Abstract
Opportunistic viral infections are common in simian immunodeficiency virus–infected rhesus macaques and include simian polyomavirus 40 (SV40), which causes interstitial nephritis, pneumonia, meningoencephalitis, and progressive multifocal leukoencephalopathy and rhesus cytomegalovirus (Macacine herpesvirus-3), which is associated with many pathologic manifestations, including the formation of neutrophil-rich gastrointestinal masses. Herein we report the findings of a simian immunodeficiency virus–infected rhesus macaque that presented to necropsy with multiple nodular masses restricted to the proximal jejunum. Histologically, the masses within the lamina propria were composed of abundant, loosely organized, mesenchymal tissue forming broad interlacing whorls and sheets admixed with variable numbers of neutrophils. Cells within the mesenchymoproliferative nodules contained numerous basophilic, intranuclear inclusion bodies with only scattered cytomegalic cells. Immunohistochemistry for rhesus cytomegalovirus and SV40 demonstrated variable numbers of immunopositive cells within the affected nodules. This report is the first description of SV40-associated pathology in the small intestine of a rhesus macaque and highlights the role that opportunistic viral infections can have on gastrointestinal pathology in immunosuppressed rhesus macaques.
Rhesus macaques (Macaca mulatta) infected with simian immunodeficiency virus (SIV) are the preeminent animal model for HIV pathogenesis. A similar spectrum of viral, bacterial, fungal, and protozoal opportunistic infections develop in both human acquired immunodeficiency syndrome (AIDS) patients and macaques with simian AIDS (SAIDS). Opportunistic viral infections in affected macaques include simian polyomavirus (SV40), Macacine herpesvirus-3 or rhesus cytomegalovirus (rhCMV), and rhesus adenovirus, with only the latter 2 viruses known to cause gastrointestinal pathology in SAIDS macaques.
SV40 is a natural infection in Asian macaques, where it establishes latency and persists in the kidney without pathologic effect. SV40 is closely related to the human BK and JC polyomaviruses, in which there is high seroprevalence but typically little disease unless there is immunosuppression. 19,22,24 With immunosuppression, BK virus reactivates and is associated with renal failure due to acute interstitial nephritis, distal ureteral stenosis, hemorrhagic cystitis, pneumonitis, upper respiratory tract infections, and meningoencephalitis. 24 Similarly, with immunosuppression, JC virus causes progressive multifocal leukoencephalopathy (PML) affecting up to 5% of AIDS patients. 22,24 Both BK and JC viruses are oncogenic in experimental animals, but their role as potential carcinogens in humans has not been established. 1,9 Although some of the polyomavirus manifestations noted in humans are absent in macaques, SV40 is typically associated with profound nephritis, pneumonia, and PML in immunosuppressed animals with SIV infection. 22,24 Histologically, SV40-infected cells have viral changes associated with the accumulation of virions in the cell nucleus that appear as glassy to smudgy, basophilic inclusions that enlarge the nucleus replacing or displacing chromatin. 6
Similar to polyomavirus, human cytomegalovirus, a betaherpesvirus, has a high seroprevalence affecting up to 100% of adults. 7,10 This ubiquitous virus remains latent in immunocompetent hosts, but it is one of the most common opportunistic infections in patients with immunosuppressive conditions such as AIDS, organ transplantation, and cancer chemotherapy. 10 With reactivation, it can affect many organs, including the alimentary tract causing erosive to ulcerative lesions. It has also been associated with mass lesions in the gastrointestinal tract of AIDS patients. 13 Rhesus CMV is the most common opportunistic viral infection in SIV-infected macaques and is associated with the development of meningitis, pneumonia, lymphadenitis, hepatitis, splenitis, orchitis, neuritis, and arteritis as well as proliferative lesions in the alimentary tract. 2,13 Although lymphoma is the most common cause of gastrointestinal tract masses in rhesus macaques infected with SIV, rhCMV-associated mass lesions are commonly noted and are characterized by hyperplastic glandular tissue and marked neutrophilic infiltrates. 13,14,16 Characteristic histologic findings in CMV infections include the presence of karyomegaly, intranuclear and intracytoplasmic inclusion bodies, and variably neutrophilic inflammatory infiltrates. 2
SV40 virus infection in rhesus macaques has no known gastrointestinal pathology, although virus infected cells have been noted in the enteric smooth muscle of cynomolgus macaques. 24 Herein, we describe the morphologic and immunohistochemical characterization of an unique mesenchymoproliferative enteropathy dually infected with rhCMV and SV40 in an SIV-infected rhesus macaque and discuss the implications of concomitant viral infections in immunosuppressed macaques.
