Abstract
Strains of Newcastle disease virus (NDV) have different abilities to elicit neurologic signs. To determine the capacity of different NDV strains to replicate and cause lesions in the brain, independently of their peripheral replication, 1-day-old chickens were inoculated in the subdural space with 7 NDV strains of different virulence (4 velogenic, 2 mesogenic, 1 lentogenic). Velogenic strains induced severe necrotizing and heterophilic ventriculitis and meningitis, as well as edema of the neuroparenchyma, and replicated extensively in the nervous tissue by day 2 postinfection, as demonstrated by immunohistochemistry, when all infected birds died. Clinical signs, microscopic lesions, and viral replication were delayed (days 3 and 4 postinfection) with mesogenic strains. Velogenic and mesogenic NDV strains replicated mainly in neurons, and immunolabeling was first detected in surface-oriented areas (periventricular and submeningeal), possibly as a reflection of the inoculation route. The lentogenic NDV strain did not cause death of infected birds; replication was confined to the epithelium of the ependyma and choroid plexuses; and lesions consisted of lymphoid aggregates limited to the choroid plexuses. Results show that extensive NDV replication in the brain is typical of velogenic and mesogenic, but not lentogenic, NDV strains. In addition, this study suggests that differences in the rate of NDV replication in nervous tissue, not differences in neurotropism, differentiate velogenic from mesogenic NDV strains. This study indicates that intracerebral inoculation might be used as an effective method to study the mechanisms of NDV neuropathogenesis.
Newcastle disease, first reported in 1926 in Java, Indonesia, is often regarded as the most important infectious disease of poultry due to its severe economic impact on the industry worldwide. 1,20 The disease is caused by virulent strains of Newcastle disease virus (NDV), also known as avian paramyxovirus serotype 1, a member of the genus Avulavirus, subfamily Paramyxovirinae, family Paramyxoviridae, and order Mononegavirales. 17 In the United States, virulent NDV (vNDV) strains are synonymous with exotic NDV, since these strains are not present in the poultry population. The NDV genome consists of a single-stranded, negative-sense RNA molecule that contains 6 genes encoding for 6 structural proteins, namely from 3′ to 5′: nucleoprotein, phosphoprotein, matrix, fusion, hemagglutinin-neuraminidase, and large polymerase. 18,20
NDV strains have worldwide distribution and may infect a range of domestic and wild avian species. 1,6,20 Based on the severity of clinical signs induced in infected chickens, NDV isolates are classified into 4 categories (from the least to the most virulent):
Velogenic strains can be divided into velogenic viscerotropic NDV (VVNDV) and velogenic neurotropic NDV (VNNDV). In susceptible birds such as poultry, VVNDV strains cause severe hemorrhagic lesions in multiple organs and up to 100% mortality, while VNNDV strains commonly induce neurologic signs and have lower mortality (50%). 1,6,20
Pathogenicity tests, such as the intracerebral pathogenicity index (ICPI) and the intravenous pathogenicity index, are used to classify the virulence of NDV isolates according to standard parameters. 6,22 The ICPI is the internationally recognized system to assess NDV virulence, and it consists of a weighted score of clinical signs (for an 8-day period) in groups of 10 one-day-old chicks after intracerebral inoculation with NDV strains. Potential scores for ICPI range from 0 and 2, with most virulent isolates closer to 2. 22 NDV isolates are classified as nonvirulent when the ICPI score is <0.7 and virulent when ≥0.7. 22 Velogenic (VVNDV and VNNDV) and mesogenic NDV strains are classified as vNDV, whereas lentogenic and asymptomatic enteric strains are considered of low virulence (loNDV). 6,20,22
Neurologic signs are reported only upon infection with vNDV strains and include hypermetria, tremors, opisthotonus, paralysis/paresis, and wing droop. 1,3,6 Animal experiments conducted in chickens inoculated via oropharyngeal or conjunctival route showed that vNDV strains can successfully replicate in the central nervous system, with immunohistochemical (IHC) staining limited to scattered neurons throughout the brain. 3,6,11,15,16,29 In the same studies, predominant microscopic lesions associated with virus replication in the brain consisted mostly of multifocal neuronal necrosis, nonsuppurative encephalitis characterized by lymphocytic perivascular cuffing, plump vascular endothelium, and multifocal astrogliosis. 3,6,11,15,16,29 In chickens, vNDV strains that induce neurologic signs (VNNDV and mesogenic) cause lower mortality rates and less severe to no visceral lesions and have limited virus spread when compared with the most virulent vNDV strains (VVNDV). 3,6 This is supported by evidence that VVNDV strains have higher ICPI than mesogenic and, albeit with some exceptions, VNNDV strains. 6,14 Furthermore, when chickens are inoculated with VNNDV or mesogenic strains, neurologic signs are a relatively late event in the progression of the disease, becoming obvious between days 5 and 14 postinfection (pi). 3,6,29 In contrast, VVNDV-infected chickens usually die or are humanely euthanized by day 5 pi, showing systemic illness without neurologic signs. 3,6,7,9,27 –29 When inoculated through the mucosal surfaces (eg, eye drop instillation), the ability of NDV strains to reach the nervous tissue depends heavily on multiple factors, such as peripheral replication, ability to cross the blood-brain barrier and reach the cerebrospinal fluid, and efficiency of replication and spread in neurons and glial cells.
To better assess the ability of different NDV strains to replicate and to cause lesions in the central nervous system of infected chickens, we utilized an approach that would allow the virus to reach the central nervous system immediately upon infection, eliminating those constraints related to peripheral replication. For this purpose, we adopted the internationally accepted ICPI technique, which consists of inoculating NDV strains directly into the subdural space of naïve day-old specific-pathogen–free chicks. Using the ICPI inoculation method, we compared the ability to replicate and to cause lesions in the central nervous system of 7 NDV strains spanning all pathotypes (4 velogenic, 2 mesogenic, 1 lentogenic). This is the first study to describe the pathologic characteristics of brains from chicks infected with NDV through the ICPI method.
Material and Methods
Ethics Statement
The experiment was conducted in rigorous accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. In addition, the protocol was approved by the Institutional Animal Care and Use Committee (Animal Welfare Act, No. A4298-01; expiration date, May 31, 2018) of the Southeast Poultry Research Laboratory (SEPRL; Agricultural Research Service, US Department of Agriculture, Athens, GA).
Chickens and Viruses
This study used 120 day-old chicks, obtained from the SEPRL specific pathogen–free White Leghorn flock. Birds were maintained in negative pressure isolators under biosafety level 2 (loNDV and control groups) and 3 (vNDV groups) enhanced conditions at SEPRL with food and water provided ad libitum.
Seven NDV strains were used to infect chicks with the ICPI method. All NDV strains in the study were from the virus repository of the SEPRL. Full names, biological characteristics, and published references for the viruses are reported in Table 1. Overall, 6 vNDV strains were used in this study: 2 wild type velogenic (SA60, TxGB), 2 recombinant velogenic (rLS-SA60v, rLS-VF), 1 wild type mesogenic (Tx-450), 1 recombinant mesogenic (rLS-Tx450v), and 1 loNDV lentogenic strain (LS-wt). Methods for recombinant NDV virus production and rescue are described elsewhere. 4,5,26 Virus stocks were propagated in specific pathogen–free 9- to 10-day-old embryonated chicken eggs. Virus titers were determined by limiting dilution in eggs and expressed as EID50 (embryo infectious dose 50%). 2,19
NDV Strains Used in the Study and Their Published Characteristicsa.

aVelogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
Abbreviations: F, fusion; ICPI, intracerebral pathogenicity index; HN, hemagglutinin-neuraminidase; NDV, Newcastle disease virus; pi, postinfection; VNNDV, velogenic neurotropic NDV; VVNDV, velogenic viscerotropic NDV.
Animal Experiment
Day-old chickens were randomly allocated in 8 experimental (infection) groups, each group with 10, 15, or 20 birds, depending on the planned sampling schedule (based on the expected time for each virus to kill all birds). Five birds were sacrificed each scheduled day as follows:
SA60, rLS-SA60v, and TxGB groups (n = 10): 5 birds sampled at days 1 and 2 pi rLS-VF group (n = 15): 5 birds sampled at days 1, 2, and 3 pi rLS-Tx450v, Tx-450, and LS-wt groups (n = 20): 5 birds at days 1, 2, 4, and 8 pi Control group (sterile allantoic fluid; n = 10): 3 birds at days 1 and 2 pi and 4 birds at day 8 pi
Intracranial inoculation was performed as prescribed for the ICPI method, following a standard protocol. 2,22 To inoculate each group with the same virus dose, before the inoculum for injection was prepared, aliquots of virus stocks were prediluted with allantoic fluid to reach the same concentration as the virus with the lowest-titer stock (Tx-450, which was not prediluted). These normalized stocks were then diluted 1:10 (target dose = 104 EID50/bird) into phosphate-buffered saline, filtered, and inoculated in the right frontal subdural space of each chick. The mock-infected (control) birds were inoculated with 50 μl of filtered allantoic fluid. Animals were monitored daily for clinical signs and disease progression over 8 days. Five birds of each group were euthanized according to the sampling schedule for each experimental group (see above). Birds with significant clinical signs were euthanized regardless of the planned schedule.
Although the ICPI scores for the NDV strains used in this study have been published, new ICPI scores were calculated again according to the results of the present work. Because of scheduled sampling, animals that were euthanized without clinical signs were not included in the final tally, and animals that were euthanized in extremis were considered sick (score = 1) for that day and dead for the next (score = 2). 22
Immediately after euthanasia, the head and spleen from each chick were harvested and fixed in 10% neutral-buffered formalin for 48 hours. Thereafter, heads of chicks were decalcified in Kristensen’s decalcification solution for 20 minutes and all samples transferred to 70% ethanol. To allow assessment of all encephalic tissues, the head of the bird was cut in the median plane and a sagittal section from the right side (including skull cap and brain tissues) obtained. Sections from heads and spleens were routinely processed and embedded in paraffin, and 3-μm sections were cut for hematoxylin and eosin (HE) stain and immunohistochemistry (IHC).
Immunohistochemistry
Immunohistochemistry was performed in all samples of brain and spleen via a primary anti-NDV antibody and a high-performance polymer enzyme detection system, Novolink Polymer DS (RE7150-K; Leica, Newcastle, UK), according to the manufacturer’s protocol. Briefly, unstained paraffin sections were deparaffinized in Hemo-De (Fisher Scientific, Pittsburgh, PA) and subjected to antigen retrieval by microwaving for 10 minutes at minimum power in preheated unmasking solution (Vector Laboratories, Burlingame, CA). Nonspecific protein and endogenous peroxidase were blocked by incubation for 5 minutes with blocking reagents from the polymer kit. The primary antibody, a polyclonal rabbit anti-NDV nucleoprotein (raised against the synthetic peptide TAYETADESETRRIC), 16 was diluted at 1:8000 in phosphate-buffered saline with 0.02% Tween 20, and sections were incubated for 1 hour at 37°C. All steps following the primary antibody incubation were done with reagents from the polymer kit and according to the manufacturer’s directions. The antigen-antibody complexes were visualized with 3,3′-diaminobenzidine. Sections were then counterstained with hematoxylin and coverslipped with Permount (Fisher Scientific).
Grading of Microscopic Lesions and IHC Signal
The system for grading microscopic lesions (HE) and IHC labeling for NDV nucleoprotein is reported in Table 2. For all the slides, grading was carried out simultaneously by 2 veterinary pathologists (V.M.B.D.M., L.S.).
Grading System to Score Microscopic Lesions (Hematoxylin and Eosin) and Extent of Nucleoprotein Immunolabeling (Immunohistochemistry).a
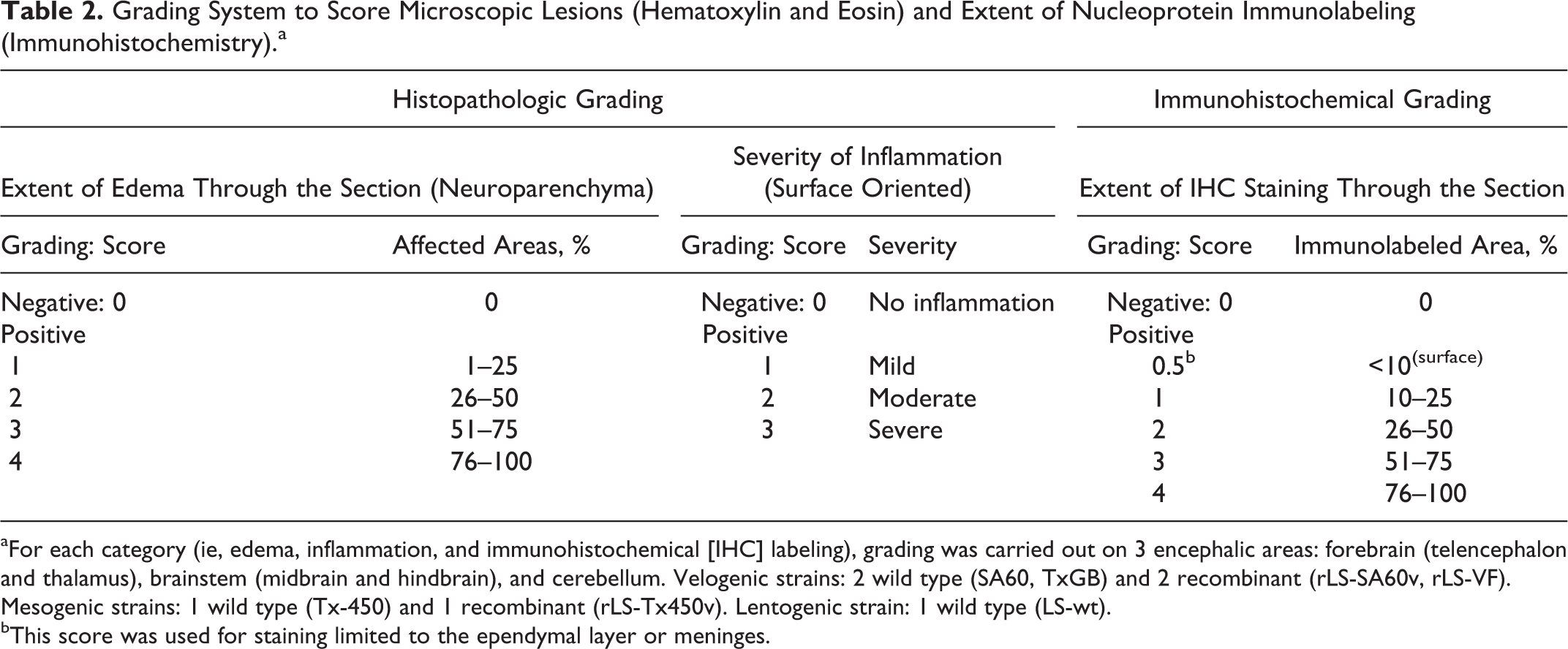
aFor each category (ie, edema, inflammation, and immunohistochemical [IHC] labeling), grading was carried out on 3 encephalic areas: forebrain (telencephalon and thalamus), brainstem (midbrain and hindbrain), and cerebellum. Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
bThis score was used for staining limited to the ependymal layer or meninges.
