Abstract
Laguna Negra virus (LNV) is a New World hantavirus associated with severe and often fatal cardiopulmonary disease in humans, known as hantavirus pulmonary syndrome (HPS). Five hamster species were evaluated for clinical and serologic responses following inoculation with 4 hantaviruses. Of the 5 hamster species, only Turkish hamsters infected with LNV demonstrated signs consistent with HPS and a fatality rate of 43%. Clinical manifestations in infected animals that succumbed to disease included severe and rapid onset of dyspnea, weight loss, leukopenia, and reduced thrombocyte numbers as compared to uninfected controls. Histopathologic examination revealed lung lesions that resemble the hallmarks of HPS in humans, including interstitial pneumonia and pulmonary edema, as well as generalized infection of endothelial cells and macrophages in major organ tissues. Histologic lesions corresponded to the presence of viral antigen in affected tissues. To date, there have been no small animal models available to study LNV infection and pathogenesis. The Turkish hamster model of LNV infection may be important in the study of LNV-induced HPS pathogenesis and development of disease treatment and prevention strategies.
Keywords
Many New World hantaviruses of the Bunyaviridae family (genus Hantavirus) are potential zoonotic pathogens associated with hantavirus pulmonary syndrome (HPS) or hantavirus cardiopulmonary syndrome. 2 HPS was first reported in 1993 following an outbreak in North America, where the disease primarily affected healthy adults between the ages of 20 and 50 years (mean age, 38 years). 11 The causative agent, Sin Nombre virus (SNV), was isolated from indigenous wild rodents (Peromyscus maniculatus), which were identified as the source of infection. 14 Several species of the subfamily Sigmodontinae and Neotominae (Rodentia, Cricetidae) have been found to serve as asymptomatic hantavirus reservoir hosts, resulting in wide geographic distribution of the virus across North America, Central America, and South America. 1,15 Generally, hantaviruses are transmitted through infected rodent excretions/secreta or bites or via the inhalation route, although rare instances of human-to-human transmission have been described for Andes virus (ANDV). 12 SNV is the most common cause of HPS in North America, while ANDV, LNV, and Rio Mamore virus are frequently seen in South America. 4 LNV was first associated with an outbreak of HPS in the Chaco region of Paraguay in 1997. Since then, infections have been reported in Brazil, Chile, Bolivia, and Argentina. 23 The small vesper mouse (Calomys laucha) was initially identified as the main rodent reservoir. 6 More recently, LNV has also been found in the large vesper mouse (Calomys callosus) in Argentina, suggesting extensive dissemination of this virus throughout South America. 4,10 Incubation times with a median of 14 to 17 days have been documented for SNV; a similar incubation range has been found in human cases of ANDV infection. 24,26 Currently, there are no specific vaccines or therapies for HPS, other than supportive care. Of the South American hantaviruses, ANDV case fatality rates are approximately 40%, 1 whereas LNV has a lower fatality rate of approximately 15%. 8 –10
HPS caused by hantavirus infection in humans is primarily characterized by an influenza-like prodrome with rapidly progressive respiratory disease, pulmonary edema, cardiogenic depression, and shock. 7,8,26,27 Hallmark pathologic features of HPS include interstitial pneumonia, edema, and perivasculitis in the lungs and generalized infection of endothelial cells. Different hantaviruses associated with HPS have shown great variability in virulence. 4,27 The reasons for this variability are not well understood, and the specifics of hantavirus pathogenesis in humans have been difficult to study as a result of the sporadic nature of the disease, rapid onset of clinical signs, and high case fatality rates. 1 In addition, mechanistic insight is hindered by the limited number of small animal models available for the study of HPS pathogenesis. Currently, there is only 1 small animal model of HPS, which uses a hantavirus known to cause disease in humans. This model involves infection of the Syrian hamster (Mesocricetus auratus) with ANDV. 5,22 Maporal virus, a Venezuelan hantavirus, has been shown to cause disease in Syrian hamsters; however, it is not known to be pathogenic in humans. 13 Syrian hamsters do not succumb to SNV infection, the causative agent of HPS in North America. 16,19
Investigations of HPS pathogenesis could be improved by expanding the scope of surrogate animal models used to study the disease and host responses. We hypothesized that additional members of the Cricetidae family might be susceptible to infection with 1 or more of the distinct hantaviruses. We found that LNV can cause disease in Turkish hamsters (Mesocricetus brandti). Here we describe a novel small animal model for LNV infection in Turkish hamsters where ensuing disease exhibits some major hallmarks of human HPS.