Materials and Methods
A 4-year-old rhesus macaque was acquired from the Caribbean Primate Research Center and housed at the New England Primate Research Center in a biosafety level 3 facility in accordance with National Research Council’s Guide for the Care and Use of Laboratory Animals (8th edition) and the standards of the Harvard Medical School Standing Committee on Animals and the Association for the Assessment and Accreditation of Laboratory Animal Care. Upon arrival, the animal tested negative for Mycobacterium tuberculosis and was screened negative for Campylobacter sp, Shigella sp, Giardia sp, Cryptosporidium parvum, and Entamoeba sp. Animals were tested negative yearly for Macacine herpesvirus 1, 4, 5, SRV, and M. tuberculosis. The animal was vaccinated 3 times using a single-cycle SIV vaccine, followed 106 days later by intrarectal SIVmac251 challenge. The macaque was humanely euthanized 710 days postchallenge due to the development of weight loss, mild dyspnea, and equivocal neurologic signs. The animal had significant reductions in tissue CD4+ T cells as measured in rectal biopsies by flow cytometry several weeks prior to euthanasia. These reductions were consistent with chronic SIV infection and immunosuppression. Necropsy was conducted within 2 hours of euthanasia, and representative sections of all major organs were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and stained using hematoxylin and eosin.
Standard immunoperoxidase staining for SV40, CMV, smooth muscle actin, Ki-67, vimentin, latency-associated nuclear antigen (LANA), and double labeling for SV40/CMV and SV40/CD68 was performed on formalin-fixed, paraffin-embedded sections of multiple jejunal nodules (Table 1). All sections were deparaffinized and