Two main microscopic lesions were graded: (1) inflammation and necrosis of surface-oriented areas (ventriculitis [including inflammation of choroid plexuses] and meningitis) and (2) edema of the neuroparenchyma (gray [neurons] and white matter). Inflammation and necrosis were graded as negative, mild (25% of ependymal or meningeal surface affected), moderate (26%–75% of ependymal or meningeal surface affected), and severe (>75% of ependymal or meningeal surface affected). Edema was graded per the extent (percentage) of tissue involved in the process (5 possible grades). Immunolabeling in brain sections was scored as an approximate percentage of positive areas over the total area of the brain section (6 possible grades). Grading for histopathology and immunohistochemistry was performed on 3 distinct encephalic areas: forebrain (telencephalon and thalamus), brainstem (midbrain and hindbrain), and cerebellum (Supplemental Fig. S1). Preliminary statistical analysis (nonparametric Kruskal-Wallis 1-way analysis of variance) showed no significant differences among those areas at any time point for each infection group (data not shown); therefore, all presented data represent aggregates from all 3 brain locations. For each infection group, scoring results for inflammation, edema, and IHC signal were averaged over the number of birds at each time point.
Lesions in the spleen were graded according to severity: mild (lymphocyte depletion and prominent macrophages in the splenic ellipsoids), moderate (multifocal to coalescent areas of splenic necrosis), or severe (diffuse splenic necrosis); IHC staining in the spleen was graded simply as either positive or negative.
Data Analysis
To compare the scores of microscopic lesions and viral distribution (IHC) among experimental groups, data were analyzed with the nonparametric Kruskal-Wallis 1-way analysis of variance, followed by pairwise Wilcoxon tests (JMP Pro 11.2). The Benjamini and Hochberg correction was used to account for multiple comparisons, with a cutoff P value of .05. Scores were statistically compared only for inflammation and IHC staining at days 1 and 2 pi, when all strains were represented.
Results
Clinical Disease, Macroscopic Findings, and ICPI Scores
Details of clinical signs, mortality, gross lesions, as well as ICPI scores are reported in Table 3. Birds infected with the VVNDV SA60 strain were severely depressed at day 1 pi, and all were found dead in the cage at the time of sampling at day 2 pi. With other velogenic virus-infected birds (rLS-SA60v, TxGB, and LS-VF), depression was present at day 1 pi and culminated at day 2 pi, when all birds spontaneously died (not euthanized) or were sacrificed in extremis. By day 2 pi, chicks infected with the mesogenic rLS-Tx450v strain began to present clinical signs, which consisted of depression, ataxia, and paralysis. By day 3 pi, all rLS-Tx450v-infected birds had died spontaneously. Birds infected with the mesogenic Tx-450 strain were all depressed at day 3 pi and spontaneously died by day 4 pi. Enlarged and mottled spleen or diffuse splenic yellow discoloration (necrosis) was the gross lesion observed in birds infected with all vNDV strains, at all sampling times (1, 2, 3, and 4 days pi). No clinical signs or macroscopic lesions were observed in chickens infected with the lentogenic LS-wt strain and control birds. ICPI scores based on the present experiment (Table 3) showed that all velogenic strains had values >1.5, mesogenic strain >0.7 and <1.5, and LS-wt = 0.0, which is the expected scoring for these pathotypes. 6
ICPI Scores, Clinical Signs, Mortality, and Macroscopic Lesions in Day-Old Chicks Inoculated With NDV via the ICPI Method.a
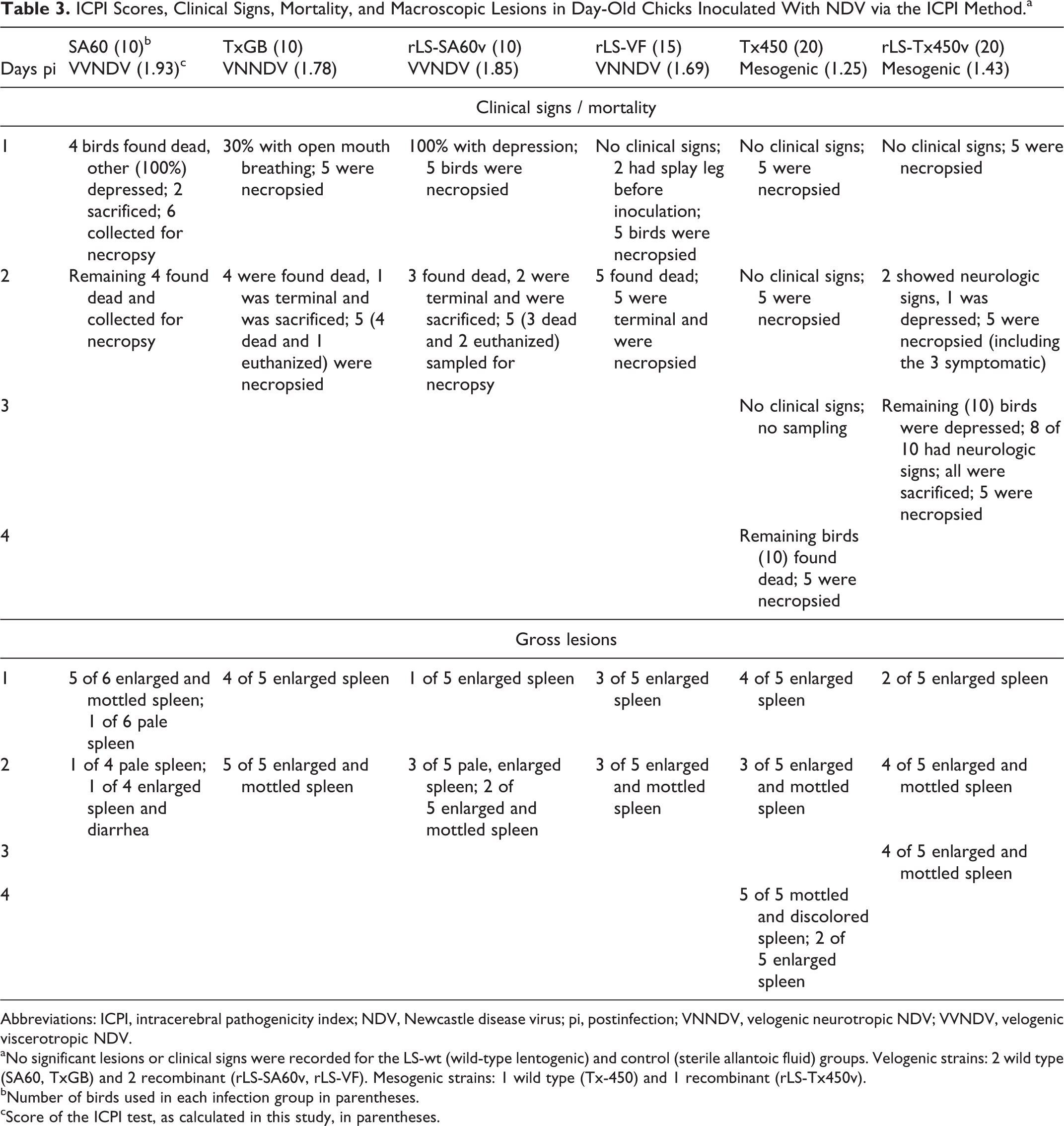
Abbreviations: ICPI, intracerebral pathogenicity index; NDV, Newcastle disease virus; pi, postinfection; VNNDV, velogenic neurotropic NDV; VVNDV, velogenic viscerotropic NDV.
aNo significant lesions or clinical signs were recorded for the LS-wt (wild-type lentogenic) and control (sterile allantoic fluid) groups. Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v).
bNumber of birds used in each infection group in parentheses.
cScore of the ICPI test, as calculated in this study, in parentheses.
