Abstract
Aleutian disease virus (ADV, Amdovirus, Parvoviridae) primarily infects farmed mustelids (mink and ferrets) but also other fur-bearing animals and humans. Three Aleutian disease (AD) cases have been described in captive striped skunks; however, little is known about the relevance of AD in free-ranging carnivores. This work describes the pathological findings and temporospatial distribution in 7 cases of AD in free-ranging striped skunks. All cases showed neurologic disease and were found in a 46-month period (2010–2013) within a localized geographical region in California. Lesions included multisystemic plasmacytic and lymphocytic inflammation (ie, interstitial nephritis, myocarditis, hepatitis, meningoencephalitis, pneumonia, and splenitis), glomerulonephritis, arteritis with or without fibrinoid necrosis in several organs (ie, kidney, heart, brain, and spleen), splenomegaly, ascites/hydrothorax, and/or encephalomalacia with cerebral microangiopathy. ADV infection was confirmed in all cases by specific polymerase chain reaction and/or in situ hybridization. The results suggest that AD is an emerging disease in free-ranging striped skunks in California.
Striped skunks (Mephitis mephitis) are distributed widely throughout North America and are part of the human-domestic animal-wildlife interface. 4 Aleutian disease virus (ADV, genus Amdovirus, family Parvoviridae) is a parvovirus that commonly infects captive mink and ferrets; its significance in free-ranging members of the order Carnivora is largely unknown. 9 Serologic studies have demonstrated that a virus with shared epitopes to ADV infects not only captive and wild mustelids but other fur-bearing animals, including bobcats, raccoons, and striped skunks. 3 Although not typically considered zoonotic, Aleutian disease (AD) has been documented in humans and is referred to as an occupational disease of mink farmers. 5 AD is caused by chronic antigenic stimulation that leads to high levels of circulating antigen-antibody complexes, which deposit in tissues and cause a type III hypersensitivity reaction. 6 ADV infection in mink causes infiltration of plasma cells in multiple tissues, hypergammaglobulinemia, glomerulonephritis, arteritis, splenomegaly, hepatitis, meningoencephalitis, and death. 6,9 Despite documented ADV infection and disease in captive striped skunks, 2,8 as well as serologic evidence of infection with a related virus in wild striped skunks, 3 little is known about the pathogenicity of the virus in skunks or the relevance of infection in free-ranging animals. 9 This study describes lesions in 7 free-ranging striped skunks with AD and their temporospatial distribution.
The case database of the Davis laboratory of the California Animal Health and Food Safety (CAHFS-Davis) Laboratory System was searched from January 1, 2010, to November 1, 2013, and 7 confirmed cases of AD in free-ranging striped skunks were identified (case Nos. 1–7). Locations of skunk capture, clinical histories, and gross and histologic findings were reviewed. The straight-line distances between capture locations were calculated using the proximity tools for ArcGIS version 10 (ESRI, Redlands, CA). Histologic evaluation was performed on samples of the brain, lungs, liver, kidneys, heart, skeletal muscle, trachea, pancreas, spleen, lymph node, adrenal gland, esophagus, urinary bladder, stomach, small and large intestines, uterus, and skin in all cases. Formalin-fixed, paraffin-embedded sections of kidney from case Nos. 4 and 6 were processed for detection of ADV nucleic acid by in situ hybridization (ISH) using a commercially available DNA probe (Roche Applied Science, Indianapolis, IN) based on the referenced protocol. 8 Fresh-frozen samples of kidney, as well as either pooled or individual samples of liver, lung, spleen, brain, and/or urine, from case Nos. 1 to 5 and 7 were submitted to either the Wisconsin Veterinary Diagnostic Laboratory (University of Wisconsin, Madison, WI; case Nos. 1 and 2) or the Infectious Disease Laboratory at the University of Georgia (IDL, Athens, GA; case Nos. 3–5 and 7) for polymerase chain reaction (PCR) testing for ADV. A sample of peritoneal fluid from case No. 5 was submitted to the Cornell University Animal Health Diagnostic Center (Ithaca, NY) for protein immunoelectrophoresis (ie, total proteins; albumin; total α- and β-globulins; α1-, α2-, β1-, and β2-globulins; and γ-globulin concentrations). As part of the diagnostic investigation on each case, other ancillary tests were performed (see Supplemental Materials).
