Abstract
In veterinary medicine, anemia without an appropriate compensatory hematopoietic response is termed
Exact definitions of
Nonregenerative anemia is recognized as a common entity in clinical veterinary medicine, occurring frequently as a complication of various types of nonhematologic disease and less often as a result of primary hematopathology. The objectives of this article are to review nonregenerative anemia in domestic animals, emphasizing mechanisms of disease, and to review some additional conditions associated with nonregenerative anemia in people. This review acknowledges some causes of nonregenerative anemia in the context of pancytopenia but is not intended to summarize anemia occurring as a result of more generalized impairment of hematopoiesis.
Laboratory Diagnosis and Classification of Anemia
Laboratory diagnosis of anemia is straightforward and often supported by other findings such as pallor, lethargy, or exercise intolerance. The automated hemogram, or complete blood count (CBC), is available through reference laboratories and in many veterinary practice settings worldwide, and the erythrogram component routinely includes the tests used to diagnose anemia (ie, erythrocyte concentration, hematocrit, and hemoglobin concentration). Erythrocyte concentration influences the other two variables, so all 3 are below reference limits in anemic individuals except in marginal cases. In situations where an automated hematocrit is not an option (eg, in field or practice settings with sparsely equipped laboratories, or if analyzing samples from nonmammalian species with nucleated erythrocytes), the packed cell volume assay may be used to determine the proportional volume of erythrocytes in the blood by centrifugation.
Enumeration of reticulocytes is used to assess the bone marrow response to anemia in most species. Reticulocyte counts are performed automatically in most modern laboratories, including many in-clinic veterinary laboratories, but may be performed manually based on microscopic examination of blood stained with a supravital stain such as new methylene blue. 179 In the absence of a reticulocyte count, regeneration can be assessed subjectively based on the degree of polychromasia in a conventionally stained blood smear. As with all laboratory tests, what is considered normal depends on many factors, including species, reference sample group, laboratory methodology, and statistical methods used to determine reference limits. 58
The main advantage of classifying anemia as regenerative or nonregenerative is that in most cases, it effectively identifies the cause of anemia as being either increased loss or destruction of erythrocytes (regenerative) or impaired production of erythrocytes (nonregenerative). Exceptions to this reticulocyte-based classification system exist. Equids release no or very few reticulocytes into circulation, even in the context of physiologically regenerative compensatory bone marrow erythroid hyperplasia.
98
In these species, assessment of the erythroid response is usually based on serial CBCs but may also be done by examining a bone marrow sample. Another exception is anemia that is “preregenerative”—that is, when the time required to translate the compensatory bone marrow response into peripheral reticulocytosis (usually 3 or 4 days) has yet to elapse. In addition, some pathologic states are characterized by nonregenerative anemia despite persistent bone marrow erythroid hyperplasia—a condition known as