Descriptive Histopathology
Results of histopathologic grading for inflammation and edema in vNDV-infected groups at each time point are shown in Figure 1 and Supplemental Figure S2, respectively. All vNDV strains used in this study produced histologic changes in the brain of infected chicks, with the extent and intensity of encephalic lesions being strain and time dependent.
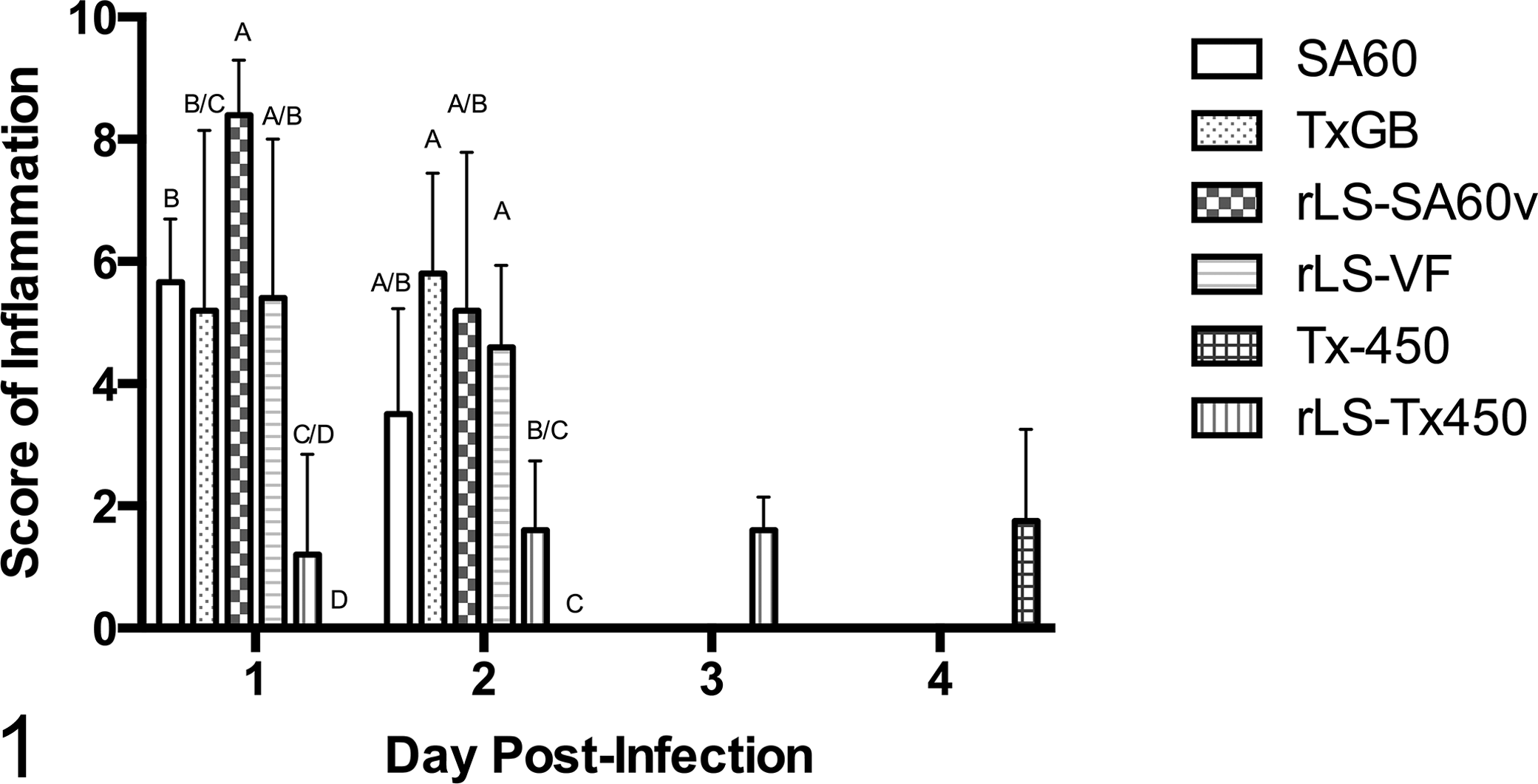
Intensity of inflammation over time in the brain of infected day-old chickens. In general, inflammation was more severe in velogenic strains without significant differences between velogenic viscerotropic and velogenic neurotropic Newcastle disease virus strains. Statistical analysis was conducted only for days 1 and 2 postinfection (see text). Data columns represent mean + 1 SD scores. Letters overlying the columns represent the statistical groupings (groups with statistical differences will not share a letter). Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
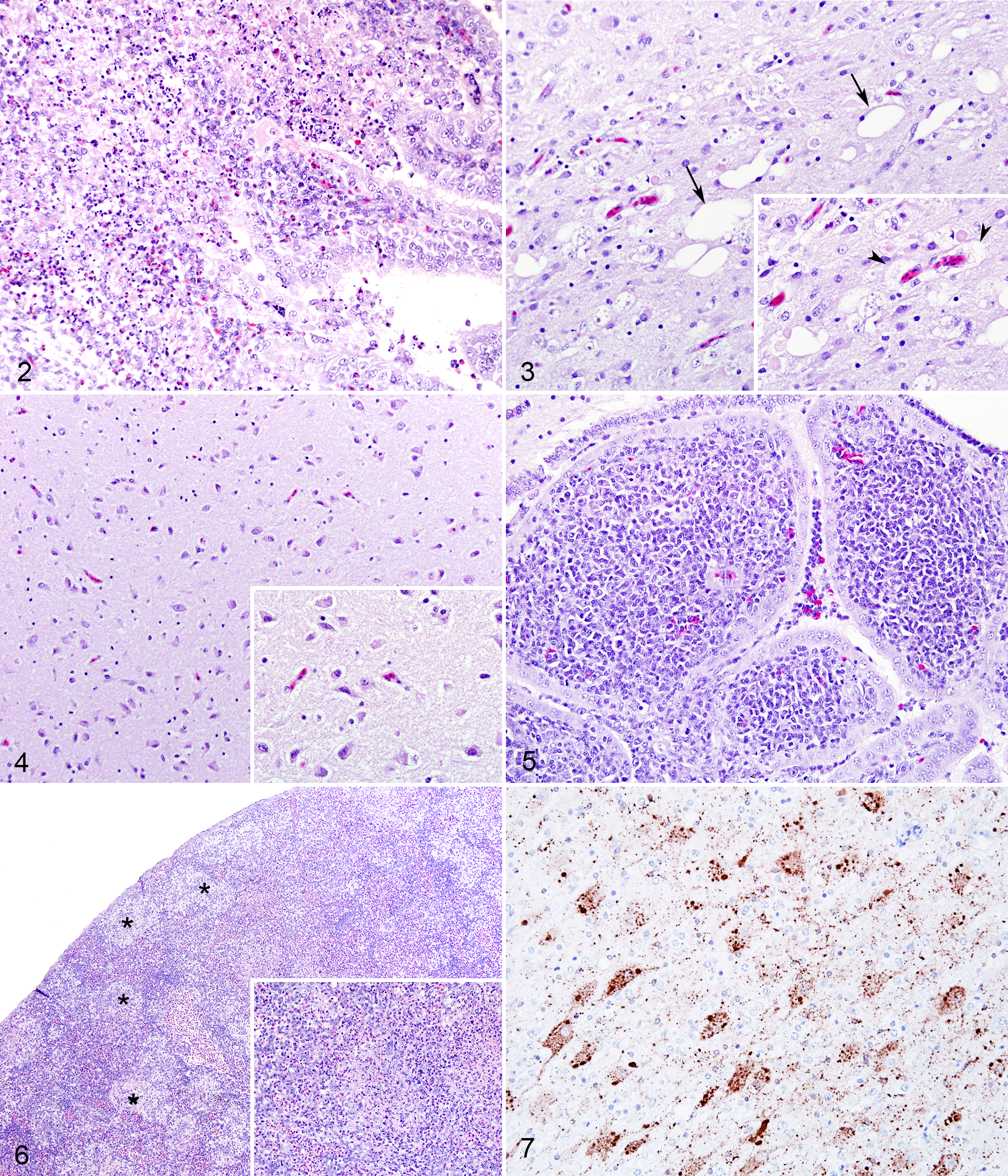
The main microscopic findings in birds infected with vNDV strains consisted of inflammation of surface-oriented areas (ependyma, choroid plexuses, and meninges) and edema in both white and gray matter, which were also the categories used in the grading system. Overall, lesions were similar in nature, and variation was mainly due to intensity and timing. The 4 velogenic strains (SA60, rLS-SA60v, TxGB, and LS-VF) caused moderate to severe inflammation of surface-oriented structures at days 1 and 2 pi. Lesions were characterized by attenuation and loss of ependymal or meningeal cells, associated with subependymal and meningeal accumulation of heterophils, macrophages, and necrotic debris. In the choroid plexuses, inflammation tended to be more severe, with marked heterophilic infiltrate (Fig. 2). Occasional syncytia in the ependymal cells were observed in rLS-SA60v-infected birds. Scores for inflammation were high with all velogenic strains as early as day 1 pi, confirming the high virulence of these strains, and slightly subsided at day 2 pi (when all velogenic-infected birds died; Fig. 1).
Day-old White Leghorn chicken infected with Newcastle disease virus (NDV) strains of various virulence. Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
Among the velogenic strains, edema was caused only by VVNDV SA60 and rLS-SA60v strains. Edema in both gray and white matter was conspicuous at day 2 pi and was variably characterized by ballooning of neurons and glial cells, associated with marked vacuolation of the white matter and neuropil (status spongiosum). Occasionally, ballooned cells and vacuoles in the white matter became confluent to create larger areas filled with low-protein edema fluid and scattered cellular debris. Within these areas of edema, single-cell necrosis of neurons or spheroids was occasionally observed. Scores for edema were highest with SA60 strain (diffuse and extensive to all section), while rLS-SA60v-infected birds presented a multifocal distribution of edematous areas.
