Abstract
Cytochrome P450 1B1 (CYP1B1) is highly expressed in human and murine ocular tissues during development. Mutations in this gene are implicated in the development of primary congenital glaucoma (PCG) in humans. Mice deficient in Cyp1b1 (Cyp1b1–/– ) present developmental abnormalities similar to human primary congenital glaucoma. The present work describes the ultrastructural morphology of the iridocorneal angle of 21 eyes from 1-week-old to 8-month-old Cyp1b1–/– mice. Morphometric and semiquantitative analysis of the data revealed that 3-week-old Cyp1b1–/– mice present a significantly (P < .005) decreased amount of trabecular meshwork (TM) collagen and higher TM endothelial cell and collagen lesion scores (P < .005) than age-matched controls. Collagen loss and lesion scores were progressively increased in older animals, with 8-month-old animals presenting severe atrophy of the TM. Our findings advance the understanding of the effects of CYP1B1 mutations in TM development and primary congenital glaucoma, as well as suggest a link between TM morphologic alterations and increased intraocular pressure.
Keywords
Glaucoma is a leading cause of vision loss in the adult human population. It represents a large, diverse group of intraocular pressure (IOP)–dependent neurodegenerative disorders that result in loss of normal function and integrity of the retinal ganglion cells and their axons in the optic nerve, ultimately causing vision loss. 20 Increased IOP is considered a major risk factor for development of glaucoma and thus has been the main focus of treatment. Defects in the aqueous humor drainage are the main cause of elevated IOP. 22 The aqueous humor outflow system is dependent primarily on the trabecular meshwork (TM), a sponge-like, 3-dimensionally arranged tissue composed of connective tissue beams lined by endothelial-like cells (trabecular cells) that is circumferentially present and expanded at the base of the iris and peripheral cornea (iridocorneal angle). The TM is responsible for draining the aqueous humor from the anterior chamber into the venous system via Schlemm’s canal and collector channels, maintaining homeostasis of the IOP. 19
Although less prevalent than the adult forms, primary congenital glaucoma (PCG) causes a devastating loss of vision in children. The pathogenesis of the disease is associated with developmental defects of the TM, but the underlying mechanisms are not well understood. 19 Genetic linkage analyses and mutation studies have identified that mutations on the cytochrome P450 family 1 subfamily B polypeptide 1 (CYP1B1) are a common cause of PCG in humans. 12,15,16,21 CYP1B1 and other genes of the CYP450 family catalyze NADPH-supported monooxygenation of diverse xenobiotics and endogenous molecules. 13,18 Strong expression of CYP1B1 in the fetal human and murine eye indicates that CYP1B1 plays an important role in ocular development and function. 3,6,17 Recently, our group demonstrated that Cyp1b1 deficiency significantly impaired trabecular cell function and oxidative homeostasis of the TM tissue in mice. Expression of oxidative damage markers was increased in TM tissues from Cyp1b1–/– mice, and cultured TM cells from the same animals presented increased apoptotic rates and decreased viability under oxidative stress and enhanced adhesion to extracellular matrix (ECM) proteins such as fibronectin, vitronectin, collagen I, and collagen IV compared with Cyp1b1 +/+ TM cells. 23 Multiple measurements of IOPs from 3 weeks to 10 months of age showed that Cyp1b1–/– mice presented significantly elevated IOP at all time points compared with those of Cyp1b1 +/+ mice. 23 Here we determine the detailed ultrastructural morphology of the postnatal iridocorneal angle of Cyp1b1–/– mice and demonstrate progressive abnormalities in the ECM of the trabecular meshwork.
Materials and Methods
Animals
All experiments were carried out in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Institutional Animal Care and Use Committee of the University of Wisconsin School of Medicine and Public Health. Cyp1b1–/– mice expressing a temperature-sensitive simian virus 40 (SV40) large T antigen (Charles River Laboratories, Wilmington, Massachusetts) in a C57BL/6 background were generated by targeted disruption of the coding exons 2 and 3 of the Cyp1b1gene in embryonic stem cells, as previously described. 23 The right globe of 42 female mice, 21 wild-type (WT) and 21 Cyp1b1–/– mice, were collected at 1, 2, 3, and 6 weeks and 3, 7, and 8 months of age (3 Cyp1b1–/– and 3 WT eyes at each time point). Enucleated globes were immersion fixed in 2% paraformaldehyde (PFA) and 2.5% glutaraldehyde in 0.1 M phosphate-buffered saline (PBS; pH 7.4) at 4°C overnight. Four areas of the trabecular meshwork were sampled representing the dorsal, ventral, temporal, and nasal regions of the globe. Samples were processed for routine transmission electron microscopy (TEM). Briefly, fixed globes were treated with 1% OsO4 in PBS for 2 hours at room temperature followed by three 10-minute washes with 0.1 M sodium acetate buffer. Tissues were then stained with 2% uranyl acetate in sodium acetate buffer for 1 hour at room temperature, washed in buffer, dehydrated in a graded ethanol series (40%–100%), and infiltrated with propylene oxide–812 resin (1005 Embed 812; EMS, Fort Washington, Pennsylvania). The samples were embedded with fresh 100% 812 resin in molds and polymerized in a 60°C oven for 36 hours. Ultrathin sections (90 nm) were analyzed using a JEOL 100CX electron microscope (JEOL Ltd, Tokyo, Japan).