Materials and Methods
Animals
Adult male and female hamsters from 5 species—Turkish, Djungarian, Siberian, Chinese, and Armenian hamsters—were selected from closed breeding colonies maintained within a National Institutes of Health facility. The colonies undergo quarterly health screening for common infectious agents affecting hamsters, including a viral serology panel. All animal experiments were approved by the Institutional Animal Care and Use Committee of the Rocky Mountain Laboratories and were performed following the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International in an accredited facility. All work with infected hamsters and potentially infectious materials derived was conducted in the Integrated Research Facility at biosafety level 4 at the Rocky Mountain Laboratories, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health. Sample inactivation and removal were performed according to standard operating procedures approved by the Institutional Biosafety Committee.
Viruses and Inoculation
SNV strain 77734 and Maporal virus strain 9702105 were kindly provided by Brian Hjelle (University of New Mexico, Albuquerque, NM) and Stuart Nichol (Centers for Disease Control and Prevention, Atlanta, GA), respectively. LNV strain 510B and ANDV strain Chile-9717869 were kindly provided by Connie Schmaljohn (US Army Medical Research Institute of Infectious Diseases, Ft Detrick, MD). Each virus was propagated and titered using previously described methods 18 and diluted to the appropriate dose in sterile Dulbecco’s Modified Eagle Medium (DMEM). Hamsters in experiments 1 and 2 were inoculated via the intraperitoneal route, with 100 focus-forming units of a single hantavirus. This was based on experience with the equivalent dose and route of administration of ANDV, which demonstrates uniform lethality in Syrian golden hamsters. 17,18 Control animals received an equal volume of sterile DMEM alone. Hamsters were observed for clinical signs twice daily and weighed every 2 days. Any animals reaching previously determined experimental end points—which included piloerection, hunched posture, >15% preinfection weight loss, and/or marked respiratory distress—were euthanized by exsanguination via cardiac puncture under deep sedation.
Hematology
In experiment 2, hematologic parameters—including white blood cell (WBC), lymphocyte, neutrophil, platelet, reticulocyte, and red blood cell counts; hematocrit values; and mean cell volume, corpuscular volume, and corpuscular hemoglobin concentrations—were obtained from EDTA blood using a Hemavet 950FS (Drew Scientific, Waterbury, CT).
Antibody Assay
To test for seroconversion, in both experiments a recombinant SNV nucleoprotein-based ELISA was used, as there is high cross-reactivity among hantavirus nucleoproteins. 1 Convalescent sera from terminal blood samples of hamsters were collected either at predetermined time points or upon reaching humane end points and were analyzed by ELISA for hantavirus antibodies using previously described methods. 3
Histopathology
Samples of all major tissues were collected at necropsy of animals in experiment 2—including brain, spinal cord, cervical lymph nodes, heart, lung, liver, kidney, adrenal gland, gonads, spleen, esophagus, stomach, duodenum, jejunum, ileum, and cecum. Sections of each tissue were fixed in 10% neutral buffered formalin, changed twice, for a minimum of 7 days. Tissues were processed routinely and histologic section stained with hematoxylin and eosin. Slides were evaluated and scored in a masked manner by a veterinary pathologist. Scoring for hematoxylin and eosin–stained lung specimens were as follows:
0 = No lesion. 1 = One to three small foci of interstitial pneumonia characterized by alveolar septa minimally thickened by congestion, edema, and very small numbers of macrophages and neutrophils. 2 = One to three larger foci of interstitial pneumonia with alveolar septa mildly thickened by congestion and edema with few macrophages and neutrophils. In some instances, the inflammatory infiltrate was predominantly histiocytic. Multifocal lymphohistiocytic cuffing of pulmonary vasculature was present. 3 = Multifocal to coalescing interstitial pneumonia with alveolar septa moderately thickened by congestion, edema, and moderate numbers of macrophages and neutrophils. In some instances, the inflammatory infiltrate was predominantly histiocytic. Multifocal lymphohistiocytic cuffing of pulmonary vasculature was present. 4 = Diffuse interstitial pneumonia with alveolar septa markedly thickened by congestion, edema, fibrin, and large numbers of macrophages and neutrophils. In some instances, the inflammatory infiltrate is predominantly histiocytic. Coalescing to diffuse lymphohistiocytic cuffing of pulmonary vasculature is present. Edema and fibrin extended into the alveolar spaces along with moderate numbers of macrophages and neutrophils.