In the mesogenic-infected groups, inflammatory lesions were less severe but similar in nature when compared with those observed with velogenic strains, with a predominantly heterophilic and heterophilic/lymphocytic inflammatory infiltrate, respectively, in rLS-Tx450v- and Tx-450-infected birds. Inflammation (ventriculitis and meningitis) in rLS-Tx450v-infected birds was milder than that observed for velogenic strains and was fairly constant from day 1 through day 3 pi (when all birds were terminated). Birds infected with Tx-450 strain showed no to minimal inflammation at days 1 and 2 pi, while moderate degree of inflammation was observed only at day 4 pi, when all birds in this group were terminally ill. Edema lesions first appeared at days 2 and 3 pi for Tx-450- and rLS-Tx450v-infected birds (Fig. 3), respectively, and progressed up to day 4 pi in birds infected with Tx-450 strain (Supplemental Fig. S2). Brains were fixed immediately after euthanasia, and artifactual vacuolation of nervous tissue was minimal (for comparison, Fig. 4 shows a TxGB-infected bird with an edema score = 0).
Birds infected with the lentogenic LS-wt strain showed a lesion pattern that was not observed with velogenic or mesogenic NDV strains and therefore was not graded in the same system. Histopathologic changes were characterized by accumulation of often-perivascular lymphoid aggregates in subependymal areas and in the choroid plexuses (Fig. 5). No loss of ependymal cells, accumulation of cellular debris, or infiltration of heterophils was observed. Lesions were first detected at day 2 pi and progressed in intensity at day 4 pi, to slightly decrease at day 8 pi (data not shown).
Splenic lesions were observed in all birds infected with vNDV strains and were absent in the spleen of those infected with the loNDV strain (LS-wt). Splenic lesions had different severity and spanned from lymphocyte depletion to multifocal to coalescing areas of necrosis and fibrin accumulation that could become diffuse to the whole spleen (Fig. 6). These lesions were present with all vNDV strains and were more severe with those displaying higher ICPI scores (data not shown).
Descriptive Immunohistochemistry
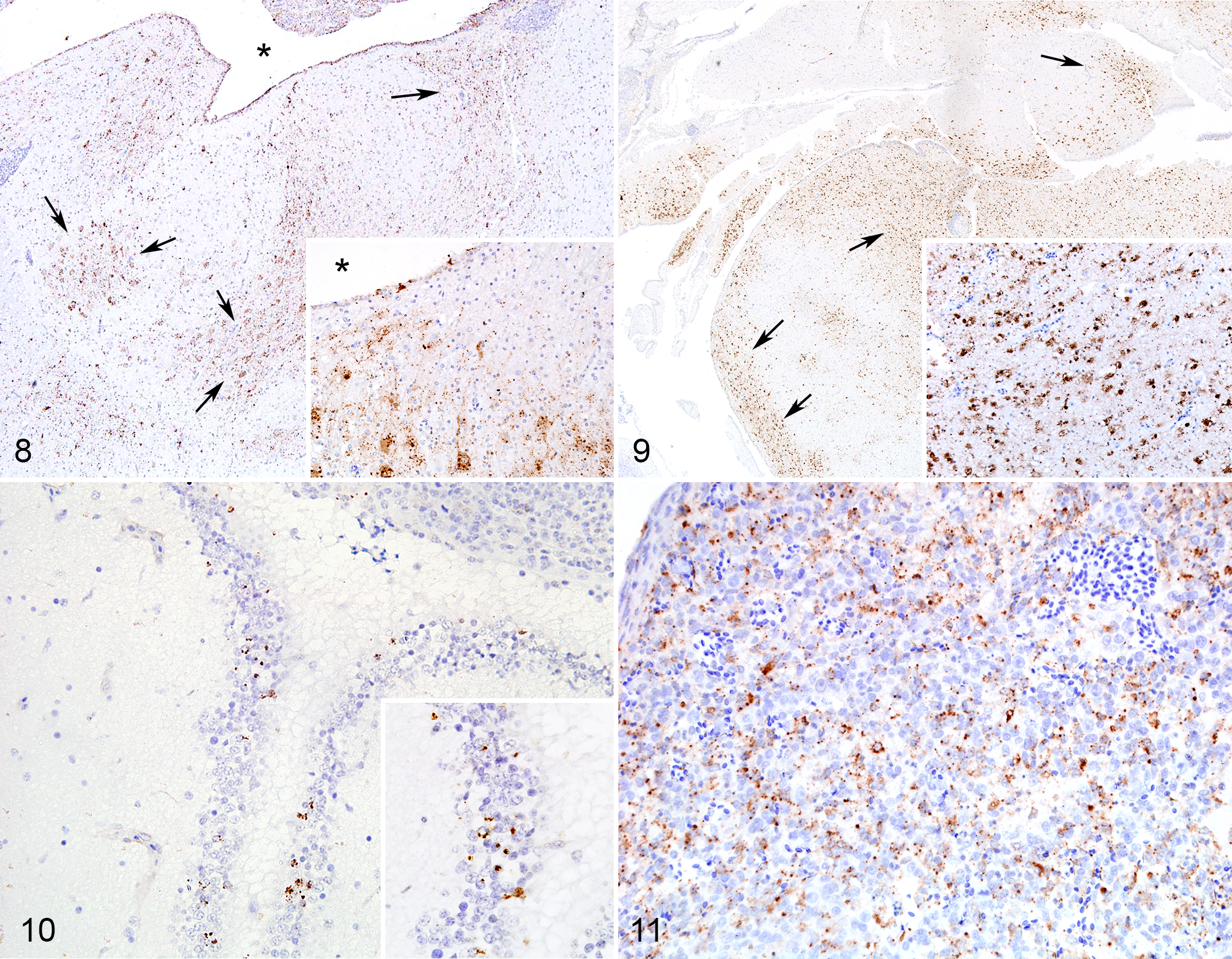
IHC signal was observed in the brain of birds infected with all tested NDV strains (vNDV and loNDV). Scores of IHC labeling—as percentage of area involved—in each experimental group over time are reported in Figure 12.
