Abstract
In research studies using rats or mice, the cause of death is often not evaluated or reported. An analysis of the causes of death is particularly valuable for aging and carcinogenesis studies. Comparing causes of death among the study groups is often an important adjunct to the biochemical, molecular, clinical, and histopathologic findings. The methods for evaluating causes of death, contributing causes of death, and comorbidities have been suggested in several publications. Surprisingly, in important mouse aging studies, causes of death are often not reported. Cause-of-death assignment in preclinical rodent model aging research suffers from a lack of a standardized approach and an understanding of the value that it can add to longevity and interventional studies. While assigning single cause of death may facilitate data analysis, defining and publishing data on contributing causes (comorbidities) provides more information on associated underlying chronic conditions and health span in mouse models. This article reviews factors that affect determination of cause of death and the methods for evaluating causes of death and comorbidities. The proposed systematic pathology analysis includes assigning cause of death and comorbidities to define total disease burden. The combination of pathology with in vivo data will fully characterize the effect of tested interventions on multiple chronic diseases and health span of aging mice with improved translation to human aging and age-associated lesions.
In medicine, patients who die must have a cause of death determined and an official death certificate completed. Cause of death often is assigned on the basis on medical history and previous health records because autopsy is performed in a minority of deaths (currently <8%). 28 This may affect accuracy of the death certificate, as studies comparing clinical and autopsy-based cause of death disagree in approximately 30% of cases. 40,68,74 Death certificates emphasize preexisting disease or causation, convey information about chronic conditions (contributory or underlying causes of death) as well as immediate cause of death, and form the basis of epidemiologic data on disease prevalence. 25,39,51,58,74
In veterinary medicine, as in human medicine, 28 the gold standard to determine cause of death is complete postmortem examination, which is often performed at referral institutions in cases of academic interest. 15,38 As with human patients, the rate of necropsy of veterinary species has declined. 15,38 Many cause-of-death databases for veterinary species rely on clinical records rather than necropsy findings. 20,31,62,63 Previous studies show a disparity between clinical and pathologic diagnoses in 15% to 30% of cases. 15,38 The lack of comprehensive pathologic examination in veterinary species may affect data on disease prevalence 86,89 and overemphasize immediate cause of death. 84
In preclinical research studies, assigning cause of death provides useful translational information when comparing outcomes with those of the human population. Defining cause of death is especially important when evaluating potentially toxic compounds, in aging studies using methods to prolong life span, and when phenotyping novel strains. Rodents are valuable in experimental studies because of the availability of complex genetic models, reagents, and well-defined strains and because large cohorts can be maintained in a controlled environment in a relatively cost effective manner. 65,77 The experimental research setting, with its emphasis on recording data and with subjects readily available for necropsy, provides an optimal opportunity to understand cause of death. Cause of death is a topic of interest in the toxicologic pathology community, and guidelines for determining cause of death in toxicity studies have been proposed, especially in carcinogenesis studies. 46 In these recommendations, cause of death is defined not only as the proximate cause of death but as “the overall process that leads to the proximate cause.” 46
End Points Affecting Cause-of-Death Determination
End points other than death are used in rodent studies for humane, legal, experimental, and practical reasons.
65
From a practical standpoint, tissues may be nondiagnostic within hours following death due to autolysis.
65
In previous studies examining end-of-life pathology and cause of death, approximately 10% to 15% of mice were autolyzed to the extent that histologic evaluation could not be performed, even when mice were checked daily to twice daily and necropsied immediately upon being found dead.
32,33,54
Defining humane and experimental end points and euthanizing prior to death is emphasized in the National Institutes of Health’s
End point criteria for moribundity vary on the basis of experimental manipulation but may include body weight loss, cold body temperature, pronounced inactivity or immobility, hunched posture with a matted or unkempt coat, ulcerated and bleeding tumor, and increased respiratory effort. 10,13,21,54,64,65 Studies in which animals develop neoplasia or ascites may result in weight gain rather than weight loss, in which case body condition score may be used as an end point in addition to weight loss. 88 However, euthanizing rodents at a moribund state may be difficult in practice. Aging mice may not show obvious clinical signs of disease, and in 1 study, 81 of 105 aging mice died without severe premonitory signs of illness, a result suggestive of limited clinical monitoring. 65
Role of the Aged Rodent Phenotype in Cause-of-Death Determination
When developing end-of-life criteria and when evaluating tissues histologically to determine cause of death, researchers need to consider aspects of normal aging rodent phenotype and aging pathology. 12,64 Clinically, old rodents in good health may have sparse or discolored hair coats, pallor, mildly increased respiratory rate, and reduced body condition and may move more slowly as compared with younger rodents as a result of mild osteoarthritis. 12,64 Those with underlying disease are more likely to have scruffy pelage. 64 Ad libitum–fed rodents may be overweight, 61 although muscle wasting and weight loss also are possible with age, especially with underlying disease.
