Abstract
Semenogelins are proteins originating in the seminal vesicle and are useful markers for the presumptive identification of human semen. Detection of semenogelin can be done with a commercially available membrane test. In this study, a commercially available membrane test for human semenogelin proteins was used to assess for cross-reactivity in dog bodily fluids to allow for the potential utilization for detection of human semen in dog bodily fluids. The authors analyzed canine semen and other bodily fluids, including urine, saliva, vaginal secretions, fecal material, and blood. They also examined the distribution of human semenogelin I transcripts in the canine testis, prostate, and several bodily fluids by reverse transcription polymerase chain reaction. No cross-reactivity was observed in the canine bodily fluids tested except for a single rectal swab, which was negative on a second test. Further testing should be done to validate the use of this kit for screening samples from dogs suspected to have been victims of sexual abuse.
Keywords
Animal sexual abuse is uncommonly reported in the scientific literature. A review of 448 cases of animal abuse revealed 28 cases of sexual abuse toward animals. 1 In a multicenter study looking at the association of sex with animals and penile cancer, 171 of 489 human subjects reported to have had sex with animals. 17 Individual cases of sexual abuse toward dogs, cattle, horses, and sheep have been reported in the veterinary literature and the media. 1,3,4,7,8,15 Types of animal sexual abuse can vary and include masturbation of the animal, performance or receiving of vaginal or anal intercourse, and/or insertion of foreign bodies into the vagina or rectum. In some cases, there may be human penile involvement; in such cases, ejaculation with deposition of seminal fluid in or on the animal victim may occur.
In cases of sexual abuse toward humans, evaluation for ejaculate including spermatozoa is of utmost importance, as this can assist with corroborating a victim’s or witness’s statement and can provide material for DNA analysis. 10,11,16 The confirmatory test for human ejaculate is microscopic identification of spermatozoa. 11 Analysis of short tandem repeat loci from the Y chromosome (Y-STR) and mass spectrometry-based proteomics have also been used as a confirmatory test for semen. 2,6,9,13 In some cases, spermatozoa are not identified in sexual assault victims, and screening tests are necessary to detect the presence of semen or seminal fluid. Possible reasons for lack of spermatozoa detection in forensic samples include lack of ejaculation, aspermia due to testicular disease, or surgical sterilization (ie, vasectomy). Screening tests employed for seminal fluid detection include acid phosphatase test, assay for prostate-specific antigen (PSA or p30), or semenogelin I and II (SgI and SgII). 10,11,16
Mass spectrometry–based proteomics can be used as an exploratory or confirmatory technique for identification of various biologic matrices, including semen. 13 This technique allows for the detection of multiple biomarkers, such as Sg (semen), alpha-amylase 1 (saliva), hemoglobin subunit beta (human blood), and cornulin (vaginal secretion). 13 Analysis of Y-STR can be employed on suspect fluid, including aged dried samples, which ultimately will allow for the identification of a DNA profile to allow for comparison with a suspect. A proposed workflow methodology incorporates Y-STR analysis for semen analysis if the sample in question is positive for the screening tests (Sg and/or PSA). 6 In cases where spermatozoa are not detected, other methods can be employed for the presumptive detection of seminal fluid and include assays for acid phosphatase, PSA, or Sg. 10,11,16 Acid phosphatase testing, although not specific for semen, is currently used to avoid underestimation of the presence of semen. 6
The human vesicular gland produces SgI and SgII, which are secreted in human semen. SgI is a nonglycosylated protein composed of 439 amino acid residues, and SgII is a protein composed of 559 amino acid residues. 5,14 In ejaculated semen, SgI and SgII interact noncovalently by disulphide bridges to form a coagulum. 5,11,14 PSA is a prostatic serine protease that breaks down Sg into fragments, liquefying the coagulum within a few minutes of its formation. 11 SgI and SgII transcripts have both been identified in multiple human tissues, including vesicular gland, vas deferens, prostate, epididymis, trachea, skeletal muscle, kidney, mammary gland, salivary gland, and gastrointestinal tract. 5,18 Immunohistochemically in human tissues, Sg antigen has been identified within the basal cell layer of the prostate, trachea, and bronchi; luminal cells of the seminal vesicle and vas deferens; scattered cells in the central nervous system; and skeletal myocytes. 5 Semenogelin is found in human seminal fluid and in the ejaculate of the common marmoset, a New World primate. 14
Two studies tested ejaculate from various animal species for cross-reactivity for Sg utilizing a membrane strip test 10,11 but were limited to 1 to 2 semen samples per animal species, and cross-reactivity with other animal bodily fluids was not assessed. The purpose of this study was to evaluate the usefulness of a commercially available membrane strip test as a possible method to screen for Sg proteins in human ejaculate in the domestic dog, by assessing the potential cross-reactivity to various bodily fluids, including ejaculate.