rehydrated, followed by blocking with 3% hydrogen peroxide in phosphate buffered saline. A wash of tris-buffered saline followed each step. Following pretreatment, an avidin-biotin block (Invitrogen Corporation, Frederick, Maryland) and then a Dako Protein block (DakoCyomation, Carpinteria, California) were conducted on all sections.
Antibody Source, Dilution, and Antigen Retrieval for Immunohistochemistry Protocols
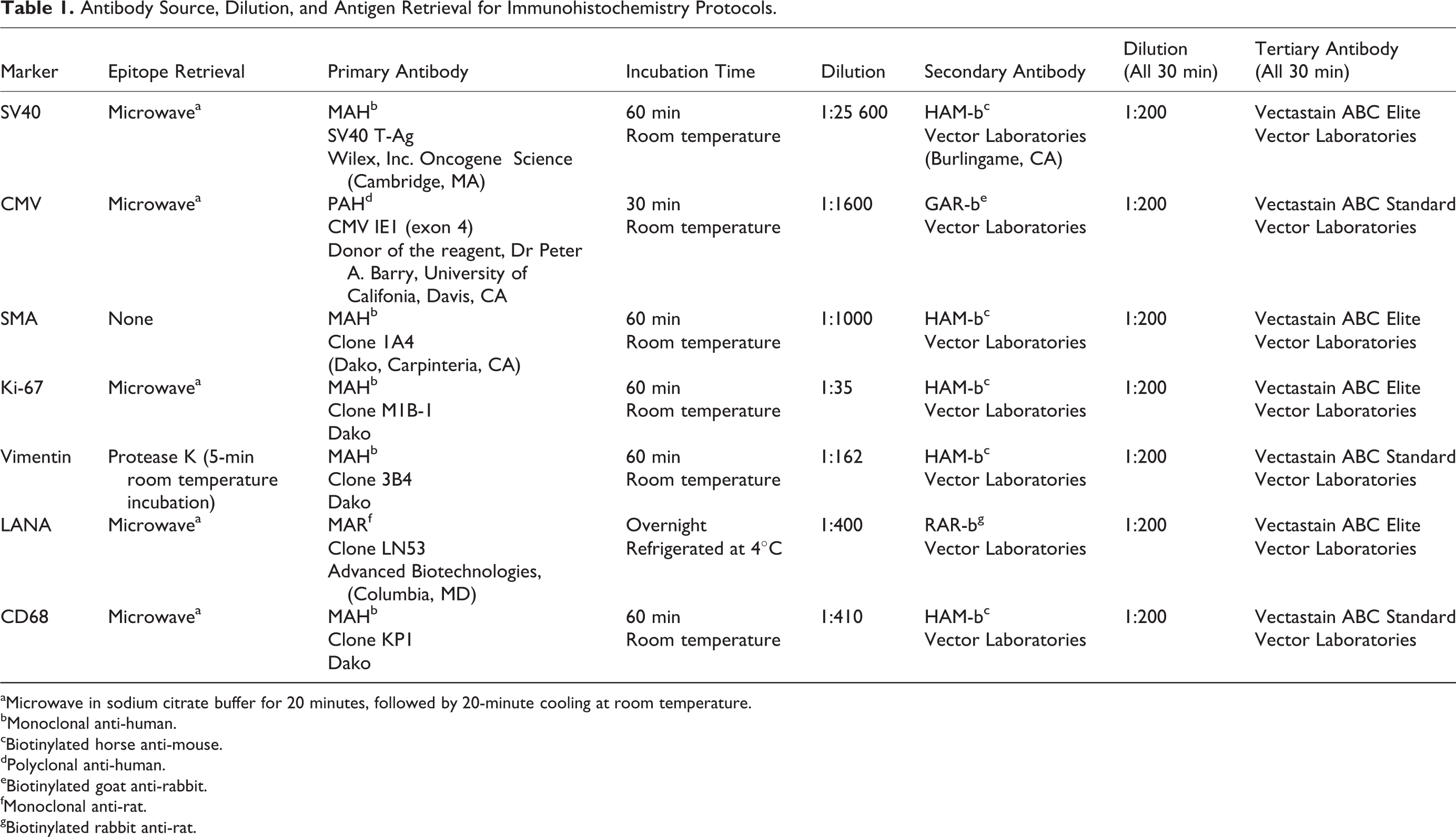
aMicrowave in sodium citrate buffer for 20 minutes, followed by 20-minute cooling at room temperature.
bMonoclonal anti-human.
cBiotinylated horse anti-mouse.
dPolyclonal anti-human.
eBiotinylated goat anti-rabbit.
fMonoclonal anti-rat.
gBiotinylated rabbit anti-rat.
Antigen–antibody complex formation was detected using diaminobenzidine (DAB; DakoCyomation) for all the single-labeled immunohistochemistries, while antigen–antibody complex formation was detected using Vector Blue and Vector Red (Vector Laboratories, Burlingame, California) for both double-labeled protocols. Irrelevant, isotype-matched primary antibodies were used in place of the test antibody as negative controls in all immunohistochemical studies. Positive control tissues consisted of archived rhesus macaque brain stem (SV40), lung (CMV), small intestine (SMA, vimentin), spleen (CD68, Ki-67), and gastrointestinal stromal cell tumor (LANA).
Results
Gross and Histopathologic Features
Grossly, within the jejunum, the animal had multiple mucosal nodules ranging from 0.2 to 1.0 cm in diameter projecting into the lumen that were variably tan and mildly reddened. The only other gross finding was multicentric lymphoma that involved the adrenal glands, kidneys, central and peripheral lymph nodes, heart, bone marrow, and spleen.