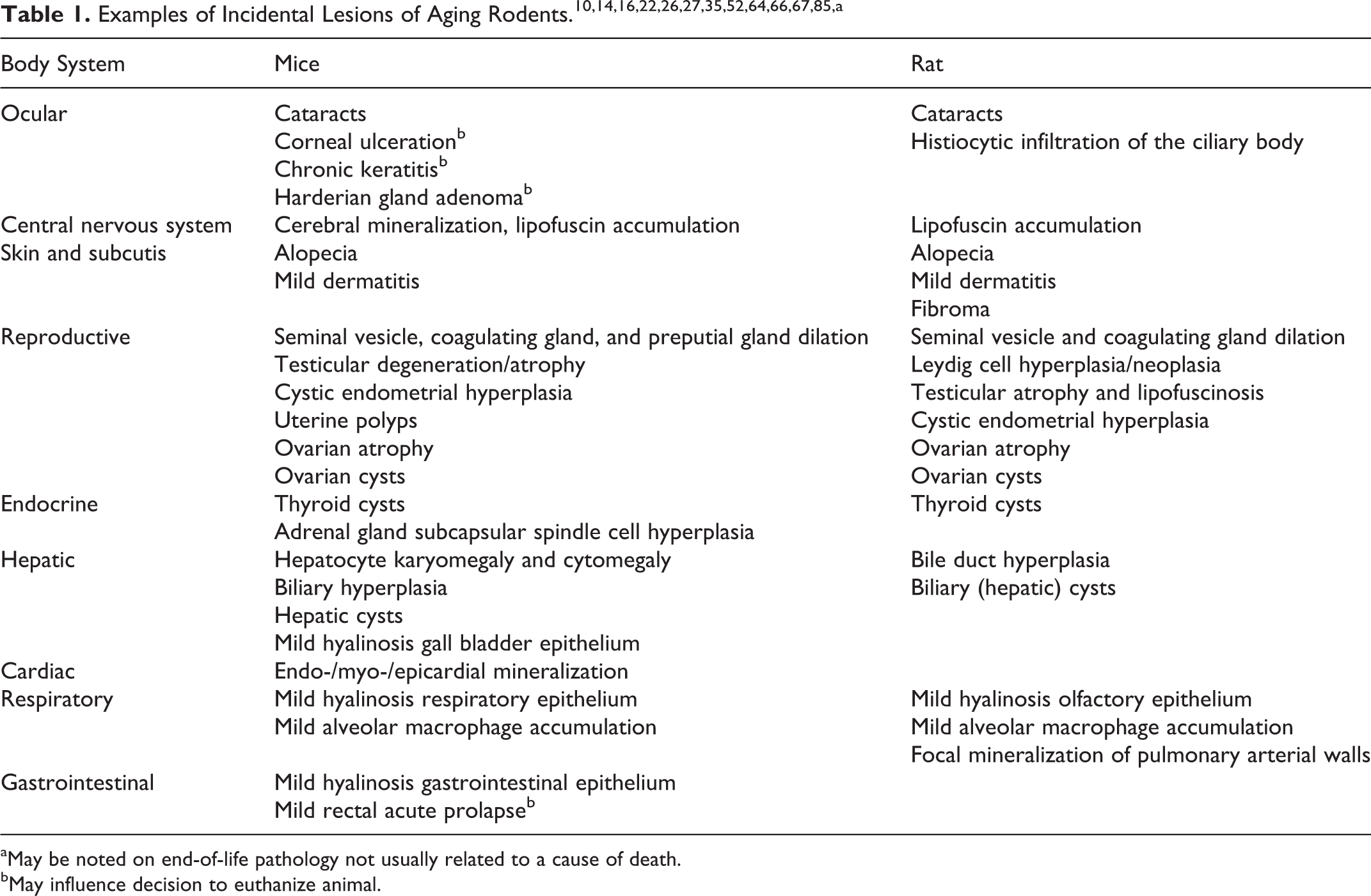
For the purpose of cause-of-death assignment, gross and histologic abnormalities can be divided into 3 categories: incidental lesions, lesions that may result in euthanasia but are not likely to cause or contribute significantly to death, and age-related or induced lesions that most likely contribute to morbidity and mortality. Examples of these 3 categories of lesions are provided for the mouse (Figs. 1–9) and rat (Figs. 10–15).
