Abstract
Genetic and environmental factors both play a role in the occurrence of age-related disease. To examine the genetic contribution to the development of spontaneous lesions in aging animals, a complete range of tissues was comprehensively analyzed by histopathology from 180 individually housed ad libitum–fed or 40% calorically restricted 24-month-old male and female mice of 2 parental strains—DBA/2NNia (D2) and C57BL/6NNia (B6)—and the F1 cross B6D2F1/NNia. Several strain- and diet-dependent patterns of lesions were identified. Many lesions were genotype dependent and exhibited recessive phenotypic expression, defined as being common in 1 parental strain but infrequently observed in the F1 cross (eg, glomerulonephritis in B6 mice), while others were maintained from 1 parental strain to the F1 with similar frequencies (eg, reproductive tract leiomyoma in D2 mice). Other lesions were common regardless of genotype (osteoarthritis, periodontitis). Only rare lesions were more common in the F1 but underrepresented in the 2 parental strains. Furthermore, F1 mice had a lower number of overall total lesions and a lower number of tumors than either parental strain. Caloric restriction reduced the total number of lesions and neoplasms regardless of genotype but differentially affected genotype-dependent lesions in B6 and D2 mice, with B6 mice more sensitive to the effects of caloric restriction than D2 mice. In summary, genetics and environmental factors (eg, dietary restriction) both substantially contribute to the pattern of lesions that develop as animals age.
Aging is a complex and multifactorial process that leads to progressive functional decline and frequently manifests as structural changes observable grossly or by routine histopathology. These structural changes may be of little clinical consequence (eg, alopecia) or may result in the death of the animal (eg, malignant neoplasms). Many studies of aging seek to identify biomarkers or measures of aging that can be used to unravel its underlying molecular mechanisms. 14,20,23 These studies frequently utilize the mouse model due to several advantages as compared with other animal model systems—namely, the mice’s relatively short life span, small size, and easy husbandry. In addition, genetic variables can be easily controlled through the use of inbred or recombinant strains, and environmental factors can be tightly regulated.
Caloric intake is 1 environmental variable receiving considerable attention with respect to its positive effects on aging and life span. In several models—including yeast, worms, flies, rodents, and primates—caloric restriction (CR) of up to 40% on diets that maintain nutrient quality delays the onset of age-associated lesions, lengthens life span, and (in mammalian models) decreases tumor incidence. 7,10,18,27 The mechanisms underlying this phenomenon have also been investigated and may include inflammatory pathways, oxidative stress, and aspects of membrane, mitochondrial, and lipid biology. 1,6,25
The average life span of mice demonstrates strain-related variability, with an average life span of 30 months in B6 mice, 20 months in D2 mice, and 33 months in the F1 cross. 5,12 Examination of the interaction of genetic background and responsiveness to CR finds that the effects of CR on life span, however, are not universal. B6 mice consistently demonstrate a 15% to 25% increase in median life span with CR. 2 In contrast, although in previously published studies using the cohort of D2 mice described herein showed a 18% (males) to 56% (females) life span increase, several more recent studies have failed to show increased life span, and D2 mice actually have reduced life span when shifted from ad libitum (AL) feeding to CR later in life. 11,19 The reasons for the strain-dependent differences in response to dietary conditions and the difference between the current cohort and those in other studies are not currently understood.
The goals of this study were to further explore the genetic and environmental components of aging by systematically documenting the lesions present in mice near the end of their laboratory life span, to expand on a previous study in this area. 5 The genotype groups utilized include the common laboratory strains C57Bl/6 (B6) and DBA/2 (D2) mice, as well as the F1 cross B6D2F1. F1 mice are genetically homogenous—that is, each genomic locus has 1 allele from B6 and 1 from D2—and they were included to explore the phenotypic stability of genotype-dependent pathology. In addition, the development of lesions was examined in each genotype group fed either AL or 40% CR to explore the interaction between genetic and environmental variables.
Materials and Methods
The National Center for Toxicological Research (NCTR; Jefferson, AR, USA) program of animal care and use has been accredited by the Association for Assessment and Accreditation of Laboratory Animal Care since 1977, and Tufts University/Tufts Medical Center, Boston Campus, has been accredited by the association since 1966. All procedures used in this study (1989–1990), including euthanasia, were Institutional Animal Care and Use Committee approved and in accordance with relevant local and federal guidelines. The experimental husbandry and tissue-processing conditions have been described. 5,28 Briefly, in 1989–1990, 30 male and 30 female age-matched C57BL/6NNia (B6), DBA/2NNia (D2), or B6D2F1/NNia (F1) mice obtained from the National Institute on Aging (180 mice total) were raised at the NCTR. Following weaning at 3 weeks of age, animals were singly housed in polycarbonate mouse cages. The air supply was HEPA filtered at 72°F with 40% to 60% humidity and a 12-hour light cycle. 28 Beginning at 14 weeks of age, half the mice were fed NIH-31 Open Formula Mouse/Rat Sterilizable Diet AL, and half were fed at 60% the caloric level of their AL counterparts with maintenance of vitamin and mineral quality. At 24 months of age, mice were euthanized by exsanguination following 0.6 mg/g of intraperitoneal tribromoethanol (Avertin) administration and were processed for histologic examination either at the NCTR or, in some cases, within 48 hours of live shipment to Tufts Veterinary Diagnostic Laboratory (Boston, MA, USA). Fixation procedures were vascular infusion and several weeks of postfixation in Bouin fixative or in formalin–acetic acid–alcohol, followed by decalcification in nitric acid. Standard paraffin tissue embedding, 6-μm sectioning, and hematoxylin and eosin–staining procedures were utilized.