For vNDV strains, IHC staining was multifocally present in the ependymal layer, choroid plexuses, and meninges and throughout the whole neuroparenchyma, predominantly in neuronal soma and axons (Fig. 7). Immunolabeling was also observed in cells morphologically consistent with astrocytes, especially when staining was diffuse in large areas of the section. At day 1 pi, IHC staining appeared to be present in the ependymal layer, meninges, and adjacent neuroparenchyma, while by day 2 pi, staining progressed to deeper areas of the brain. Similar staining pattern was observed for mesogenic strains, although with a slight delay. Compared with birds infected with velogenic strains, rLS-Tx450v- and Tx-450-infected birds showed low IHC scores at days 1 and 2 pi; however, they became comparable to those in the velogenic-infected groups at days 3 and 4 pi, respectively (Figs. 8, 9). IHC signal in LS-wt-infected birds was confined to few scattered ependymal cells in the ependymal layer and choroid plexuses; no staining was observed in the neuroparenchyma (Fig. 10). Birds infected with vNDV strains showed immmunolabeling in splenic cells with abundant cytoplasm and open nuclear chromatin (macrophages) at days 1 and 2 pi (Fig. 11). Birds infected with the lentogenic LS-wt strain did not show any IHC signal in the spleen. In the brain and spleen, IHC signal was finely to coarsely granular and intracellular. Occasional extracellular signal was observed in areas of necrosis, especially in the spleen.
Day-old White Leghorn chicken infected with Newcastle disease virus (NDV) strains of various virulence. Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
Quantative Analysis of Microscopic Lesions and IHC Scoring
Preliminary statistical analysis for inflammation and immunohistochemistry in each infection group were statistically compared (nonparametric Kruskal-Wallis 1-way analysis of variance) among forebrain, brainstem, and cerebellum. No significant differences were detected among those areas at any time point for each infection group. While this does not exclude the possibility of undetected differences, all further data represent aggregates from all 3 brain locations.
The grading scores for inflammation and edema were compared at days 1 and 2 pi only among vNDV infection groups (Fig. 1 and Supplemental Fig. S2, respectively), since lesions caused by the lentogenic LS-wt strain (loNDV) were very different in nature and not included (see description above). The grading scores of IHC staining were compared at days 1 and 2 pi among all infection groups (vNDV and loNDV; Fig. 12). In any case, data from days 3 and 4 pi were not included into the statistical analysis, because only some of the infection groups were present at those time points.
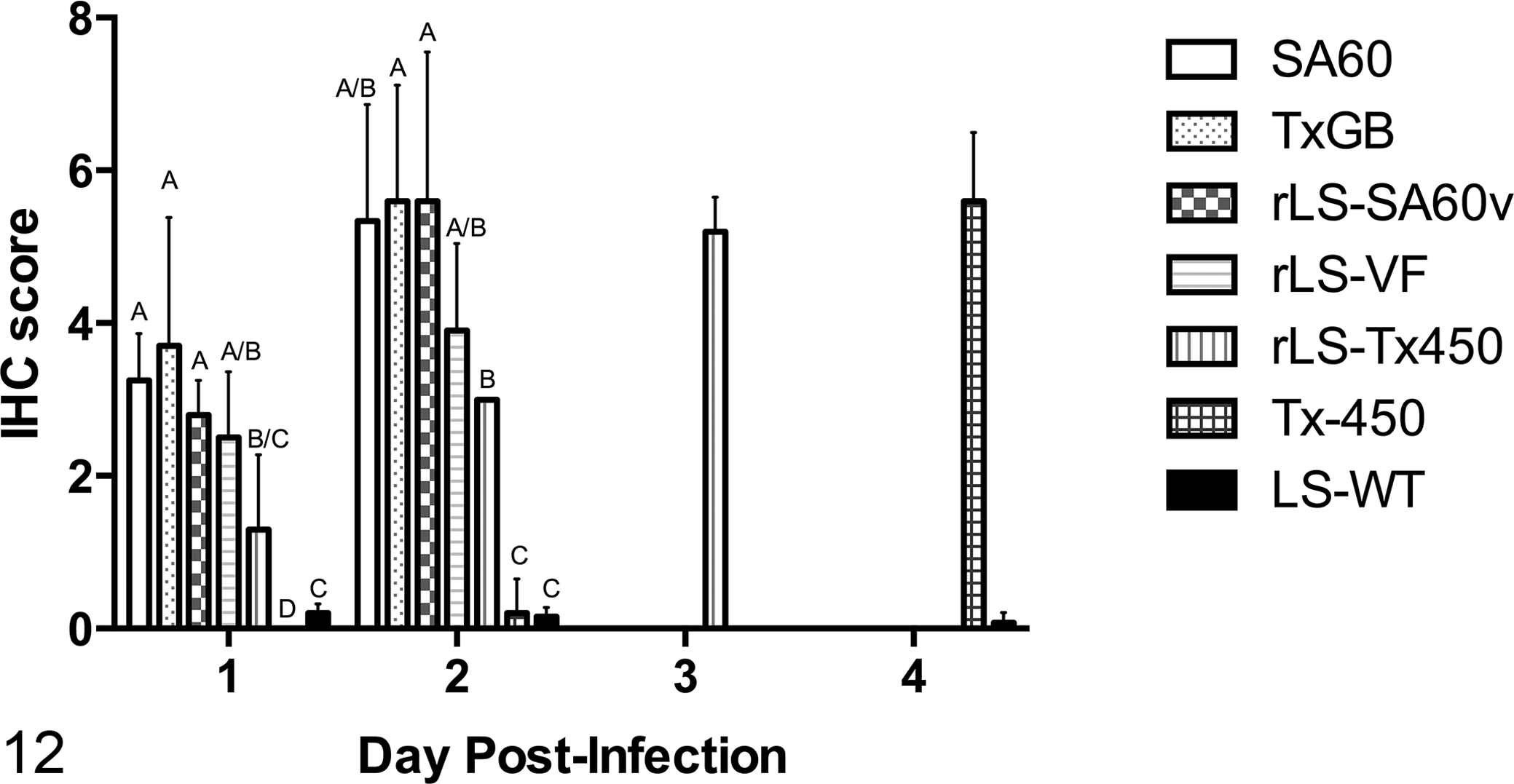
Intensity of immunohistochemical score over time in the brain of infected day-old chickens. In general, velogenic strains had higher viral antigen loads in the brain early in the disease course as compared with mesogenic strains. Viral replication was very limited in the lentogenic strain. Statistical analysis was conducted only for days 1 and 2 postinfection (see text). Data columns represent mean + 1 SD scores. Letters overlying the columns represent the statistical groupings (groups with statistical differences will not share a letter). Velogenic strains: 2 wild type (SA60, TxGB) and 2 recombinant (rLS-SA60v, rLS-VF). Mesogenic strains: 1 wild type (Tx-450) and 1 recombinant (rLS-Tx450v). Lentogenic strain: 1 wild type (LS-wt).