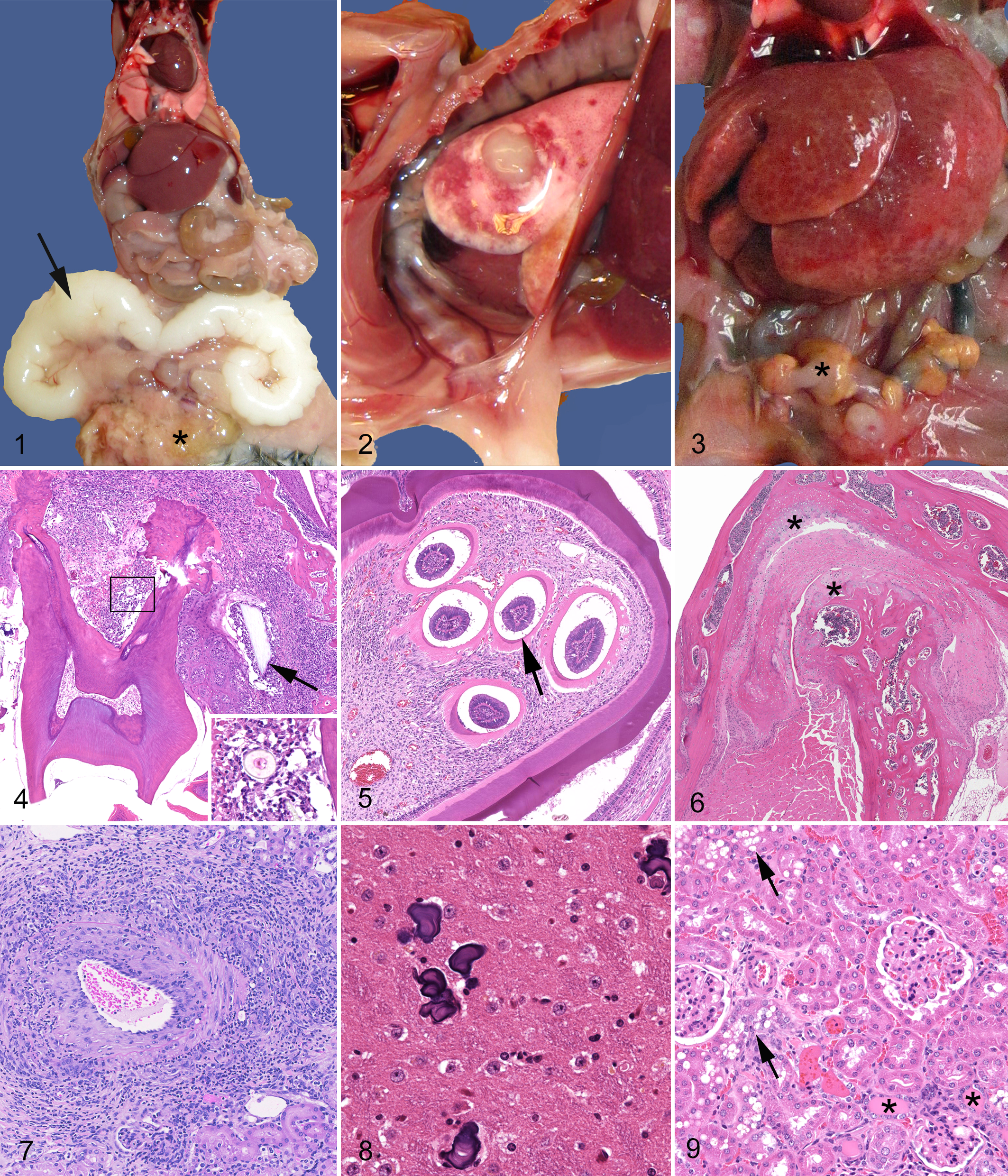
Incidental Lesions
Examples of benign degenerative and inflammatory lesions in aging rodents are presented in Table 1. 14,16,27,52,64,78,85 Incidental lesions also include benign proliferative and neoplastic lesions that increase in frequency with age, such as small masses that do not affect organ function and are not malignant (Table 1). 22,27,49,53
Lesions Contributing to Decision to Euthanize but Unlikely to Cause Death
Mice may develop gross lesions that contribute to a decision to euthanize the animal, such as prolapsed rectum, ocular abnormalities (periorbital abscess and corneal ulceration), and skin abnormalities (cutaneous masses and chronic dermatitis with or without ulcers). 10,53,64,81 In some rat studies, benign and malignant cutaneous and subcutaneous neoplasms are a common cause of death, 19 and rodents with large and/or ulcerated skin tumors generally require euthanasia for humane reasons, although these tumors may be localized and benign. 10 Neoplasms of the Harderian gland are also usually benign 3,10 but may lead to a decision to euthanize given the visible deformity of the orbit on physical examination. 64
Lesions Contributing to Cause of Death
Age-related lesions (as well as induced lesions) likely to contribute to morbidity and mortality are judged by the pathologist on the basis of experimental and clinical history, laboratory results, and gross and histologic examination. Examples include severe renal 47 or pulmonary 30 disease, disseminated neoplasia or neoplasms compromising vital organ function, 23,75 and sepsis. Included lesions are generally severe and therefore considered likely to impair the rodent’s ability to maintain homeostasis.
