Abstract
Cyprinid herpesvirus 2 is a pathogen of goldfish, inducing a disease referred to as herpesviral hematopoietic necrosis. The disease is described so far in Japan, North America, Taiwan, Australia, the United Kingdom, and recently also Italy. Here the authors describe histologic lesions in clinically affected fish in comparison with clinically normal but virus DNA–positive goldfish in Switzerland. While necrosis or enhanced single-cell necrosis in the hematopoietic tissue in the pronephros or mesonephros was evident in dead and sick animals, in clinically normal goldfish, only single-cell necrosis was observed. Virus DNA was demonstrated in dead as well as clinically affected and subclinically infected goldfish by polymerase chain reaction and in situ hybridization. This study identifies the presence of goldfish herpesvirus in Switzerland and highlights the fact that the virus might be more widespread than assumed, as clinically normal goldfish can also carry cyprinid herpesvirus 2, showing histologically similar lesions but of lesser extent and severity.
Keywords
The genus Cyprinivirus belongs to the Alloherpesvirus family and comprises 4 herpesvirus species: cyprinid herpesvirus 1, 2, and 3 (CyHV-1, CyHV-2, CyHV-3) as well as herpesvirus anguillae. Common carp and koi (Cyprinus carpio L) are susceptible to infections with CyHV-1 (carp pox virus) 13 or CyHV-3 (koi herpesvirus). 11 CyHV-2 is described as a pathogen of goldfish (goldfish herpesvirus), inducing a disease referred to as herpesviral hematopoietic necrosis (HVHN). This disease was first described in western Japan in the 1990s, causing high mortality in goldfish and occurring during the spring and autumn. 10 Since then, CyHV-2 has been reported in North America 5,8 as well as Taiwan 2 and Australia. 14 In Europe, the virus was first detected in the United Kingdom in 2006, 12 followed by Italy in 2013. 4 There are indications of the presence of the virus in central Europe, such as Germany and Poland; however, these observations are not published yet (Bergmann, personal communication). Clinically, affected goldfish display signs such as apathy, pale gills, increased respiratory rate, and ascites. Histopathologically, necrosis of the hematopoietic tissue in kidney and spleen represents a consistent finding in CyHV-2-infected goldfish in all reports so far; therefore, the disease was named HVHN. However, reports of branchial, cutaneous, intestinal, and cardiac lesions are inconsistent. Hyperplasia and hypertrophy along with variable degrees of necrosis of the branchial epithelium were reported in goldfish from Australia, 14 North America, 8 and the United Kingdom. 9,12 Only 2 studies reported lesions within the heart: In Taiwan, necrosis and mild infiltration of the myocardium had been observed in affected animals, 2 and a North American study found mild inflammation of the endocardium and myocardium. 5 Within all these studies, intranuclear eosinophilic to amphophilic inclusion bodies with marginated chromatin were reported within the lymphohematopoietic tissue or epithelial cells or both.
Here we describe the detection of CYHV-2 in central Europe. Furthermore, the manifestation of the disease in dead/sick goldfish, as well as the characterization of possible early lesions in clinically normal but polymerase chain reaction (PCR)–positive goldfish, is described.
In Switzerland, in the vicinity of Bern during summer 2010, high mortality in mature goldfish was observed by a private owner keeping goldfish in an outdoor pond with koi. Mortality persisted until autumn 2010. Clinically, the only finding was a mild infestation with Gyrodactylus sp and Dactylogyrus sp, which was treated. After moving the fish to an indoor pond (water temperature, 18°C), low mortality continued over the winter. One dead, 6 moribund, and 45 clinically normal goldfish were submitted to the Centre for Fish and Wildlife Health (CFWH) for diagnostic examination. The clinically normal goldfish were divided in 2 groups and euthanized within a 1-week interval (group 1: 34 fish, group 2: 11 fish). As reference, 3 live mature goldfish from 2 locations other than the one described above were investigated at the CFWH. These reference goldfish were kept in outdoor ponds; in one location with koi; in the other, only with goldfish. Fish were presented in spring and early summer at a water temperature >15°C showing clinical signs as increased mucus production and increased respiratory rate.
