Abstract
The virulence of morbilliviruses for toothed whales (odontocetes) appears to differ according to host species. In 4 species of odontocetes, morbilliviruses are highly virulent, causing large-scale epizootics with high mortality. In 8 other species of odontocetes, including white-beaked dolphins (Lagenorhynchus albirostris), morbilliviruses have been found as an incidental infection. In these species, the virulence of morbilliviruses is not clear. Therefore, the admission of 2 white-beaked dolphins with morbillivirus infection into a rehabilitation center provided a unique opportunity to investigate the virulence of morbillivirus in this species. By phylogenetic analysis, the morbilliviruses in both animals were identified as a dolphin morbillivirus (DMV) most closely related to that detected in a white-beaked dolphin in Germany in 2007. Both animals were examined clinically and pathologically. Case No. 1 had a chronic neural DMV infection, characterized by polioencephalitis in the cerebrum and morbillivirus antigen expression limited to neurons and glial cells. Surprisingly, no nervous signs were observed in this animal during the 6 months before death. Case No. 2 had a subacute systemic DMV infection, characterized by interstitial pneumonia, leucopenia, lymphoid depletion, and DMV antigen expression in mononuclear cells and syncytia in the lung and in mononuclear cells in multiple lymphoid organs. Cause of death was not attributed to DMV infection in either animal. DMV was not detected in 2 contemporaneously stranded white-beaked dolphins. Stranding rate did not increase in the region. These results suggest that DMV is not highly virulent for white-beaked dolphins.
Keywords
Morbilliviruses are recently detected pathogens of marine mammals that cause epizootics with high mortalities. Virulence and susceptibility for morbillivirus as well as epidemiology differ among species. For example, phocine distemper virus causes mass mortality in harbor seals (Phoca vitulina), whereas gray seals (Halichoerus grypus) do not develop clinical signs of disease. 27 For many odontocete species, including the white-beaked dolphin (Lagenorhynchus albirostris), virulence and epidemiology of morbillivirus are unclear.
The virulence of morbillivirus for odontocetes appears to differ according to host species. They have been responsible for epizootics with high mortalities in bottlenose dolphins (Tursiops truncatus), common dolphins (Delphinus delphis), long-finned pilot whales (Globicephala melas), and striped dolphins (Stenella coeruleoalba).3,8,12,22,30 Morbillivirus infection causes lesions in odontocetes in 2 ways. First, it is a lymphotropic virus, which is highly immunosuppressive and thereby allows preexisting infections to be exacerbated and secondary infections to spread. Second, from the lymphoid tissues, the virus may spread to other tissues, such as the brain and the epithelia of multiple organs, where it is characterized by viral inclusions both in the cytoplasm and the nucleus. Particularly in the brain, infection is also associated with inflammation, necrosis, and demyelinization. 9 Clinical signs in cetaceans have been observed in striped dolphins and common dolphins. In striped dolphins, poor body condition, disorientation, apathy, muscle tremors, abnormal respiratory rates, ulcers in the mouth, and increased parasite burden of the skin were found. In common dolphins, seizures, uncontrolled trembling, and dyspnea have been observed. 27 In contrast, to date, morbilliviruses have been observed as incidental pathogens in 3 species: harbor porpoises (Phocoena phocoena), 18 white-beaked dolphins,24,34 and a pygmy sperm whale (Kogia breviceps). 35 In addition, serologic evidence for contact with morbillivirus has been found in 5 species of odontocetes: Risso’s dolphin (Grampus griseus), Atlantic white-sided dolphins (Lagenorhynchus acutus), Fraser’s dolphins (Lagenodelphis hosei), Atlantic spotted dolphins (Stenella frontalis), and false killer whales (Pseudorca crassidens). 10 The virulence of morbillivirus for these latter 8 species, including the white-beaked dolphin, is not clear.
In odontocetes, morbillivirus infections have resulted in epizootics with high mortalities and have occurred as unique events in bottlenose dolphins and common dolphins involving a single species.3,21,22 In the Mediterranean Sea, recurrent epizootics have occurred in striped dolphins with simultaneous infection of long-finned pilot whales.8,12,30 In these epizootics, it is assumed an infection of an immunonaive population has occurred. The source of infection is not identified in any of the epizootics in odontocetes. Morbillivirus needs a large population to remain endemic. Infections of immunonaive populations should therefore be expected from large populations where morbillivirus is endemic. The population structure and serologic status of white-beaked dolphins is therefore important. White-beaked dolphins inhabit the North Atlantic Ocean, and based on genetic analysis, there are 2 separate populations in the eastern North Atlantic. 1 One large population resides in the North Norway-Barents Sea (up to 100 000 animals) and a smaller one occurs around the United Kingdom and in the North Sea (22 000 animals). Vagrant animals occur as far south as the Strait of Gibraltar and the Mediterranean Sea. 5 There is no knowledge of the epidemiology of morbillivirus in white-beaked dolphins.
