Abstract
Streptococcosis caused by Streptococcus iniae has become one of the most serious marine and freshwater aquatic diseases in the past decade, causing large losses in farmed and wild fish worldwide. In this study, we performed an ultrastructural study of major lesions in gilthead seabream Sparus aurata and red porgy Pagrus pagrus experimentally infected with the S. iniae IUSA-1 strain, isolated in a natural outbreak in Spain in the mentioned species. The transmission electron micrographs revealed the resistance of this pathogen inside the phagosome, indicating that the macrophage may provide a significant bacterial reservoir for continuing infection, disease dissemination, and tissue injury by crossing the blood-brain barrier.
Streptococcus iniae has become one of the most serious aquatic pathogens in the past decade and is considered a reemerging disease affecting a variety of wild and cultured fish throughout the world, causing large losses with mortality rates of up to 50%. 8 The first isolation of this pathogen was recorded in a captive Amazon River dolphin Inia geoffrensis in 1976. S. iniae has caused outbreaks of disease in marine and freshwater fish in Asia, Australia, the United States, and Europe. 1,3 Clinical signs and gross external lesions of S. iniae infection include loss of orientation, lethargy, ulcers, and exophthalmia, and the most common histological lesion is meningoencephalitis. 1 S. iniae also causes fulminant soft-tissue infection in humans, with associated bacteremia, following percutaneous injury acquired while handling whole fish. 8
There are several mechanisms that microorganisms deploy to evade the host immune systems, including inducing host cell death or necrosis. S. iniae is reportedly able to withstand the bactericidal activity of macrophages. 9 And most recently, the capsule has been described to play an important role in resistance to phagocytic clearance by teleost macrophages. 7 We have recently described the first report of infection by S. iniae in red porgy (Pagrus pagrus) and as well the infection of gilthead seabream (Sparus aurata) in Europe. 3 The aim of this study is to describe ultrastructural lesions caused by S. iniae in gilthead seabream and red porgy after an experimental challenge.
The S. iniae IUSA-1 strain used in this study was previously isolated in a natural outbreak in gilthead seabream and red porgy in Spain. 3 The strain was cultured on trypticase soy agar at 22°C for 24 hours. Bacterial cultures were grown overnight at 22°C in trypticase soy broth (TSB) supplemented with 0.5% (v/v) yeast extract (TSBY) without agitation. Overnight cultures were diluted 1:100 in fresh TSBY medium, incubated at 22°C, and harvested in the mid-logarithmic phase. Bacteria were pelleted by centrifugation at 1500 g for 5 minutes, washed in fresh phosphate-buffered saline (PBS), repelleted, and resuspended in PBS to achieve the desired optical density at 600 nm by spectrophotometry (BioPhotometer; Eppendorf Hamburg, Germany).
Twenty juveniles of each fish species, red porgy (average 50 g body weight [BW]) and gilthead seabream (average 20 g BW), were obtained from a local fish farm and maintained in a closed seawater flow circuit with water at a temperature of 24°C and a salinity of 37‰ under a 12-hour light/12-hour dark cycle. The fish were intraperitoneally inoculated with 100 μl of the virulent strain S. iniae IUSA-1 at 105 colony-forming units/fish. Inoculated and control fish were monitored for clinical disease and mortalities for the duration of the study (10 days). Mortality was attributed to the inoculated bacterium if the injected organism was recovered in pure culture from the internal organs. Inoculated and control fish were analyzed by transmission electron microscopy (TEM).
In the TEM study, samples were fixed in suspension with 2% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4), postfixed with osmium tetroxide, and dehydrated with increasing concentrations of acetone before being infiltrated and embedded in resin. Ultra-thin sections were cut at 50 nm and contrasted with lead citrate before being observed under a Zeiss (Jena, Germany) EM 910 transmission electron microscope at the Electron Microscope Service of the University of Las Palmas de Gran Canaria.
In red porgy, the mortality rate was over 50%, while in gilthead seabream, the mortality rate was 40% over a period of 10 days. Clinical signs and gross external lesions in infected fish began 2 days after inoculation and consisted of lethargy, anorexia, darkness of skin, abnormal swimming, exophthalmia, and corneal opacity. Macroscopically, this group of fish had petechial hemorrhages, abdomen distended with ascitic fluid, hemorrhages around the anus, splenomegaly, and hepatomegaly with pale internal organs. Control fish had no clinical signs or macroscopic lesions.
Macrophages with abundant membranous material together with numerous intracellular bacteria within phagolysomes (Fig. 1) were observed by TEM. Some of these bacteria had intact cell walls (Fig. 2). Others showed cellular degradation with dissolution of cells walls and other cellular debris (Fig. 3). In the gilthead bream, free bacteria were found in splenic vessels (Fig. 4) as well as within the cytoplasm of macrophages in the head kidneys (Fig. 5). Numerous free bacteria that were surrounded by a microcapsule were seen in the brain (Fig. 6). This microcapsule was limited to brain (free and intracellular bacteria) and was not seen in the other infected tissues.