Lesions in tissues that contribute to death by interfering with the functioning of adjacent or distant tissues are presented in Table 2. In addition, diseases must be considered that may contribute meaningfully to moribundity or death in old mice and rats but are not traditionally considered “cause of death,” such as dental disorders 17,18,64 that decrease the ability of the animal to eat and neurologic 6,11,43,48,55,77 and musculoskeletal diseases 5,8,52,57,64 that decrease mobility.
Specific Leading Causes of Death of Laboratory Rodents.
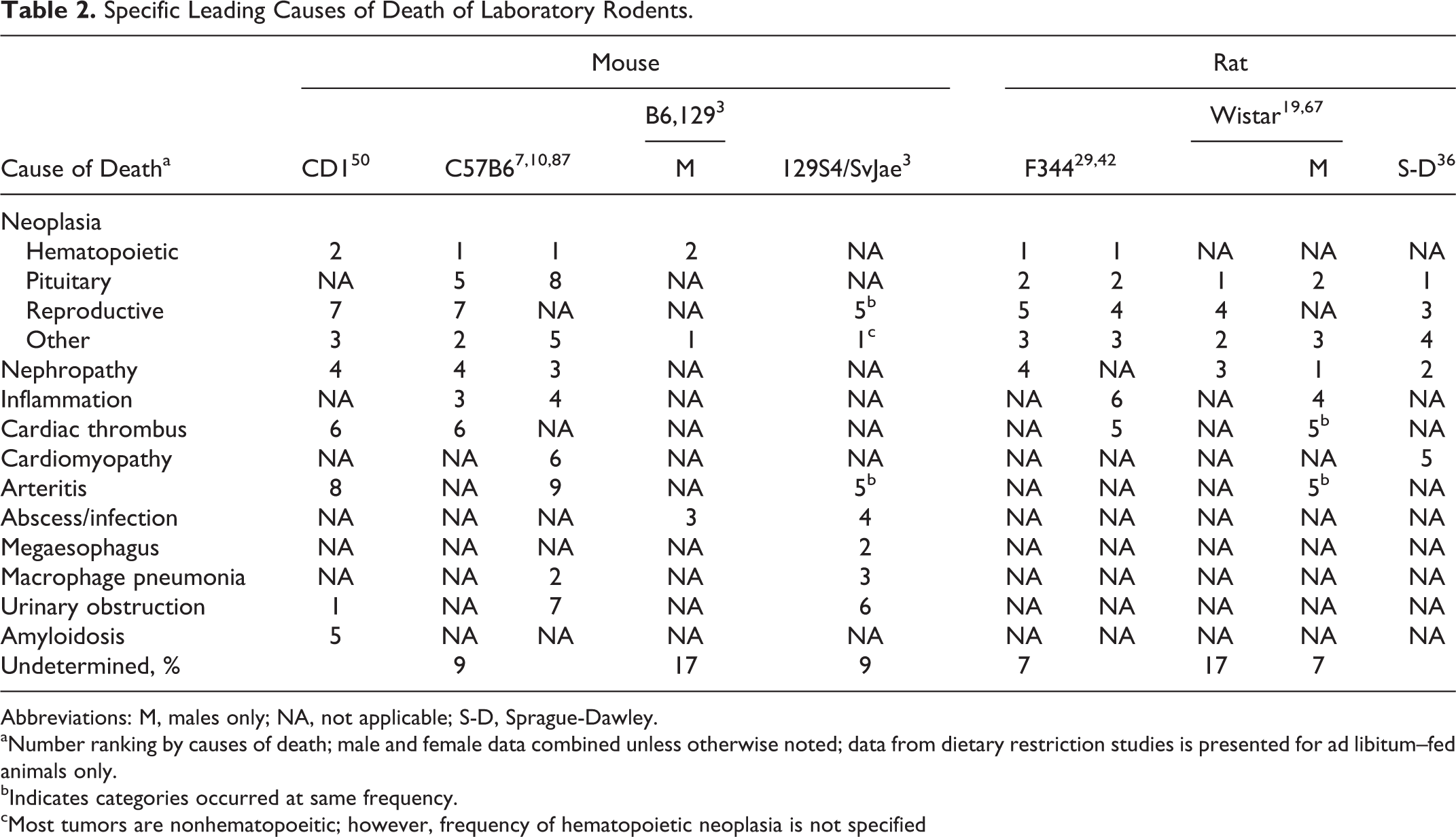
Abbreviations: M, males only; NA, not applicable; S-D, Sprague-Dawley.