Archived hematoxylin-and-eosin slides were examined to detect any deviation from normal without regard to clinical significance, severity, or contribution to mortality. All such deviations are termed “lesions.” A lesion was defined as common if it occurred in tissues from ≥5 mice in any sex, genotype, or diet group. This number was chosen to enable statistical detection of differences while minimizing sample numbers. Tissues were examined regardless if there were no gross lesions, and they included the following: adrenal, urinary bladder, bone (mandible with maxilla considered separately), bone marrow, brain (in situ following Bouin fixation and/or dissected), ear and spiral ganglion, exorbital gland, eye, fat, intestine (all segments considered together), Harderian gland, heart, joints (generally intervertebral, temporomandibular, and/or stifle), kidney, liver, lung, lymph nodes, mammary glands, muscle, nerves, ovary, pancreas, pituitary gland, preputial/clitoral glands, prostate, salivary glands, seminal vesicles, spleen, stomach, thyroid, teeth, testis and epididymis, trachea, uterus, vagina, and blood vessels throughout tissues. One of or both paired organs were sampled. Lesion classification was based on the work of Bronson. 4 A 2 × 2 contingency table and Fisher exact test were used to determine the statistical significance of differences in occurrence of common lesions between diet and genotype groups. To account for sampling error and differences in the numbers of tissues available from animals examined among groups, data are presented as a percentage of (1) the number of that organ or (2) the total number of tissues examined overall within a diet and/or genotype group (Suppl. Table 2).
Results
Total Lesion Burden and Neoplasia
Animals were scored for 143 independent lesions in all major organ systems (Figs. 1 –4). A total of approximately 3650 lesions in 10 200 tissues were observed, with 36% of organs containing ≥1 lesions overall. The genotypes and dietary groups differed in total lesion burden, with B6 AL and D2 AL animals equivalent to each other and containing significantly more lesions than the F1 AL (P < .01; Fig. 1). These differences were erased with CR (P > .05) in females but were preserved in males. In addition, within a genotype, CR significantly reduced total lesion burden for all groups (P < .05). Between the sexes, female B6 CR, F1 AL, and F1 CR mice had significantly more lesions than males of their respective groups, while male D2 CR mice had significantly more lesions than female D2 CR.
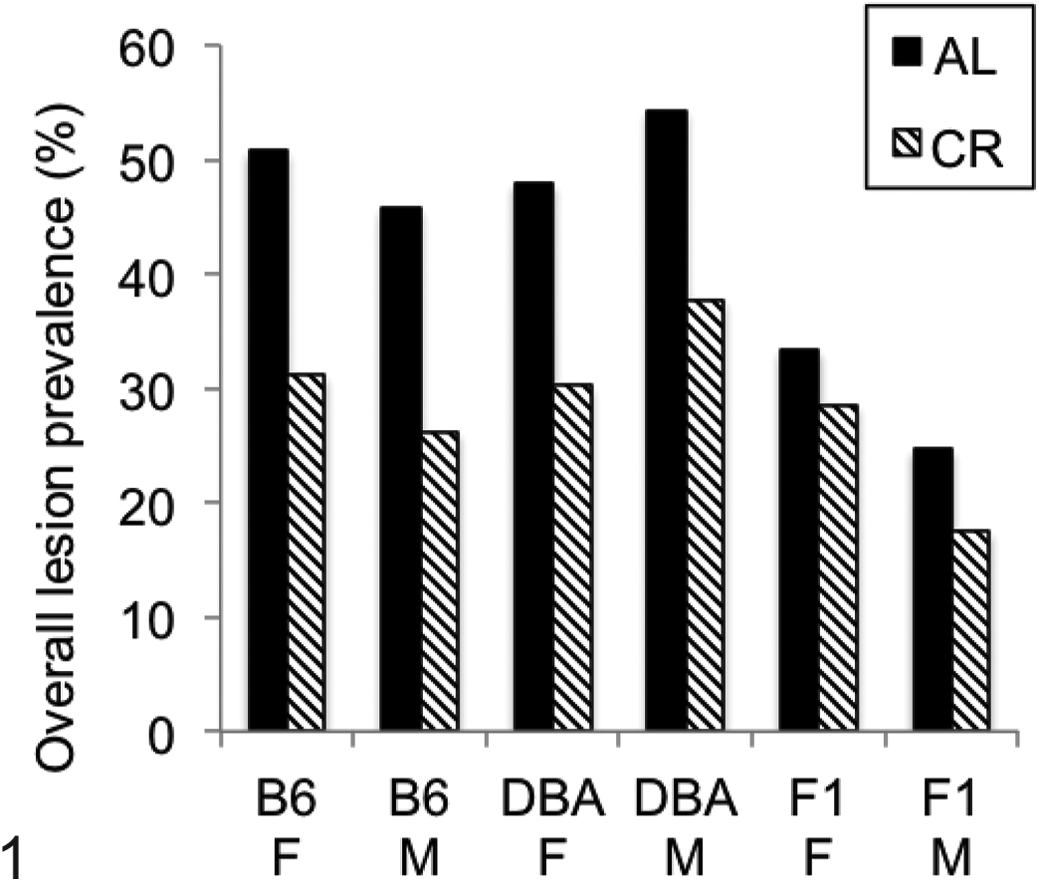
Total lesion burden. Data are shown as number of lesions/total number of tissues examined in all mice of each genotype/diet group to account for differences in numbers of animals examined. Both sexes of all genotypes showed a significant reduction in total lesion number with calorie restriction (P < .05). AL, ad libitum fed group; B6, C57Bl/6NNia mice; CR, 40% calorically restricted group; DBA, DBA/2NNia mice; F, female; F1, B6D2F1/NNia mice; M, male.