Classification of Anemia According to Presence or Absence of Reticulocytosis.
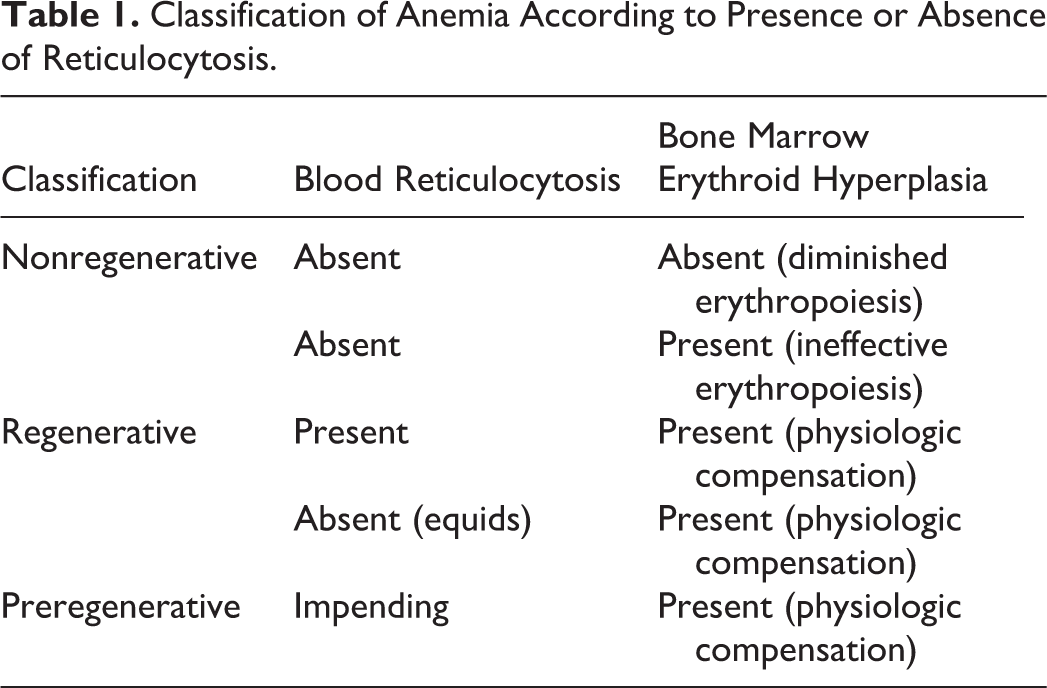
Anemia also may be categorized according to other criteria. A system originating in the 1930s that classifies anemia based on erythrocyte mean cell volume (MCV) and mean cell hemoglobin concentration (MCHC) is still in wide use. 171,192 MCV and MCHC indices are components of virtually every hemogram, and some patterns correlate with certain pathologic conditions. For example, patients with severe anemia and a robust regenerative response are often macrocytic (supranormal MCV) and hypochromic (subnormal MCHC), and those with advanced iron deficiency anemia are typically microcytic (subnormal MCV) and hypochromic. Macrocytic, hypochromic regenerative anemia occurs when the proportion of circulating reticulocytes, which are larger and have lower hemoglobin concentration than mature erythrocytes, is sufficiently high to cause MCV and MCHC to be outside the reference intervals. However, classifying anemia based on these mean values has inherent limitations and may be misleading. In fact, many if not most cases of regenerative anemia are normochromic and normocytic (ie, the same pattern that typifies nonregenerative anemia) because the magnitude of the reticulocytosis is not great enough to cause the MCV and MCHC to be outside of reference limits. 11,79 Moreover, prolonged storage of blood in routine EDTA anticoagulant tubes is associated with erythrocyte swelling, which can cause artifactual macrocytosis and hypochromasia even in the absence of reticulocytosis. 62
Normal Erythropoiesis
Erythropoiesis, the formation and development of erythrocytes, is a complex, highly regulated process that, in healthy adult animals, takes place primarily in the bone marrow. Mature erythrocytes, like other blood cells, arise from pluripotent hematopoietic stem cells and differentiate and proliferate under the influence of various growth factors and cytokines. Readers are referred to other sources for more detailed information about normal erythropoiesis. 76,86
Basic Mechanisms of Nonregenerative Anemia
Nonregenerative anemia occurs as a result of decreased or ineffective erythropoiesis. Diminished erythropoiesis is associated with various specific mechanisms—for example, impaired hormonal stimulation, diminished availability of nutrients, and toxic insult to the bone marrow—and is a recognized sequela of a wide array of primary diseases. Ineffective erythropoiesis, a relatively uncommon condition, is characterized by anemia despite normal to increased erythropoietic activity—a situation recognized to occur in veterinary species in certain types of neoplasia and immune-mediated disease.
Nonregenerative Anemia Associated With Various Disease Processes
Inflammation
Inflammation causes anemia via multiple downstream effects of inflammatory cytokines. Anemia of inflammation is typically mild to moderate and accompanied by hypoferremia and normal to increased tissue iron stores.