Histologic Analysis
One-micron-thin sections of the trabecular meshwork samples were stained with toluidine blue and examined under a light microscope (Olympus BX43; Olympus, Melville, New York). The morphology of the ciliary cleft, trabecular meshwork beams, Schlemm’s canal, and aqueous veins was accessed.
Morphometric Analysis
To quantify the relative amount of collagen in the trabecular meshwork, we have applied a computer-based segmentation analysis to TEM images of the trabecular meshwork. Five nonconsecutive 15 000× TEM images of the anterior, mid, and posterior TM were processed and analyzed using ImageJ software (National Institutes of Health, Bethesda, Maryland; http://fiji.sc/Fiji). Briefly, the image’s scale bar was used to set up the system’s spatial calibration. A background subtraction was performed on the images (Process>Subtract Background option) followed by segmentation of the collagen fibers (Image>Adjust>Threshold tool option). Using the threshold tool, the collagen fibers were manually selected and the amount of collagen in the image was measured (Analyze>Measure tool) and expressed as a percentage of the image. To standardize the data, we avoided imaging the intratrabecular spaces, focusing predominantly on the trabecular beams.
Semiquantitative Analysis
A semiquantitative ordinal scoring system was devised to access the alterations in the ultrastructural morphology of trabecular cells and distribution of the collagen lesions in the TM. TM collagen lesion distribution score ranged from 0 to 4, where 0 indicates no lesion; a score of 1 indicates collagen fiber disarrangement (irregularly oriented and fragmented collagen fibers) affecting <25% of the TM; score of 2, 25% to 50% of the TM; score of 3, 50% to 75% of the TM; and score of 4, >75% of the TM. Trabecular cell morphology was analyzed by accessing the presence of the following ultrastructural lesions (criteria): irregular cell surface, loss of contact with the basement membrane, cytoplasmic vacuolization, cell swelling, and the presence of irregular cytoplasmic material. Samples were scored from 0 to 4, where 0 indicates no lesion, 1 indicates 1 criterion present, 2 indicates 2 criteria, 3 indicates 3 to 4 criteria, and 4 indicates all 5 criteria were present.
Statistical Analysis
Statistical analyses were performed using the 2-tailed unpaired Student t test for collagen morphometric data and the Wilcoxon–Mann-Whitney 2-sample rank sum test for the semiquantitative analyses. Collagen quantification data are reported as mean ± SEM, and semiquantitative analysis data are reported as median ± range. Differences were regarded as significant when P ≤ .005.
Results
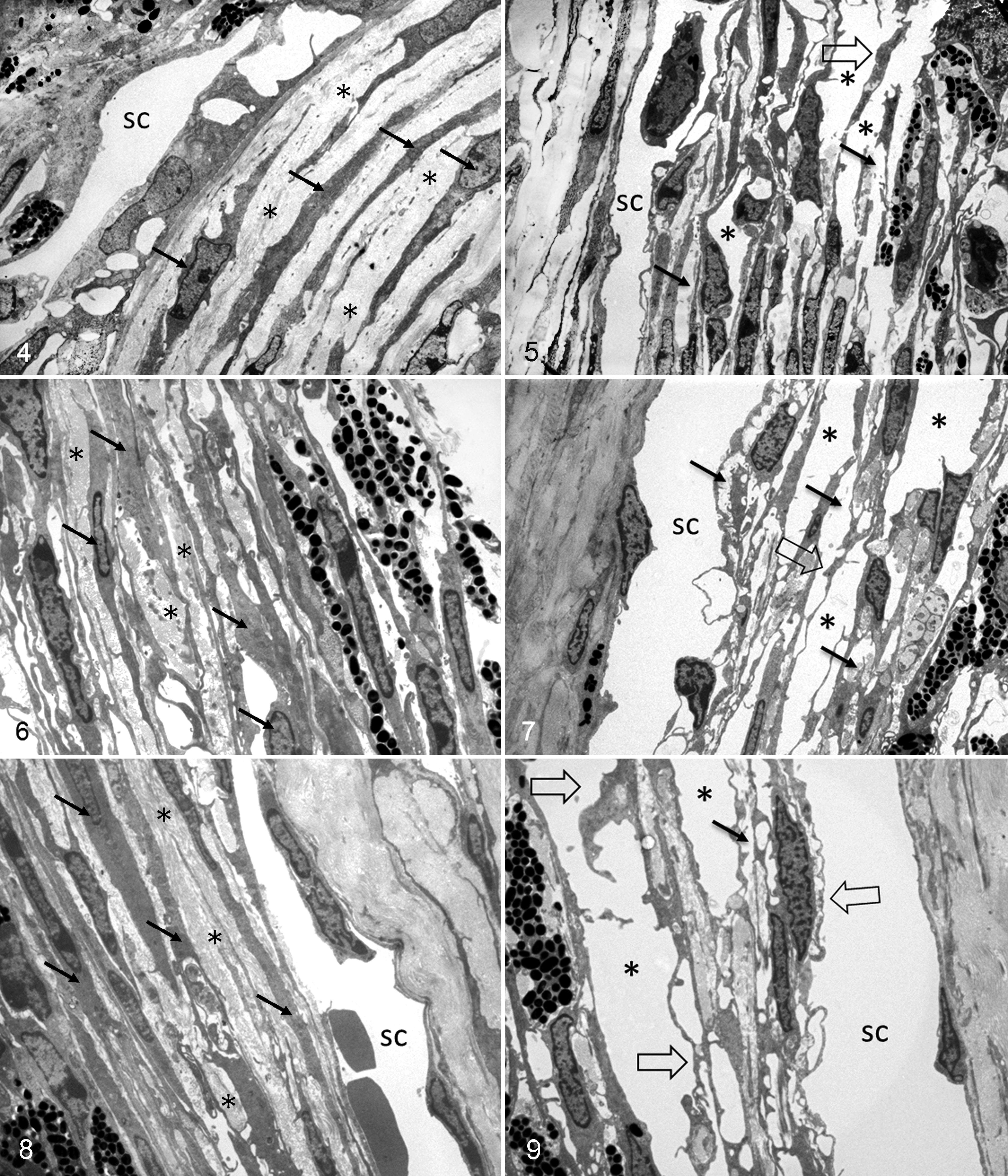
Histologic analysis of the iridocorneal angles of Cyp1b1–/– animals showed that all the animals exhibited an open ciliary cleft and no abnormalities of Schlemm’s canal or aqueous veins (Fig. 1). Ultrastructural lesions were noted in the extracellular matrix of the trabecular meshwork. One- and 2-week-old