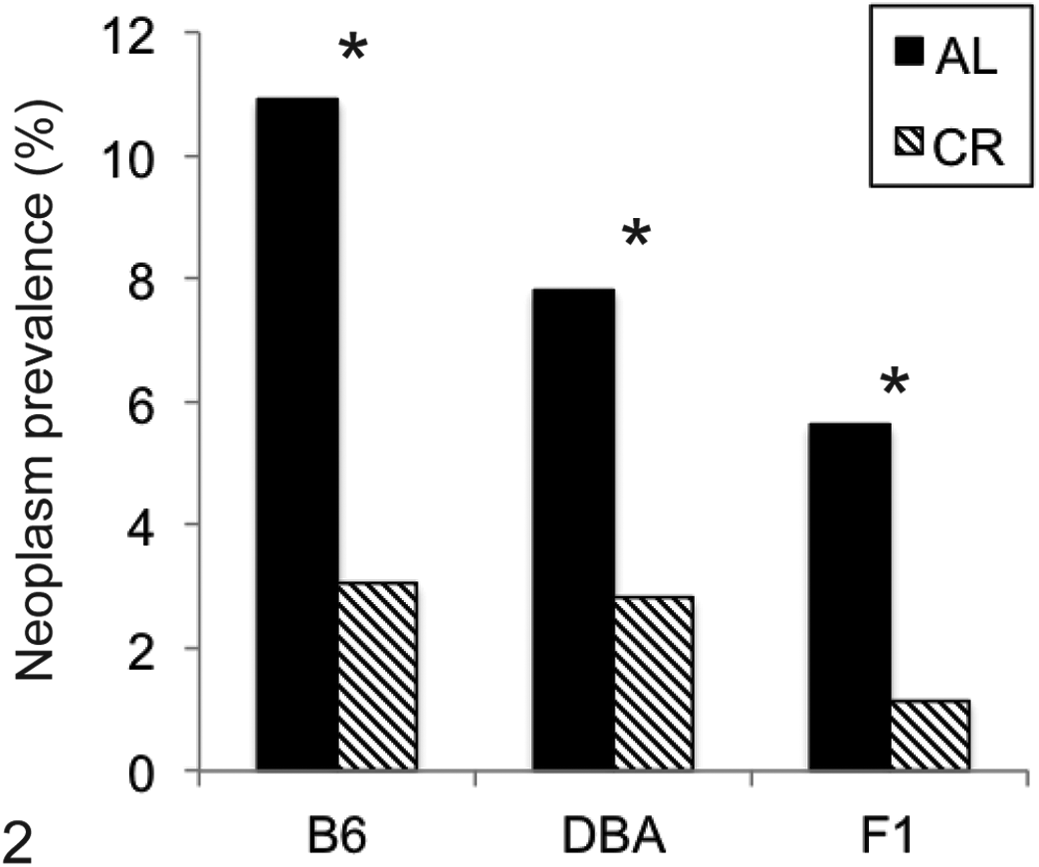
Overall prevalence of neoplasia. Data are shown as number of lesions/total number of tissues examined in all mice of each genotype/diet group. Males and females had statistically equivalent number of tumors and are pooled. C57Bl6/NNia (B6) ad libitum (AL) mice had more tumors as compared with B6D2F1/NNia (F1) AL mice. All groups showed a significant reduction in total neoplasia with calorie restriction. *P < .05. CR, 40% calorically restricted group; DBA, DBA/2NNia mice.
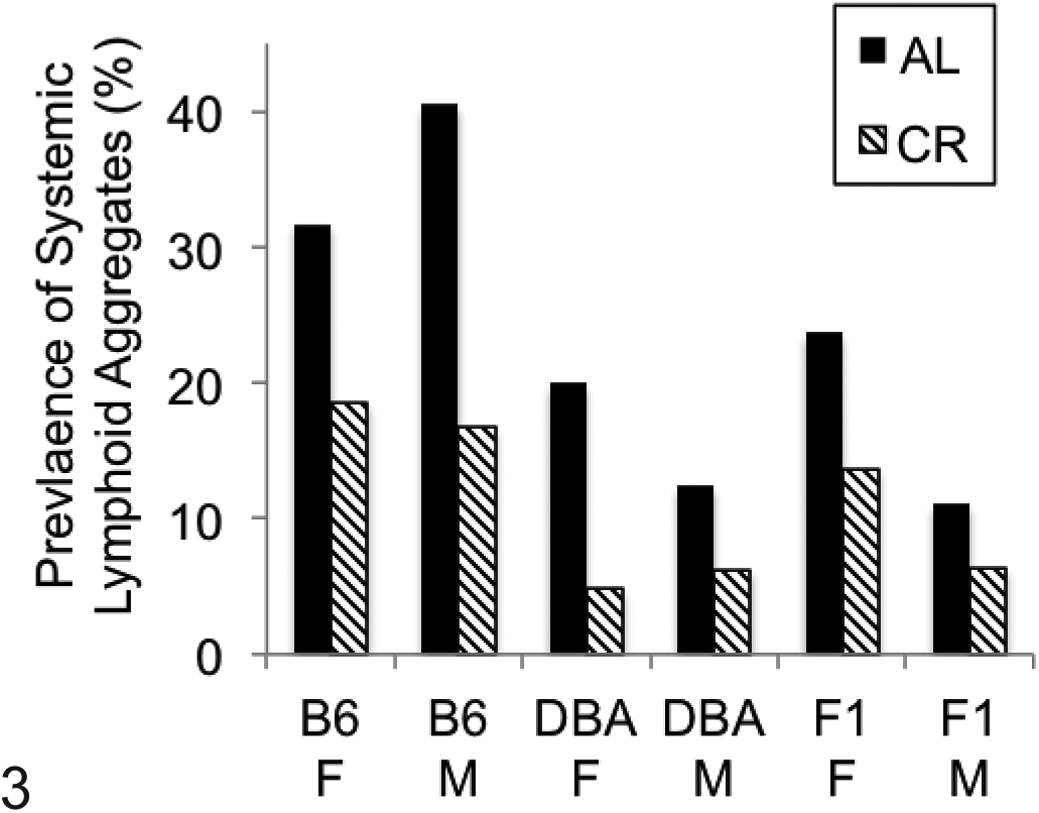
Overall prevalence of systemic lymphoid aggregates. Data are shown as number of tissues with lymphoid aggregates/total number of tissues examined in all mice of each genotype/diet group (see also Suppl. Table 2). Significant reduction in lymphoid aggregates by caloric restriction (P < .05) was observed for all genotype and sex groups. AL, ad libitum fed group; B6, C57Bl/6NNia mice; CR, 40% calorically restricted group; DBA, DBA/2NNia mice; F, female; F1, B6D2F1/NNia mice; M, male.