Within the 46-month period, 25.9% (7/27) of all striped skunks submitted for necropsy were diagnosed with AD. All 7 animals had neurologic signs, including increased diurnal activity, depressed mentation, docility, ataxia, and/or seizures. Animals were captured and either immediately euthanized or brought to wildlife rehabilitators, where they were given supportive care and were either euthanized or died naturally (see Supplemental Materials for clinical histories). All cases were found in a relatively restricted geographical range; 6 cases occurred within a circular area of a 30-km radius in Marin (4) and Alameda (2) Counties, and 1 case occurred in Santa Cruz County. The maximum distance between case locations was 134 km (Suppl. Fig. S1).
At necropsy, the most common gross lesions were diffuse renal pallor (n = 4; Fig. 1), ascites and/or hydropericardium (n = 4), splenomegaly (n = 3), and chronic passive hepatic congestion (enhanced reticular pattern characteristic of “nutmeg liver”; n = 2, Fig. 2). Histologic lesions (Table 1) included varying degrees of multisystemic, plasmacytic, and lymphocytic inflammation, which was most common in the kidney (n = 7, 100% of cases), liver (n = 6, 86% of cases), brain (n = 6, 86% of cases), heart (n = 5, 71% of cases), and lungs (n = 5, 71% of cases). Most cases had lymphoplasmacytic, perivascular infiltrates in at least 1 other organ (ie, tongue, pericardium, thyroid, urinary bladder, mammary gland, trachea, lymph node, spleen, mesentery, and dermis), although each of these organs was affected in only 1 or 2 cases. Renal lesions included glomerulonephritis, lymphoplasmacytic interstitial nephritis (Fig. 3), and arteriolitis (with or without fibrinoid necrosis). In 1 case (No. 4), tubular epithelial cells occasionally contained round to ovoid 3- to 5-micron basophilic intranuclear inclusion bodies with margination of the chromatin (Fig. 3). Microscopic mineralization of the gastric mucosa and/or tongue, suggestive of chronic renal failure, was seen in 2 animals. Myocarditis (n = 5) was frequently associated with fibrinoid arteritis (4/5 cases; Suppl. Fig. S2) and was accompanied by centrilobular hepatic congestion/fibrosis (case Nos. 1, 5, and 7; Suppl. Fig. S3) or pulmonary edema (case No. 3), suggestive of congestive heart failure. Lymphoplasmacytic meningoencephalitis was frequent (n = 6) but mild. In 2 cases, there were either focal or extensive areas of encephalomalacia associated with microangiopathy (arteriolar wall hyalinization) of the adjacent cerebral microvasculature (Suppl. Fig. S4). The histologic correlate of splenomegaly was congestion and lymphoplasmacytic arteriolitis (3/3 cases) with or without follicular hyperplasia (1/3 cases) or diffuse plasma cell infiltration (1/3 cases).