The white-beaked dolphin is the second most common species to strand on the North Sea coasts of the Netherlands and adjacent countries. Autopsy programs exist and stranding data are collected in the Netherlands, Belgium, and Schleswig-Holstein. Schleswig-Holstein is the northernmost state of Germany, close to the Netherlands and the state where a white-beaked dolphin infected with dolphin morbillivirus (DMV) was found in 2007. 34 From 2000 to 2012, 4.6 animals stranded on average annually on these coasts. Around 25 of these carcasses were autopsied and evaluated for presence of morbillivirus (U. Siebert, T. Jauniaux, and T. Kuiken, personal communications, 2013).
In 2011, 2 white-beaked dolphins stranded alive on the Dutch coast and died or were euthanized after spending a period of time at the rehabilitation center SOS-Dolfijn in Harderwijk. The diagnosis of morbillivirus infection in both animals provided a unique opportunity to gain information both on clinical and pathological aspects of morbillivirus infection in this species.
The primary goal of our research was to fully investigate the virulence of morbillivirus in white-beaked dolphins. This report describes the clinical signs, gross autopsy, histology, immunohistochemistry, bacteriology, and serology of the 2 dolphins admitted into the rehabilitation center. Two more white-beaked dolphins stranded on the Belgian and Dutch coasts within a month of the last arrival in the rehabilitation center. We investigated samples of these last 2 animals for the presence of morbillivirus infection and found they were both uninfected. The secondary goal was to investigate the epidemiology of morbillivirus in white-beaked dolphins and other odontocetes by comparing the RNA sequence of the morbillivirus of white-beaked dolphins with RNA sequences of previously found odontocete morbilliviruses.
Materials and Methods
Signalment
Case No. 1 was a juvenile male that was 229 cm long and weighed 153 kg. It stranded alive on the island Ameland, the Netherlands, on June 12, 2011, and died on December 12, 2011, while in rehabilitation. Based on length and sexual immaturity, its age was between 4 and 8 years old. 14
Case No. 2 was a juvenile female that was 158 cm long and weighed 49 kg. It stranded alive at Den Helder, the Netherlands, on December 4, 2011, and was humanely killed the next day. Based on her length, it was estimated to be 18 months old. 14
Case No. 3 was a subadult male that was 257 cm long and weighed 255 kg. It stranded dead at Zoutelande, the Netherlands, on January 3, 2012. The carcass was fresh.
Case No. 4 was an adult female that was 211 cm long and weighed 240 kg. It stranded alive at Koksijde, Belgium, on December 17, 2011. Due to the presence of severe lesions, the animal was euthanized.
Clinical Evaluation
SOS-Dolfijn is a rehabilitation center for small odontocetes. It has two 50-m3 pools with fresh water from a local source, to which salt is added. Salinity was kept between 20 and 30 parts per thousand (ppt) until September 23, 2011, and between 25 and 35 ppt afterwards. Water treatment is by high-rate sand filtration with a turnover time of approximately 1 hour for the entire volume of each pool. Water temperature varies from 11 to 21°C. Upon admittance, new animals are subjected to a full veterinary examination by an experienced marine mammal veterinarian. The monitoring of complete blood count and clinical chemistry of blood samples is done daily after admittance and thereafter according to perceived necessity. In the first period of rehabilitation, animals are observed continuously day and night. Multiple observations on respiration rate, defecation, cramps, food intake, and body temperature are recorded. In addition, a log book is kept in which swimming behavior, alertness, and other observations that are presumed potentially relevant are recorded. As the animal improves, observation and care diminish to a minimum of 9 hours per day. Case No. 1 was continuously observed after admittance for 10 days. Case No. 1 was moved to a larger outdoor pool (120 m3) on September 14, 2011, in preparation for its release. Case No. 2 was continuously observed from admittance until euthanasia.