Beside a single dead animal, goldfish were euthanized with buffered 3-aminobenzoic acid ethyl ester (MS 222; Argent Chemical Laboratories, Redmond, WA, USA). The dead animal was shipped immediately after death. The tissue was still in good condition, judged by the red color of the gills and the histologically intact cell morphology. Each fish was examined for the presence of parasites by microscopic evaluation of fresh mounts from the skin, gills, and intestinal content. A bacteriologic examination of liver, spleen, and kidney was performed with blood agar plates (bioMérieux, Lausanne, Switzerland). At necropsy, pathologic findings of internal organs were recorded. Representative samples of gill, skin, pronephros, mesonephros, heart, spleen, and liver were collected and fixed in 10% buffered formalin. Fixed specimens were processed routinely, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin for routine histologic examination. Special stains were performed as appropriate—including Gram to detect possible bacterial infections, Giemsa to detect parasites, Ziehl-Neelsen for mycobacteria, and periodic acid–Schiff for fungi.
Lesions within the different organs were classified as minimal (1), mild (2), mild to moderate (3), moderate (4), marked (5) and severe (6). Different criteria were selected to grade and compare the lesions of the different organs (see Suppl. Table 1). For molecular analysis, reference goldfish and dead and clinically sick goldfish (n = 6) were analyzed individually, and clinically normal goldfish (n = 45) were analyzed as pools of 4 fish each (one with 5 fish). Gill, spleen, and kidney samples were fixed as a pool in isopropanol (100%) and stored at 4°C until further analysis; 25 to 30 mg of alcohol-fixed tissues were then used for DNA extraction via the DNA Mini Kit (Qiagen, Hilden, Germany) according to manufacturer’s instructions. DNA was eluted with 50 µl of elution buffer provided by the kit. CyHV-2 and a particle-associated nucleic acid cyprinid herpesvirus PCRs were performed. 5,6 PCR products were eluted from agarose gels and purified with the QIAquick Gel Extraction Kit (Qiagen) for direct sequence analysis. DNA was sequenced with the Prism BigDye Terminator v1.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA), with primer pairs for the viral polymerase gene. 3 Sequence data were assembled and edited with GCG 11.1 (Accelrys Inc, San Diego, CA, USA).
Two types of negative controls were used for the PCR: (1) no-template controls consisted of PCR water used as contamination and (2) tissue materials (gills, kidney) obtained from healthy goldfish (Riemserort and Greifswald, Germany). To exclude cross-reaction, all samples were additionally analyzed by PCR for the presence of CyHV-1 and CyHV-3. 1
For in situ hybridization (ISH), total DNA obtained from 3 samples of Prussian carp (Carassius gibelio) naturally infected by goldfish herpesvirus (Friedrich-Loeffler-Institut samples) were labeled by digoxigenin-11-2′-deoxy-uridine-5′-triphosphate (DIG-dUTP; 30%) by substitution of 2′-deoxythymidine 5′-triphosphate (70%) during PCR according to the “Nonradioactive In Situ Hybridization Application Manual” (Roche, Basel, Switzerland). Primer pair CyHVpolforint and CyHVpolrevint recognizing parts of the viral DNA polymerase was used to produce DIG-dUTP-labeled probes. 3 As a negative control, an irrelevant DIG-dUTP-labeled probe developed against Lactococcus sp was used. Tissue sections from the dead goldfish as well as from 5 clinically normal, virus DNA–positive goldfish were framed by PapPen (Merck, Darmstadt, Germany), treated with proteinase K, and fixed by 100% ethanol. After air-drying, sections were framed by PapPen again and covered by hybridization mixture and incubated for 1 hour at 42°C. DIG-dUTP-labeled probes (5 µl in 200 µl of hybridization mixture) were applied to each section, placed on the in situ plate of a thermal cycler (Mastergradient; Eppendorf, Hamburg, Germany), and heated to 95°C for 5 minutes for denaturation of the DNA. Slides were incubated overnight at 42°C for hybridization. For removing unspecific bound probes, slides were incubated in 0.4× single-strength citrate at 42°C for 10 mintues. Sections were counterstained with Bismarck-Brown Y.