Cyp1b1–/– animals presented a relatively normal trabecular meshwork, characterized by multiple trabecular beams composed of a core of collagen fibers interspersed with small amounts of elastic fibers covered by trabecular cells (Figs. 2, 3). Starting at 3 weeks of age, Cyp1b1–/– animals presented marked disruption of the anterior and posterior trabecular meshwork characterized by multifocal atrophy of the trabecular beams with accentuation of the intertrabecular spaces. The collagen fibers on the collagenous layer of the trabecular beams were markedly fragmented and irregularly distributed compared with control groups. The TM cells covering the trabecular beams exhibited irregular cytoplasmic processes, loss of contact with basement membranes, variable cell swelling with cytoplasmic vacuolization, and intracytoplasmic accumulation of organelle debris (Figs. 4, 5). The 6-week-old and 3-, 7-, and 8-month-old Cyp1b1–/– animals presented progressively more severe disarrangement of trabecular collagen and consequently more dramatic overall architectural changes of the TM (Figs. 6–9). At the latest time point, 8-month-old Cyp1b1–/– animals presented a complete collapse of the TM with formation of 1 atrophic and irregular trabecular beam spanning the anterior and posterior TM (Figs. 10–12).
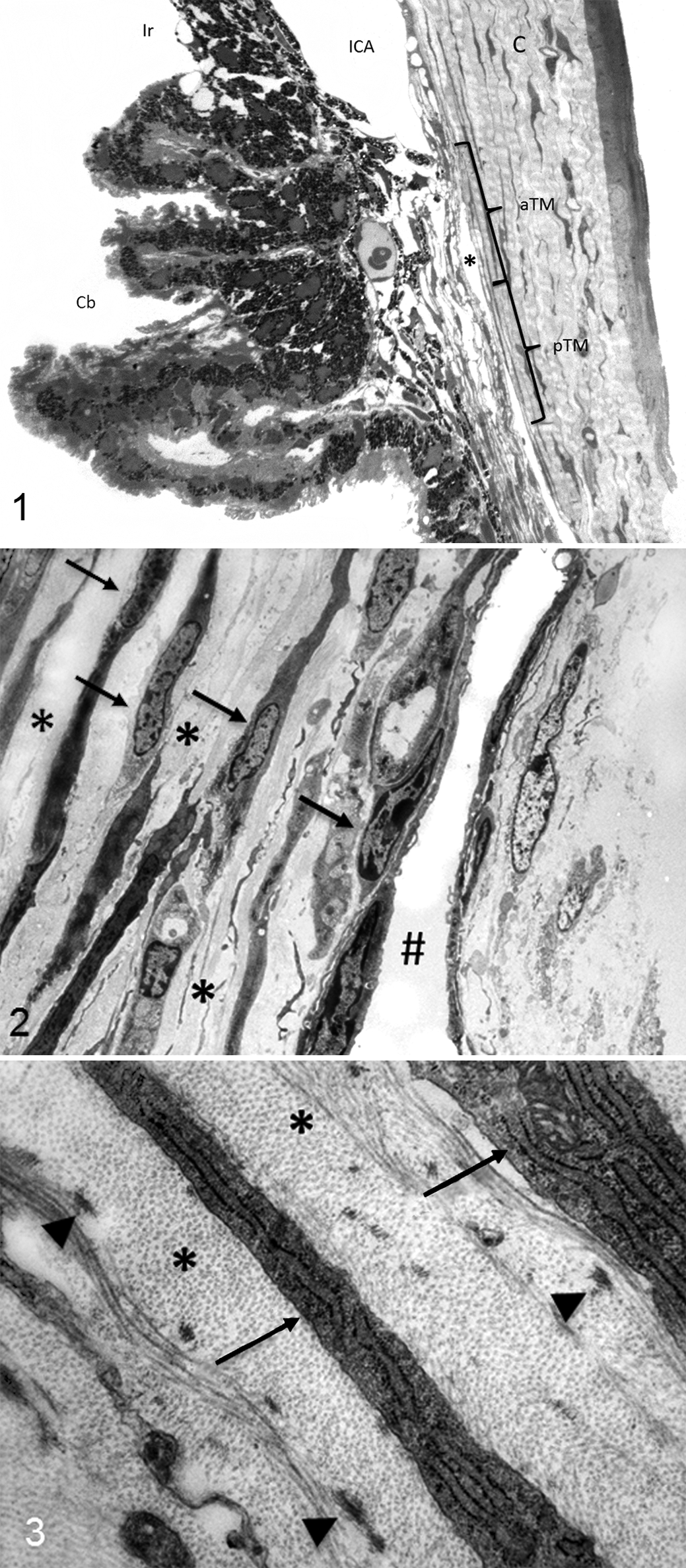
Trabecular meshwork, wild-type and Cyp1b1–/–
mice.
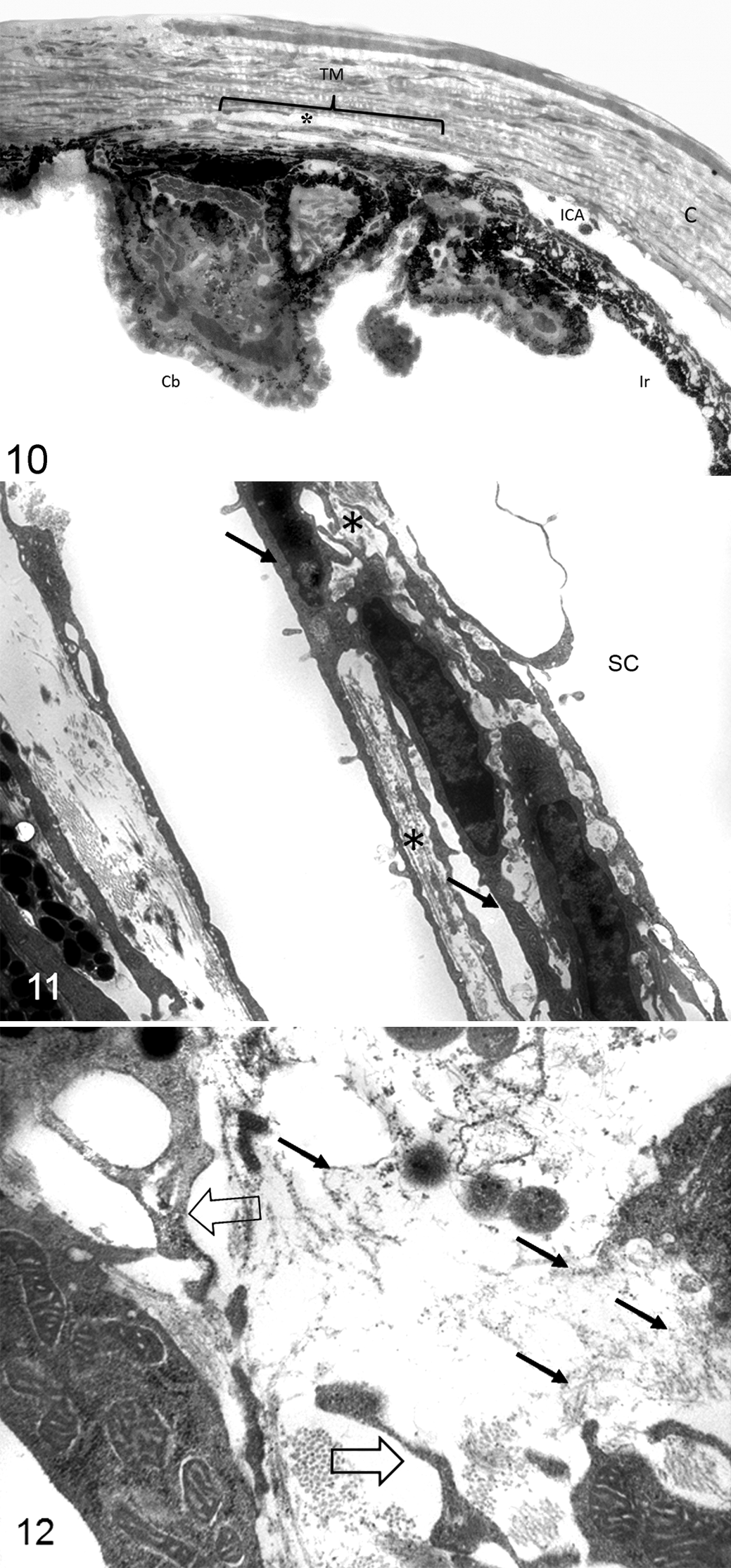
Iridocorneal angle and trabecular meshwork, 8-month-old Cyp1b1–/–
mice.