Materials and Methods
Specimens
Domestic dog ejaculate samples were collected by digital massage and pressure from 10 dogs with owner consent. Saliva samples were collected by swabbing the oral cavity of 10 dogs with sterile cotton-tipped swabs; samples were collected from 5 male and 5 female domestic dogs. All dogs were >1 year old. Testis samples were collected from the medical waste of 10 healthy domestic dogs following surgical castrations at a nonuniversity institution. All dogs were originally stray animals, and 5 dogs were identified as >1 year old. Sample collections from live animals received owner consent and were approved by the University of Illinois at Urbana-Champaign Institutional Animal Care and Use Committee protocol.
The following samples were collected within 24 hours of euthanasia from animals euthanized for medical purposes. Rectal swabs were collected with sterile cotton-tipped swabs from 5 dogs (age range: 3 weeks to 14 years; 2 intact males, 2 spayed females, and 1 intact female). Swabs of the vaginal canal were collected with sterile cotton-tipped swabs from 6 dogs (age range: 3 months to 11 years; 3 spayed females and 3 intact female). No information regarding the estrus cycle was available for these animals. Prostate samples were collected from 3 dogs all >1 year of age with 2 intact males and 1 castrated male. Urine samples were collected by cystocentesis from 5 dogs (age range: 3 months old to 14 years old; 1 intact male, 2 castrated males, 1 spayed female, and 1 intact female). Cardiac blood samples were collected from 4 dogs (age range: 3 months to 14 years; 1 intact male, 1 castrated male, 1 spayed female, and 1 intact female).
Materials
Membrane Test Assay
The rapid stain identification semen test (RSID-Semen; Independent Forensics) was used for this study. The RSID-Semen test is an immunochromatographic assay that uses 2 monoclonal antibodies specific for SgI and SgII. 10 The dog samples were tested according to manufacturer instructions for suspect human-origin forensic samples. Briefly, the samples were placed in the provided extraction buffer for 30 minutes before being pipetted and placed in the sample window. The test result was read 10 minutes after placement in the sample window. Two lines appear for a positive result, and 1 control line appears for a negative result. The control line must appear for a test to be valid. Prior to testing, liquid samples, with the exception of semen, were transferred to a sterile cotton swab by dipping in the sample containers. For semen samples, 1 μl of semen was transferred to the swab tips.
RNA Extraction From Tissues
Canine semen, blood, saliva, vaginal fluid, rectal swabs, prostate, testis, and urine specimens were analyzed. Approximately 200 μl of canine semen was centrifuged at 6000 × g for 1 minute. Supernatants were discarded, and pellets were resuspended in 110 μl of RLT Buffer (Qiagen Inc, Valencia, CA, USA). Vaginal, rectal, and salivary swabs were soaked in 500 μl of 1× phosphate-buffered saline (PBS) and squeezed, and the liquid was stored at –20°C until further use. Approximately 25 to 30 mg of prostate or testicular tissues were placed in 1 ml of RNALater (Life Technologies, Grand Island, NY, USA) and homogenized with 2.3-mm zirconia beads on a Mini Bead Beater-16 (Biospec Products; Fisher Scientific, Pittsburgh, PA, USA). Urine was centrifuged at 6000 × g for 10 minutes, and the pellet was resuspended in 110 μl of 1× PBS or RLT Buffer. For each sample of whole blood, 500 μl was centrifuged at 400 × g for 5 minutes, and the pellet was resuspended in 600 μl of RLT Buffer. A total of 110 μl of each processed sample was used for RNA extraction on a magnetic particle processor (Biosprint; Qiagen) with Qiagen One-for-All Vet Kit, per the manufacturer’s recommended protocol. Nucleic acid was eluted in 60 to 75 μl of elution buffer.