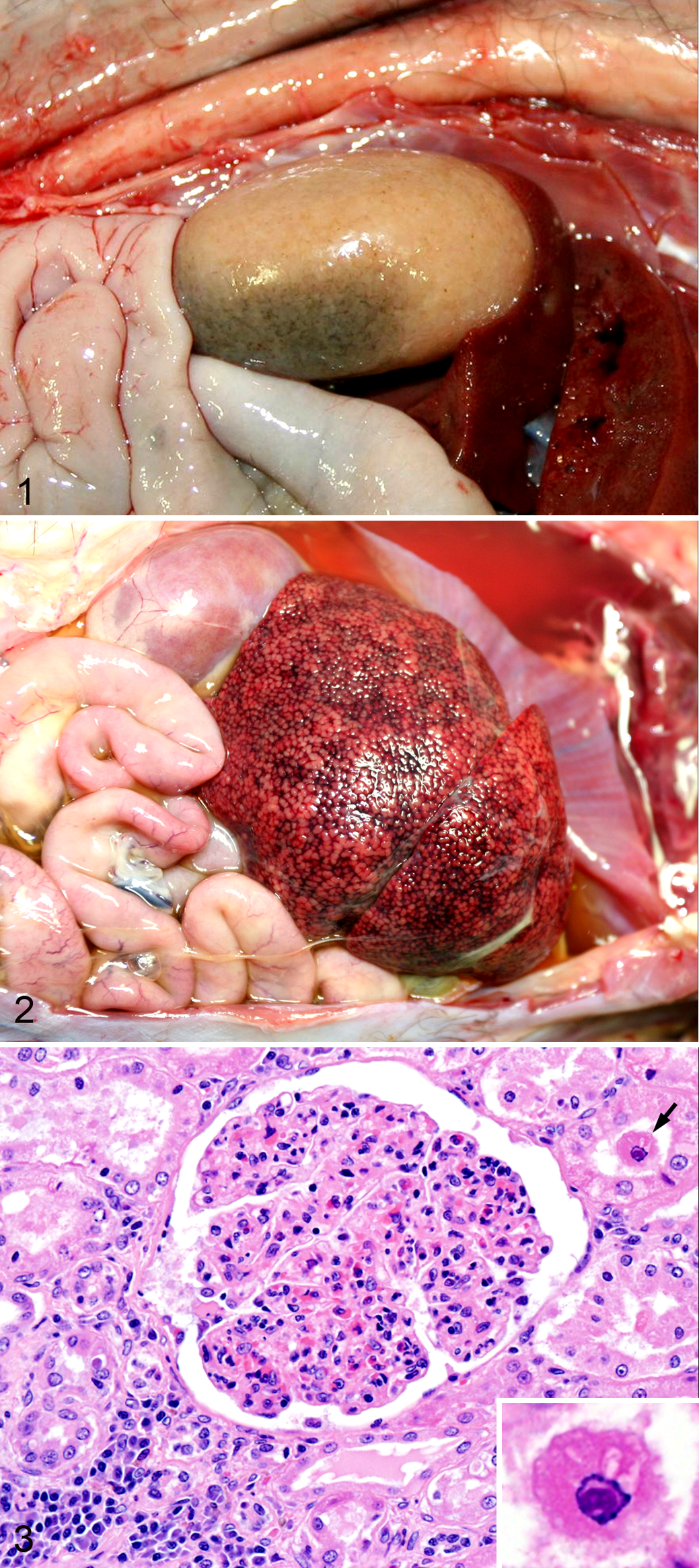
Aleutian disease, skunk.
Summary of Histologic Lesions in Striped Skunks (Mephitis mephitis) With Aleutian Disease.a

GN, glomerulonephropathy; LP, lymphoplasmacytic.
aSeverity: –, absent; +, mild; ++, moderate; +++, severe.
bThe lungs in case Nos. 1 and 7 had concomitant conditions that may have contributed to LP interstitial pneumonia. Case No. 1 had necrosuppurative bronchopneumonia with mixed bacterial infection; case No. 7 had pulmonary nematodiasis.