The morphometric quantification of the relative amount of collagen occupying the trabecular meshwork is represented in Fig. 13. Starting at 3 weeks of age, Cyp1b1–/– mice presented significantly (P < .005) less collagen in the trabecular beams (11.1% ± 3.8%) than age-matched WT controls (35.2% ± 6.7%) as well as younger 2-week-old WT (44.9% ± 4.3%) and Cyp1b1–/– (39% ± 8.4%) mice. The relative amount of TM collagen in affected animals progressively decreased with age and Cyp1b1–/– mice had significantly less TM collagen than age-matched WT animals from 6 weeks of age through the course of the study (Fig. 13).
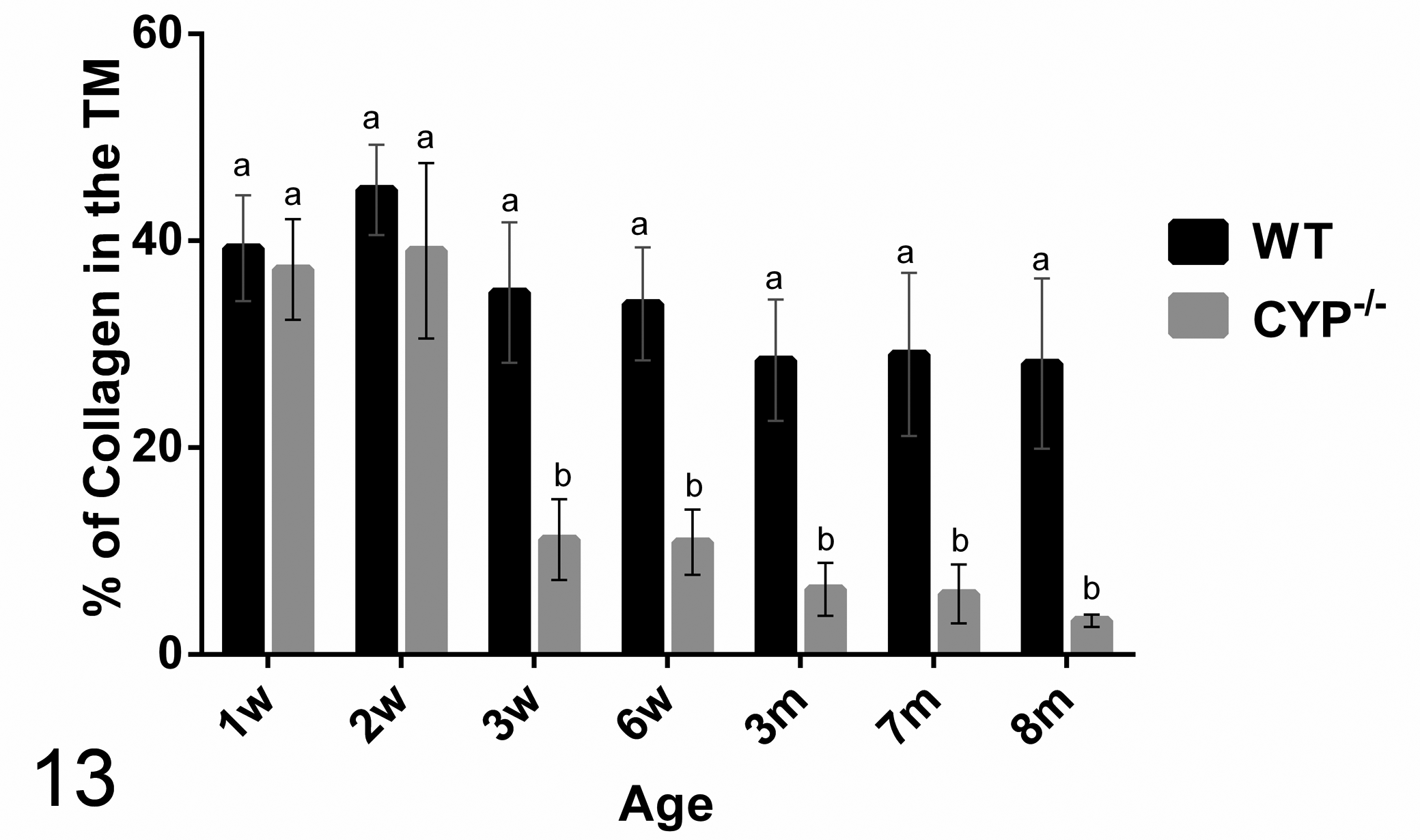
Morphometric quantification of the relative amounts of collagen in the trabecular meshwork (TM) of Cyp1b1–/– mice. Three-week-old Cyp1b1–/– mice presented significantly less collagen in the trabecular meshwork than did 3-week-old wild-type mice and younger wild-type and Cyp1b1–/– mice. TM collagen loss progressed with age. Means with different letters are significantly different (n = 42 [3/group/age], mean ± SD, Student t test, P < .0005). m, month; w, week; WT, wild type.