65
The condition has been recognized for many decades and often has been referred to as “anemia of chronic disease”; however, in recent years, as the underlying molecular pathways have been elucidated, usage of the term
A hallmark feature of anemia of inflammation is decreased concentration of plasma iron, which reduces its availability to developing erythroid cells for hemoglobin synthesis. This condition often occurs in conjunction with increased storage of iron in the tissues, consistent with sequestration and functional deficiency of iron. On the molecular level, anemia of inflammation involves the hormone-ligand interaction of hepcidin, a peptide produced mainly in the liver, and its receptor, ferroportin, a membrane protein that enables efflux of iron stored within macrophages and other cells, or absorbed into enterocytes from the diet, into the plasma. In addition to being a hormone, hepcidin is a type II acute-phase protein, and its expression increases dramatically in response to inflammatory cytokines, especially interleukin (IL)–6. 121 Hepcidin-ferroportin binding causes ferroportin to be internalized and degraded, thus trapping iron within cells. 120 This interaction effectively restricts plasma iron by sequestering iron stored within the reticuloendothelial system and by limiting absorption of dietary iron. Anemia of inflammation is suspected to have evolved as a defense mechanism to limit iron availability to pathogenic microbes. 89
Inflammation has been linked to other effects on iron metabolism that may contribute to development of anemia. For example, treatment of murine macrophages with interferon (IFN)–γ and lipopolysaccharide (LPS) caused a nitric oxide (NO)–mediated increase in ferritin synthesis 141 ; rats treated with repeated injection of Freund’s complete adjuvant had decreased transferrin receptor (TfR) expression in bone marrow cells and increased ferritin in serum (changes that were also NO-mediated), in addition to anemia and hypoferremia 177 ; treatment of human erythroleukemic cells and reticulocytes with α1-antitrypsin interfered with transferrin binding to the TfR and inhibited growth and proliferation of early progenitor cells 68 ; and treatment of murine reticulocytes with a NO donor caused inhibition of heme synthesis and iron uptake via the transferrin-TfR pathway. 110
In addition to altering iron metabolism, inflammatory cytokines are thought to contribute to anemia by other direct effects on developing erythroid cells and by blunting erythropoietin (EPO) production. For example, treatment of human hematopoietic cell lines with tumor necrosis factor (TNF)–α caused rapid, irreversible inhibition of erythroid progenitors, 151 and subsequent studies have shown TNF-α to inhibit EPO-mediated erythroid differentiation and decrease hemoglobin synthesis. 26 Other studies have demonstrated inhibition of EPO production by IL-1, 52 TNF-α, 52 IFN-γ, 175 and, to a lesser degree, transforming growth factor (TGF)–β. 52 LPS and IL-1 have been shown to suppress renal EPO gene expression in rats during hypoxia. 56 Also, IL-6 and TNF-α are thought to contribute to EPO resistance. 66
Decreased erythrocyte survival time also likely contributes to anemia of inflammation. Erythrocytes of healthy humans have been shown to have a decreased life span when transfused to patients with anemia of inflammation; conversely, erythrocytes of patients with anemia of inflammation had a normal life span when transfused to healthy individuals. 30 Significantly reduced erythrocyte survival time has also been demonstrated in dogs 146 and cats 185 with turpentine-induced abscesses, rabbits with hyperthermia, 84 and rats given TNF, IL-1, or endotoxin. 111 Proposed mechanisms for inflammation-associated decreased erythrocyte survival time include macrophage activation, which may stimulate premature removal of erythrocytes from the blood, 64 and premature clearance of erythrocytes with altered antigenicity or structure resulting from oxidative or other injury. 91
Immune-Mediated Disease
Selected immune-mediated causes of nonregenerative anemia that are of particular relevance to clinical veterinary medicine are considered in this section. Readers are referred to other reviews regarding the role of the immune system in development of aplastic pancytopenia, myelodysplasia, and hematopoietic disorders. 102,103,133
Immune-mediated hemolytic anemia
Immune-mediated hemolytic anemia (IMHA) occurs as a result of lysis of erythrocytes with surface-bound immunoglobulins. The condition is classified as primary (ie, idiopathic) or secondary (ie, occurring as a sequel to an identified underlying disease) and may involve targeted erythrocytes being phagocytosed by macrophages (extravascular hemolysis), lysed in the blood circulation (intravascular hemolysis), or both. Complement cascade activation, cell-mediated pathways, and anti-EPO antibodies may also contribute to the pathophysiology of IMHA in some cases. 53