Statistical analysis of inflammatory lesions at days 1 and 2 pi showed that all velogenic NDV strains caused significantly more severe inflammation when compared with the mesogenic Tx-450 strain. At day 1 pi, the mesogenic rLS-Tx450v strain caused inflammatory lesions significantly less severe compared to all velogenic strains except for TxGB. At day 2 pi, lesions caused by rLS-Tx450v were significantly less severe than those of the velogenic TxGB and rLS-VF but not SA60 and rLS-SA60v strains. Severity of inflammation was not significantly higher for rLS-Tx450v versus Tx-450 (both mesogenic strains) at either day 1 or day 2 pi. At days 1 and 2 pi, wild-type and recombinant velogenic viruses caused statistically similar inflammatory lesions (clustering in the same group designation; Fig. 1), with the exception of only 2 comparisons at day 1 pi (SA60 vs rLS-SA60v and TxGB vs rLS-SA60v). Statistical analysis for edema scores showed no significant differences at day 2 pi among SA60-, rLS-SA60v-, and Tx450-infected groups (Supplemental Fig. S2).
Statistical analysis for IHC at days 1 and 2 pi showed that all velogenic strains caused significantly more extensive labeling than the mesogenic Tx-450 and the lentogenic LS-wt strains. Mesogenic rLS-Tx450v strain induced significantly less IHC labeling compared to the velogenic SA60, rLS-SA60v, TxGB, but not rLS-VF, strains at day 1; at day 2 pi, the mesogenic rLS-Tx450 strain caused significantly less immunolabeling than the velogenic TxGB and rLS-SA60v but not SA60 and rLS-VF strains. The amount of IHC signal for rLS-Tx450v strain was significantly higher as compared with that of the Tx-450 strain (both mesogenic) at days 1 and 2 pi and significantly higher versus that of the lentogenic LS-wt strain at day 2 pi but not day 1 pi. At days 1 and 2 pi, wild-type and recombinant velogenic viruses tended to induce significantly higher IHC scores than those of the mesogenic and lentogenic strains (clustering in the same group designation; Fig. 12). The lentogenic LS-wt strain showed significantly higher IHC staining versus the mesogenic Tx-450 strain at day 1 pi.
Discussion
In the present work, we assessed virus distribution and microscopic lesions caused by NDV strains of various virulence after intracranial inoculation in day-old chickens. To our knowledge, this study is the first to investigate NDV neuropathogenesis through this system.
According to a recent retrospective study that used immunohistochemistry to assess virus distribution in the brains of chickens infected via eye drop instillation, 11 VNNDV and mesogenic strains showed widespread replication in the brain parenchyma at days 5 and 10 pi, with lesions most severe at days 9 and 10 pi. In the same study, VVNDV strains also showed immunolabeling in neurons starting at day 5 pi; however, all birds in that group had died within day 5 pi, and lesions could not be assessed at later time points. These data suggest that differences in the ability to replicate in the neuroparenchyma, to cause microscopic lesions in the brain, and to induce neurologic signs may be influenced by the peripheral rate of virus replication and the ability of the virus to spread into the nervous tissue. For instance, VNNDV strains are able to display the classical “neurotropic” phenotype, most likely because birds survive the visceral lesions, allowing sufficient time to develop neurologic lesions on day 5 to 10 pi, while VVNDV strains that may be capable of infecting the brain kill infected birds before day 5 pi. In this study, direct intracranial inoculation of NDV was deployed to study the inherent ability of NDV strains to replicate and cause lesions in the brain, by partially eliminating confounding factors such as systemic virus replication and immunity. The intracranial inoculation system has been variously used to assess the neurovirulence/neuropathogenesis of several viruses and possibly could be used as a model for other neurovirulent avian pathogens.
All vNDV strains killed the animals within day 2 (velogenic) or 3 and 4 (mesogenic) pi, which is the time frame expected for these viruses, confirming that the ICPI inoculation procedure was carried out successfully. 6,22 In fact, the ICPI scores derived from the present study are very similar to those reported in the literature for the tested strains. Our results demonstrate that predilution of virus inoculum, to infect birds with the same viral dose, did not significantly change the mortality patterns of infected birds, suggesting that the ICPI is a reliable method using several concentrations of virus inoculum.
The vNDV strains (velogenic and mesogenic) deployed in this study caused a severe inflammatory reaction of the surface-oriented areas in the brain: ependyma, choroid plexuses, and meninges. Inflammation was characterized by multifocal areas of erosion/ulceration of the surface lining, accumulation of necrotic debris, and infiltration with variable numbers of heterophils, macrophages, and scattered lymphocytes. These lesions developed earlier and were more severe in velogenic, as compared with mesogenic, infected birds. It is unclear why inflammation caused by vNDV strains was present only in the ependyma and meninges and not in the neuroparenchyma. It is possible that the rapid course of the disease (48 hours) might have been too acute for development of inflammatory lesions. Typical nonsuppurative encephalitis with perivascular cuffing is usually a late event in chickens inoculated with either mesogenic or VNNDV strains through eyelid inoculation. 3,6,29 Similarly, in distemper or measles encephalitis, perivascular cuffings usually develop in the subacute stage of the disease, after the initial virus-induced lymphopenia subsides. 12,25 Alternatively, day-old chickens might have an immature immune system that does not allow severe lymphocytic inflammation, as is observed in mature birds. Edema of the neuroparenchyma was also observed in vNDV-infected birds and was characterized by multifocal to coalescing areas with swollen neurons and severe vacuolation of the neuropil and white matter. Among velogenic-infected birds, edema was caused by only the VVNDV SA60 and rLS-SA60v strains and appeared to be a later event when compared with inflammation, becoming conspicuous at day 2 pi. Compared with velogenic strains, mesogenic strains caused a delayed appearance of edema lesions at days 3 and 4 pi; however, the extent of edema was comparable to and more severe than that caused by other velogenic strains, with the exception of SA60.
Results of IHC showed that that the velogenic strains replicated faster than the mesogenic strains, the latter having a delay of 24 to 48 hours. In fact, only at days 3 and 4 pi, mesogenic rLS-Tx450v and Tx-450 strains, respectively, had viral antigen magnitude similar to those of velogenic strains at day 2 pi. Similar to inflammatory lesions, IHC labeling of vNDV strains appeared to progress from the surfaces (ependyma, choroid plexuses, meninges) toward the neuroparenchyma. Surface orientation of initial IHC staining and inflammation is consistent with the intracranial inoculation method, which likely caused the inoculum to readily spread following the path of the cerebrospinal fluid in the ventricular system and in the subdural space. As replication of vNDV strains progressed from the surfaces to the neuroparenchyma at days 2, 3, and 4 pi (highest IHC scores), edema ensued, albeit not in every case. The edema, extremely severe with the SA60 strain, was possibly the result of intracellular viral replication and increased permeability of cell membranes. This is supported by the fact that highest edema scores coincided with highest virus replication (as shown by IHC). Still, it remains unclear why not all the vNDV strains that replicated in the neuroparemchyma were able to induce edema. It is possible that this might have been the consequence of strain-specific features or that our scoring system or collection time intervals might not have been sensitive enough to detect differences in virus replication that might have been important for edema development.