Reverse Transcription Polymerase Chain Reaction
Reverse transcription polymerase chain reaction (RT-PCR) testing was performed with the Qiagen OneStep RT-PCR Kit (Qiagen). SgI primers (5′ SgI primer: 5′-GCAGACACCAACATGGATCTCA-3′; 3′ SgI primer: 5′-CTGAGGTCAACTGACACCTTGA-3′; 180–base pair amplicon) were used for the detection of SgI transcripts, and Rpl32 primers (rpl32-Fwd: 5′-ACCTCTGGTGAAGCCCAAG; rpl32-Rev: 5′-GGGATTGGTGACTCTGATGG; 100–base pair amplicon) were used to detect the transcripts of an internal reference gene associated with the 60S ribosomal subunit. 12,14 The RT-PCR mix consisted of 1× Qiagen OneStep RT-PCR Buffer, 400 nM of each dNTP, 0.6 μM each of forward and reverse primers, 0.5 μl of OneStep RT-PCR Enzyme Mix, 7 U of RNAseOut (Life Technologies), and 1 μl of nucleic acid template (100 ng of total RNA) in a final reaction volume of 13 μl. RT-PCR reactions were carried out on a Bio-Rad S1000 Thermal Cycler (Bio-Rad, Hercules, CA, USA) with the following cycling parameters: reverse transcription at 50°C for 30 minutes; initial denaturation of cDNA at 95°C for 15 minutes, followed by 40 cycles of 94°C for 1 minute, 55°C for 1 minute, and 72°C for 1 minute; and a final extension at 72°C for 10 minutes. Amplification products were separated by electrophoresis on 3% agarose gels (Bio-Rad) visualized on a ultraviolet transilluminator and photographed with PhotoDoc-It 60 Imaging System (UVP, Upland, CA, USA). Human Prostate Total RNA (No. 636550; Clontech, Mountain View, CA, USA) was used as positive control for amplification of SgI and rpl32 transcripts. A nucleic acid extraction control performed with 1× PBS and a RT-PCR amplification control with RNAse- and DNAse-free water served as negative controls. SgI and rpl32 amplicons were excised following electrophoresis, and DNA was purified with the Qiagen Gel Extraction Kit (Qiagen) according to the manufacturer’s recommendations (Fisher Scientific). Amplicon sequencing was performed by ACGT Inc (Wheeling, IL, USA) using the SgI and rpl32 primers. Sequence data were analyzed with BLAST.