ADV nucleic acid was detected by PCR in all tested samples except the renal sample from case No. 3. ADV nucleic acid was detected in the kidney of case Nos. 4 and 6 by ISH (Suppl. Fig. S5). Results of the immunoelectrophoresis of the peritoneal fluid in case No. 5 were as follows: total protein, 4.2 g/dl; albumin, 0.93 g/dl; total α-globulin, 0.69 g/dl (α1-globulin, 0.19 g/dl; α2-globulin, 0.5 g/dl); total β-globulins, 1.04 g/dl (β1-globulin, 0.19 g/dl; β2-globulin, 0.85 g/dl); and γ-globulin, 1.54 g/dl. The albumin/globulin ratio was 0.28, and albumin represented 22% of the total protein. Although normal values (reference intervals) for peritoneal fluid in striped skunks are not established, these results were compatible with increased γ-globulin levels compared with normal serum values in ferrets, 7 suggesting either exudation or localized antibody (immunoglobulin) production. Results of the other ancillary diagnostic tests are shown in the Supplemental Materials.
All AD cases were found within a relatively small geographical region in California and represented more than a quarter of all skunks submitted to CAHFS-Davis within the 46-month study period. As AD has not previously been reported in free-ranging striped skunks, this finding suggests an emerging disease in California. Reports describing pathologic findings in skunks with AD are limited to 3 cases diagnosed in captive/pet individuals in Tennessee, Georgia, and Ohio. 2,8 All referenced cases described multisystemic plasmacytic and lymphocytic inflammation, as seen in the present cases. Encephalomalacia associated with cerebral microangiopathy has not been previously described in skunks with AD. Splenomegaly, renal pallor, and peritoneal effusion have been described in captive skunks with AD and were observed in the present cases; these features may increase the pathologist’s index of suspicion for AD at necropsy. 2,8 Inclusion bodies have been described in type II pneumocytes of mink experimentally inoculated with ADV, but viral inclusions have not been described in other tissues in AD. 1 Inclusions were identified in only 1 case in this series and were not confirmed to be viral by transmission electron microscopy. Several of the animals had comorbidities (eg, bronchioloalveolar carcinoma, bronchopneumonia, hepatitis, endometritis, and/or respiratory, gastrointestinal, or muscular parasitism; data not shown), which may have exacerbated AD lesions, but only 1 comorbidity was deemed significant (hepatic salmonellosis; case No. 5).
AD may be underdiagnosed in free-ranging populations of striped skunks. A survey of parasitic and infectious diseases in 2 rabies-free populations of wild striped skunks in Illinois reported frequent multisystemic lymphoplasmacytic inflammation, including glomerulonephritis or interstitial nephritis (43% of cases), interstitial (43% of cases) or bronchointerstitial (62% of the cases) pneumonia, meningoencephalitis (29% of cases), and myocarditis (29% of cases), all of which were of unconfirmed etiology. 4 It was suggested that these lesions were due to canine distemper virus, leptospirosis, and/or macroparasites, and ADV testing was not performed. Given the type and distribution of lesions in our case series, we suggest that ADV infection be considered in any striped skunk with glomerulonephritis, interstitial nephritis, and/or multisystemic lymphoplasmacytic lesions involving the kidneys, heart, brain, liver, and lungs. In this study, PCR was the method of choice to confirm ADV infection. In case No. 4, ADV nucleic acid was detected in kidney and urine by PCR and in kidney by ISH, suggesting viral shedding in urine.
In conclusion, AD is an emerging disease in free-ranging striped skunks in California and should be suspected in individuals with systemic lymphoplasmacytic inflammation. PCR and ISH allow for etiologic confirmation in suspect cases. Additional studies are warranted regarding the prevalence and geographic distribution of AD in skunks, impact on populations of skunk and other mustelids in California, and the possible zoonotic risk.
Footnotes
Acknowledgements
We thank Terrence Wildman, Joe Grismer, Karen Sverlow, and Scott Fish from CAHFS-Davis, as well as Lora Konde from the California Department of Fish and Wildlife for their technical assistance, and Rebecca Dmytryk of Wildlife Emergency Services for capture and submission of a skunk.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for necropsies and diagnostic testing was provided by the California Department of Fish and Wildlife, and WildCare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