aNumber ranking by causes of death; male and female data combined unless otherwise noted; data from dietary restriction studies is presented for ad libitum–fed animals only.
bIndicates categories occurred at same frequency.
cMost tumors are nonhematopoeitic; however, frequency of hematopoietic neoplasia is not specified
Can Cause of Death Be Determined in Rodent Studies?
In many rodent studies that examine the effect of treatment or experimental/genetic manipulation on life extension or health span, cause of death is not reported. 21,60,70,71,79,90 This may be related to cross-sectional study design in some cases, for which cause-of-death assignment is not appropriate and fewer fatal lesions are expected. 11 Additionally, this may be related to the fact that animals often are found dead in life span studies and pathologic analysis is not pursued. Even in cases for which diagnostic samples and funds are available, cause-of-death assignment may be problematic. As discussed previously, many conditions that lead to euthanasia may limit the quality of life but would not rapidly result in death. 10 Therefore, the decision of whether a lesion is responsible for death requires clinical (antemortem) as well as pathologic (postmortem) information. 19 Such detailed clinical information may not be collected during the study or may not be available to the pathologist during cause-of-death determination.
Assigning cause of death in the rodent research literature is a contentious topic. 19 In many studies, a major cause of death is listed, with additional information provided on incidence of neoplasms and other systemic diseases identified grossly or histologically in the population of interest. 7,50 Other authors classify end-of-life pathology as “fatal, probably fatal, incidental, and probably incidental.” 19 A report from the National Center for Toxicological Research also proposed a scheme for cause-of-death assignment. 42 If a severe and likely fatal lesion is identified, then this is deemed the “probable” cause of death. 42 If no fatal lesion is identified, then additional “contributory” causes of death may be listed for lesions that are significant but not likely to be the singular cause of death. “Equivocal,” or possible, causes of death may also be identified. Finally, if no cause of death is apparent, then the cause of death is “unknown.” 42 Most causes of death were assigned a status of “probable” or “contributory,” and no cause of death was identified in 1% to 12% of cases. 42 In a survey of rodent studies examining cause of death, the rate of unknown cause of death ranged from 7% to 17% (Table 2).
Considerations in Animal Strain, Sex, and Number in Cause-of-Death Assignment
There are differences contributing to cause of death among the different strains in mice 10,27,50,82 and rats, 45,61,67 and even within a given strain, rodents are notorious for variation in disease prevalence (Table 2). For this reason, each study examining cause of death must contain its own controls. Changes in specific pathogen–free conditions over time and facility and strain/substrain variations may be associated with different mortality rates and causes of death. For example, when a variant strain of Sprague-Dawley rat was introduced in 1 report, a decrease in life span and incidence of certain malignant neoplasms was noted. 61 In addition to mice that will be found dead and not useful for pathology evaluation, numbers of control and experimental mice used must take into account those that will be humanely euthanized for conditions not meeting end-of-life criteria, such as severe dermatitis. In a previous study involving 592 end-of-life mice, 139 were used for histopathology; 269 were found dead in the cage and could not be used; and 184 were not end of life but were euthanized for humane reasons per the institutional and protocol requirements. 87
Sufficient males and females should be used so that the sexes can be identified separately, as disease incidence and cause of death vary by sex. 10,27 For example, lymphomas are often found in 30% to 50% higher incidences in female mice, and lung tumors are more common in male mice. 27 Cause of death also varies depending on the age at which animals die. 59,76 For example, at 20 weeks of age, no female CD-1 mice died of neoplastic disease, whereas 40.6% of CD-1 females up to 50 weeks of age died of neoplasia. 76 Groups should therefore contain similar numbers of young and old mice, and all mice should be necropsied. If necropsy is performed only on a defined set of the mice, then the true data on diseases and causes of death will not be determined. When mice are discarded or only those moribund are sacrificed, data are skewed as well.