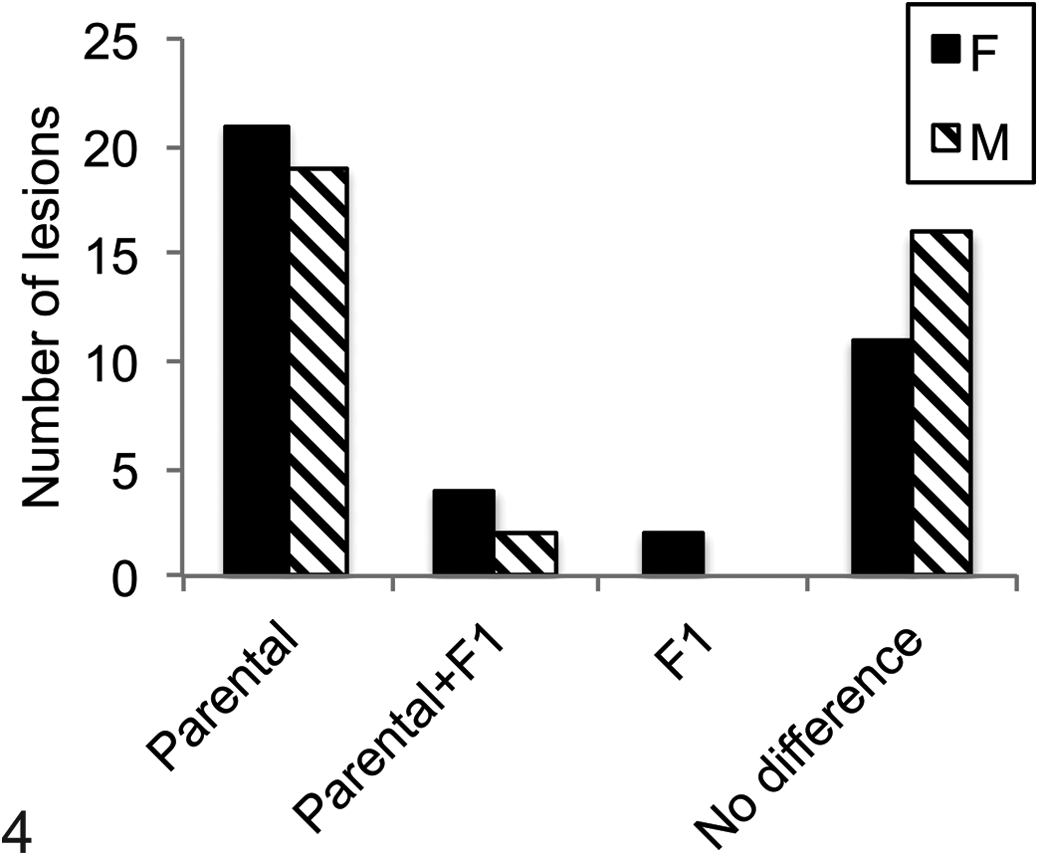
Genotype-specific patterns of lesions. Most commonly, a lesion was present in 1 parental strain, either C57Bl/6NNia or DBA/2NNia, significantly more often than in the other parental strain or the F1 cross (Parental). Less commonly, lesions were more prevalent in 1 parent and the F1 cross as compared with the other parental strain (Parental+F1). Only 2 lesions were significantly more common in the F1 cross versus either parent (F1). Many other lesions did not differ among the 3 genotypes studied and were considered genotype independent (no difference). F, female; M, male.
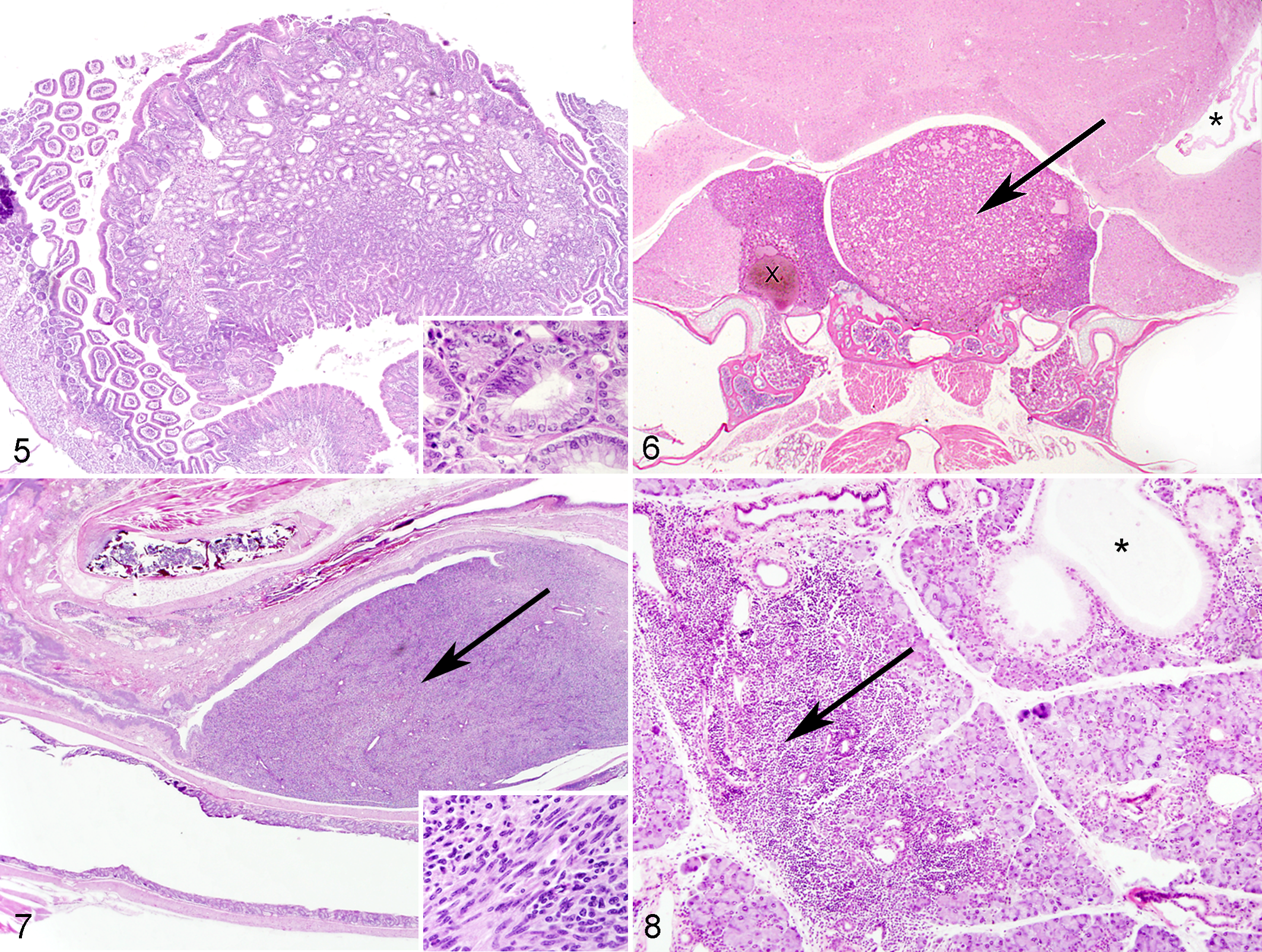
The total number of tumors was affected by both genotype and diet (Fig. 2, Table 1). B6 AL animals had significantly more tumors overall than F1 AL animals (P = .02–.03) for females and males, and CR significantly reduced total tumor burden within each genotype (P < .01). The most common tumor types observed were hematopoietic tumors (lymphoma, histiocytic sarcoma), pituitary adenomas in females (Fig. 5), and liver tumors in males. These tumors were each primarily seen in B6 mice and (with the exception of lymphoma in female B6 mice) were all decreased with CR. The only tumors seen more often in D2 mice than B6 were intestinal adenomas (Fig. 6) and reproductive tract leiomyomas (Fig. 7), neither of which was affected by CR.