Most cases of IMHA involve the peripheral destruction of mature erythrocytes in circulation—by extravascular hemolysis (macrophage phagocytosis, especially in the spleen), intravascular hemolysis, or both—and are associated with a strong regenerative response. However, nonregenerative forms of IMHA also occur. Nonregenerative IMHA has been described in dogs, 167 cats, 88 and cattle. 119 An inadequate or absent regenerative response in cases of IMHA may occur because of underlying disease that inhibits erythropoiesis or because of targeted destruction of developing erythroid cells. 167
Nonregenerative IMHA in veterinary species usually is diagnosed based on exclusion of other causes of anemia and on response to immunosuppressive therapy. Bone marrow findings in the erythroid lineage range from rare or absent erythroid precursors (as in PRCA), to the absence or decreased proportions of later stage cells, to marked generalized erythroid hyperplasia (signifying ineffective erythropoiesis); one study in dogs found erythroid hyperplasia in most patients with nonregenerative IMHA. 167 In theory, the stage at which developing erythrocytes express the antigen(s) being targeted immunologically will affect bone marrow findings: if the target antigen is expressed on early precursor cells, PRCA or erythroid hypoplasia is likely; if the target antigen is not expressed until the reticulocyte stage, then erythroid hyperplasia is likely. Moreover, various other bone marrow dyscrasias—including dysmyelopoiesis, myelonecrosis, myelofibrosis, inflammation, edema, hemorrhage, and hemophagocytic syndrome—have been found in dogs and cats with nonregenerative IMHA and marrow erythroid hyperplasia, suggesting additional mechanisms contributing to anemia. 181 Note that PRCA can also occur as a result of feline leukemia virus (FeLV) subgroup C infection in cats, probably because of infection of erythroid precursor cells (see the section on viral diseases), and is a component of Diamond Blackfan anemia (see the section on congenital disorders).
Hemophagocytic syndrome
Hemophagocytic syndrome is a condition characterized by hypercytokinemia, in which excessive stimulation and proliferation of nonneoplastic macrophages results in unrestrained phagocytosis of blood cells. 72 It has been extensively described in humans and also reported in several veterinary species, including dogs, 183 cats, 48,176 cattle, 117 and a macaque. 40 Nonregenerative anemia is a frequent feature in veterinary species and is thought to be due to bone marrow suppression secondary to hypercytokinemia and/or phagocytosis of erythroid precursors. 183 An underlying cause (eg, infection, systemic disease) is suspected to trigger the disease in both animals 183 and people 72 ; however, a primary (inherited) form is also described in humans. 72
Bovine neonatal pancytopenia
Over the past 5 to 6 years, bovine neonatal pancytopenia (BNP), sometimes called “bleeding calf syndrome,” has become recognized as an important emerging disease in Europe. 2,127 The syndrome is characterized by pancytopenia and bone marrow aplasia in neonatal calves that frequently present with clinical signs related to hemorrhagic diathesis. Similar, perhaps related, cases have also been reported in Canada 67 and Japan. 61 Although other etiologies were once suspected, there is strong evidence now that bovine viral diarrhea vaccine-induced maternal alloantibodies in colostrum mediate destruction of blood and bone marrow cells in calves, resulting in the disease. 13,85 Investigators of a syndrome in dromedary camels that has features similar to that of BNP suggest a possible link between the diseases in the 2 species. 188
Neoplasia
The mechanism of cancer-related anemia (CRA) is often multifactorial and includes both direct and indirect effects of tumors, as well as treatment-related factors. Anemia in patients with cancer may be nonregenerative or regenerative, depending on the specific mechanisms involved. The following sections focus on mechanisms of cancer-related nonregenerative anemia in veterinary species.
Inflammation
Cancer and inflammation are strongly associated. Inflammation is a risk factor for the initiation and progression of many cancers, 114 and cancer frequently causes secondary inflammation. In fact, inflammation is generally considered the most common cause of anemia in human 19 and veterinary 14 patients with cancer, and discussions of anemia of inflammation and CRA sometimes use the terms in tandem or even interchangeably. Hepcidin 148 and many inflammatory cytokines, in particular TNF-α, 27 IL-1, 45 IL-6, 73 and IFN, 45 have been implicated in the pathogenesis of CRA, emphasizing the role of anemia of inflammation in these patients.