Integration of Pathology and Cause-of-Death Assignment in Rodent Studies
In many rodent studies, methods by which the authors assign cause of death are either not reported 7,50 or not consistent among studies. Methods that assign primary cause of death, causes contributing to death, and equivocal or incidental cause of death most closely resemble assignment of cause of death in humans. Studies investigating consistency of diagnosis among pathologists in rodent studies generally report good agreement. 33,42 In 1 study, 2 independent pathologists agreed on major cause of death in >90% of cases. 33 If the pathologists did not agree or if lesions identified were deemed insufficient to cause death, then the cause of death was listed as unknown. 33
Determining cause of death requires a good gross necropsy examination so that tissues are systematically collected and lesions are trimmed. 44 Histopathologic evaluation is critical. 4,72,80 In many research institutions, gross examinations are performed by staff with varying degrees of training in rodent pathology, and histology slides prepared from these samples are subsequently read by an experienced pathologist. Individuals performing the gross examination should be trained by the pathologist, and salient lesions should be photographed for future reference. 9,44 Gross lesions can be helpful for cause-of-death assignment, especially for neoplasms such as pituitary adenomas that can be measured and, if above a certain size, are presumed to cause life-threatening compression of adjacent nervous tissue. 19
Not only should gross lesions be harvested for histology, but a standardized base set of tissues should be evaluated. For example, hematopoietic neoplasia may be detected histologically in tissues that appear grossly normal. 13 Histology can also distinguish among diseases that have a similar gross appearance, or they may identify the presence of systemic contributory causes of death, such as arteritis, inflammation, and amyloidosis, which may not be apparent grossly. 64 Furthermore, examining a standard set of tissues allows for improved comparison of lesion incidence and severity among studies. In existing studies, various numbers of tissues are evaluated. Some manuscripts provide a list of tissues examined, 13,50 whereas others list the number of tissues evaluated (eg, “45 tissues and all gross lesions”). 7 A suggested minimal tissue set that includes approximately 25 tissues for histologic evaluation is provided (Table 3).
Suggested Minimum Histopathologic Data Set (Mouse) by Paraffin Block.a

aTotal of 25 tissues: soft tissues (muscle) of skull, bone and bone marrow, skin, eye and associated adnexa, teeth, brain, ear and associated adnexa, salivary gland, lung, heart, thymus, lymph node, liver, gall bladder (mouse), kidney, pancreas, adrenal, spleen, stomach, small intestine, large intestine, adipose tissue, reproductive tract, mammary gland (female) or accessory sex glands (male), bladder. Additional tissues may be examined; tissues may be excluded.
In an attempt to standardize cause-of-death assignment and capture comorbidities, lesions could be graded and the extent of involvement (eg, neoplasia) recorded. The importance of consistent grading has been reviewed, 24 and many grading schemes 33,47,87 have been reported for rodent pathology. In 1 scheme, 32,33 neoplasms were divided into 4 grades: grade 1, 1 site only; grade 2, primary site plus metastasis within that organ or to 1 other organ; grade 3, present in 2 or 3 organs; and grade 4, present in ≥4 organs or in 2 or 3 organs with associated pathology, such as pleural effusion or ascites. Neoplastic lesions that were either grade 3 or 4 were considered cause of death. 33 In contrast, the Peto model 1 divides neoplasms into those observable in life (mortality independent), incidental (not contributing to death), rapidly fatal, and not rapidly fatal. The pathologist is responsible for distinguishing between rapidly fatal and not rapidly fatal neoplasms, and this distinction may be subjective. 1
A Proposed Method of Cause-of-Death Assignment in Rodent Studies
A proposed method for cause-of-death assignment in rodent studies (especially studies of aging) that builds on past work
42,87
assigns a major cause of death followed by contributory causes. Contributory causes (also termed
In an effort to maximize correlation between clinical data and pathologic examination, clinical status should be monitored, with results available to the pathologist. Rodents euthanized for nonfatal conditions—including dermatitis, mild rectal prolapse, ocular abnormalities, or subcutaneous neoplasms—that remained active and in adequate body condition score would not be included in end-of-life analysis. 87 Rodents euthanized with these conditions and moderate to severe signs of systemic illness would be included in end-of-life analysis.