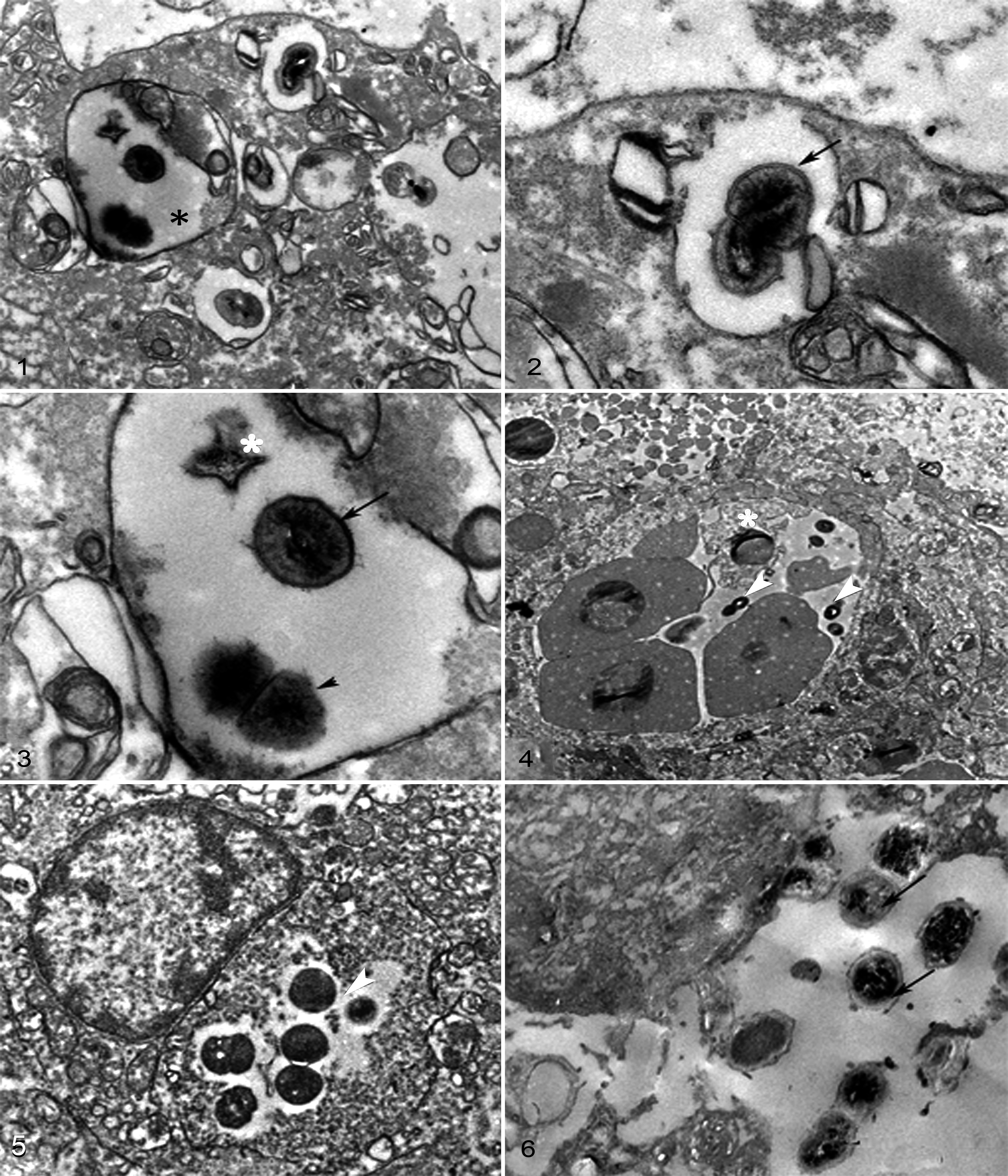
One of the most devastating diseases in warm water finfish aquaculture is caused by S. iniae. The first worldwide isolation of S. iniae in cultured red porgy, as well as the first European isolation in gilthead seabream, was reported recently. 3 The strain causing this outbreak, S. iniae IUSA-1, was used in this study to reproduce the disease and scrutinize injuries by TEM analysis in gilthead seabream and red porgy. The lesions are similar in both fish, despite the difference in the rate mortality. Infected fish presented clinical signs and gross lesions described in other streptococcal infections. 2 These lesions can be correlated with the clinical symptoms of lethargy and loss of orientation seen in our study.
The ultrastructural study revealed the presence and persistence of this bacterium in the phagosome within macrophages located in the lesions, which may facilitate its hematological dissemination. 10 S. iniae remained intact within the phagocytic vacuoles, and bacterial replication was evident as described for Streptococcus pyogenes. 6 In our study, we indicate that the inoculated strain, S. iniae IUSA-1, is capable of crossing the blood-brain barrier as free bacteria or associated with monocytes or phagocytes, which has been described previously in the genus Streptococcus. 1
Previous in vitro studies have demonstrated that S. iniae IUSA-1 invades fibroblastic cells of fish and that once bacteria become intracellular, they can survive, replicate, and remain viable for many days. 4 The bacterium is capable of residing within the macrophages, using the host cells for dissemination, and this ability may play a significant role in the events following primary transepithelial translocation. 5 Bacteria located in the nervous system had morphological changes, expressing an extracellular microcapsule, described previously in S. iniae and S. pyogenes as an M-protein layer. 1,6 The presence of this microcapsule has been demonstrated in most serotypes of Streptococcus, which might be implicated in the pathogenicity of this organism, particularly its recognized antiphagocytic activities in the blood. 1,6
TEM indicated that the macrophage may provide a significant bacterial reservoir for continued infection and tissue injury. The regulation of virulence-related metabolic factors such as capsular polysaccharides and exopolysaccharides synthesis can play a critical role in bacterial survival strategies.
Footnotes
Acknowledgements
We thank the Spanish Agency for International Development Co-operation (AECID) by their program of PhD scholarships for foreign students for F. El Aamri, as well as Dr Javier Roo (ICCM) for making animals available for the experiments used in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