Direct marrow effects
Both primary and secondary tumors may proliferate in the marrow, resulting in myelophthisis, destruction of normal hematopoietic cells, and alteration of the marrow microenvironment through inhibition of growth factors, induction of cytokines, and, potentially, induction of fibrosis. 109 Cytopenias and ineffective hematopoiesis are classic features of myelodysplastic syndrome; ineffective hematopoiesis occurs at least in part because of increased apoptosis. 169 These conditions tend to result in other cytopenias in addition to anemia. 109 Neoplastic tissues may also produce other hormones and factors that interfere with erythropoiesis. Iron-refractory anemia associated with excess production of hepcidin by hepatic adenomas has been reported in humans. 194 Pure red cell aplasia has been reported in a variety of cancer types in humans, including thymoma, in which there appears to be a relatively strong association. 104
Nutritional deficiencies
Multiple, nonspecific mechanisms may contribute to nutritional deficiencies that depress erythropoiesis in patients with cancer. 69 Chronic hemorrhage may result in whole-body iron deficiency. 14,69 Inadequate dietary intake or malabsorptive diseases may cause deficiencies in cobalamin and folate. Deficiencies in erythropoietic nutrients are commonly associated with neoplasia in humans. 12 The incidence of nutritional complications in veterinary oncology patients has not been thoroughly evaluated, although decreased serum levels of cobalamin and folate may be associated with lymphoma (in particular gastrointestinal lymphoma) in cats. 162 In dogs, hypocobalaminemia was found in 16% of 58 patients with multicentric lymphoma and was associated with a poor outcome. 37 Hematologic changes relating to cobalamin and folate deficiencies in veterinary species are discussed further in the section on congenital disorders.
Impaired renal function
Human and veterinary oncologists are keenly aware of the impact that cancer and its treatment may have on renal function. The kidney may be a primary site of tumor development and is considered a relatively common site of metastasis. Paraneoplastic biochemical (eg, hypercalcemia) and hemodynamic (eg, marked hyperproteinemia) abnormalities may also contribute to renal damage. 14 Diverse renal lesions (eg, glomerulonephritis, renal vasculitis, and hemolytic-uremic syndrome) abnormalities have been associated with various malignancies in human patients. 22 Similar lesions in veterinary patients with cancer are rarely cited 126,191 but may be underreported. Various medical and radiotherapies used in cancer treatment may also have renal-damaging side effects. Mechanisms of anemia associated with renal disease are included in Table 2.
Nonregenerative Anemia Associated With Pathology of Specific Organ Systems.

EPO, erythropoietin; GI, gastrointestinal; PTH, parathyroid hormone.
Cancer therapy
A comprehensive discussion of the side effects of various cancer therapies is beyond the scope of this review, but when evaluating the anemic oncology patient, the potential of chemotherapeutic agents, 182 radiation therapy, 180 and erythropoiesis-stimulating agents 136,137 to cause or exacerbate anemia should be considered.
Infectious Disease
Anemia secondary to infection may be regenerative or nonregenerative, depending on the disease mechanism, and often occurs because of associated inflammation, organ damage, blood loss, or misdirected immune response. However, some infectious agents cause anemia more directly—for example, by infecting erythroid cells or generating products that inhibit erythropoiesis. This section discusses the prevalence and pathogenesis of nonregenerative anemia associated with selected infectious agents of particular relevance to veterinary medicine.
Viral disease
FeLV is recognized to cause a spectrum of hematopathological alterations in cats. The prevalence, severity, and nature of anemia vary depending on the viral subgroup, the stage of infection, and host-related factors. One large study identified anemia in 11% of infected cats. 41 In most anemic FeLV-infected cats, anemia is nonregenerative. 96 FeLV infects hematopoietic, lymphoid, and stromal cells in the marrow. 96 Of the 3 clinically important FeLV subgroups, FeLV-C has a unique erythrocytopathic effect. While all FeLV subgroups have a similar hematopoietic cell tropism and may cause 1 or multiple cytopenias, FeLV-C is the causative agent of FeLV-associated PRCA. The host’s cellular receptor for FeLV-C (which binds to the FeLV-C’s unique surface envelope glycoprotein) may be necessary for erythropoiesis. Viral infection is suspected to alter the functional capacity of this receptor, thereby inhibiting erythropoiesis, possibly blocking the ability of blast-forming unit-erythroid progenitors to mature to the colony-forming unit-erythroid stage. 44,96 FeLV-C–induced PRCA is thought to occur in less than 1% of viremic cats 83 ; however, PRCA in an FeLV-infected cat strongly suggests infection with the C subgroup. 160 FeLV-C-induced PRCA is associated with normal myeloid and megakaryocytic precursors. 160 FeLV can also cause nonregenerative anemia via other mechanisms. Notably, FeLV infection is a risk factor for the development of myelodysplastic syndrome, lymphoma, and other hematologic malignancies. 78,96
Anemia is a less prominent feature of feline immunodeficiency virus (FIV) infection. Anemia (typically nonregenerative) has been reported in 31% of cats naturally infected with FIV and in 44% of cats with FIV and FeLV coinfection. 161 Blood cytopenias most frequently occur in the symptomatic stage of FIV infection, but anemia occasionally occurs in asymptomatic cats. 60 Unlike FeLV, FIV does not appear to directly infect hematopoietic precursor cells. Rather, marrow suppression likely results from viral or viral antigen-related factors or infection of stromal cells, altering the hematopoietic microenvironment. 96 Nutritional deficiencies, immune suppression and opportunistic infections, and other factors also likely contribute to anemia in many symptomatic patients.