Immunohistochemistry
Tissues evaluated for immunohistochemistry from animals in experiment 2 were fixed and processed as described above. Immunoreactivity to Laguna Negra viral antigen was detected using a cross-reactive anti-N protein rabbit polyclonal antibody raised against Sin Nombre hantavirus at a 1:27 000 dilution. Tissues were then processed for immunohistochemistry using the Discovery XT automated processor with a DAB Map kit (Ventana Medical Systems) using Biogenexbiotinylated anti-rabbit SS Link as the secondary antibody. Digital images were captured using cellSens Dimension 1.4.1 software (Olympus).
The presence of viral antigen in tissues was evaluated by a veterinary pathologist and quantitatively scored in a masked manner as follows: 0 = no immunopositivity, + = 1% to 25% of tissue immunopositive, ++ = 26% to 50% of tissue immunopositive, +++ = 51% to 75% of tissue immunopositive, ++++ = 76% to 100% of tissue immunopositive.
Results
Experiment 1 evaluated clinical signs consistent with HPS when groups of 6 hamsters of 5 species (Turkish, Djungarian, Siberian, Chinese, and Armenian) were intraperitoneally inoculated with 100 focus-forming units of SNV, ANDV, Maporal virus, and LNV. All animals were monitored twice daily for 28 days postinfection (dpi). At the end of the experiment, regardless of disease status, all infected animals demonstrated hantavirus seroconversion with antibody titers ranging from 1:100 to 1:6400. In this experiment, Djungarian, Siberian, Chinese, and Armenian hamsters did not show any signs of disease upon infection with any of the viruses tested. Clinical signs indicative of HPS (piloerection, hunched posture, and dyspnea) were observed in all 6 Turkish hamsters (M. brandti) infected with LNV. Two of 6 hamsters succumbed to disease, at 15 and 16 dpi (Fig. 1). A second experiment was performed in which 20 animals were infected as above, and 5 control animals were mock infected with comparable volumes of DMEM. At predetermined time points (5, 8, and 14 dpi), groups of 4 infected animals and 1 control animal were euthanized for blood and organ sampling. The remaining 8 infected animals were intended for clinical monitoring until the end of the experiment at 28 dpi or until reaching humane end points.
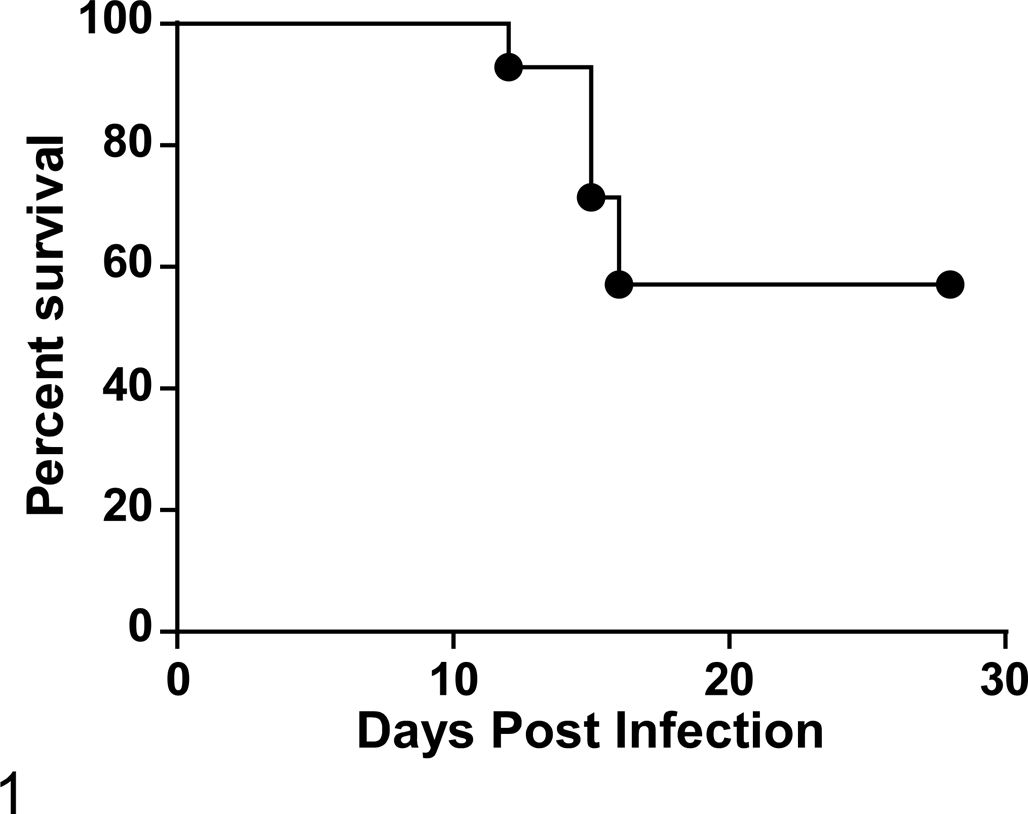
Fourteen Turkish hamsters over 2 experiments were infected with Laguna Negra virus and showed a 43% fatality rate. All deaths/humane end points occurred between 12 and 16 days postinfection.
