Abstract
The formation of biofilms on surgical implants is thought to play a major role in chronic infection and wound-healing disorders and has been rarely described in veterinary medicine. Due to poor and unreliable results from bacterial culturing, histology may be an economic tool for the detection of biofilms. In this study, the prevalence of biofilms on surgical suture materials and swabs with chronic wound-healing complications in dogs, cats, and horses was assessed by histologic examination using hematoxylin and eosin, Gram, and Giemsa stains, as well as periodic acid–Schiff reaction. Of the 91 tissue samples with intralesional suture material or swab residues associated with inflammation, only 2 contained bacterial colonies arranged in an extracellular polymeric matrix consistent with a biofilm. The results of this study suggest that biofilms on suture material may occur in veterinary medicine.
Bacteria may grow and cause wound infections either in the commonly observed planktonic form (defined as nonsessile individual rapidly multiplying and dispersing bacteria) or in biofilms. 1 Biofilms differ from the planktonic bacterial growth by 3 criteria. First, biofilms are bacterial colonies with obligatory adherence to each other and may reach a thickness up to 1.000 μm. 10 Second, these colonies are attached to an interface between 2 substances, typically a solid phase and a liquid or soluble phase. Third, a biofilm is composed of approximately 20% to 25% viable bacteria and 70% to 75% of extracellular polymeric matrix (EPM), which consists mainly of water, polysaccharides, and proteins. 1 Unequivocal identification of a biofilm thus typically requires histologic analysis to confirm the fulfillment of these criteria. The arrangement of bacteria in a biofilm and, more specifically, the synthesis of an EPM is commonly regarded as a stress response and is thought to serve as protection against adverse environmental conditions, including antibiotics, thus making them a major cause of nosocomial infections in humans. 3 In addition, biofilm-associated infections are often identified after temporary or long-term implantation of medical devices, including suture material. 4 However, the incidence and relevance of biofilms on implanted surgical devices, including suture material and surgical implants, have rarely been analyzed for any nonhuman species.8,9,13 The aim of our retrospective study was therefore to assess the prevalence of biofilms in cases of chronic inflammation surrounding suture material or swabs taken from wounds of dogs, cats, and horses and to correlate their presence with the type of foreign material as well as the intensity and morphology of the surrounding inflammation.
Ninety-one tissue samples from dogs, cats, and horses that contained inflammatory lesions associated with suture segments or swabs were selected from the institute’s archive of paraffin-embedded biopsies by a database search using the search terms suture material and inflammation. Of these, 68 tissue samples originated from dogs, 15 from cats, and 8 from horses. Fifty-three tissue samples were collected from skin, mucosa, or mammary gland; 3 from skeletal muscle; 7 from the intestine; and 28 tissue samples from the urogenital tract. Four serial paraffin sections of each sample were hematoxylin and eosin (HE), Gram, and Giemsa stained, and 1 section per sample was used for a periodic acid–Schiff (PAS) reaction according to standard protocols.
Identification of a biofilm was based on the fulfillment of the following 3 criteria modified by Parsek and Singh. 7 First, HE staining was used to identify an interface between the surgical material and the biologic tissue as described before. Second, a PAS reaction was used to identify deposits consistent with extracellular polymeric matrix (EPM) at the interface between the surgical material and the biologic tissue. Third, Giemsa and Gram stains were used to identify nucleic acid or Gram-positive bacterial organisms in the EPM.
In 79 of 91 cases, polyfilic suture was identified intralesionally. Monofilic suture was detected in only 2 feline tissue samples. Residues of cotton swabs were present in 8 canine tissue samples and 2 equine tissue samples. Of these, only 2 cases had intralesional PAS-positive EPM, which was arranged around and between individual fibers of the polyfilic suture (Fig. 1). Both PAS-positive cases also contained Gram-positive and Giemsa-positive bacteria in a laminar formation closely attached to the suture material and the EPM (Fig. 1). Only one additional case also had Gram- and Giemsa-positive bacteria directly associated with the foreign body structure but no PAS-positive EPM and was thus not considered a biofilm. Both biofilms were found in canine tissue samples and contained exclusively cocci. One case was a severe chronic-active suppurative and granulomatous inflammation following surgical skin incision adapted with polyfilic suture. The second showed a severe chronic-active lymphoplasmacytic and granulomatous inflammation at the stump of an ovariohysterectomized uterus also sutured with polyfilic material.
