Abstract
This report describes atypical cases of yersiniosis in squirrel monkeys in which aberrant forms of Yersinia pseudotuberculosis were seen. There were 2 outbreaks due to yersiniosis in squirrel monkeys in Japan. The monkeys had systemic necrotizing and hemorrhagic lesions with Gram-negative rod-shaped bacilli and microthromboembolism in the kidneys. Some lesions contained filaments, globular bodies, and other pleomorphic forms of bacteria. All forms were usually seen in the same lesions, and those with pleomorphic morphology appeared to be an intermediate form between the rod-shaped bacteria and the filaments or globular bodies. In addition, they had strong immunolabeling for Y. pseudotuberculosis, as did the rod-shaped bacteria. Therefore, the globular bodies, filaments, and others are strongly suspected to be shape-changed bacilli of Y. pseudotuberculosis. These morphologically altered bacteria could cause errors in diagnosis since they resemble fungi or protozoa, and special staining techniques, including immunohistochemistry, can be helpful in establishing the correct diagnosis.
In June and July 2003, 8 of 20 captive young squirrel monkeys (Saimiri sp) kept in a zoological park in Japan exhibited a moribund state or died suddenly. When the fourth monkey died, amoxicillin (AMPC) (40 mg/kg, once a day [SID]) was administered orally to the surviving monkeys for a week. However, following the initiation of antibiotic therapy, 4 more monkeys (animal Nos. 1–4) died 1, 7, 13, and 17 days later, respectively. In addition, in March 2010, 3 of 20 monkeys aged 2 years (Nos. 5–7) kept in the same cage became moribund with loss of appetite; the monkeys received fluid replacement and were treated with intramuscular cefazolin (CEZ) (40 mg/kg, SID) administration for 3 days in Nos. 5 and 6, and for 10 days in No. 7, but died within a few days. In November 2010, a squirrel monkey (No. 8) aged 4 years in the same cage was found moribund and administered CEZ (40 mg/kg, SID) for a day but died thereafter. These monkeys originated from breeding in captivity in the zoological park.
Complete necropsies were performed on the 8 monkeys (Nos. 1–8). Histologic sections were prepared and stained with hematoxylin-eosin (HE), Gram stain, periodic acid–Schiff (PAS) reaction, Grocott’s methenamine silver (GMS) stain, Fungiflora Y fluorescent stain (Trust Medical Co. Ltd., Hyogo, Japan), and phosphotungstic acid–hematoxylin (PTAH) stain according to routine methods. Immunohistochemistry was performed using a commercial rabbit anti–Y. pseudotuberculosis serum set (O1, O2, O3, O4, O5, and O6) (Denka-Seiken Co., Tokyo, Japan). Special staining and immunohistochemistry were performed on all tissue from the 8 monkeys (Nos. 1–8). For transmission electron microscopy (TEM), the formalin-fixed hepatic and splenic tissues from 2 monkeys (Nos. 1 and 2) were routinely processed. Bacterial examinations of the liver and spleen were performed for 3 monkeys (Nos. 3, 4, and 6) as described by Iwata et al. 5 Briefly, the samples collected from the monkeys that died were homogenized and plated on Yersinia selective irgasan-novobiocin (IN) agar plates. Colonies morphologically similar to those of Yersinia spp were tested for biochemical examination, virulence-associated autoagglutination test, and serotyping.
At necropsy, all monkeys had moderate bloody ascites and a small amount of pleural effusion. Mucosal petechial hemorrhages were scattered in the small intestine. Peyer patches were swollen with hyperemia, and solitary lymphoid follicles in the cecum were clear, accompanied by hemorrhage. There was significant enlargement of the mesenteric lymph nodes and spleen with a large number of fine white nodules. Liver was discolored, and there were a small number of scattered fine white nodules. There were pulmonary edema and petechiae. There were also petechial or ecchymotic hemorrhages in the kidney in Nos. 1, 2, and 6 to 8. Subepicardial petechial to ecchymotic hemorrhages were seen in the heart in Nos. 1, 2, 6, and 8.