Autopsy, Histology, and Immunohistochemistry
Autopsies were done on case Nos. 1 and 2 according to a standard protocol. 19 The following tissues were sampled for histology: adrenal gland, bronchus, cerebellum, cerebrum, colon, duodenum, esophagus, forestomach, fundic stomach, gonads, heart, jejunum, kidney, liver, lung, mesenteric lymph node, muscle, pancreas, pulmonary lymph node, pyloric stomach, skin, spleen, thymus, thyroid, trachea, tracheobronchial lymph node, and urinary bladder. Tissue samples were fixed in 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. The 3-μm-thick sections were mounted on glass slides and stained with hematoxylin and eosin (HE) for light microscopy.
For detection of morbillivirus, 3-μm-thick sections of all tissues sampled for histology were stained with an immunohistochemical technique as follows. After antigen retrieval (boiling for 15 minutes in citric acid 10 mM, pH 6.0), slides were incubated with 3% hydrogen peroxide in phosphate-buffered saline solution (PBS) for 10 minutes to inactivate endogenous peroxidase. To block nonspecific antibodies, the sections were incubated with 0.1% bovine serum albumin (BSA) in PBS for 10 minutes. Then the slides were incubated with a monoclonal antibody directed against the nucleoprotein of canine distemper virus (MoAb CDV-NP isotype IgG2b; VMRD, Pullman, WA) 1:400 in PBS/0.1% BSA for 1 hour at room temperature. This antibody is known to cross-react with DMV antigen and has been used previously in DMV-infected tissues of cetaceans. 26 After being washed with PBS-Tween, the sections were incubated for 30 minutes at room temperature with a biotinylated secondary antibody (α-mouse-bio; DAKO, Glostrup, Denmark), 1:100 in PBS/0.1% BSA. After washing, the slides were incubated with ABComplex (DAKO) for 1 hour at room temperature. Antibody binding was visualized using 3-amino-9-ethylcarbazole (AEC) and hydrogen peroxide. The sections were counterstained with hematoxylin. In the staining procedure, an isotype IgG2b control was included as a negative control and a sample of the prostate of a canine distemper virus–infected Caspian seal (Pusa caspica) was included as a positive control.
Bacteriology
For bacteriological examination, samples of lung, kidney, liver, spleen, pulmonary lymph node, and adrenal gland were frozen at –20°C and transferred on dry ice to Inverness, Scotland, where they were cultured according to a standard protocol. Briefly, each tissue was plated on Columbia sheep blood agar (CSBA) (Oxoid, Basingstoke, UK), MacConkey agar (Oxoid), and Farrells medium, 11 which was set up specifically for the recovery of Brucella ceti. 13 CSBA and Farrells plates were incubated aerobically plus 5% CO2 and examined daily for 14 days, whereas MacConkey agar plates were incubated aerobically without added CO2 at 37°C for 48 hours.
RT-PCR, Sequencing, and Phylogenetic Analysis
Tissue samples were fixed in RNAlater solution (Life Technologies Corporation, Carlsbad, CA) until analysis. The following tissues were tested: for case No. 1, adrenal gland, prescapular lymph node, trigeminal nerve, spleen, lung, kidney, urinary bladder, liver, and brain; for case No. 2, urinary bladder, kidney, trigeminal nerve, liver, spleen, lung, brain, adrenal gland, tongue, pulmonary lymph node, stomach ulcer, blowhole swab, tongue ulcer swab, and genital slit swab; for case No. 3, lung, urinary bladder, cerebellum, and kidney; and for case No. 4, lung, urinary bladder, kidney, spleen, pulmonary lymph node, and brain.