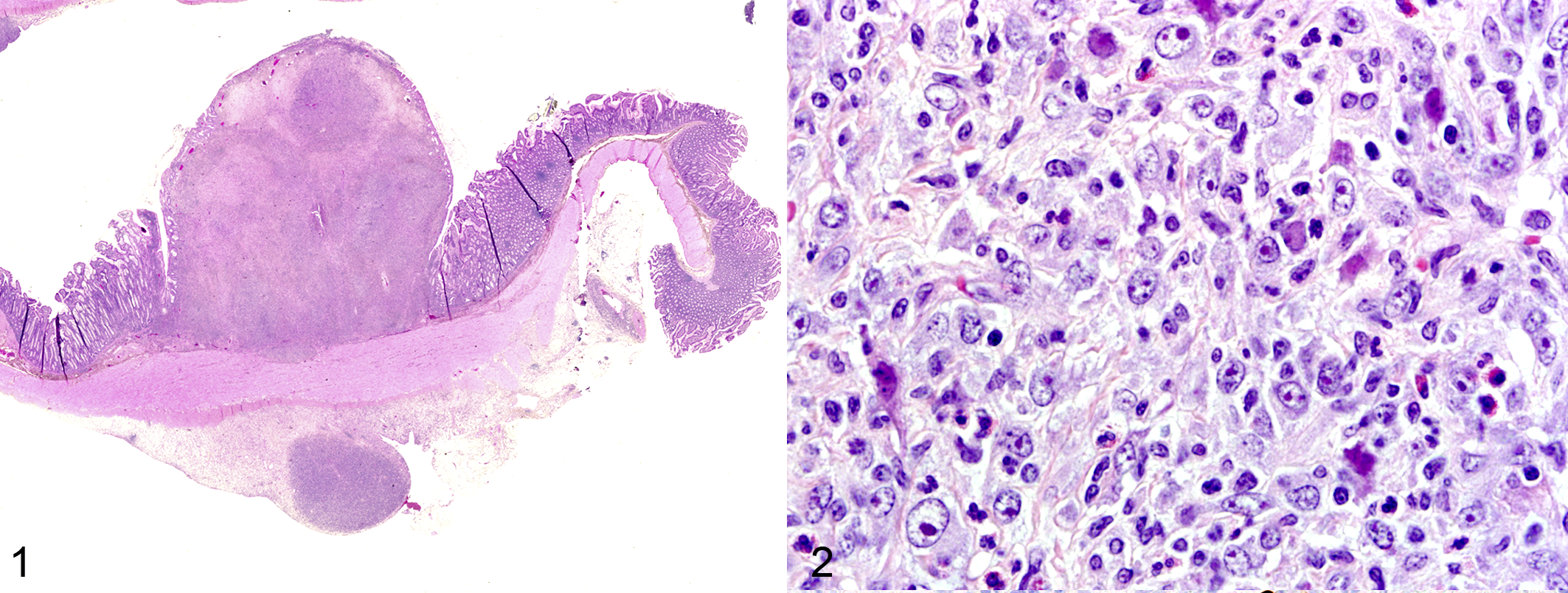
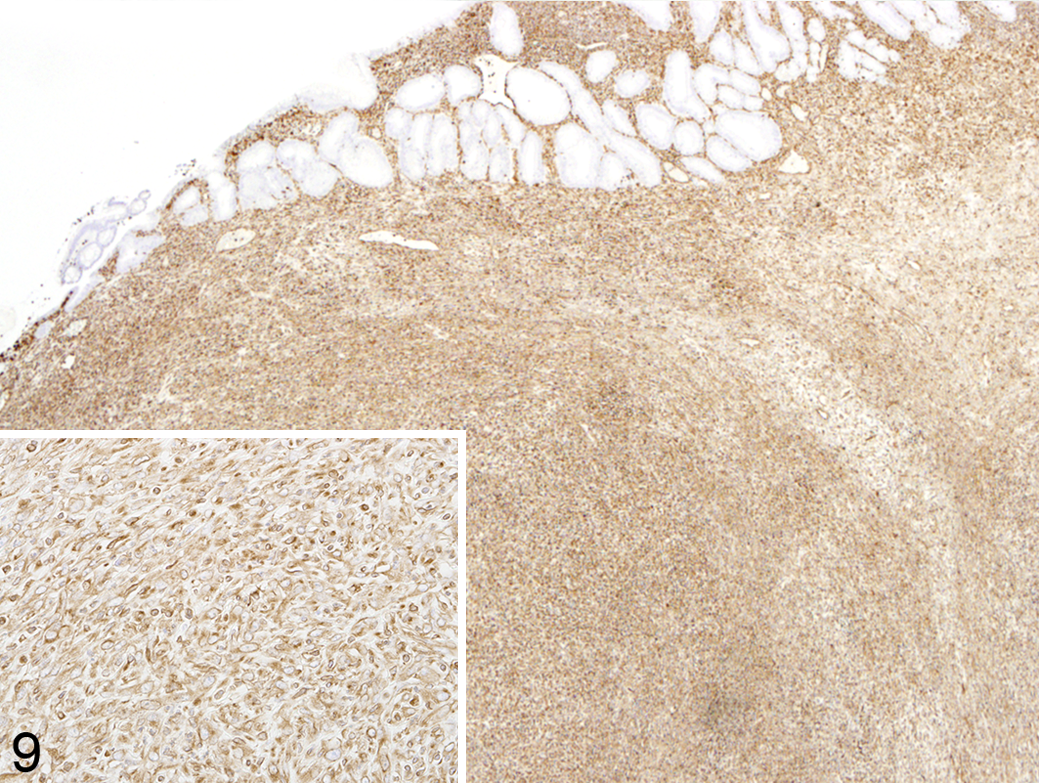
Histologically, the largest nodule focally and markedly expanded the lamina propria of the jejunum and was characterized by a large, moderately cellular, unencapsulated, discrete proliferative nodule composed of loosely arranged mesenchymal cells encompassed in abundant fibrous tissue that formed broad whorls and sheets subsequently infiltrated with marked numbers of viable neutrophils admixed with fewer macrophages, lymphocytes, and eosinophils (Figs. 1, 2). The apical portions of the villi overlying the mass were ulcerated and overlain with fibrin, necrotic debris, and a mixed population of bacteria and neutrophils. Scattered throughout the mass were mesenchymal cells containing smudgy, 20-μm, irregularly shaped, amphophilic to basophilic intranuclear inclusion bodies that occasionally marginated chromatin. Rarely, cytomegalic cells were present. No mitotic figures were present. Diffusely, within the adjacent normal tissue, the lamina propria was mildly expanded by small numbers of viable neutrophils. Goblet cells were increased in the overlying villi. Four other nodules in the jejunum were smaller in size but possessed similar histologic characteristics in the lamina propria (Fig. 3). No additional lesions attributable to SV40 were noted in either the kidneys or the brain. There were no other significant comorbid histologic findings other than the multiorgan lymphoma.
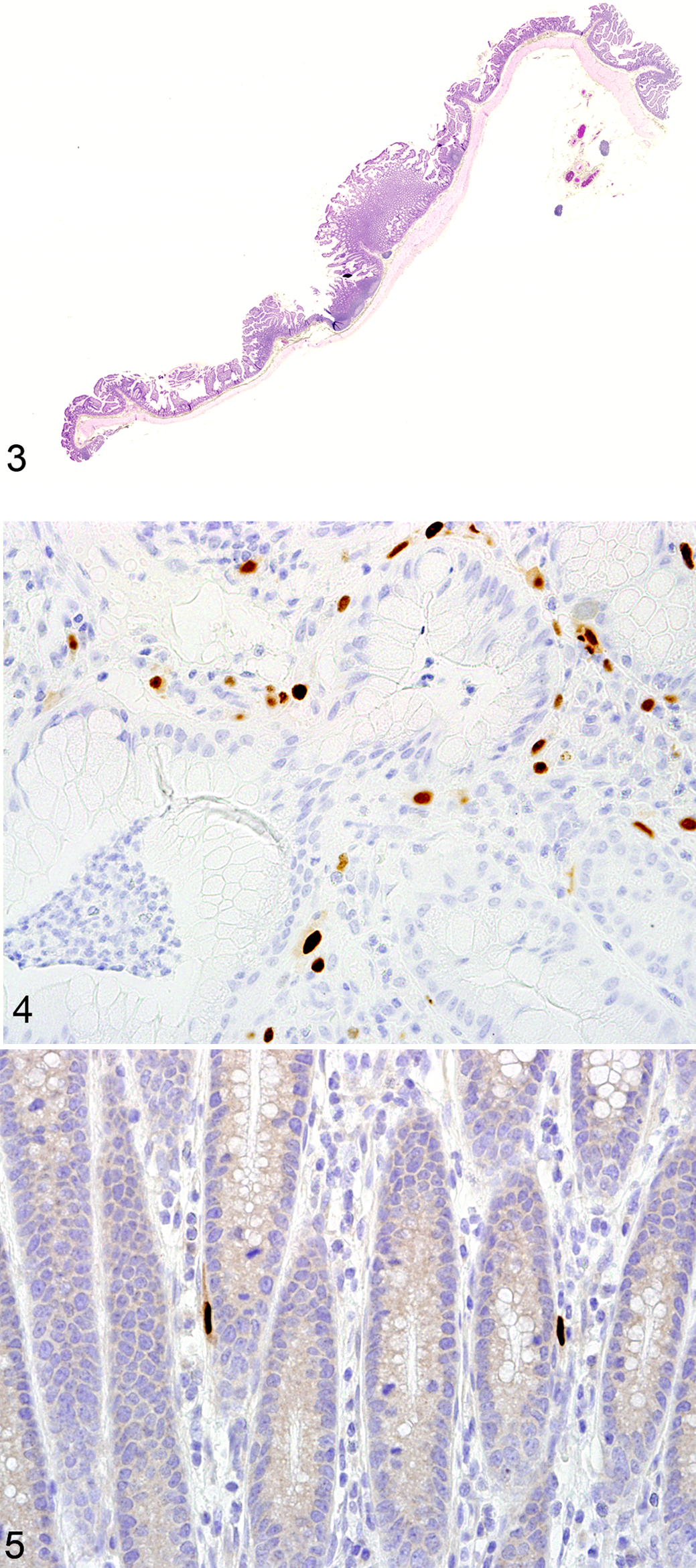
Immunohistochemical Features