At the 14 dpi time point, 2 animals began to exhibit clinical signs and were euthanized. An additional 4 animals reached experimental end points: 1 at 12 dpi, 2 at 15 dpi, and 1 at 16 dpi (Fig. 1). These 6 animals demonstrated reduced mobility and increased respiratory effort and were thus designated as the clinical group. Four infected animals reached the end of the experiment (28 dpi) and appeared clinically normal throughout with no mild or transient clinical signs; these were designated as survivors. All control animals were clinically normal over the course of the experiment. Supplemental Table 1 summarizes the outcome of experiments 1 and 2.
Changes in body weight from baseline to 12 dpi were significantly different among clinically affected animals (n = 6 hamsters), survivors (n = 4), and controls (n = 3) for which data were available (P = .003, exact Kruskal-Wallis test). Animals in the clinical group showed significantly greater loss of body weight compared to surviving (P = .019) and control animals (P = .024) at 12 dpi (exact Wilcoxon rank-sum test). There was no significant difference between survivors and controls (P = .5) at this time point (Supplemental Table 2).
Serology
Animals surviving to 28 dpi in experiment 2 demonstrated seroconversion. Antibody titers ranged from 1:1600 to 1:6400. All control animals were negative for anti-hantavirus nucleoprotein antibodies.
Hematology
Hematologic parameters were determined from animals in experiment 2, including hamsters euthanized at 5, 8, and 14 dpi (n = 4 per group), animals that reached humane end points (clinical group), 2 of the surviving infected animals, and all uninfected control animals (Figs. 2–5). Hematology data were not obtained from 1 animal found dead at 16 dpi. WBC counts were significantly lower in the clinical group compared with controls, survivors, and the groups of animals not clinically affected that were sampled at 5 and 14 dpi (Fig. 2; P < .05, unpaired t test). WBC