Anemia in horses with equine infectious anemia virus (EIAV) develops as a result of both hemolysis and inhibition of erythropoiesis. 159 However, immunofluorescent studies did not identify EIAV antigen in erythroid precursors, and the impaired bone marrow response in affected animals is associated with findings consistent with anemia of inflammation. 159
Parvoviruses cause disease in a variety of mammalian species and generally demonstrate a tropism for mitotically active cells, particularly hematopoietic and intestinal epithelial cells. Parvoviral-induced hematopathology is not limited to the erythroid lineage but may include erythroid-specific effects. 24,122 Cytotoxic viral proteins are thought to be responsible for the death of erythroid progenitors in human parvovirus B19, 34 and a similar direct toxic effect on marrow precursor cells in dogs with parvoviral enteritis has been suggested. 20 A relationship between the parvovirus vaccine and immune-mediated hemolytic anemia and erythroid aplasia has also been suspected. 46 Parvovirus B19 is a particularly relevant cause of PRCA in human medicine. This strain of the virus specifically targets erythroid cells. 24 Simian parvovirus is similarly erythrotropic, 122 and PRCA in monkeys with experimental parvoviral infection has been documented. 123
Hemophagocytic syndrome (discussed further in the section on immune-mediated disease) has been associated with several different human viruses, including parvovirus B19, human immunodeficiency virus (HIV), herpesvirus, and others. 153 A connection between hemophagocytic syndrome and simian retrovirus in a macaque with pancytopenia has been suggested. 40
Chicken anemia virus also targets and destroys erythroid progenitor cells, causing severe nonregenerative anemia. 8
Protozoal disease
Hematoprotozoal infections often cause hemolytic disease and are associated with regenerative anemia. However, some notable exceptions exist.
The pathogenesis of anemia in malaria has been investigated intensively, given the disease’s far-reaching effects in humans. The majority of this work has explored the mechanisms of anemia caused by
Fungal disease
Inflammation, renal disease, nutritional deficiencies, and other nonspecific mechanisms may contribute to nonregenerative anemia secondary to mycotic disease. Generalized myelotoxicity and pancytopenia are associated with some fungal diseases (eg,
Parasitic disease
Chronic infection by hemophagous parasites may cause iron deficiency anemia in veterinary species. Most commonly, these are infections of the skin (eg, fleas, lice 115 ) or intestinal tract (eg, Strongylid infection 75 ), but iron deficiency anemia secondary to parasitic infestation of other organs (eg, liver flukes in cattle 99 ) is also possible.
Toxicity
Anemia may be the result of an adverse reaction to a therapeutic or environmental agent. The character of the anemia is variable, as are the underlying pathophysiologies. While some agents may induce regenerative anemia (eg, via blood loss or hemolysis), others may damage erythroid precursor cells or the hematopoietic environment, causing a nonregenerative anemia. The presence and severity of anemia may be idiosyncratic or dose-dependent, and patient factors, such as species or breed characteristics, individual genetic susceptibility, and preexisting conditions, may influence whether a given patient will develop anemia. A list of the specific therapeutic (Table 3) and environmental (Table 4) agents that have been associated with the development of nonregenerative anemia in veterinary species is provided. A discussion of the pathophysiology and phenotype of anemia associated with each agent is beyond the scope of this article.