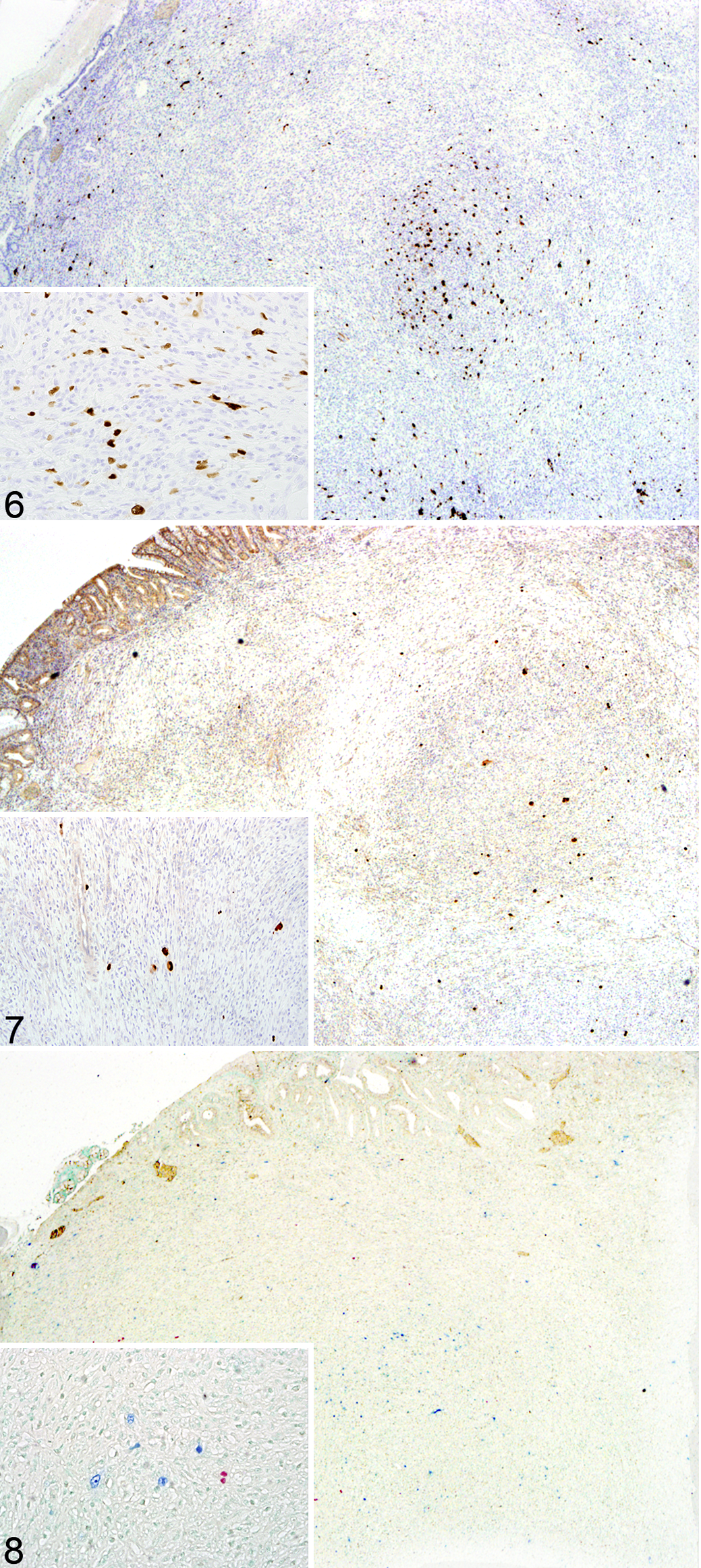
All of the mesenchymal proliferative regions identified in this case possessed frequent cells expressing intense nuclear SV40 immunoreactivity. These cells were randomly located within the expanded regions of the lamina propria in all of the jejunal nodules but were absent from all other regions of the intestine. Positive staining cells averaged 20 per high-power field throughout the nodule. Within the largest of the jejunal nodules as well as within 2 of the smaller proliferations, randomly scattered cells had intensely positive nuclear immunoreactivity to CMV, averaging 3 per high-power field (Figs. 4 –8). CMV nuclear immunoreactivity was present within occasional cells in the lamina propria of the more normal tissue adjacent to the largest nodule; however, positive staining cells were more numerous in the proliferative regions than in other areas of tissue. To investigate the origin of the spindle-shaped cells composing the bulk of the nodules, immunohistochemistry for smooth muscle and mesenchymal cells was performed using smooth muscle actin (SMA) and vimentin, respectively. Reactivity to SMA antibodies was limited to cells lining variably sized capillaries and the underlying muscularis mucosa (data not shown). Proliferative regions of all of the nodules were diffusely and strongly positive for vimetin (Fig. 9). Ki-67 was expressed in approximately 30% of the cells composing the nodules (data not shown). The cells composing the proliferative regions had no immunoreactivity to LANA (data not shown). 