory components. Different populations of inflammatory cells were observed in each of the 3 IPMD stages. The stage 1 polyps appeared to contain more B cells as compared with stage 2 and 3 polyps, which contained more neutrophils. The reduced amount of epithelial tissue seen in the higher-stage IPMD may be explained by the decreased epithelial cell mitotic activity (Fig. 29), excessive granular tissue formation, and increased neovascularization observed in these polyps (Fig. 11). 21 Thus, these findings could be the main histopathologic changes associated with the progression of IPMD.
Furthermore, we examined whether there were changes indicative of neoplastic transformation in IPMD. The well-differentiated epithelial cells in the normal upper crypt of the intestine (ie, enterocytes, goblet cells, and endocrine cells) express CK20. 6,7 In humans, high-grade adenocarcinomas exhibit decreased CK20 positivity. 3 In the present canine study, the epithelial cells that composed the IPMD expressed CK20, while the neoplastic epithelial cells of the adenomas and adenocarcinomas were CK20 negative. Therefore, IPMD could be distinguished from adenoma and adenocarcinoma according to their differentiation—that is, their CK20 expression. Beta-catenin, a component of the Wnt/beta-catenin signaling pathway, plays an important role in intestinal tumorigenesis in humans and animals. 16 Beta-catenin is expressed in the nuclei of adenoma and adenocarcinoma cells, and mutation of the beta-catenin gene induces intestinal polyp formation. 4,10,12,14,20 However, in the IPMD examined in the present study, beta-catenin was only expressed in the cytoplasm. In contrast, beta-catenin was expressed in the nucleus and cytoplasm in the adenomas and adenocarcinomas. These observations support the nonneoplastic nature of IPMD.
COX-2 expression is induced by macrophages, fibroblasts, and other cells. 9,24 It is known that the neoplastic cells found in canine mammary tumors and intestinal tumors express COX-2. 8,15 The present study revealed that the epithelial cells of IPMD express COX-2, indicating that COX-2 plays a role in the development and/or progression of IPMD. COX-2 is known to induce the expression of FGF-2, which is a major cytokine that activates fibroblasts. 1,5,13,18,28 FGF-2 is involved in neovascularization and contributes to granulation tissue formation during ulcer healing. 23 Therefore, COX-2 and FGF-2 expression may play a role in the excessive granulation tissue seen in the IPMD.
In the present study, we described the histopathologic features of IPMD. Immunohistochemical examinations of CK20 and beta-catenin expression indicated that the proliferating epithelial cells seen in IPMD are well differentiated and nonneoplastic. However, in the progressive stage of IPMD, foci of dysplastic epithelial cells are occasionally observed, and these foci exhibit nuclear beta-catenin expression, which is indicative of the neoplastic transformation of IPMD to adenoma or adenocarcinoma. This notion is supported by the high incidence of adenoma in Miniature Dachshunds. Last, our findings regarding the COX-2 and FGF-2 expression patterns of IPMD, adenoma, and adenocarcinoma indicate that COX-2 may play a role in the development and/or progression of IPMD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Japan Society for the Promotion of Science (grants 15K14863 and 26660236).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