Therapeutic Agents Associated With Nonregenerative Anemia (and Absence of Other Cytopenias) in Veterinary Species.

Environmental Toxins Associated With Nonregenerative Anemia (and Absence of Other Cytopenias) in Veterinary Species.a

aThe association of certain mycotoxins with the development of nonregenerative anemia in veterinary species is discussed in the section on infectious diseases.
Selected Congenital Disorders
Dyserythropoiesis of English Springer Spaniels
Congenital dyserythropoiesis has been described in English Springer Spaniels. 81 Affected animals had moderate, microcytic, normochromic nonregenerative anemia with markedly abnormal erythrocyte morphology. Marrow evaluation revealed erythroid hyperplasia and dyserythropoietic changes, including lobulated nuclei, binucleation, bizarre mitoses, and cytoplasmic vacuolation. The syndrome was associated with polymyopathy, megaesophagus, and cardiac disease in all 3 dogs. The specific molecular lesion has not been identified.
Congenital dyserythropoiesis of polled Hereford cattle
Congenital anemia associated with progressive alopecia, dyskeratosis, and, often, death occurs in some polled Hereford calves. 164 Anemia in these cases is normochromic, normo- to macrocytic, and nonregenerative, and circulating macrocytes and/or variable numbers of nucleated erythrocytes may be identified in circulation. Marrow findings include erythroid hyperplasia and evidence of dyserythropoiesis. It is suspected to be of genetic origin 165 ; the specific molecular lesion has not been identified.
Inherited defects of cobalamin absorption and metabolism
Congenital disorders affecting cobalamin absorption and transport in humans include inherited intrinsic factor (IF) deficiency, Imerslund-Gräsbeck syndrome (caused by a mutation in either the cubulin or amnionless gene), and transcobalamin deficiency. Cobalamin is an essential cofactor in the formation of physiologic folate, which is required for normal DNA synthesis. Insufficient cobalamin and/or folate causes base-pair mismatch errors in the growing DNA strand, which are subsequently excised by DNA repair mechanisms. An inability to repair these errors leads to multiple DNA strand breaks, thwarting cytopoiesis. 10 In humans, inherited disorders causing hypocobalaminemia are associated with dyserythropoiesis, nonregenerative anemia, and other clinicopathologic changes. 190 Similar conditions have been reported in Giant Schnauzers, 63 Border Collies, 125 a Beagle, 55 and a cat. 173 Although an inherited defect of cobalamin absorption was supported in all cases, a mutation (in amnionless) was demonstrated only in affected Giant Schnauzers. In humans, anemia secondary to cobalamin deficiency is most often macrocytic (although the pathophysiologic mechanism of this change is not clear 10 ). In the veterinary cases, macrocytosis was not a consistent finding.
Hypocobalaminemia associated with gastrointestinal or other diseases does not typically cause anemia in veterinary patients. However, the previously cited cases of suspected cobalamin malabsorption in dogs and a cat were associated with a normocytic, normochromic, nonregenerative anemia. As discussed above, anemia in humans with hypocobalaminemia is typically macrocytic. While macrocytosis was not observed in the veterinary cases, mild megaloblastic changes were found in the blood and/or marrow in a few of the dogs. 63,113,125
Hereditary cobalamin deficiency in Chinese Shar Pei dogs 70 and a breed predisposition to cobalamin deficiency in several other breeds 71 have also been described, although hematologic changes in these cases have not been fully characterized.
Diamond Blackfan anemia
Diamond Blackfan anemia (DBA) is an inherited PRCA in humans that causes a macrocytic, normochromic, nonregenerative anemia and marked marrow erythroid hypoplasia.
57
Animal models of this disease include mice
106
and zebrafish
168
and, although not an established disease in dogs, a DBA-like syndrome was described in 1 dog.
112
Furthermore, there may be a relationship between DBA and FeLV. FLVCR1, a heme exporter and cell surface receptor for FeLV-C, is important in early erythropoiesis.