counts for the clinical group were not significantly different from those of the 8-dpi group (P = .06). Neutrophil and lymphocyte counts in blood were significantly lower (P < .05, unpaired t test) in the clinical group compared with controls, survivors, or those animals sampled at 5 and 14 dpi (nonclinical), but no significant difference was seen between clinical animals and the 8-dpi group (Figs. 3, 4). The percentage of lymphocytes and neutrophils was not significantly different among groups. Platelets were significantly lower in all infected animal groups compared to uninfected controls, except in the 14-dpi group (Fig. 5; P < .05).
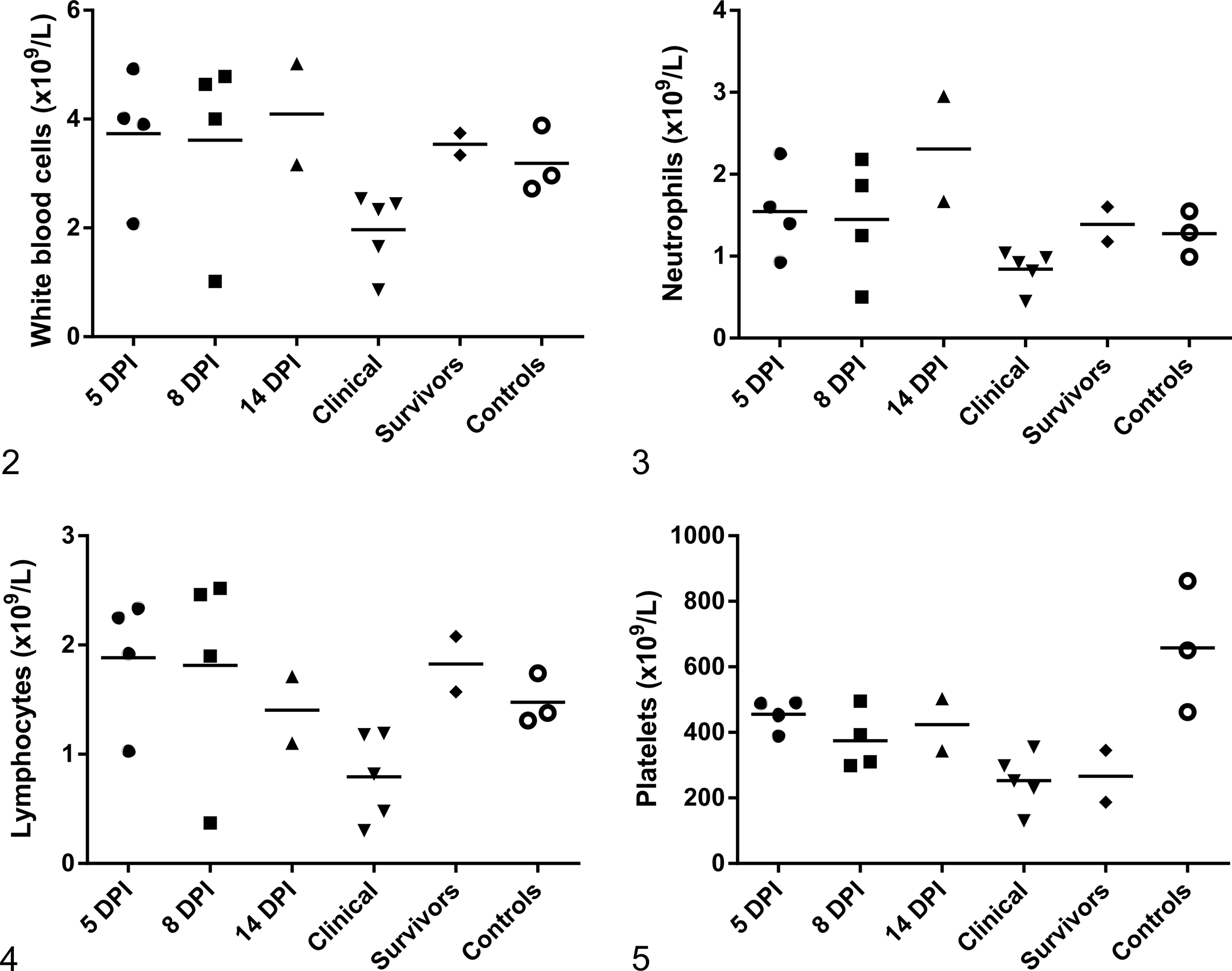
Hematology findings in Turkish hamsters inoculated with Laguna Negra virus (LNV) and uninfected controls (experiment 2). Each symbol represents an individual animal. Horizontal bars represent mean values. Five, 8, and 14 days postinfection (dpi): clinically normal animals euthanized at predetermined end points. Clinical: clinically affected animals that succumbed to infection. Survivors: clinically normal animals that survived to 28 dpi.
Histopathology and Immunohistochemistry
Histologic pulmonary lesions consisted of acute multifocal interstitial pneumonia with perivasculitis characterized by alveolar septal thickening with numerous neutrophils and macrophages, as well as small amounts of hemorrhage, fibrin, and edema (Figs. 6–13). The inflammatory cells, fibrin, and edema frequently extended into alveoli. In addition, there were scattered pulmonary vessels bounded by small to moderate numbers of lymphocytes; this cuffing was most pronounced in animals necropsied at later time points. Pathologic changes were observed as early as 5 dpi but were all more pronounced in animals at later time points up to 16 dpi. By the overt clinical stage of disease, lesions had progressed to diffuse subacute interstitial pneumonia, with abundant fibrin deposition in alveolar and interstitial spaces.