4,20 Cells expressing CD68 were not concurrently positive for SV40, indicating that the SV40 inclusions were not present in macrophages (data not shown). Similarly, double labeling for CMV and SV40 showed that these 2 viruses did not coinfect the same cells (Fig. 8).
Discussion
This report is the first description of SV40-associated pathology in the small intestine of an immunocompromised rhesus macaque and expands the tissue tropism and pathology associated with this virus. The histologic characteristics and immunoreactivity for vimentin are consistent with a proliferation of mesenchymal cells. The immunosuppressive effects of SIV infection undoubtedly played a significant role in the reactivation of rhCMV and SV40 in this case and likely contributed to the development of this mesenchymoproliferative enteropathy.
Pathology associated with SV40 infection in SIV-infected, immunosuppressed rhesus macaques may be as a result of either primary infection or reactivation of a latent infection. 22 SV40-associated nephritis is characterized by tubular necrosis and tubulointerstitial nephritis with prototypical intratubular intranuclear inclusion bodies. 6 Interstitial fibrosis, glomerulosclerosis, and glomerular atrophy may also be present with persistent infection. 6,12,21 Two distinct central nervous system lesions are associated with SV40 infection in SIV-infected macaques. PML is a demyelinating central nervous system disease in which SV40 primarily infects the oligodendrocytes. 