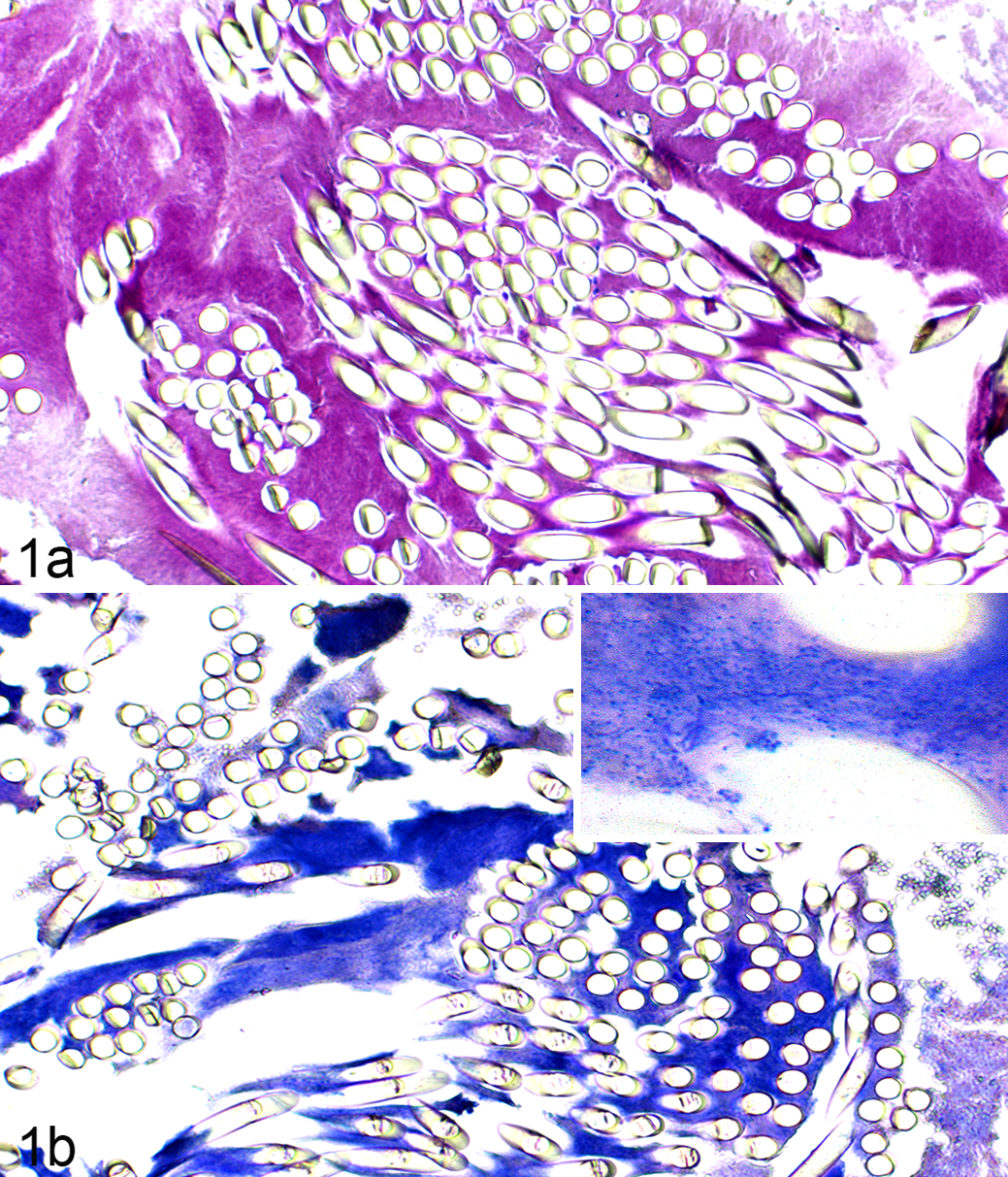
Dog No. 68. Polyfilic suture segments in a chronic-active inflammation associated with biofilm formation (ovariohysterectomized uterus stump). (a) Periodic acid–Schiff (PAS)–positive extracellular polymeric matrix of a biofilm associated with the suture material, PAS reaction. (b) Gram-positive cocci arranged around and between the fibers of the polyfilic suture material. Inset: Higher magnification of Gram-positive cocci. Gram stain.
The low number of histologically detectable biofilms in this study stands in contrast to previous reports on the occurrence of biofilms in human cases of postoperative surgical site infection (SSI) in which between 66% and 100% of human patients with suture segment–associated SSI had culturable bacteria capable of biofilm formation. 2 However, methodologic approaches for the detection of biofilms were different, and no different or definitive conclusions can be drawn. So far, it thus remains questionable whether our histologic approach is sufficiently sensitive to allow for general conclusions.
In general, the identification of biofilms is a diagnostic challenge due to several reasons. Most important, in contrast to their planktonic counterparts, bacteria arranged in biofilms are considered poorly culturable. 6 Furthermore, detection of specific bacterial gene sequences associated with the bacterial strain’s ability to grow in biofilms is associated with 2 major problems. First, the genes associated with biofilm growth vary tremendously between bacterial strains and species, and it is therefore necessary to initially identify the bacterial species involved in the putative biofilm and then to specifically detect the relevant genetic markers for these species. Second, it has been shown that biofilm-associated genes may also be present in planktonic bacteria that may have the potential to form a biofilm but are currently not organized as such. 12 Polymerase chain reaction (PCR) assays are therefore highly sensitive to detect specific bacterial DNA but generally unable to confirm the presence of a biofilm. An additional morphologic diagnosis with localization of bacteria attached to a surface and arranged in an EMP is thus currently the only accepted method to diagnose biofilms in situ. The identification of biofilms by histologic staining methods has been described, mostly employing a combination of HE and Gram stains. 11 Sufficient sensitivity of the histologic approach to biofilm identification has been confirmed by comparing HE staining with the highly sensitive method of fluorescent in situ hybridization (FISH) for bacterial gene detection, which has revealed a similar sensitivity for both methods. 5
Both biofilms detected in our study were located between single threads of multifilic suture. Thus, polyfilic suture might be a predisposing factor for the formation of biofilms, although the small number of cases in this study does not allow for a reliable evaluation. Similarly, the low number of cases identified in this study also impaired the analysis of possible correlations between the presence of biofilms on surgical implants and the extent, duration, and character of the induced inflammation.
The bacterial shapes, sites, and Gram-positive stainings were suggestive of staphylococci or streptococci as the predominant bacteria in the 2 biofilms. The retrospective character of this study, which employed formalin-fixed and paraffin-embedded tissues, however, excluded a systematic microbiological identification of the associated bacteria. Further studies are needed to better characterize the bacteria involved in veterinary medicine.
Footnotes
Acknowledgements
We thank Petra Nehrig, Alexandra Harder, Anke Kmetsch, and Gabriele Hahn for excellent technical assistance.
Authors’ Note
The manuscript has been prepared in the Uniform Requirements format. The present study is part of the Dr. med. vet.-thesis of L.K.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