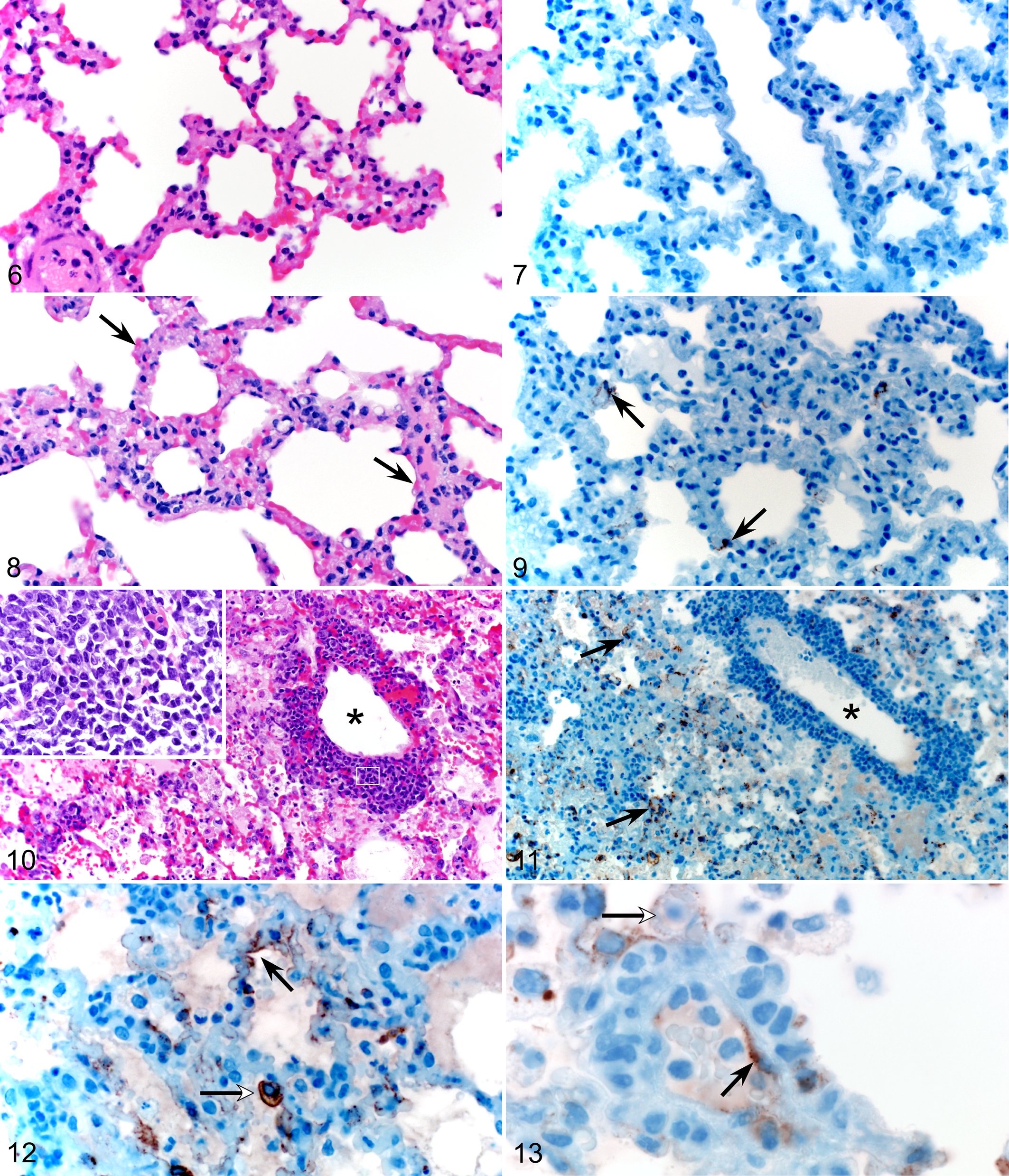
Normal lung, uninfected Turkish hamster.
Average lung histopathology scores increased from 5 to 14 dpi and were greatest in the animals succumbing at 12 to 16 dpi. Lung histopathology scores at 5, 8, and 14 dpi were 0.5 ± 1, 0.5 ± 1, and 2.25 ± 1.7, respectively, while that in the clinical group was 3.5 ± 0.8. No lesions were observed in the lungs of infected animals that survived to 28 dpi; these animals showed no clinical signs throughout the course of the experiment. No lesions were detected in the lung tissues of control animals.
Immunohistochemistry of lung tissues revealed viral antigen in type I pneumocytes, pulmonary macrophages, and endothelial cells (Figs. 7–13). This was a consistent finding at time points from 8 to 16 dpi. Immunohistochemistry scores for viral antigen in lung and other major tissues were 2.2 ± 1.5 between 12 and 16 dpi in the clinical group, compared with 1.5 ± 1.5 and 1.0 ± 1.7 in the 8- and 14-dpi groups, respectively. No viral antigen was detected in tissues of control animals or infected animals that survived to 28 dpi.
Immunohistochemistry revealed viral antigen in hepatocytes and lymphocytes in the marginal zone of splenic follicles. Viral antigen was also present in splenic, hepatic, renal, and cardiac endothelial cells (Figs. 14–17) with increasing distribution in these tissues over the time course of infection (Table 1). Moderate subacute myocarditis with myocardial degeneration, necrosis, and mineralization was identified in 8 of the 20 infected hamsters.