Tissue pools containing gill, kidney, and spleen from clinically affected animals (n = 7) were incubated on CCB (common carp brain) cells and KF-1 (koi carp fin) cells; cells were incubated for 7 days at 20°C and 26°C, respectively, before subculturing. Together 5 passages were run. Cultures were controlled daily for cytopathic effect. To determine significant differences in the intensity of histologic lesions between reference and clinically normal goldfish and between clinically normal and clinically sick goldfish, a 2-sided Wilcoxon rank-sum test was performed. Because all samples tested were small (n < 50) and contained ties, exact P values could not be calculated; therefore, normal approximation was used. In addition, nonparametric 95% confidence intervals were calculated for each test. P values <.05 with nonparametric 95% confidence intervals not containing or reaching zero were considered statistically significant.
During summer 2010, the only abnormal clinical sign observed by the owner was increased respiratory rate, after which the affected fish died 1 or 2 days later. Abnormal clinical signs found during autumn and winter (indoors at 18°C temperature) included distended abdomen and protruding scales, indicative for ascites and edema of scale pockets (Fig. 1). At the CFWH, no parasites were found in dead and sick goldfish. In the clinically normal goldfish, the parasitologic examination revealed a mild to moderate infestation with Ichthyobodo sp on the gills and/or skin of 6 fish. In 1 goldfish, a mild infestation with Dactylogyrus sp was found within the gills. In 1 of the 3 reference fish, a mild infestation with Ichthyobodo sp on the skin and a mild infestation with Trichodina sp on the gills were diagnosed. No external parasites were found on the other 2 animals.
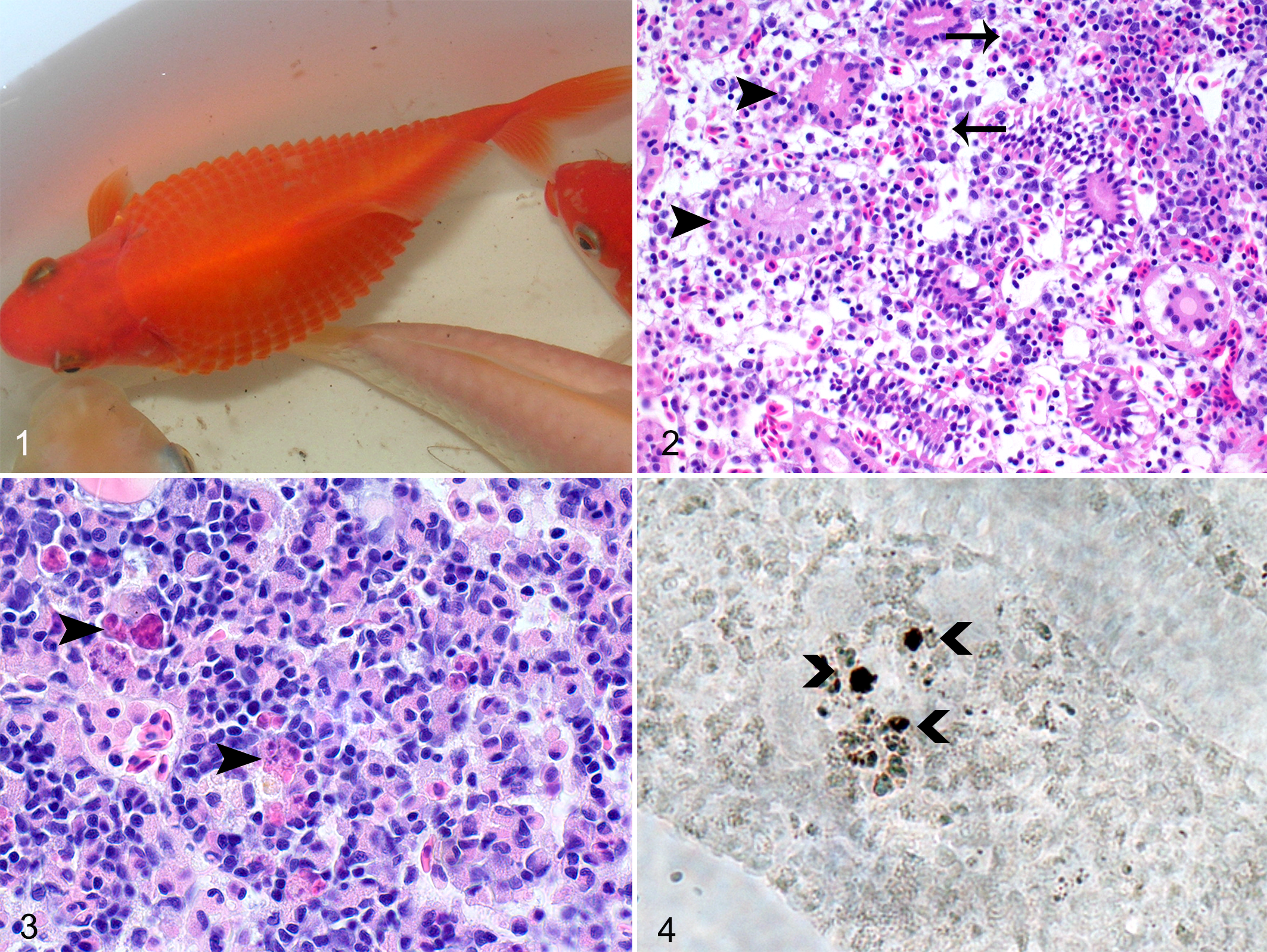
Herpesviral hematopoietic necrosis, Carassius auratus auratus.
No bacterial pathogens were cultivated from any of the goldfish.