12 SV40 may also cause meningoencephalitis affecting cerebral gray matter, a separate and distinct process than PML. 22 Less often SV40 has been associated with proliferative interstitial pneumonia characterized by the presence of the typical inclusion bodies and type 2 pneumocyte hyperplasia. 21 A novel polyomavirus infection in the muscularis of the small intestine of an immunosuppressed cynomolgus macaque was recently reported, but this infection caused apoptosis and inflammation not a proliferative response. 24
CMV infection is the most common opportunistic viral infection in HIV-infected humans as well as in SIV-infected macaques and can be disseminated. 13 Productive infection leads to histopathologic lesions characteristic for this betaherpesvirus—namely, variable numbers of large cytomegalic cells containing large intranuclear inclusion bodies surrounded by a clear halo. A predominantly neutrophilic infiltration often accompanies infection. 16 The brain, lung, lymph nodes, liver, spleen, intestine, testicle, nerves, and arteries are the tissues most often involved in CMV infections in both humans and macaques. 2 Gastrointestinal lesions typically associated with CMV infection consist of focal erosions/ulceration with dense sheets of neutrophils or the expansion of the lamina propria by neutrophils, macrophages, and lymphocytes accompanied by prototypical cytomegalic cells. Although the current case grossly had proliferative nodules similar to those seen in CMV-associated enteric disease, the profound proliferation of spindle cells is atypical in CMV-induced gastrointestinal disease.