The semiquantitative analysis of the trabecular cell and TM collagen lesions presented a similar spatial distribution pattern between the ages. Trabecular cell and collagen median scores of Cyp1b1–/– were significantly (P < .005) more severe than respective controls in all ages (Figs. 14, 15). There was a statistically significant increase in the median collagen score between 2- and 3-week-old Cyp1b1–/– animals and 6-week-old and 3-month-old Cyp1b1–/– animals. A statistically significant increase in the median trabecular cell score between 2- and 3-week-old Cyp1b1–/– animals was also observed.
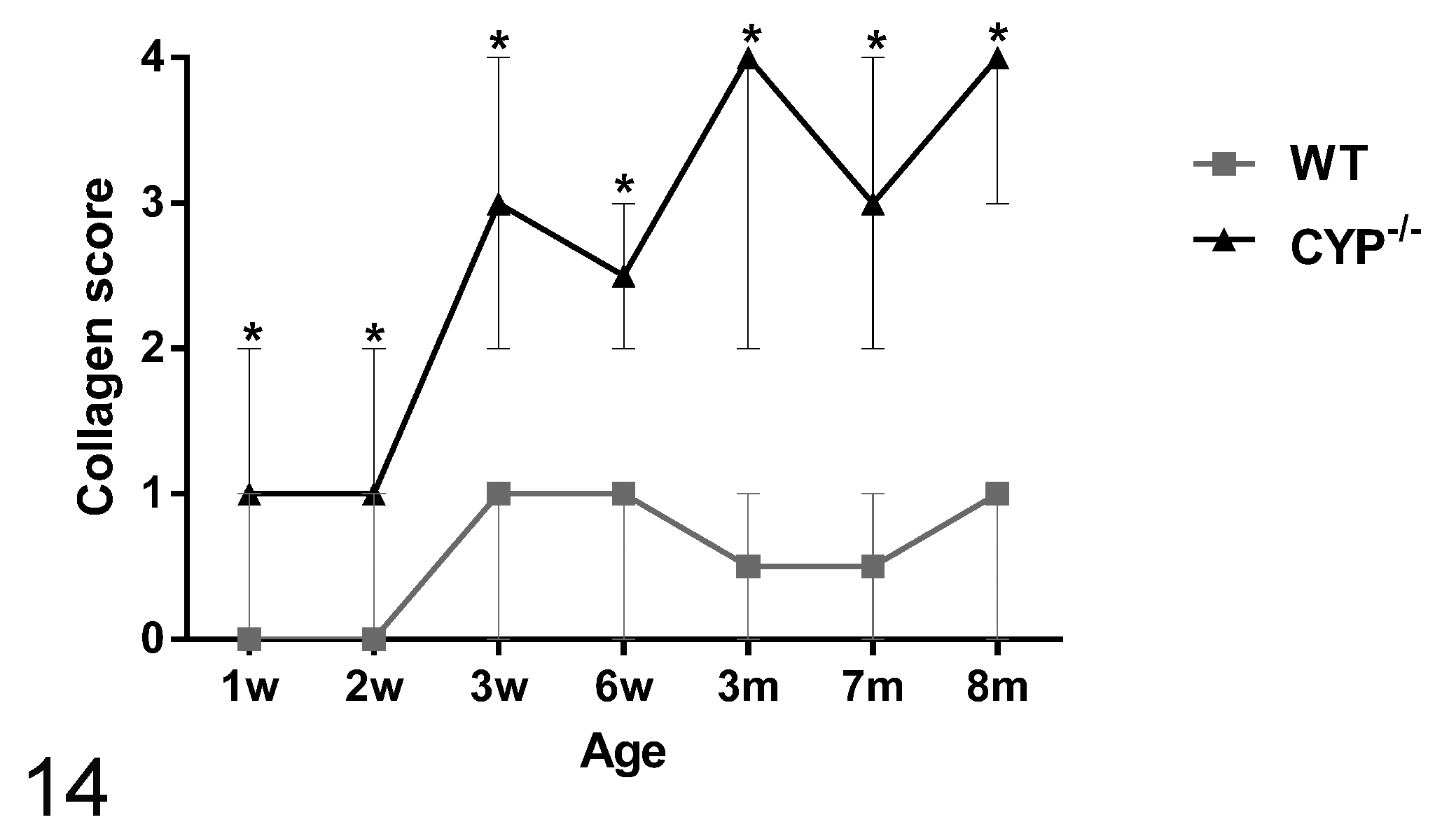
Semiquantitative scoring analysis of the distribution of the collagen lesions in the trabecular meshwork of Cyp1b1–/– mice. Cyp1b1–/– mice presented significantly higher median scores than did wild-type mice at all ages (*P < .005). There was a significant increase in median score between 2- and 3-week-old and 6-week-old and 3-month-old Cyp1b1–/– mice (*P ≤ .005) (n = 42 [3/group/age], Wilcoxon–Mann-Whitney, median ± range). m, month; w, week; WT, wild type.