134
Physiologic and Environmental Factors
Aging and pregnancy
Healthy humans develop increased EPO concentrations with age. 50 Failure of this normal rise in EPO levels is suspected to play a role in unexplained anemia of elderly patients. 174 The physiologic erythrokinetic changes of geriatric animals are not well characterized.
Anemia associated with pregnancy in humans is highly prevalent and associated with a variety of factors, including iron or other micronutrient deficiency, infectious disease, a maternal congenital hemoglobinopathy, and aplastic pancytopenia 94 (the latter perhaps due to estrogen excess or another hormonal imbalance or to the presence of other myelotoxic factors 35 ). Pregnancy-associated anemia has not been as extensively studied in veterinary species; however, a physiologic decrease in hematocrit during late pregnancy and early lactation is described in some species. 15,28,116 The changes are sometimes significant enough to cause mild nonregenerative anemia.
Sports anemia
Exercise can lead to increases in total hemoglobin and erythrocyte mass, presumably as an adaptive measure to increase oxygen-carrying capacity, leading some investigators to suggest that exercise may be a warranted treatment for some forms of anemia. 82 However, strenuous exercise may also be associated with anemia. First, intense exercise can cause excessive iron losses, and human athletes are commonly diagnosed with iron deficiency, which can lead to anemia. 129 Mechanisms of iron loss associated with strenuous exercise are well established and include sweating, hemolysis, hematuria, and gastrointestinal (GI) hemorrhage. 129 More recently, the invocation of an inflammatory state has been implicated in the development of sports anemia. Increased circulating levels of IL-1, TNF-α, and, in particular, IL-6 have been identified in humans following intensive exercise. 124,128 IL-6 has been shown to be released from working muscle and at higher rates with increasing intensity. 54 Strenuous exercise in rats also induced inflammation that was associated with increased circulating IL-6, increased hepatic hepcidin expression, and anemia. 97
Plethora
Researchers studying physiologic responses in astronauts have discovered a predictable 10% to 15% decrease in red cell mass associated with space travel. 172 Upon entry into microgravity, blood in the extremities is redistributed, resulting in an acute central pooling of blood. This localized hypervolemia (known as acute central plethora) causes further redistribution of plasma to interstitial tissues, causing edema and an increased central hematocrit. The raised hematocrit leads to a profound inhibition of EPO secretion, causing suppression of erythropoiesis and initiation of neocytolysis (the selective hemolysis of young erythrocytes, aka neocytes). The end result is a decrease in red cell mass that is adaptive for space but not suitable for terrestrial conditions. A similar red cell response has been identified in polycythemic, high-altitude dwelling individuals upon descent to sea level. 145
A suggested mechanism of neocytolysis is that EPO suppression causes decreased endothelial cell production of TGF-β (and thus decreased macrophage deactivation). Concurrent macrophage production of thrombospondin may stimulate bridge formation between adhesion molecules on young erythrocytes, targeting them for phagocytosis. 170 To the authors’ knowledge, neocytolysis associated with plethora and EPO suppression has not been studied in domestic animals. However, the mechanisms that underlie this phenomenon have implications for changes in red cell mass associated with renal disease, polycythemia, “blood-doping” in athletes, oxygen therapy, and the hemolytic anemia associated with pyruvate kinase deficiency. 143,144
Other environmental factors
Environmental stressors have rarely been described to cause anemia in animals. Increased population density and other environmental stress may cause anemia in fish, 36 and nonregenerative anemia has been identified in herring gulls in captivity. 80
Nonregenerative Anemia Associated With Pathology of Specific Organ Systems
The information above about mechanisms of nonregenerative anemia is organized by disease process; information organized by pathology of specific organ systems is presented in Table 2.
Conclusion
Various types of disease and disorders of specific organ systems are recognized to cause nonregenerative anemia, via either decreased or ineffective erythropoiesis. Multiple mechanisms often contribute to diminished erythropoiesis. Many conditions associated with diminished erythropoiesis in people have parallel descriptions in animals; the prevalence of nonregenerative in animals is described better for some of these conditions (eg, chronic renal failure) than others (eg, inflammation). Other conditions linked to impaired erythropoiesis in people—for example, heart failure and aging—are poorly understood as causes of anemia in animals. Nonregenerative anemia in animals remains a fertile area for investigation, from the molecular to the epidemiologic level.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