Microscopically, nodules seen in the liver, spleen, and mesenteric lymph nodes were focal necrosis accompanied by the infiltration of neutrophils and macrophages with fibrin, containing bacterial colonies consisting of rod-shaped or pleomorphic bacilli. In the liver of all monkeys and the spleen of Nos. 1, 2, and 5 to 7, there were basophilic globular bodies associated with focal necrosis and rod-shaped bacilli (Fig. 1). The globular bodies were round to oval and 4 to 5 μm in diameter and sometimes showed bizarre forms and a fine granular structure. In addition, small numbers of basophilic filaments were seen in the same lesions (Fig. 1). These rod-shaped bacteria, globular bodies, and filaments were usually seen in the same lesions, and those with pleomorphic morphology appeared to be an intermediate form between the rod-shaped bacteria and the filaments or globular bodies (Fig. 1). In the small intestine, colon, and cecum, mild to moderate infiltration of neutrophils, macrophages, and lymphocytes; severe congestion; and focal necrosis containing numerous bacterial colonies consisting of rod-shaped or pleomorphic bacilli were seen. In particular, lesions in both small and large intestines near the Peyer patches and solitary lymphatic nodules were severe, accompanied by ulceration and erosion of the mucosa, hemorrhage, and the accumulation of nuclear debris. In the intestine of Nos. 5 and 7, there were numerous basophilic elongated filaments associated with necrosis and desquamated epithelia with colonies of rod-shaped bacteria (Fig. 3). The lengths of these filaments were 5 to 40 μm (about 20 μm on average) and 1 to 2 μm in width. The filaments were randomly branched and sometimes partly swollen. In the kidney, bacterial colonies consisting of rod-shaped or mildly pleomorphic bacilli were seen in the glomerular loop and interstitial blood capillary of Nos. 1 to 4 and 6 to 8. Necrotic foci were scattered and micro-thromboemboli in the glomerular loop were seen in Nos. 