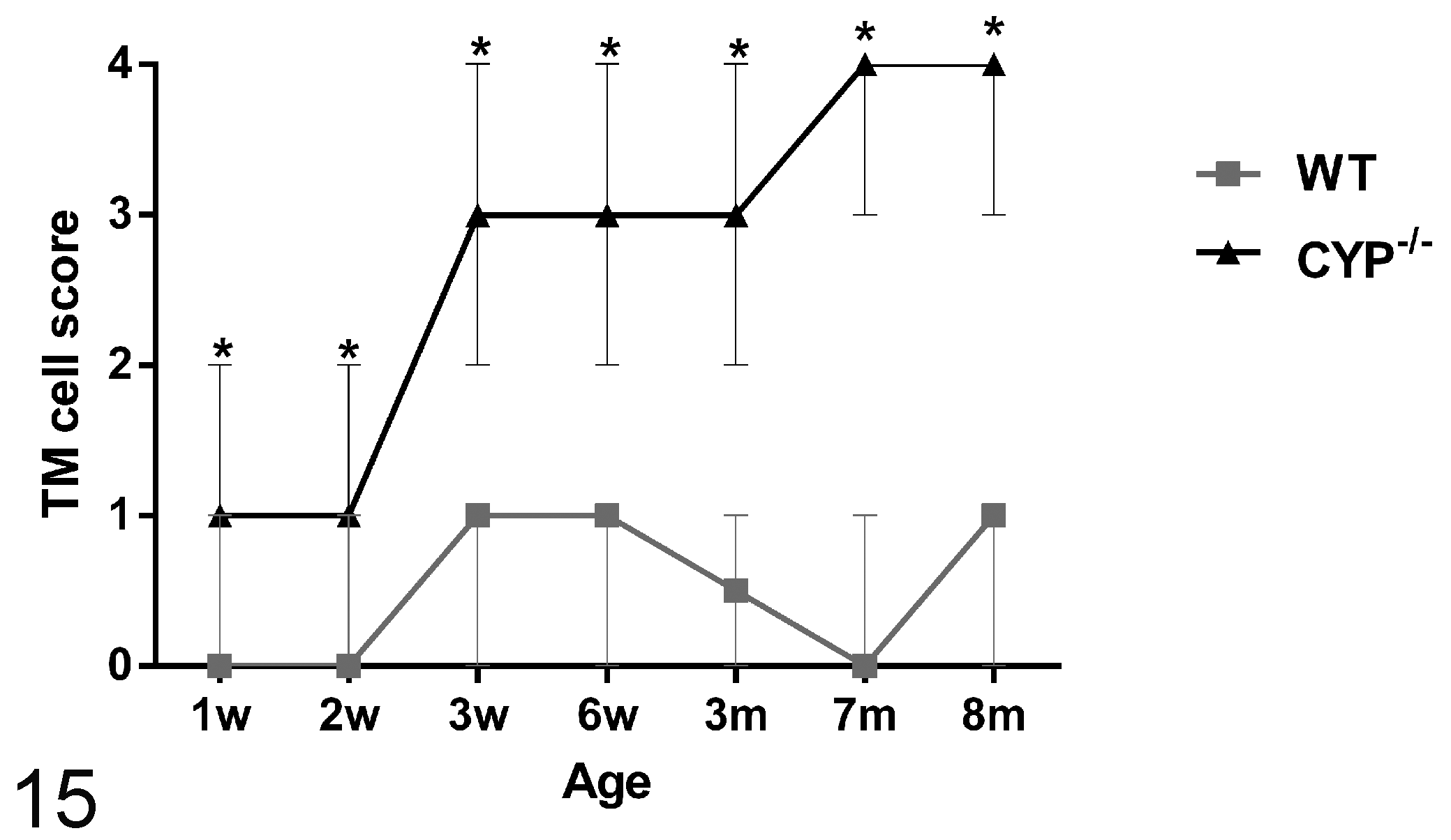
Semiquantitative scoring analysis of the trabecular cell ultrastructural morphology of Cyp1b1–/– mice. Cyp1b1–/– mice presented significantly higher median scores than did wild-type mice at all ages (*P < .005). There was a significant increase in median score between 2- and 3-week-old Cyp1b1–/– animals (P ≤ .005) (n = 42 [3/group/age], Wilcoxon–Mann-Whitney, median ± range). m, month; TM, trabecular meshwork; w, week; WT, wild type.