Neoplasia: Occurrence of Neoplasms in Each Diet/Genotype Group.a
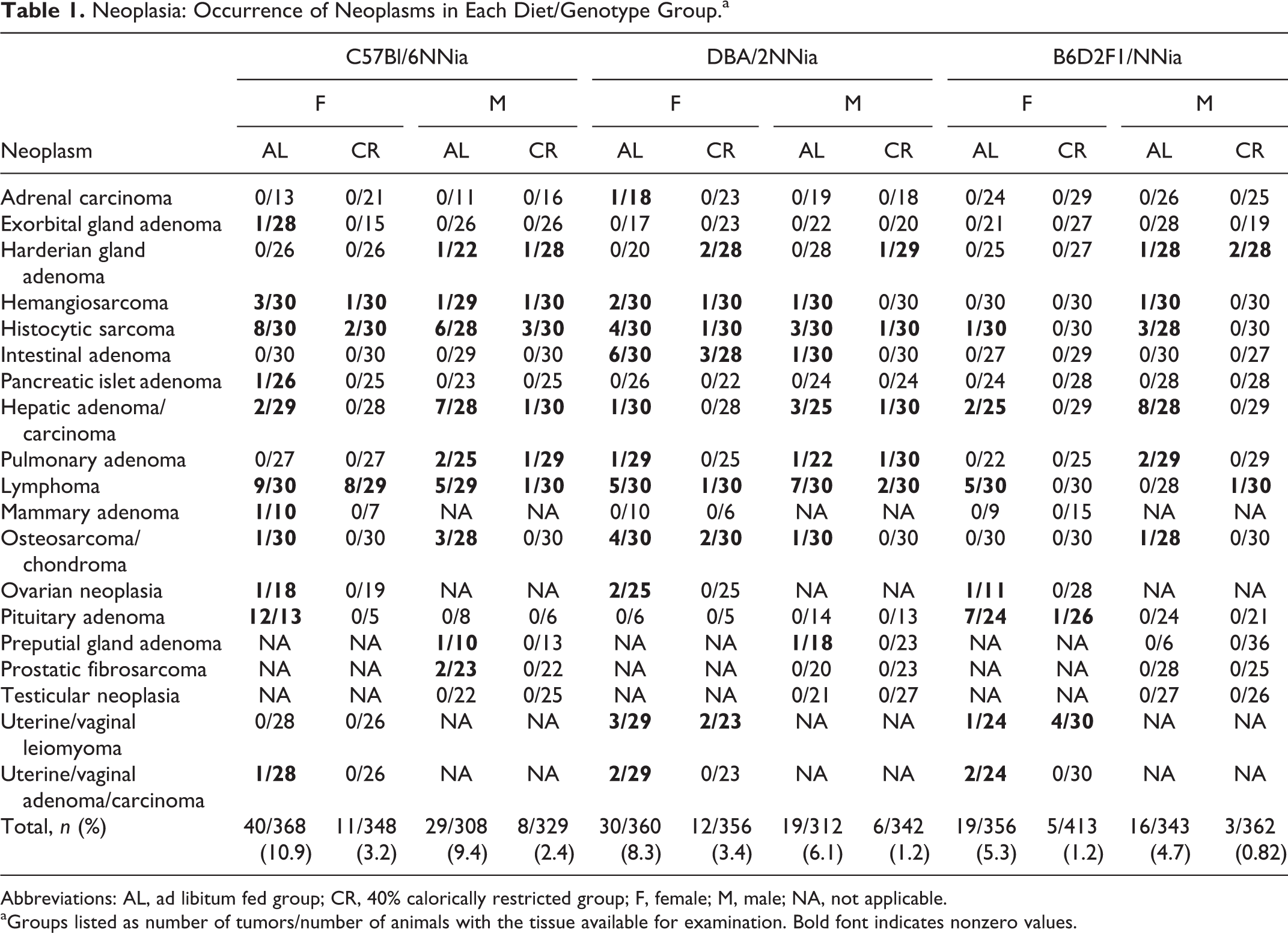
Abbreviations: AL, ad libitum fed group; CR, 40% calorically restricted group; F, female; M, male; NA, not applicable.
aGroups listed as number of tumors/number of animals with the tissue available for examination. Bold font indicates nonzero values.
Systemic Lymphocyte Aggregates
Lymphocyte aggregates were commonly observed in multiple organs (Fig. 3; Suppl. Table 1). B6 animals had significantly more aggregates in the exorbital gland, fat, liver (females only), lung, salivary gland (males only; Fig. 8), and prostate than other strains, while the observed tissue prevalence of lymphocytes in the bladder, Harderian gland, nerves/spinal nerve roots, pancreas, and stomach was equivalent regardless of genotype. When genotypes were pooled, females overall had significantly more total lymphoid aggregates than males (P = .03). CR reduced total systemic lymphocyte aggregate accumulation within each genotype, although there was variable significance of the effect of CR within an individual organ system among different genotype and sex groups (Suppl. Table 2).