Total RNA was isolated from 300 μl of a 10% organ homogenate using the High Pure Viral Nucleic Acid Kit (Roche diagnostic GmbH, Mannheim, Germany), following the protocol provided by the manufacturer. For reverse transcriptase polymerase chain reaction (RT-PCR), morbillivirus-specific primers P1: 5′-ATGTTTATGATCACAGCGGT-3′ and P2: 5′-ATTGGGTTGCACCACTTGTC-3′ were used after first-strand synthesis with specific morbilliviral primers. PCR reactions were checked on agarose gels. Automated sequencing of RT-PCR fragments was performed on an ABI 3130XL genetic analyzer with the Big Dye terminator cycle sequencing kit (ABI, Applied Biosystems, Foster City, CA) using the RT-PCR primers P1 and P2. For identification of sequenced fragments, the BLAST option of the NCBI website was used (http://www.ncbi.nlm.nih.gov/). Phylogenetic analysis and construction of phylogenetic trees was conducted using MEGA version 5. 31
Serology
Serum samples were frozen at –20°C until analysis and tested for the presence of morbillivirus antibodies by a virus neutralization assay as previously described. 32 In brief, 2-fold dilutions of heat-inactivated serum samples were incubated with 100 median tissue culture infectious dose of CDV. After a 1-hour incubation at 37°C, 104 Vero cells were added to each well. After 4 to 6 days, the plates were checked for the presence of cytopathic effect (CPE). Antibody titers were expressed as the reciprocal of the highest serum dilution with complete inhibition of CPE. Antibody titers >10 were considered positive. 23,25
Results
History and Clinical Signs
Upon arrival on June 12, 2011, case No. 1 was in poor nutritional condition and needed continuous support. Frequent tremors and occasional cramps were noted during the first month. After the first week, the dolphin managed to stay afloat without support, and its swimming behavior gradually improved. Normal swimming was observed 5 weeks after admission. Initial complete blood count and clinical chemistry indicated the dolphin had a mild anemia, muscular damage, and an inflammation based on increased number of white blood cells (WBCs), fibrinogen level, and sedimentation rate. Gastroscopy revealed that foreign materials, mainly pieces of plastic, were present in the forestomach. The animal received various antimicrobial drugs until July 1 (18 days postadmission), when it was considered healed from the previously noted inflammation. By July 22 (40 days postadmission, 21 days after initial treatment had stopped), WBC numbers had increased again. A regular tremor of the dorsal fin and signs of pneumonia, notably inactivity and forced labored breathing with sporadic exudates upon expiration, were noted. Antibiotic therapy was restarted and clinical signs subsided after 17 days. WBC levels normalized toward the end of August. Subsequent to the suspected pneumonia, a dermatitis developed in September, and antibiotic treatment continued until the skin appeared healed by December 7. The dermatitis was characterized by epidermal damage, which was irregular, multifocal to coalescing across the entire body with foci on the peduncle and head, around the dorsal fin, and between the 2 pectoral fins at the time of its severest manifestation. The dermatitis was not associated with the introduction of a new medication, nor did it improve after withdrawal of a medication. It disappeared during long-term antibiotic treatment. The animal had active behavior (Fig. 1), interest in its environment, spontaneous frequent vocalizations, and ravenous appetite with the exception of the first 2 weeks after arrival and during the start of the episode of presumed pneumonia in mid July. On December 7, it was considered completely healthy and the final preparations for release were started, including withdrawal of antibiotics. However, on December 11, it had a dramatic drop in appetite, and antibiotic treatment was resumed. The next day, the pool was drained for diagnostic examination and treatment of the dolphin, which received intramuscular injections with midazolam (10 mg) and cefovecin (1280 mg). During these injections, the animal arched its back, stopped breathing, and died.

White-beaked dolphin, case No. 1.
Case No. 2 was in very poor nutritional condition on arrival, December 4, 2011. Its breathing frequency was close to normal during transport (20/5 minutes) but became high in the rehabilitation center (43/5 minutes). Complete blood count and clinical chemistry upon admittance showed a severe leucopenia, slightly increased sedimentation rate and fibrinogen level, a mild anemia, and muscular damage. It was provided part-time support to stay afloat. Force feeding was problematic, and upon the first attempt, fish gruel was noted to be expulsed from the blowhole. The animal had interest in its surroundings and the persons accompanying it and made many vocalizations. Because of the very poor nutritional condition, leucopenia, and poor future prospects, the animal was humanely killed on December 5.
Gross Autopsy
Case No. 1 was 229 cm long (straight line from tip of snout to notch between tail flukes) and 153 kg at autopsy. Case No. 1 was in good nutritional condition based on the lack of indentation in the neck and 20-mm-thick blubber layer lateral and just cranial to the dorsal fin. There was pulmonary edema, characterized by heavy, slightly firm lungs; thick yellow-white foam in the bronchi and trachea; and white viscous fluid in the trachea and bronchi. There was edema in the mediastinum dorsal to the heart over an area of 4 by 4 cm. The gastric (64 g, 0.04% wt/body wt), left prescapular (72 g, 0.05% wt/body wt), right prescapular (82 g, 0.05% wt/body wt), and pulmonary lymph nodes (78 g, 0.05% wt/body wt) appeared enlarged. In the forestomach, plastic debris and nylon strings were found. The compressed volume of this debris was approximately half a liter. The skin had an irregular surface due to fresh and old skin peelings mainly on the belly, peduncle, and dorsal fin.