The suggested minimal tissue set would be submitted for histopathology, along with all major gross lesions. Techniques of rodent necropsy, organ sampling, and tissue trimming have been described elsewhere. 9,41,56,69,73 Histopathologic scoring would be performed as previously described, with modifications to reflect the impact of the neoplasia on the functional capacity of the tissue of origin, regional structures, and metastasis (Table 4) with a 1–4 severity scale. 33 Degenerative and inflammatory lesions would also be scored on a severity scale from grade 0 (normal) to 4 (severe). 33 In this proposed scheme, the major pathologic findings suspected to be the probable cause of death would be assigned. Contributing causes would include all identified neoplasms (irrespective of grade) and systemic diseases receiving scores of 3 or 4. 87 Examples of cause-of-death determination based on clinical cases presenting to the University of Washington Veterinary Diagnostic Laboratory are presented in Supplemental File 1.
Examples of Histopathologic Scoring.a
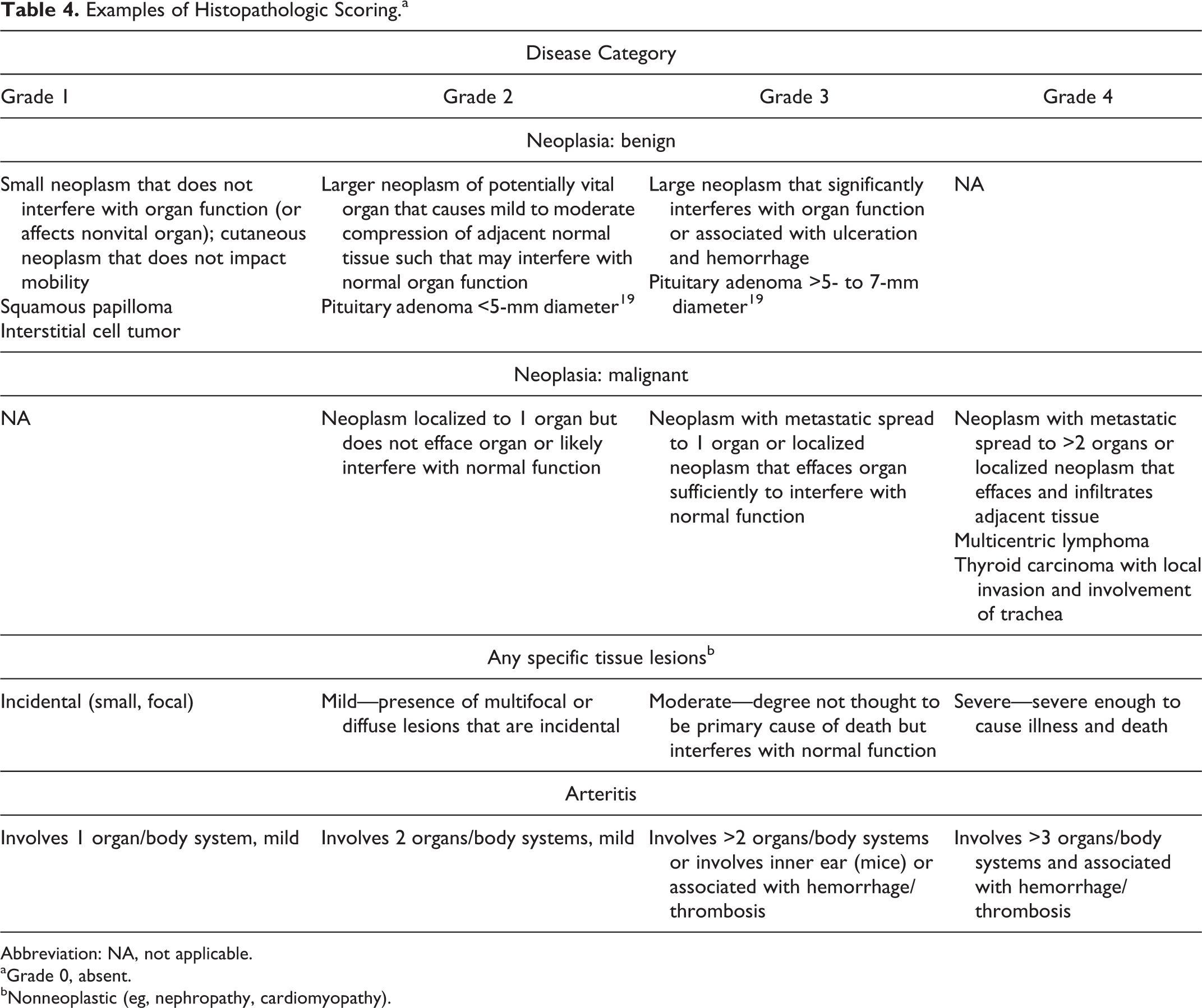
Abbreviation: NA, not applicable.
aGrade 0, absent.
bNonneoplastic (eg, nephropathy, cardiomyopathy).