The severity of histologic lesions in reference (n = 3), dead (n = 1), moribund (n = 6), and clinically normal (n = 45) fish are summarized in Supplementary Table 1.
The dead and clinically affected goldfish displayed fluid accumulation in skin and celomic cavity, indicative for scale pocket edema (Fig. 1) and ascites; enlarged pale kidneys and a pale liver with multiple pinpoint hemorrhages; and minimal petechial hemorrhages within the skin, subcutaneous tissue, and intestine. Histologic examination revealed multifocal moderate necrosis within the hematopoietic tissue and enhanced single-cell necrosis of the pronephros (examined only in dead goldfish) and mesonephros (in both dead and clinically affected goldfish), along with edema and hemorrhages within the interstitium (Fig. 2), hyaline droplet degeneration and vacuolization within the epithelial tubular cells, together with karyopyknosis (tubulonephrosis) in the dead animal (Fig. 2). In moribund fish, moderate tubular epithelial degeneration within the mesonephros was observed in all of the specimens, while necrosis of hematopoietic tissue and single-cell necrosis were only rarely found (Suppl. Table 1). Compared with clinically normal fish, tubular epithelial cell degeneration in mesonephros (P < .001) and hepatic lipidosis (P < .001) were significantly increased in the moribund fish (Suppl. Table 1). In the dead goldfish, the epicardium and endocardium were severely infiltrated with histiocytes and lymphocytes, with the bulbus arteriosus being most affected, followed by the atrium and the ventricle (Suppl. Table 1, Suppl. Fig. 1). The myocardial compact layer of the ventricle was also inflamed with multifocal areas of necrosis and edema (Suppl. Fig. 1). One of 6 moribund goldfish presented a severe lymphohistiocytic epicarditis, myocarditis of the compact layer, and endocarditis, while the changes in the heart were mild within the other goldfish (Suppl. Table 1).
In dead and moribund goldfish, there was also mild hyperplasia of the gill epithelium. Gram, Giemsa, Ziehl-Neelsen, and periodic acid–Schiff staining revealed no specific infectious agents.