1, 2, 7, and 8. In the heart, necrotic foci, including bacterial colonies consisting of rod-shaped or mildly pleomorphic bacilli, were seen in Nos. 1, 2, and 6 to 8, and hemorrhagic foci were scattered in Nos. 1, 2, 6, and 8. Other findings included bacterial colonies consisting of rod-shaped or mildly pleomorphic bacilli in the lung, pulmonary edema with hemorrhage in all monkeys, severe adrenal hemorrhage and bacterial colonies in Nos. 6 and 8, and subcapsular hemorrhage and bacterial colonies in the testis in No. 6.
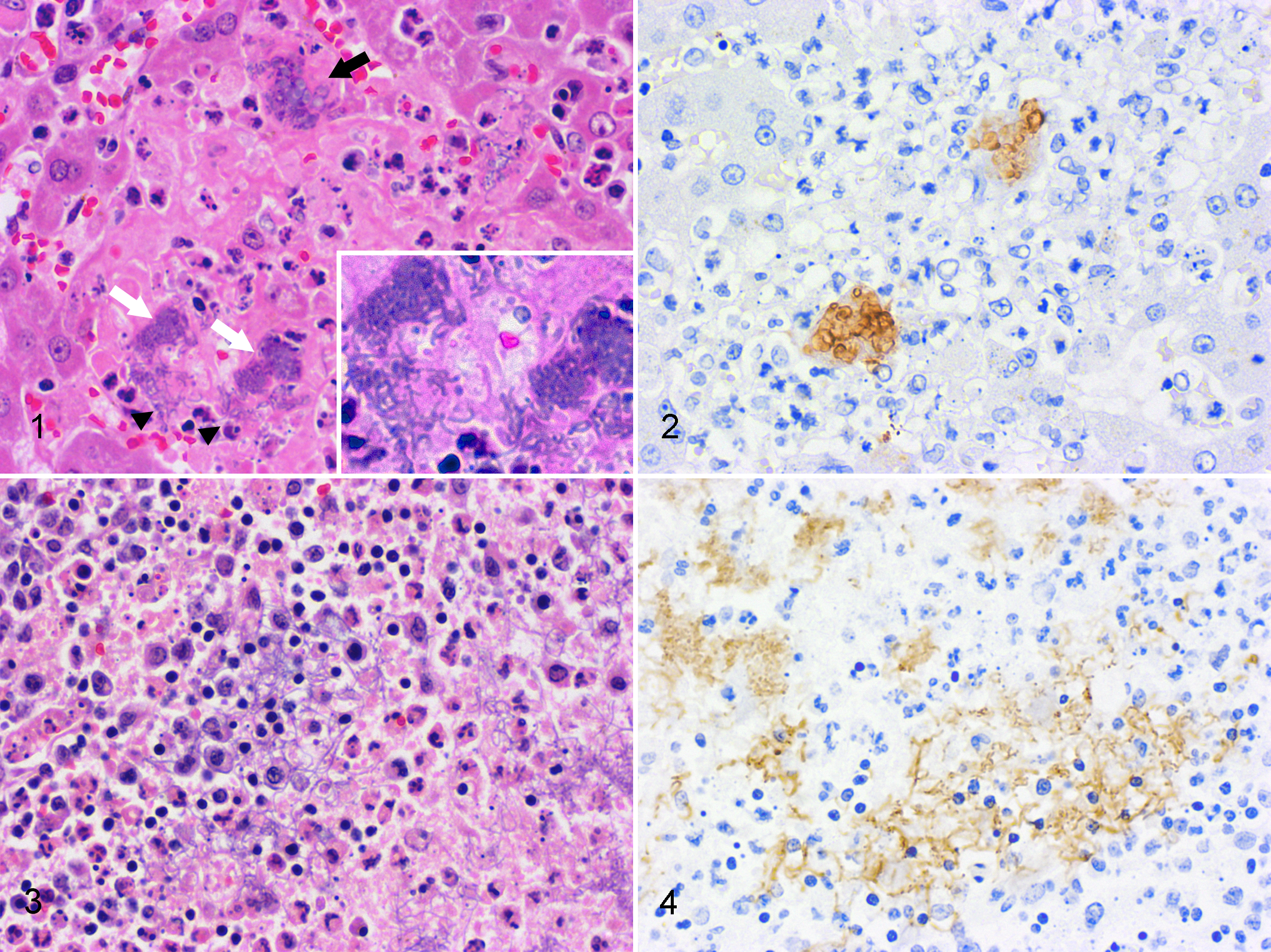
The rod-shaped bacteria, globular bodies, filaments, and other pleomorphic forms of bacteria were Gram negative; however, this was sometimes unclear in the globular bodies and filaments (Suppl. Table S1). The globular bodies and filaments were negative for GMS stain and Fungiflora Y fluorescent stain and rarely positive for PAS (Suppl. Table S1). The rod-shaped bacteria, globular bodies, filaments, and other pleomorphic forms of bacteria also had strong immunolabeling for Y. pseudotuberculosis O1 in Nos. 1 to 4, O6 in Nos. 5 to 7 (Figs. 2, 4), and O4 in No. 8.
Ultrastructurally, globular bodies in the spleen and liver were round or oval and a variety of sizes (about 2–5 μm in diameter), and there were relatively normal rod-shaped bacteria in the same lesions (Fig. 5). The rod-shaped bacteria had an outer membrane, periplasmic space, plasma membrane, and simple internal structure without a membrane-bound nucleus or organelles, endoplasmic reticulum, and mitochondria. They had ribosomes that appeared as small electron-dense particles and chromosomes as a nonstaining region in the bacterial cytoplasm (Fig. 6). Although globular bodies had a similar characteristic structure to rod-shaped bacteria, such as a membrane-bound structure, the cell wall was partly detached from the cytoplasmic membrane. In addition, fewer ribosomal granules were present in the globular bodies compared with the rod-shaped bacteria (Fig. 7).