Results
Semen samples from 10 male dogs were tested with the RSID-Semen test. All semen samples tested negative for Sg (Fig. 1a). All samples of blood, saliva, vaginal swabs, and urine tested negative for Sg. A single positive test was obtained from a rectal swab (Fig. 1b). Repeat testing from a second sample obtained from this dog tested negative.
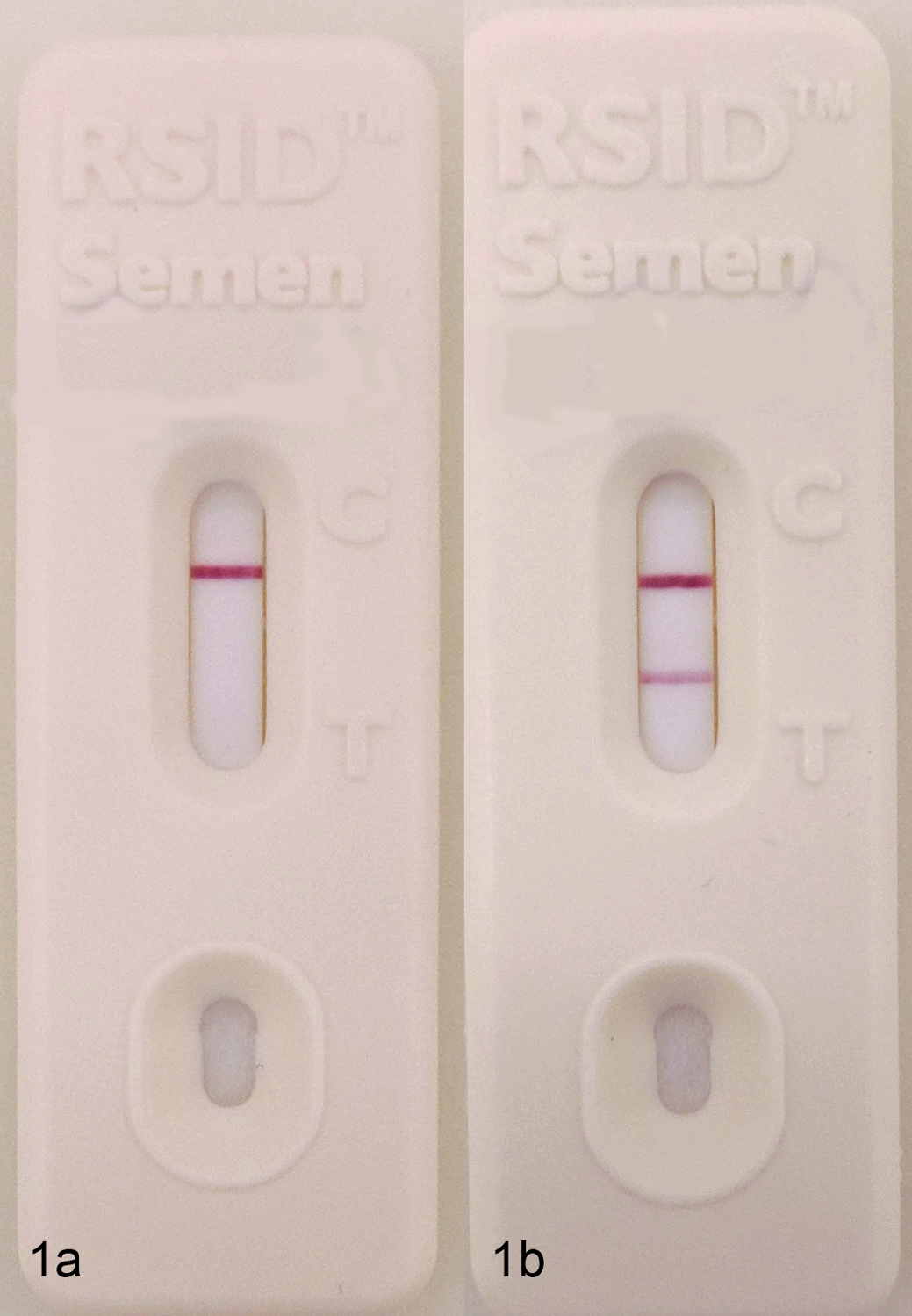
Semenogelin membrane strip test. a, Dog semen. Negative test result: only the control line (C) is observed. b, Rectal swab. False-positive result: both the control line (C) and test line (T) are observed.
RT-PCR testing was performed on semen samples; testis, prostate, and rectal swabs; vaginal swabs; and saliva, blood, and urine samples. All the samples tested negative for SgI transcript and positive for rpl32, the internal reference gene (Fig. 2). Human Prostate Total RNA was positive for SgI transcript and rpl32. Sequence analysis of SgI and rpl32 amplicons confirmed specific amplification of these 2 genes. No canine homologs were identified in this search.
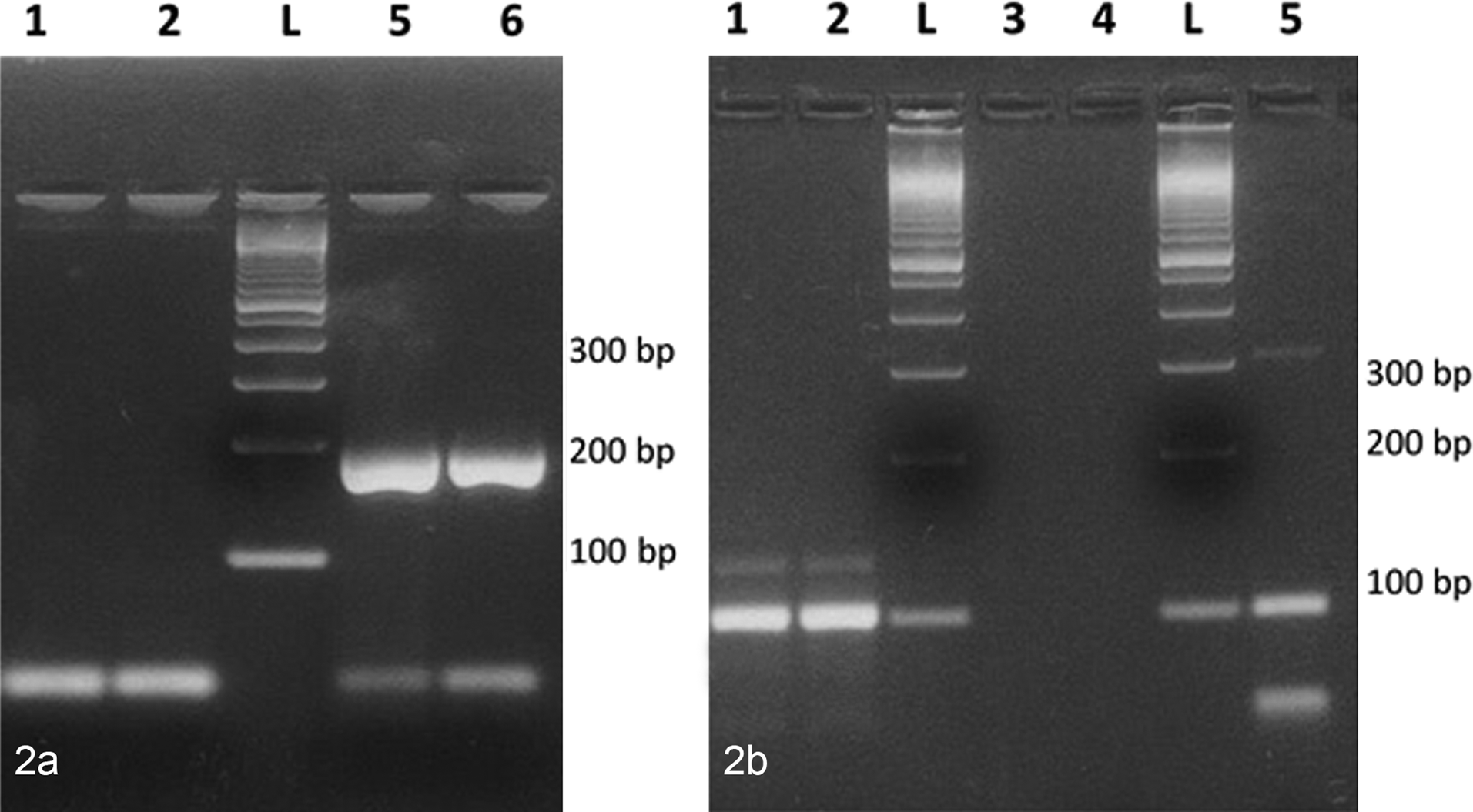
Reverse transcription polymerase chain reaction. Lanes 1 and 2: canine vaginal swabs; lanes 3 and 4: negative controls (omission of cDNA); lanes 5 and 6: human prostate; L: 100–base pair (bp) ladder. a, Semenogelin I. Note lanes 5 and 6 are positive for semenogelin I transcript. b, rpl32. Note lanes 1, 2, and 5 are positive for rpl32.
RT-PCR testing of the extract buffer containing the false-positive dog rectal sample was performed, and the sample was negative for the human SgI transcript.
Discussion
When cases of sexual assault are investigated, the conventional confirmatory test for human semen is the microscopic identification of spermatozoa. 