Among velogenic strains, no substantial differences were observed between wild-type or recombinant strains; however, it is noteworthy to mention that TxGB strain—the model neurovirulent Newcastle disease strain 3,6 —did not induce significantly more lesions or replicate more extensively than the velogenic viscerotropic strains. Between velogenic and mesogenic NDV strains, the main difference appeared to be the timing of lesion development. Although inflammatory lesions were consistently less severe than those elicited by velogenic strains, edema and virus replication were comparable to those observed in velogenic-infected groups but occurred at later time points. These results suggest that differences in neuropathogenesis between velogenic and mesogenic NDV strains may not be caused by a differential capability of the virus to infect nervous tissues. Other factors—such as faster replication rates in nonneuronal cells or increased capacity to redistribute using other cell types by strains with higher ICPI scores—may explain this outcome.
Results of this study demonstrate that vNDV strains commonly responsible for neurologic signs (VNNDV and mesogenic) 6 can replicate in the brain of intracranially inoculated chicks, but they spread at slower rates than VVNDV strains, which are not commonly considered “neurotropic.” This suggests that the outward neurotropic phenotype of VNNDV and mesogenic strains might depend—when inoculation occurs by natural route of infection—by the overall capacity of the virus to infect other organs and tissues, with more virulent strains killing the birds too early for neurologic signs and lesions to ensue and be detected. This is supported by the fact that VVNDV strains have been detected by IHC in neurons as early as day 5 pi, when inoculated through eyelid instillation, before neurologic signs were readily distinguishable. 3,6,11
Strain LS-wt, the only lentogenic NDV strain tested in this study, was able to replicate in only a limited amount in the ependymal cells, most likely due to diffusion of the inoculum through the cerebrospinal fluid, but it lacked neuroinvasiveness—that is, it failed to reach and/or replicate in the neuroparenchyma. These observations are in agreement with a previous study that demonstrated the inability of LS-wt strain to replicate in primary neuronal cell cultures at low multiplicity of infection, even after addition of allantoic fluid as a source of proteases. 13 Since lentogenic NDV strains require trypsin to produce infectious progeny, 18,21 it is possible that ependymal cells produce enough trypsin to allow limited and sustained viral replication (from day 1 to 4 pi), while neurons do not. Ependymal cells appear to produce different kinds of proteases to clear the cerebrospinal fluid, and this could explain their ability to partially maintain LS-wt replication. 8 Despite local ependymal replication, no loss of ependymocytes, necrosis, or inflammation was observed in the brain of LS-wt-infected chicks. Instead, starting at day 2 pi and increasing at day 4 pi, in subependymal locations there was accumulation of several lymphocytes often arranged in nodules. It is unclear whether these lymphocytes are a true inflammatory response or rather a local proliferation simply due to immunologic stimulation. Chickens produce large amounts of ectopic lymphoid infiltrates in multiple tissues during early life (up until 3–4 months of age), partly because of local immunologic stimulation. 23 Ectopic lymphoid tissues are also found in brain, nerves, and pineal gland. 23 Because of their different nature, LS-wt-induced lesions were considered separately from those induced by vNDV strains. Notably, change of LS-wt fusion cleavage site into a virulent one (rLS-VF) induced a neuroinvasive and neurovirulent phenotype in LS-wt, indicating that the cleavage site of the fusion protein is sufficient to allow vaccine viruses to replicate in neurons, in agreement with other studies. 30,31
Overall, recombinant velogenic strains behaved similarly to wild-type velogenic strains in both timing and severity of lesions. The VVNDV SA60 strain induced the highest extent of edema among all strains used in the study. Two mesogenic strains, 1 wild type and 1 recombinant, were used in the study: Tx-450 (a pigeon isolate) and rLS-Tx450v (LS-wt backbone with fusion and hemagglutinin-neuraminidase protein from Tx-450). Strain rLS-Tx450 had an increased virulence versus Tx-450 strain, as observed by ICPI scores, severity, and earlier development of lesions. This suggests that genes from LS-wt may have increased the fitness of Tx-450, although the contribution of each gene to this event remains to be elucidated. The RNA-dependent RNA polymerase of LS-wt has been shown to increase the virulence when inserted in the context of other NDV backbones, possibly because of a higher RNA processivity. 10,24 This could explain the higher virulence of rLS-Tx450v versus Tx-450.
The fact that no salient differences in neurotropism (type of cells infected, extent of replication) were observed in this study among vNDV strains (velogenic and mesogenic) supports the idea that the variable presentation of NDV neuropathogenicity in the field is not receptor mediated, at least for the most part. Most strikingly, results obtained with rLS-VF strain suggest that change of the cleavage fusion site is sufficient to increase the level of neuroinvasiveness of LS-wt at par with other virulent strains, showing that changes in the hemagglutinin-neuraminidase protein (the receptor of the virus 2,6 ) were not necessary to increase neurovirulence. Although it is likely that small changes at receptor level could increase NDV fitness in replicating in the nervous tissue, this does not appear to be the main mechanism driving the differences in neurologic presentation elicited among VVNDV, VNNDV, and mesogenic NDV strains during natural disease.
Multifocal to coalescing splenic necrosis associated with viral replication was observed in the spleen of vNDV-infected birds, while LS-wt-infected birds did not show lesions or virus replication in the spleen. This suggests that after intracranial inoculation (parenteral), vNDV strains can enter the bloodstream and spread systemically, whereas the lentogenic LaSota strain is not able to disseminate systemically. These results are consistent with the fact the intravenous pathogenicity index shows minimal to no virulence (score 0.00) for the LaSota strain. 6 Furthermore, in numerous pathogenesis studies carried with eyelid inoculation, lentogenic strains could replicate only locally and failed to replicate systemically. 3,6
Conclusions
First, velogenic and mesogenic NDV strains produce similar type and distribution of lesions in the brain; however, lesions caused by mesogenic strains are delayed in time. Second, extent of immunolabeling for NDV nucleoprotein showed that mesogenic strains replicate in the same brain regions as velogenic strains, but with a delay of approximately of 24 to 48 hours. Third, in this study, NDV strains commonly responsible for neurologic signs (VNNDV and mesogenic) replicated in the brain of intracranially inoculated chicks at the same or slower rates than those of VVNDV strains, which are not commonly considered “neurotropic.” This suggests that, when inoculated by natural route of infection, the neurotropic manifestations of VNNDV and mesogenic strains might depend on systemic replication in the animal, with more virulent strains killing the birds too early for neurologic signs and lesions to be detected. Finally, lentogenic NDV strains replicate minimally in the ependymal layer and cannot spread through the neuroparenchyma. However, change of the fusion cleavage site of LS-wt strains was sufficient to enable full ability to replicate in the neuroparenchyma and induce lesions.
Footnotes
Acknowledgements
We thank Tim Olivier and Jian Zhang for the excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Agricultural Research Service and Current Research Information System of the US Department of Agriculture (6040-32000-064). V.M.B.D.M. was supported by a scholarship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and by the Federal University of Goiás, Brazil.