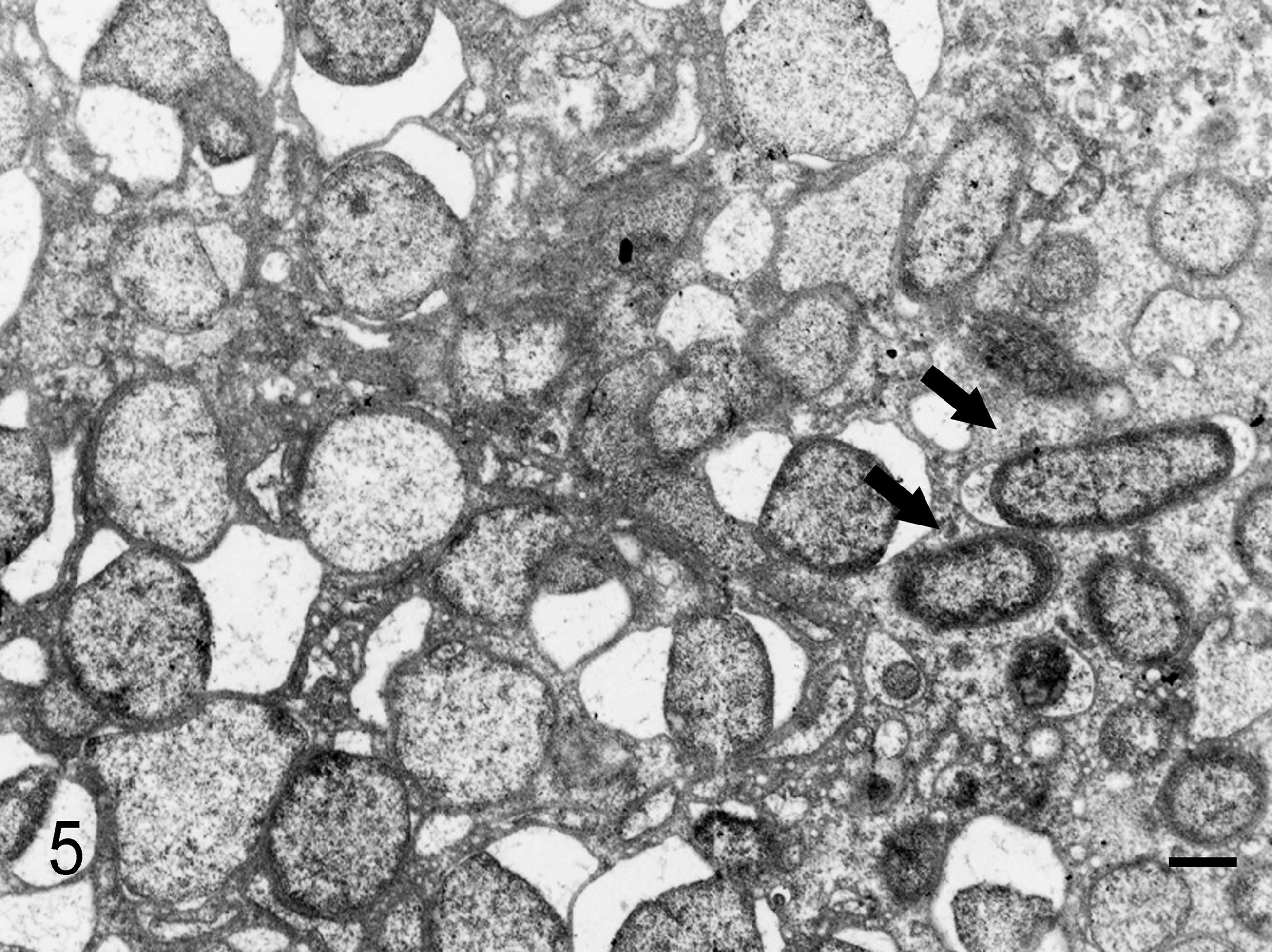
Liver; squirrel monkey No. 2. There are numerous globular bodies in the necrotic lesion with rod-shaped bacteria (arrows). The cell wall of globular bodies is partly detached from the cytoplasmic membrane. Transmission electron microscopy. Bar, 0.5 μm.