11 We demonstrated that Sg cross-reactivity was not identified in urine, vaginal fluid, semen, saliva, and blood from domestic dogs with the RSID-Semen test for Sg. A single rectal sample tested positive with the membrane strip test. Repeat testing was performed, and on the second test, the result was negative. This was considered a false-positive result, as the case history had no indication of exposure to human seminal fluid. The cause for this false-positive result was not identified, but the case highlights the need for confirmatory tests when this screening test is positive.
RT-PCR testing for the human SgI transcript was performed on canine testis, prostate, semen, saliva, blood, rectal swabs, vaginal swabs, and urine, and no transcripts were identified. A BLAST search of the canine genome with the nucleotide sequence of the human SgI gene revealed no homologues, thus corroborating the lack of amplification in the RT-PCR assay.
Routine screening of dogs suspected to be victims of sexual abuse with the RSID-Semen test could result in the presumptive identification of human ejaculate, at which point confirmatory testing for human ejaculate should be performed. Additional studies need to be performed—including inhibitory studies of canine bodily fluids on samples known to contain human ejaculate, comparison of other methods to presumptively identify human ejaculate (eg, p30), the effects of postmortem decomposition on Sg detection in canine samples, and the effects of contamination of various spermicides and lubricants on Sg detection in canine fluid samples. Lubricants and spermicides did not have adverse effects in Sg testing in human forensic samples. 11
The results of this project demonstrate a lack of cross-reactivity with various bodily fluids when the RSID-Semen test is used, with the exception of a single rectal sample, and human SgI transcripts were not identified in any sample tested. Testing with a membrane test for Sg could allow for the presumptive identification of human seminal fluid in dog bodily fluids.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the American Society for the Prevention of Cruelty to Animals Grant (2014-0029).