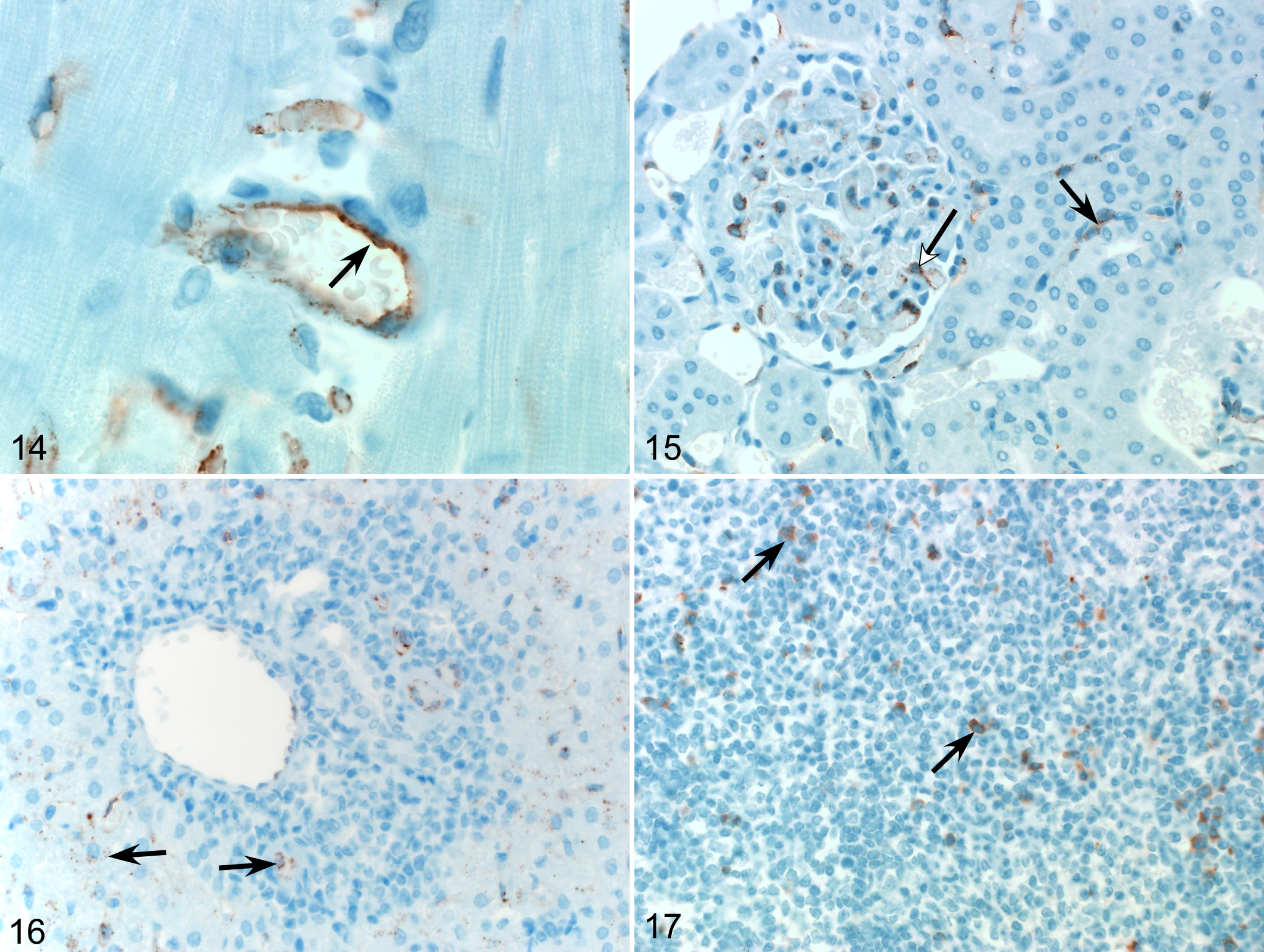
Laguna Negra virus (LNV) infection, Turkish hamster.
Immunohistochemistry scores for viral nucleoprotein in Turkish hamsters infected with Laguna Negra Virus.

Mean immunohistochemistry scores in groups of animals at the time of necropsy. The percentage of immunopostivity in these tissues increased over the time course of infection.