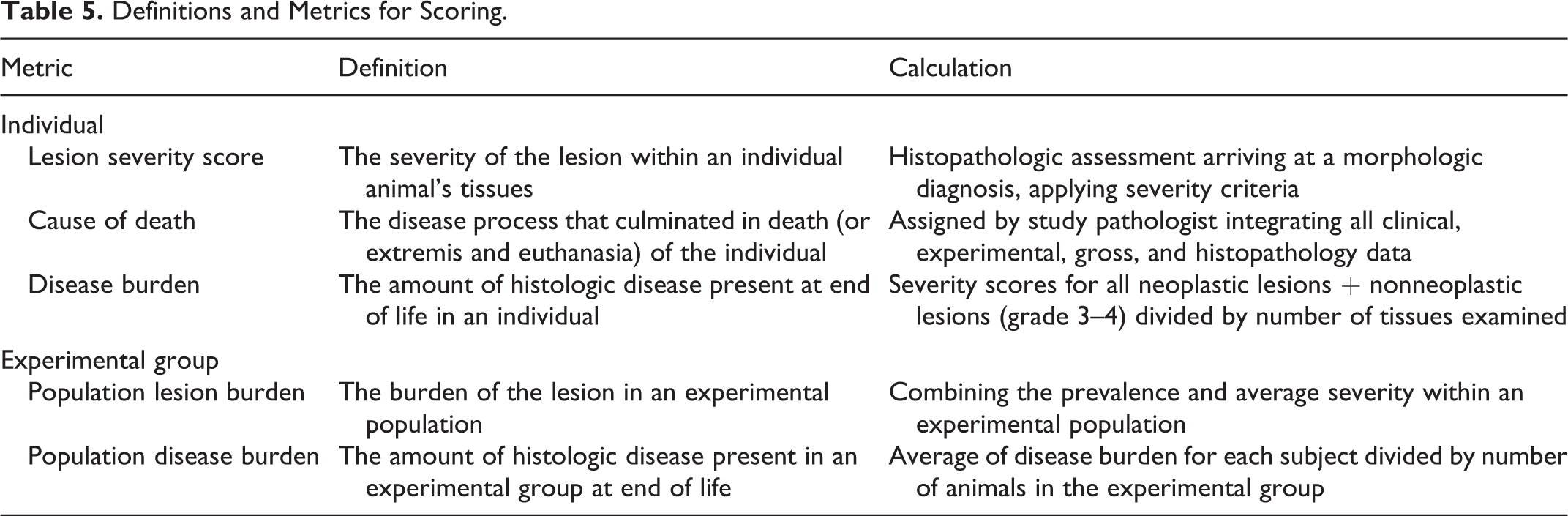
Table 5 provides definitions and metrics for the individual animal and experimental groups. The goal is to assign a cause of death and contributing causes that can be compared across experimental groups. Combined disease burden, including primary cause-of-death assignment and contributory causes, can be used to assess animal health status at end of life. 32,44 The proposed systematic pathology analysis includes assigning cause of death and comorbidities to define total disease burden. These pathology data may be compared with in vivo data and all study data correlated to determine the effect of tested interventions on multiple chronic diseases and health span of aging mice, with the goal of improved translation to human aging and age-associated chronic diseases. 34,37,83
Definitions and Metrics for Scoring.
Conclusion
Cause-of-death assignment in preclinical rodent research suffers from a lack of a standardized approach and an understanding of the value that it can add to longevity and interventional studies. While assigning single cause of death may facilitate data analysis, defining and presenting data on comorbidities provides more information on associated underlying chronic conditions and health span in mouse models. Studies designed to assign comorbidities (contributory causes) in addition to cause of death may better translate to human aging and advance the field of “geroscience.” 37
Footnotes
Acknowledgement
Figures 1, 5, and 6 and Supplemental Figure 17 were used under the terms of the Creative Commons Attribution-Noncommercial 3.0 Unported License (
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the National Institutes of Health R25OD010450, P30 AG013280 (P.M.T.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.