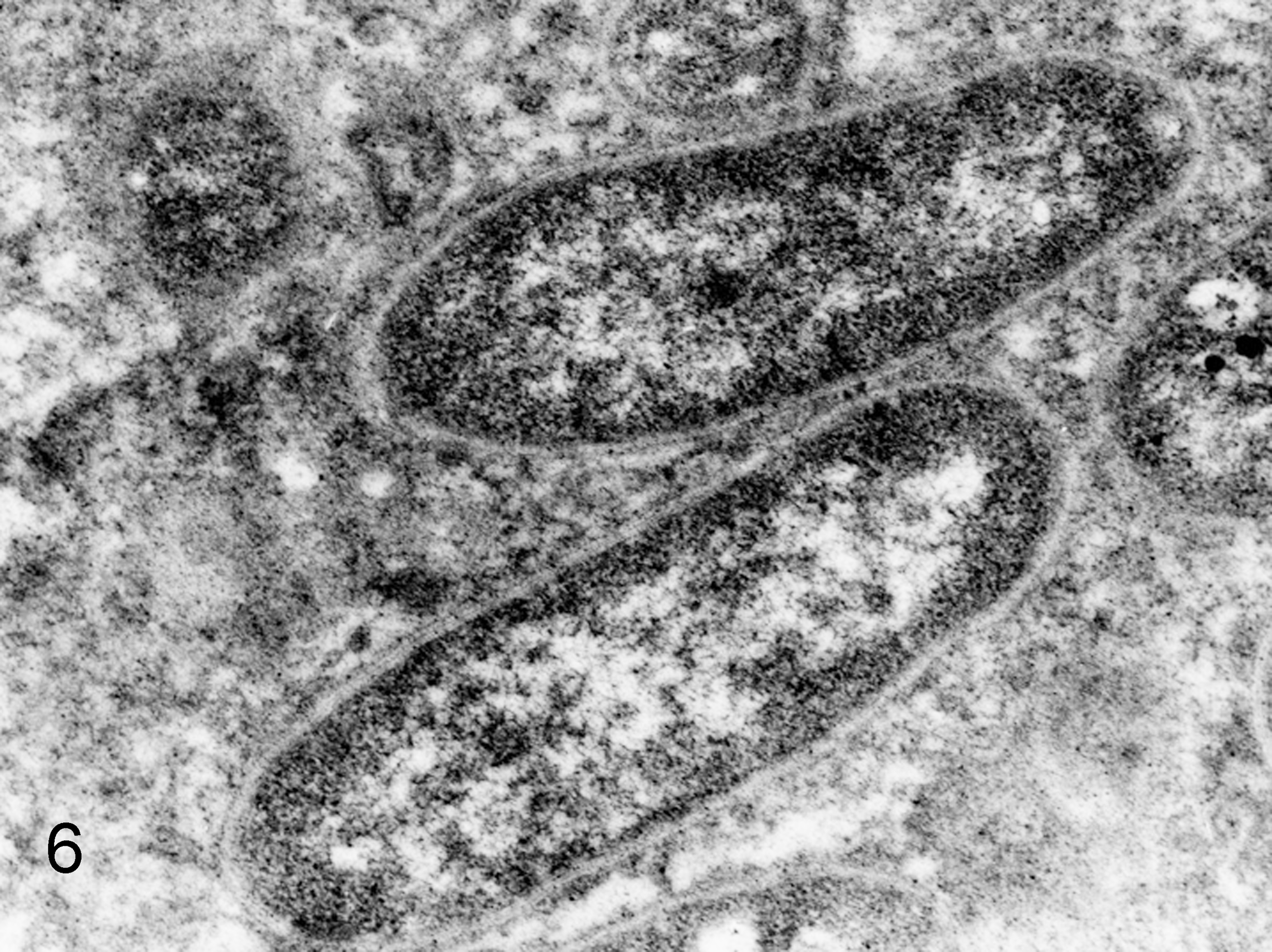
Liver; squirrel monkey No. 2. Higher magnification of rod-shaped bacteria with trilaminal cell wall and internal ribosomes that appeared as small electron-dense particles. Transmission electron microscopy.