Discussion
Clinically, HPS in humans commences with a prodrome characterized by initial fever and malaise, followed by a cardiopulmonary phase in which there is a sudden onset of severe respiratory symptoms. Patients often progress from coughing and shortness of breath to marked respiratory distress, hypoxemia, shock, and cardiac failure within 48 hours. 28 Raised leukocyte counts with marked left shift and thrombocytopenia are common findings in the cardiopulmonary phase. 27 Pathophysiologically, this stage is characterized by vascular leakage leading to interstitial pneumonia with congestion, edema, mononuclear cell infiltration, and fibrin deposition in the respiratory tissue. 7,26,28 It has been shown that the Syrian hamster (M. auratus) is an excellent model for the study of HPS caused by ANDV infection, principally because it mimics the rapid onset cardiopulmonary phase of disease, which is so characteristic of human HPS. 5,17,20 Histopathologic changes in lung tissue of hamsters infected with ANDV show marked correlation with those reported in humans. Timelines of onset and progression and the distribution of antigen are also highly comparable features.
In the lethal model of ANDV, Syrian hamsters succumb to disease from 9 to 16 dpi (range, 7–29 days), and microscopic abnormalities in lung tissue consistent with HPS are reported from as early as 5 dpi, with most marked lung pathology seen from 10 dpi. 5,20 LNV infection of Turkish hamsters produces detectable lung pathology at 5 dpi with histopathology scores increasing to maximal severity between 12 and 16 dpi when animals demonstrated clinical signs of disease. The variability in virulence and severity between different hanta viruses is poorly understood. The fact that this model is not uniformly lethal will allow further studies to address the issue of why many individuals in human populations are subclinically infected or show mild symptoms, while others progress to rapidly fatal disease.
Another confounding factor in adequately identifying and treating clinical cases of HPS in humans is the rapid onset of severe disease with nonspecific premonitory signs. 28 Small animal models of HPS are required to better understand and predict clinical outcomes. A feature of clinical disease not always reported in the Syrian ANDV model is weight loss. Animals in the LNV clinical group showed statistically significant weight loss compared to surviving and control animals. Further studies with a greater number of animals are indicated to confirm and investigate this finding, which could be helpful in studying early stages of hantavirus infection in the Turkish hamster model of LNV. Further comparison of host immune responses between the 2 models, specifically comparing previously reported immune-modulation by ANDV, 20 could also enhance understanding of hantaviral pathogenesis.
Leukocytosis is a feature of disease in the Syrian ANDV model, 5,20,25 whereas a panleukopenia was seen in Turkish hamsters infected with LNV. There was a significant difference in WBC counts between clinically affected animals and clinically normal animals euthanized at 5 and 14 dpi but not at 8 dpi (Fig. 2). In the 5- and 8-dpi groups, a single animal had a lower WBC count similar to those in the clinical group. Clinical signs were not observed until 12 dpi, so it is unknown if the animals euthanized at 5 and 8 dpi would have developed clinical signs. Platelet numbers were significantly lower in all infected animals (both clinical and surviving animals) compared with uninfected controls. Thrombocytopenia and coagulopathy are features of the Syrian ANDV model: although LNV appears to have some effect on platelets, further investigation is required to determine the significance and role of this finding in LNV pathogenesis.
Myocardial necrosis was observed in 8 of the 20 infected hamsters in the second experiment and is a reported finding in human clinical HPS cases. 21 These lesions were not associated with viral antigen based in immunohistochemistry, and further investigation is required to verify and determine the significance of this finding. Widespread endothelial cell involvement throughout the major tissue groups is consistently reported in human and Syrian hamster HPS. 5,21,25,28 This has been demonstrated clearly in the Turkish hamster model of LNV.
In this study, we have shown that LNV infection causes disease in the Turkish hamster that mimics HPS in humans. Animals demonstrated rapid onset of severe dyspnea and concomitant progressive lung pathology, ranging from multifocal to diffuse interstitial pneumonia with alveolar edema and hemorrhage and with a corresponding presence and extent of viral antigen. This novel small animal model will be a valuable tool in further studying hantavirus pathogenesis and in the development of disease treatment and prevention strategies.
Footnotes
Acknowledgments
We thank Kathleen Cordova, Sandy Skorupa, Amanda Weidow, Jayne Farris, and Kimberly Meade White for their assistance with animal care and sampling; Dan Long, Rebecca Rosenke, and Elaine Haddock for their assistance with sample processing; and Anita Mora and Austin Athman for their help with the figures (all with the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Division of Intramural Research, National Institutes of Allergy and Infectious Diseases, National Institutes of Health.