No gross lesions were noted in clinically normal goldfish. However, at the histologic examination, similar lesions were encountered within all goldfish examined (Suppl. Table 1). Single-cell necrosis of the hematopoietic tissue within the pronephros (Fig. 3) was significantly more severe in clinically normal fish as compared with reference fish (P = .009). Whereas no significant differences were obvious regarding tubular epithelial degeneration within the mesonephros, tubuloneogenesis was significantly increased in clinically normal fish versus reference animals (P = .01). Necrosis of the hematopoietic tissue was absent (Suppl. Table 1). All these goldfish exhibited a mild to moderate epicarditis and myocarditis of the compact layer and most fishes, also a mild to moderate endocarditis. The nature of the cardiac inflammation (histiocytic and lymphocytic), affecting the bulbus arteriosus more severely, was similar to that observed in the cardiac lesions of the clinically affected goldfish. Compared with that of the reference fish, epicarditis and myocarditis were significantly enhanced in clinically normal animals (P = .004). A mild epithelial hyperplasia of the gills with some single-cell necrosis was encountered (Suppl. Table 1). As an additional finding, variable numbers of granulomas along with variable numbers of melanomacrophages were observed within the pronephros, mesonephros, spleen, and liver (Suppl. Figs. 2, 3), showing no significant differences to reference goldfish. Granulomas were also scattered randomly within the myocardium of few goldfish. Special stains such as Ziehl-Neelsen revealed no acid-fast bacteria.
Reference fish were diagnosed with mild to moderate proliferative branchitis, minimal epicarditis (Suppl. Table 1, Suppl. Fig. 4), ulcerative dermatitis in 1 animal, and varying numbers of granulomas in liver, kidney, and perivisceral fat tissue in 2 animals (Suppl. Table 1). Myocardium, endocardium, and bulbus arteriosus were unaffected. In the kidney, multiple tubular epithelial cells contained eosinophilic amorphous droplets, interpreted as hyaline droplet degeneration. However, neither necrosis of the hematopoietic tissue nor single-cell necrosis in the interstitium was observed (Suppl. Table 1, Suppl. Fig. 5).
Samples from dead and sick goldfish (n = 7) were positive for CyHV-2 according to the method of Goodwin et al. 6 Pooled samples from clinically normal goldfish (pools, n = 11) were also positive for CyHV-2. Sequence analysis of selected PCR products showed 100% identity with the published sequence for CyHV-2. 5 Samples from reference goldfish were all negative for CyHV-2. Samples from the dead fish, as well as 3 of 6 samples from the sick goldfish and all pools from clinically inconspicuous fish, were tested positive with the particle-associated nucleic acid cyprinid herpesvirus PCR. PCR specifically for CyHV-1 and CyHV-3 in all samples as well as negative controls was negative.
To confirm the presence of virus DNA in the histologic lesions, ISH was performed. In the dead goldfish as well as in clinically normal goldfish, ISH revealed single CyHV-2-infected cells in spleen and gill. A higher number of positive cells were detected in the hematopoietic tissue of the kidney, namely in areas of single-cell necrosis (Fig. 4).
Cell culturing with CCB and KF-1 cells was unsuccessful. No cytopathic effect was observed over the whole incubation period.
HVHN usually occurs in spring and autumn when temperature of the water ranges between 15°C and 25°C. 8,10 Most often, it affects juvenile goldfish (<1 year old) 8,10 ; however, occasionally, all age classes can be affected, especially under stress conditions. 7,12 In the present study, we encountered mortality in adult goldfish particularly during the summer and to a lesser extent during autumn, as well as over the winter months while fish were kept in an indoor pond with water temperature of 18°C. Spawning of goldfish takes place during summer months, which might represent a physiologic stress. Additionally, it is reported that water temperature can influence clinical disease expression and mortality due to herpesvirus infections in cyprinids, like koi herpesvirus and HVHN, with increasing mortality at higher temperature. 8
The disease outbreak reported in our study shows similarities to previous reports of goldfish affected by HVHN. The clinical features observed in our study along with the gross findings of dead and moribund goldfish represent unspecific findings similar to other studies. 8,10 Histology of dead goldfish revealed necrosis within pronephros and mesonephros consistent with other reports, while in our study no necrosis was observed in the spleen. 2,5,8 –10,12,14 However, ISH revealed single CyHV-2-positive cells in the spleen. In clinically normal goldfish, no necrosis was evident within the hematopoietic tissue of the pronephros and mesonephros; instead, multiple single-cell necroses in the pronephros represented a prominent feature, significantly different to virus DNA–negative reference animals (P = .008). In fish examined in this study, eosinophilic intranuclear inclusion bodies were found neither within the hematopoietic cells nor within the epithelial cells. This is in contrast to previous reports, where intranuclear inclusion bodies with marginated chromatin were regularly found. 2,5,8 –10,12,14 However, the lack of inclusions in our study could be a result of the early stage of disease, as most investigated animals were subclinically infected. This is supported by the fact that despite the lack of inclusion bodies, CyHV-2 DNA was found in different organs by PCR as well as by ISH.