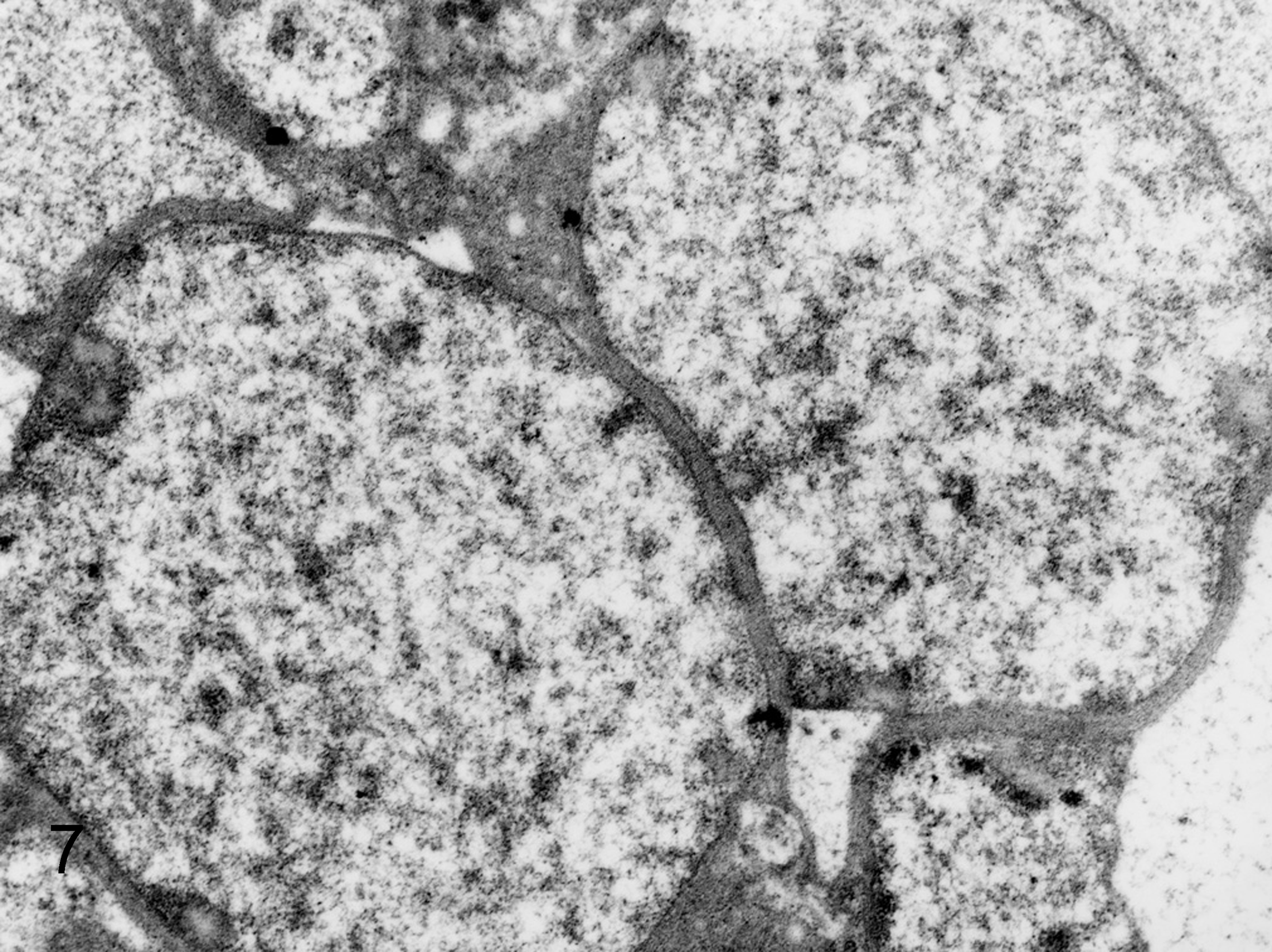
Liver; squirrel monkey No. 2. Higher magnification of globular bodies with decreased ribosomes compared with rod-shaped bacteria. Transmission electron microscopy.