Case No. 2 was 158 cm long (straight line from tip of snout to notch between tail flukes) and 49 kg at autopsy. Case No. 2 was in poor nutritional condition based on the indentation in the neck behind the skull. The blubber layer lateral and just cranial to the dorsal fin was 11 mm thick. Fibrinosuppurative bronchopneumonia was present, characterized by yellow viscous floccules (<2 mm diameter) in the trachea and bronchi. Pus extruded from airways upon pressure on the ventral parts of the caudal lung lobes. There was subdermal emphysema over 30% of the body surface. The gas bubbles were small (1–5 mm diameter) and coalesced sporadically to bubbles of 2 cm diameter. There were gas bubbles in the epidermis, which caused a split in the epidermis of 10 by 5 cm. An ulcer (4 mm diameter) was found on the ventral surface of the tongue, and on the junction between the fore- to second stomach, 4 ulcers (2–3 mm diameter) were observed in the fore-stomach. The left prescapular (23 g, 0.05% wt/body wt), right prescapular (23 g, 0.05% wt/body wt), and pulmonary (69 g, 0.15% wt/body wt), pericardial, esophageal, and subcutaneous lymph nodes appeared to be enlarged.
Histology and Immunohistochemistry
Case No. 1 had multifocal moderate polioencephalitis in the cerebrum, characterized by perivascular aggregation of lymphocytes (Fig. 2A), neuronal necrosis, neuronophagia, vacuolation of the neuropil, and diffuse gliosis (Fig. 3A). In addition to the above chronic lesion, there were acute lesions in other organs: moderate diffuse pulmonary edema, moderate multifocal gastric hemorrhage, and mild focal thymic hemorrhage.
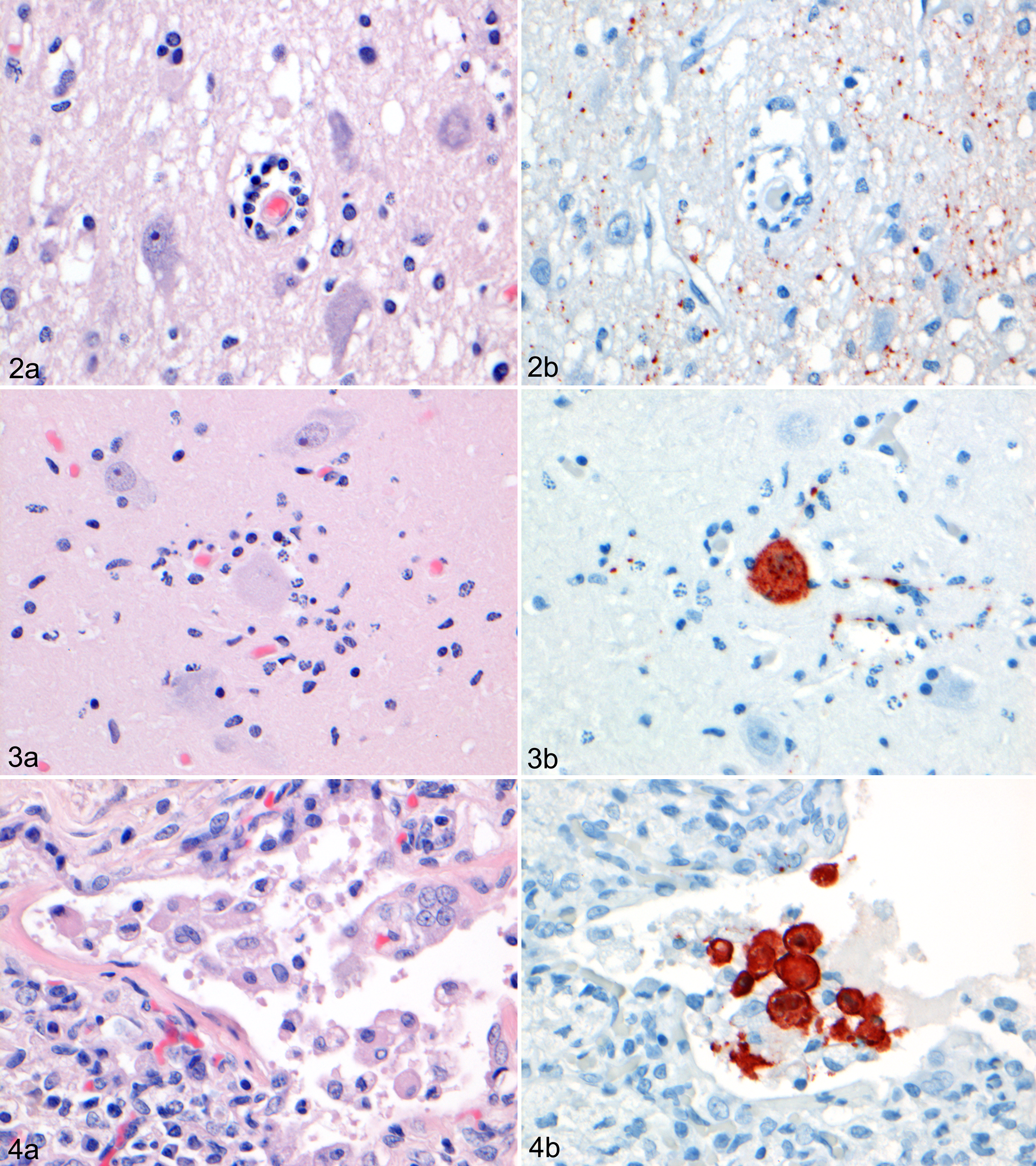
Morbillivirus antigen expression was limited to particulate matter in areas of polioencephalitis (Fig. 2B), as well as rare neurons and glial cells in the gray matter of the cerebrum (Fig. 3B). Most of these neurons appeared normal; others were degenerate and surrounded by phagocytes (neuronophagia).
Case No. 2 had multifocal mild interstitial pneumonia, characterized by multiple small aggregates of large mononuclear cells—some of which formed syncytia containing 2 or 3 nuclei—in the alveolar and bronchiolar lumina of the lung (Fig. 4A). There was marked lymphoid depletion in the spleen, pulmonary lymph node, and mesenteric lymph node. In contrast, the prescapular lymph node showed lymphoid hyperplasia, characterized by homogeneous fields of lymphocytes and lymphoblasts in the cortex.