Degenerative, Proliferative, and Miscellaneous Lesions and Effects of CR
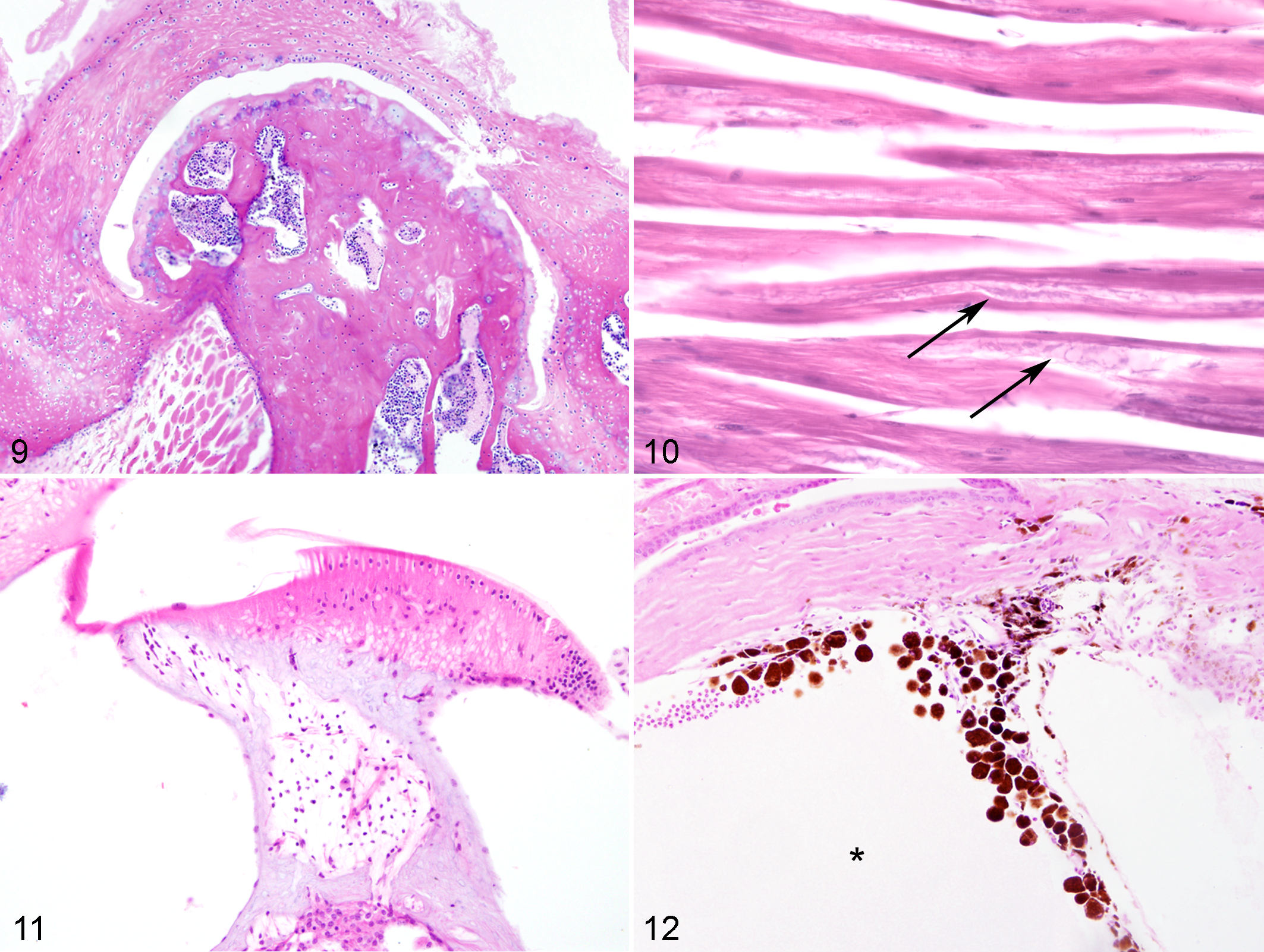
Twenty-one common genotype-independent lesions were identified (present in statistically equivalent amounts in all 3 genotypes)—including bone marrow hematopoietic atrophy with fatty replacement, dental abnormalities including periodontitis with or without impacted hairs, degenerative (osseous) hyperplasia of the jaw and hypercementosis primarily affecting molar tooth roots, arthritis (particularly of the temporomandibular joint, stifle, and vertebral joints; Fig. 9), tubular aggregates of sarcoplasmic reticulum within skeletal muscle in males (Fig. 10), atrophy and loss of spiral ganglion neurons in the ear (Fig. 11), and others (Table 2). 8 These were variably affected by CR (Table 2). Fifty-one rare lesions (occurrence <5 in all genotype-diet groups) were also observed and are omitted from Supplementary Table 2—namely, encephalitis, cerebral corpora amylacea, pneumonia (various), ectasia of lymphatics or splenic or ovarian vessels, muscle fibrosis, pyelonephritis or other nephritis, gall bladder dilation or fibrosis, accumulation of pigmented macrophages in the spleen/Harderian gland/lung, otitis media, hepatic lipidosis, hepatic sinusoidal atrophy, gastric hyperkeratosis or mucosal hyperplasia, nodular pancreatic hyperplasia, pancreatitis, uterine edema, adrenal cysts, splenic fibrosis, tracheitis, meningeal pigment, inflammation in exorbital lacrimal gland, retinal degeneration without concurrent glaucoma, keratitis, hyphema, myositis, bladder stones, hyperplasia of bladder mucosa, pulmonary hemorrhage, focus of dysplastic hepatocytes, sperm granuloma, myocarditis, cystitis, disc prolapse, abdominal nodule of necrotic fat, Harderian gland inflammation/cystic dilation, lymphadenitis, testicular cyst, epididymal inflammation or dilation, and amyloid in the adrenal and exorbital lacrimal glands, intestine, heart, and kidney.