Coinfection with CMV and human polyomaviruses, particularly with concurrent immunosuppression, has been documented in human patients as well as in vitro. For example, concomitant infections with human CMV and polyomaviruses have been reported in renal, bone marrow, and stem cell transplants in patients who are immunosuppressed. 23 It was concluded, in a study involving renal transplant patients, that BK virus infection may be permissive for the development of CMV infection, which is consistent with previous reports using in vitro studies by Kristoffersen et al. 15,23 Research conducted in vitro by Heilbronn et al identified human CMV as a potent helper virus for JC virus in human fibroblasts that were resistant to the replication of JC virus alone. They found that human CMV enhanced JC virus replication in human glioblastoma cells in vitro. 11 These findings imply an interaction and possibly synergistic relationship between polyomaviral and herpesviral replication. 18 It has been reported that the development of PML in human HIV-infected patients may be due to the release of cellular products, proteins, or cytokines from virally infected cells. These substances are believed to act as positive regulatory triggers transactivating JCV and triggering the development of PML. 3 The possibility of a synergistic relationship between viruses, especially in immunosuppressed individuals, seems likely.
Mesenchymoproliferative disorders, such as retroperitoneal and subcutaneous fibromatosis as well as gastrointestinal stromal tumors, have been associated with the development of SAIDS in macaques both positive and negative for simian retrovirus type D. 4,8 Additionally, the characteristics of these lesions are similar in many ways to human Kaposi sarcoma (KS), which is a proliferative lesion associated with HIV infection and human herpesvirus-8. 8,17 The comparison of retroperitoneal fibromatosis lesions in macaque species to KS in humans has been bolstered by the demonstration of the macaque homologue of human herpes virus 8 known as retroperitoneal fibromatosis herpes virus (RFHV) in these lesions. 20 Immunohistochemical analysis using the LN53 monoclonal antibody can be used to detect the RFHV LANA, which is also used to identify KS in human patients. 5 Bielefeldt-Ohmann et al identified colonic submucosal stromal tumors with similarities to mesenchymoproliferative disorders in an SIV-infected, immunosuppressed, macaque that was negative for simian retrovirus type D. The lesions within the colon of that animal expressed intense immunohistochemical expression of LANA-1, supporting the role of RFHV in tumor cells. However, in the case reported herein, there was no immunohistochemical expression of LANA in the proliferative nodules, indicating that neither RFHV nor KS associated herpesvirus played a role in this proliferative condition.
SV40 virus was detected in all of the proliferative regions in this case. Three of the 5 nodules in this case also possessed cells expressing rhCMV. We propose that the nodules in this case represent various stages in the progression of this unique mesenchymoproliferative enteropathy associated with SV40 viral infection. Because there were CMV positive cells in the lamina propria adjacent to the mesenchymoproliferative nodules, it is likely that rhCMV did not play a direct role in the mesenchymoproliferative lesion in this case as the proliferative gastrointestinal lesions associated with rhCMV are typically epithelial in nature. In conclusion, this report provides the first description of SV40 associated pathology in the small intestine of a rhesus macaque and expands the tissue tropism associated with this polyomavirus. Immunohistochemistry confirmed the expression of SV40 as well as CMV concurrently in this mesenchymoproliferative enteropathy, and even though SV40 and CMV were not coinfecting the same individual cells, a potential interaction of these 2 viruses is plausible and should be explored further.
Footnotes
Acknowledgement
We thank Kristen Toohey for graphic assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the National Center for Research Resources and the Office of Research Infrastructure Programs of the National Institutes of Health through grant Nos. RR07000 and RR00168.