Discussion
The Cyp1b1–/– mice presented progressive ultrastructural abnormalities in the trabecular meshwork starting at 3 weeks of age that ultimately led to architectural collapse of the tissue. These developmental abnormalities resemble those reported in human patients with primary congenital glaucoma. 1,11
Libby et al 8 described severe iridocorneal angle abnormalities in albino 129X1/SvJ (tyrosinase-deficient) Cyp1b1–/– mice and postulated that the lesions are associated with the lack of tyrosinase production in the trabecular tissues. The albino animals presented absence of Schlemm’s canal and trabecular meshwork and areas of anterior synechia of the iris. The same authors reported that the lesions on Cyp1b1–/– pigmented (129X1/SvJ X B6) mice were less severe, with animals at 13 months of age presenting an open ciliary cleft, attenuated endothelium of the Schlemm’s canal, and reduction of the TM to a singular trabecular beam. Our data present similar findings on even younger animals. The presence of severe atrophy and collapse of the TM into a single trabecular beam was observed in 8-month-old animals (Figs. 10, 11), and based on the similar appearance of the lesion described by Libby et al in mice at 13 months of age, it appears to represent a final stage of TM atrophy.
The quantitative assessment of the extracellular matrix and trabecular endothelial cell morphology demonstrated that the loss and disruption of the collagen fibers and degeneration of TM cells were increased with time. The severity and temporal distribution of the lesions also correlated with the postnatal development period of the TM in mice. At birth, the mouse TM still presents a primitive appearance, only reaching full maturity at P35 to P42. The major developmental changes on the TM occur by P18, with subsequent maturation primarily involving final enlargement of spaces in the posterior TM. 14 Our data showed that the major losses in TM collagen and most significant increases in severity of the TM endothelial cells and collagen morphology score occurred between 2 and 3 weeks of age, a period roughly analogous to the final stages of TM development in the mouse (P10–P18).
These finding along with the near-normal morphology of the trabecular meshwork of 1- and 2-week-old Cyp1b1–/– mice suggest that the lesions observed are more likely a failure of ECM remodeling than a failure of ECM deposition in the TM. The molecular mechanisms responsible for normal or abnormal development of the iridocorneal angle are not well defined. 14
The embryological development of the iridocorneal angle starts with cells of the periocular mesenchyme migrating into the developing eye to form the primitive TM. 10 As TM development proceeds, the tissue differentiates, organizes, and develops channels to produce the mature meshwork with its trabecular beams separated by intertrabecular spaces through which the aqueous percolates. 5 Since there were no morphological differences between 1- and 2-week-old Cyp1b1–/– mice and controls, a failure of cell migration during embryogenesis as a cause of the lesions is unlikely.
The trabecular meshwork forms most of the resistance to aqueous humor outflow needed for maintenance of a pressure gradient between intraocular and venous pressure. 9 The aqueous outflow facility through the drainage structures of the iridocorneal angle is dependent, among others things, on the normal spatial arrangement of the TM tissue. On this regard, the ECM of the trabecular beams is of special importance, providing tensile strength and tissue flexibility to the TM. 2,4 The maintenance of these mechanical properties relies on the composition of the trabecular ECM, where type I and III collagen fibers provide structural support in tension, and elastin fibers enable a mechanism for reversible deformation in response to cyclic changes in the IOP. 7 The ECM changes present on the TM of Cyp1b1–/– mice reported here can alter the mechanical properties of the tissue and affect its ability to respond to injury. The disruption in collagen and elastic fibers can decrease tissue elasticity, alter the shape and cytoskeleton organization of TM endothelial cells due to lack of support, and affect the proper expansion of the trabecular spaces, leading to compromised aqueous humor outflow and increase in IOP.
These findings complement previous data published by our group that demonstrated that the Cyp1b1–/– mice presented elevated IOPs, increased oxidative stress, and trabecular meshwork endothelial cell abnormalities. Cyp1b1 modulates the expression of periostin, an extracellular matrix protein essential to collagen fibrillogenesis, by suppression of oxidative stress. 23 As evidenced by our results, the suppression of Cyp1b1 activity, and consequentially periostin expression, causes marked ultrastructural abnormalities on the ECM of the TM. Since the architectural organization of the TM is essential to maintain normal aqueous outflow, we believe these abnormalities are responsible for the increase in IOP in these animals.
In summary, our results demonstrated progressive ultrastructural ECM abnormalities on the TM of Cyp1b1–/– mice that are likely associated with impairment of aqueous outflow and initiation of glaucoma. CYP1B1 mutations are associated with multiple forms of human glaucoma. 21 The Cyp1b1–/– mice present morphologic lesions similar to human cases of primary congenital glaucoma and can be a valuable tool in understanding the mechanisms by which CYP1B1 mutations affect the TM function and development, leading to possible preventive approaches for glaucoma patients with such a mutation.
Footnotes
Acknowledgments
We thank Ben August and Randall Massey for their technical assistance with electron microscopy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants EY018179, EY016995, P30-EY016665, and P30 CA014520 UW Paul P. Carbone Cancer Center Support Grant from the National Institutes of Health and an unrestricted departmental award from Research to Prevent Blindness. NS is a recipient of a Research Award from the American Diabetes Association, 1-10-BS-160. NS and CMS are supported by the Retina Research Foundation.