Genotype Dependency of Common Lesions in Male and Female Effects of CR.a
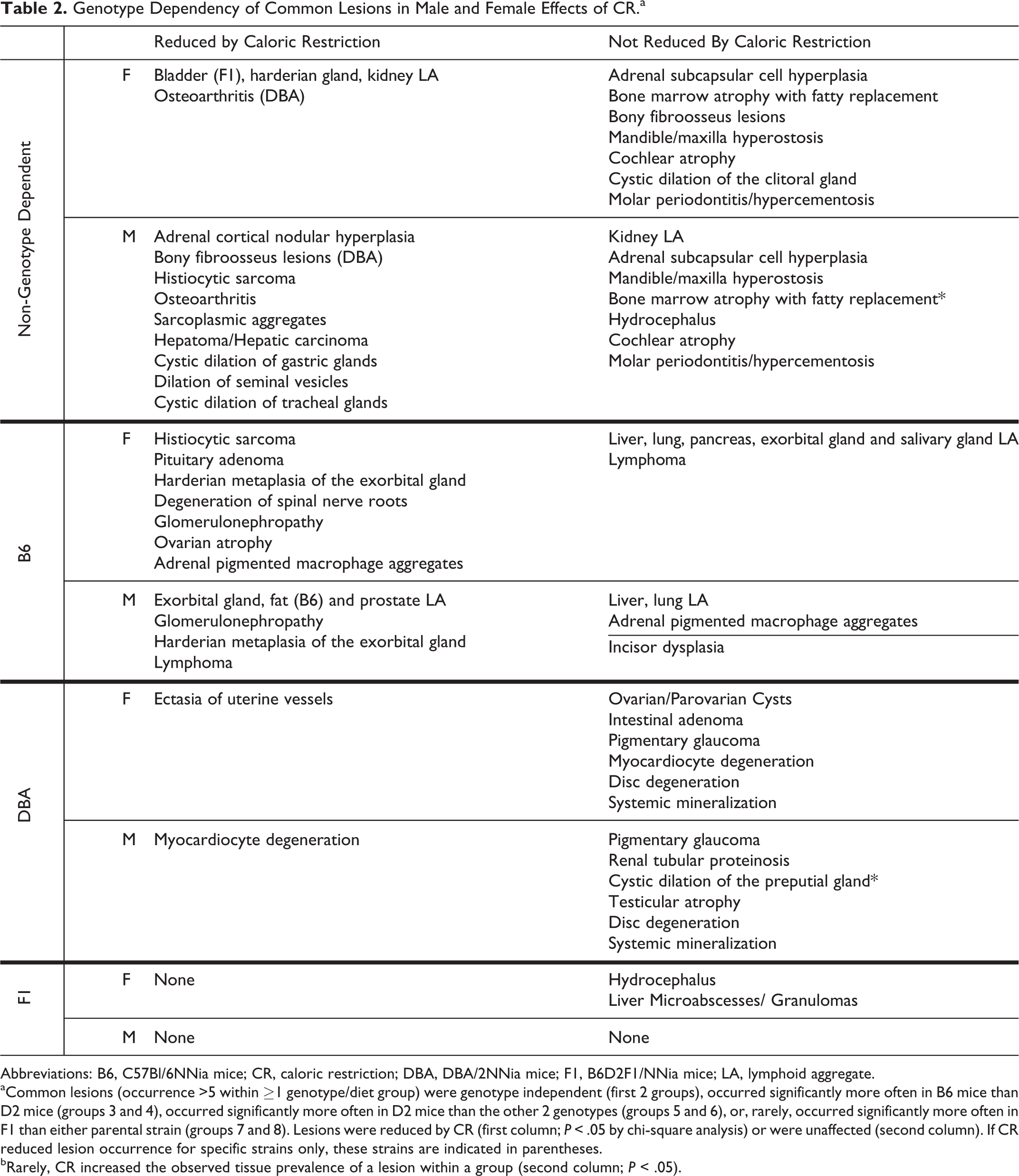
Abbreviations: B6, C57Bl/6NNia mice; CR, caloric restriction; DBA, DBA/2NNia mice; F1, B6D2F1/NNia mice; LA, lymphoid aggregate.
aCommon lesions (occurrence >5 within ≥1 genotype/diet group) were genotype independent (first 2 groups), occurred significantly more often in B6 mice than D2 mice (groups 3 and 4), occurred significantly more often in D2 mice than the other 2 genotypes (groups 5 and 6), or, rarely, occurred significantly more often in F1 than either parental strain (groups 7 and 8). Lesions were reduced by CR (first column; P < .05 by chi-square analysis) or were unaffected (second column). If CR reduced lesion occurrence for specific strains only, these strains are indicated in parentheses.
bRarely, CR increased the observed tissue prevalence of a lesion within a group (second column; P < .05).
The most frequently observed distribution pattern of common lesions (occurrence ≥5 in at least 1 diet-genotype group) was of genotype-dependent lesions that were observed significantly more often in 1 parental strain compared with the other parental strain or the F1 cross (Fig. 4). B6 genotype-dependent lesions included pituitary adenomas, hematopoietic tumors, and multiorgan lymphoid aggregates as described above. In addition, B6 mice had significantly more ovarian atrophy, membranoproliferative glomerulonephropathy, accumulation of pigmented macrophages in the adrenal gland, Harderian metaplasia of the exorbital lacrimal gland (Fig. 8), dysplastic incisor teeth with denticle formation (males), and degradation of spinal nerve roots (females) than other genotypes. With the exception of adrenal gland pigment and dental dysplasia, the observed tissue prevalence of these lesions was reduced with CR (Table 2).
D2 mice had significantly increased intestinal adenomas, multiorgan mineralization, myocardial degeneration, ovarian cysts, ectasia of uterine vessels, disc degeneration (males), renal tubular casts, testicular atrophy with loss of mature spermatids, cystic dilation of preputial glands, and ocular abnormalities related to pigmentary glaucoma (Fig. 12). Ocular findings ranged from the accumulation of melanin-laden macrophages in the filtration angle to lesions associated with advanced glaucoma, including cataracts, optic nerve cupping, loss of ganglion cell layers, corneal inflammation, dilated anterior chambers, and, in some cases, progression to phthisis bulbi. 15 Mineralization occurred in several organs, most commonly muscle (including tongue and heart), and was observed more often in males than females (Suppl. Table 1). In males but not females, mineral was also frequently observed in the kidney, lungs, and blood vessels of the testis. Among the D2 genotype–specific lesions, only ectasia of uterine vessels and myocardial degeneration (in males but not females) were reduced by CR.
Compared with lesions that were significantly more common in 1 parental strain and decreased in the F1, relatively few lesions were maintained from 1 parental strain to the F1 cross (Suppl. Table 2). Specifically, B6 and F1 mice had more ovarian pigmented macrophage aggregates and endometrial cysts than D2 mice, while female D2 and F1 mice had more disc degeneration and reproductive tract leiomyomas than female B6 mice. In addition, male D2 and F1 mice had significantly more liver microabscesses than male B6 mice. Liver microabscesses were more common in F1 females than in females of either parental strain and along with mild hydrocephalus were the only 2 lesions with this distribution pattern. Neither of the F1 genotype–dependent lesions was affected by CR.