Interestingly, severe changes were observed within the heart of dead goldfish as well as to a milder extent of clinically affected and subclinically infected animals, consistent with epicarditis, endocarditis, and multifocal necrosis of the myocardium, while endocardial and myocardial lesions were significantly enhanced as compared with reference fish (Suppl. Table 1, Fig. 4). These findings represent a rather unusual presentation of the disease, as only 2 studies have reported mild heart lesions so far. 2,5 As no virus DNA was found in the heart lesions by ISH, it remains unclear whether the heart lesions are directly correlated to the CyHV-2 infection or caused by additional stressors.
In the past, diagnosis of HVHN was not an easy task, as CyHV-2 is difficult to isolate and maintain in cell culture. 10 Previous diagnoses of HVHN have been based on histologic findings of necrosis of the renal hematopoietic tissue and electron microscopy to detect herpesvirus particles, 2,8,14 while more recent reports relied on histologic findings and PCR assay. 4,6 Our diagnosis of HVHN was based on histologic findings along with PCR results and ISH, while the polymerase gene-specific product shared 100% nucleotide sequence identity with the published sequence for the viral polymerase gene of CyHV-2. 3 Goodwin et al 7 demonstrated that virus DNA is present in most healthy goldfish populations in United States and that mortality occurs when the fishes are subjected to stress along with permissive water temperature. No histologic examination of the different organs was performed in the latter study. We demonstrated that mild to moderate pathologic findings could already be observed within the internal organs, especially in the kidney, of goldfish showing no clinical signs but being positive for CyHV-2 by PCR. The virus DNA could be confirmed by ISH in spleen, gills, and kidney. Namely in the kidney, areas of single-cell necrosis were revealed to harbor CyHV-2 DNA. In animals negative for CyHV-2 by PCR (reference animals), lesions in the renal hematopoietic tissue were not present (Suppl. Fig. 5), further indicating that these lesions are associated with the herpesviral infection. This further supports the hypothesis that goldfish can be latently infected with CyHV-2 and that these latently infected fishes are predisposed to develop apparent disease signs following stress and activation of the virus. Our results in the reference animals, however, indicated that virus might not be ubiquitously distributed in Swiss goldfish populations.
The transmission pathway of the virus between the goldfish is not yet known. Horizontal transmission by cohabitation with gills or injured skin as portal of entry could be suspected in our study. In the present study, koi were kept with goldfish. However, koi showed no signs, and PCR analyses of 2 koi from the pond did not reveal the presence of CyHV-2 (data not shown). This result may be due to the PCR sensitivity of generic primer pairs and a concentration of the CyHV-2 in those koi that might have been below the detection limit of the assay.
This study detected the presence of CyHV-2 and the manifestation of the disease in dead and sick goldfish in Switzerland. Furthermore, the presence of viral DNA, as well as the characterization of early histologic lesions with lower extension and severity, was demonstrated within clinically normal goldfish, highlighting the fact that the virus might be more widespread within goldfish populations than currently assumed. However, more work, possibly an experimental infection, needs to be done to confirm these findings and to support our hypothesis.
Footnotes
Acknowledgements
We thank our colleagues from the Centre for Fish and Wildlife Health, especially Anja Möller, for their help during the samplings and the staff of the histology laboratory of the Institute of Animal Pathology for preparation of the histologic sections. We also thank Matthias Escher from Koipraxis as well as Ralph Knüsel from Fishdoc GmbH for time and effort. Finally, we thank the owner of the affected goldfish/koi pond for his support and assistance, as well as Irina Werner (Friedrich-Loeffler-Institut) for her excellent technical work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