Y. pseudotuberculosis serotypes 1b and 6 were isolated from the liver and spleen of Nos. 3 and 4, and No. 6, respectively, in pure culture. All isolates showed a positive reaction for virulence-associated properties such as autoagglutination at 37°C.
Discussion
The necrotic lesions with Gram-negative bacilli seen in the spleen, liver, mesenteric lymph nodes, and bowels of the 8 monkeys were consistent with yersiniosis due to Y. pseudotuberculosis, 1 and the diagnosis was confirmed by immunohistochemistry and bacterial isolation. Although the globular bodies and filaments are not commonly seen in cases of yersiniosis, the characteristics of PAS, GMS and Fungiflora Y fluorescent staining indicated that these were not fungi or protozoan organisms. In addition, they had strong immunolabeling for Y. pseudotuberculosis, as did the rod-shaped bacteria. These pathogens were usually seen in the same lesions as rod-shaped bacteria, and there appeared to be transition from rod-shaped bacteria, to those with pleomorphic morphology, to spheroplasts or filaments. Therefore, the globular bodies and filaments were considered shape-changed forms of Y. pseudotuberculosis. Generally, Yersinia bacteria are observable as colonies of small bacilli or pleomorphic coccobacilli in necrotic lesions. 1 Bronson et al 2 reported that Yersinia bacteria from old cultures can become bipolar, pleomorphic, and filamentous and do not form a capsule, and although pleomorphic forms have been observed in tissues, detailed descriptions of the microscopic morphology were lacking. 1
Shape-changed bacilli such as spheroplasts and filaments often have been observed in clinical specimens from human patients with bacterial infection treated with antibiotics. 6,7 It is known that β-lactam antibiotics change the integrity of the bacterial cell envelope by binding to penicillin-binding proteins (PBPs). 3 PBPs are enzymes anchored in the cytoplasmic membranes of bacteria and are associated with the synthesis of peptidoglycan. PBPs, particularly PBP1, PBP2, and PBP3, have significant biological functions, including determination of cell shape, phage resistance, induction of capsule synthesis, and regulation of autolysis. 4,8 Although morphological change of the bacteria might be induced by the host’s inflammatory response, we have never experienced such cases despite diagnosing this disease in more than 400 monkeys (about 250 of them squirrel monkeys) in the past. For these reasons, the shape-changed bacilli may have been induced by the β-lactam antibiotics AMPC and CEZ. The stainability of these bacterial forms with PAS and Gram stain might have been unstable because the β-lactam antibiotics inhibited the cross-linking of peptidoglycan.
In this report, we described cases of yersiniosis in squirrel monkeys with aberrant forms of Y. pseudotuberculosis as spheroplasts and filaments. This could lead to an erroneous diagnosis of fungal or protozoal infection. This report should raise awareness of aberrant forms of bacteria in atypical cases of yersiniosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Research on Emerging and Re-emerging Infectious Diseases from the Ministry of Health, Labor and Welfare of Japan (H24-Shinko-Ippan-006), and Grant-in-Aid for Scientific Research (B) (#23310169).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