Discussion
The functional decline that is associated with the aging process is associated with observable structural changes. These range from presumptively clinically insignificant findings, such as lymphoid aggregates in various organs, to malignancies and degenerative lesions that result in readily apparent morbidity and mortality. The historical design of aging studies has included both cross-sectional and longitudinal studies, which allow for different sets of observations and conclusions to be formed about the aging process. Although the most common causes of death cannot be as readily ascertained in cross-sectional studies as in longitudinal ones, clinically healthy animals euthanized at scheduled time points may have early stages of those same potentially fatal lesions despite a lack of apparent functional change. In the current cross-sectional study, the 24-month point was chosen to allow for direct comparison of lesion burden, albeit without regard to lesion severity, across different genotypic and dietary groups. This age was chosen to allow expression of a fuller spectrum of lesions as compared with younger groups without excessive spontaneous loss prior to the selected time point, which may underestimate the prevalence of severe, rapidly fatal lesions.
Genotype plays a substantial role in determining predisposition to certain lesions. 3,9 Several of the common lesions observed in these studies have been described in B6 and D2 mice, such as glomerulonephropathy, hydrocephalus, and hematopoietic tumors in B6 mice and pigmentary glaucoma, multiorgan mineralization, and myocardial degeneration in D2 mice. 4,5,15,23 The occurrence of tumors, particularly in B6 mice, was also largely consistent with previous studies in which female B6 mice were prone to pituitary adenomas (and associated galactorrhea), male B6 mice were prone to liver adenomas and carcinomas, and both sexes were predisposed to hematopoietic tumors. 2,13,21,26 Other B6 genotype–specific lesions of interest not highlighted in previous studies include a higher observed prevalence of ovarian atrophy, Harderian metaplasia of the exorbital gland, and multiorgan accumulation of lymphoid aggregates (especially in females) as compared with D2 mice. D2 genotype–specific lesions of interest included testicular atrophy and intestinal adenomas (in females). Interestingly, despite the marked variability in reported life span between B6 and D2 mice, no differences in the occurrence of neoplasia or overall lesion burden were observed.
B6D2F1/NNia (F1) mice have no specific reported lesion predisposition, although in a previous longitudinal study, aged mice developed similar lesions as seen in this study, including cystic endometrial glands, testicular calcification, and cellular alterations in the adrenal cortex. 29 The F1 mice had fewer lesions overall versus B6 and D2 mice, supporting the theory of hybrid vigor and consistent with their longer life span versus either parental strain (see also http://www.research.jax.org). 5,19 Consistent with this finding, most genotype-specific lesions identified in either parental strain were observed significantly less often in the F1 cross and thus were defined as exhibiting a recessive phenotype expression. Only a few lesions were maintained from 1 parental strain to the F1 and were defined as exhibiting a dominant phenotypic expression, including endometrial cysts retained from B6 mice and uterine/vaginal leiomyomas retained from D2 mice. The specific genetics behind these patterns of phenotypic expression, such as sex linkage or locus homozygosity/heterozygosity in each strain, were not examined in this study and remain an interesting area of inquiry for future work. Just 2 lesions were more common in the F1 cross than in either parent—namely, mild hydrocephalus and foci of inflammation in the liver, both significant in females only. Spontaneous hydrocephalus has been reported in B6 mice, although the underlying developmental abnormality is unknown and was observed in 2 of 29 male B6 mice in the current study. 24 In female B6 mice, hydrocephalus occurred only in conjunction with pituitary adenomas and brain compression. The pathogenesis of the observed hydrocephalus in the F1 mice is unknown.
There were also many common lesions (11 in females and 16 in males) that did not differ among the 3 analyzed strains and are thus contenders for general measures of aging in inbred mice (Table 1). However, the conclusions that can be drawn in this area from comparisons of small numbers of strains is limited, and a wider variety of strains and ages should be evaluated before concluding this group of lesions to be truly aging related but genotype independent. 16 For example, early deafness from degeneration and loss of spiral ganglion neurons is a reported predisposition of both B6 and D2 parental strains but does not occur universally across mouse strains. Interestingly, several lesions often seen in aging mice were infrequently observed in this study—namely, multiorgan amyloidosis, crystal pneumonia, polyarteritis nodosa, lung tumors (previously reported in male B6 mice), and ulcerative dermatitis of B6 mice. 17 The reasons for these absences are unclear and may be related to reduced stress with individual housing or other unknown environmental or genetic substrain variation.
CR reduced total lesion burden and neoplasia in all genotype groups, consistent with its reported anti-aging and life span–extending effects. 2 For any given individual lesion, however, the effects of CR were inconsistent and variable, depending on strain background and sex. For example, myocardial degeneration in D2 mice was reduced by CR in males only, and lymphoma was reduced by CR in all genotype and diet groups except female B6 mice. In addition, common genotype-specific lesions in D2 mice were less often affected by CR than those in B6 mice. For example, ocular abnormalities and multiorgan mineralization in D2 mice were unaffected by CR, and myocardial degeneration was reduced only in CR males, whereas many of the genotype-specific lesions in B6 mice were reduced by CR treatment (glomerulonephropathy, ovarian atrophy, pituitary adenoma, etc). The effects of CR on glaucoma development in D2 mice are consistent with previous work showing that the treatment slightly delays the progression of ocular lesions but does not prevent it. 22 The relative insensitivity of D2 mice to CR with regard to lesion development is in line with previous reports that CR has little positive effect on overall life span, although the mechanisms behind this phenomenon remain undetermined. 11,19 CR also rarely increased the observed tissue prevalence of specific lesions—namely, accumulation of pigmented macrophages in the ovary, hematopoietic atrophy of bone marrow with replacement by fat in male mice, and cystic dilation of the preputial glands. Thus, while CR reduced lesion development and neoplasia, its effects were not universally beneficial for all lesions and across all genotypes.
The relationship between environmental variables such as CR and genetics, sex, and aging is complex. Recognizing the contribution of each component is necessary for understanding the aging process. The current study provides a platform for future studies investigating the molecular mechanisms of these lesions and the aging process in general. In addition, this work thoroughly documents systemic background spontaneous lesions in aging mice from 3 commonly used strains without regard to the clinical importance of the lesions and is thus of use for interpretation of nearly any study using these strains.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