Superimposed on the above small aggregates of mononuclear cells was a marked acute aspiration pneumonia, characterized by coalescing large foci of inflammation centered on bronchi and bronchioles and consisting of many neutrophils and macrophages, mixed with cellular debris, fibrin, and few erythrocytes. There were large segments of autolytic skeletal muscle (aspirated fish remains) and aggregates of bacilli and cocci in the lumina of affected bronchi and large bronchioles. No other significant lesions were detected in any of the tissues examined from case No. 1 or 2.
Morbillivirus antigen expression occurred in a moderate number of mononuclear cells and syncytia in alveolar and bronchiolar lumina of the lung (Fig. 4B), rare mononuclear cells in the red or white pulp of the spleen, rare mononuclear cells in the cortex of the pulmonary lymph node, rare mononuclear cells in the cortex of the mesenteric lymph node, and rare enterocytes in the mucosa and mononuclear cells in the lamina propria of the intestine. No morbillivirus antigen expression was detected in any of the other tissues examined from case No. 1 or 2.
RT-PCR, Sequencing, and Phylogenetic Analysis
The brain sample of case No. 1 contained morbilliviral RNA; the remaining samples (adrenal gland, prescapular lymph node, trigeminal nerve, spleen, lung, kidney, urinary bladder, and liver) were negative. The lung of case No. 2 contained morbilliviral RNA; the remaining samples (urinary bladder, kidney, spleen, pulmonary lymph node, and brain) were negative. All samples of case No. 3 (lung, urinary bladder, cerebellum, and kidney) and case No. 4 (lung, urinary bladder, kidney, spleen, pulmonary lymph node, and brain) were negative.
The morbilliviruses observed in case Nos. 1 and 2 were identified as DMV and were most closely related to one another and to a previously observed DMV in a white-beaked dolphin in 2007. The next closest identity of the DMV found in white-beaked dolphins was with the DMV observed in striped dolphins during the 1990 epizootic in the Mediterranean Sea. Of the 299 base pairs sequenced, the DMVs found in case Nos. 1 and 2 differed in 2 and 3 locations, respectively, from the DMV of a white-beaked dolphin in 2007 and in 5 locations from the DMV of striped dolphins in 1990 (Fig. 5).
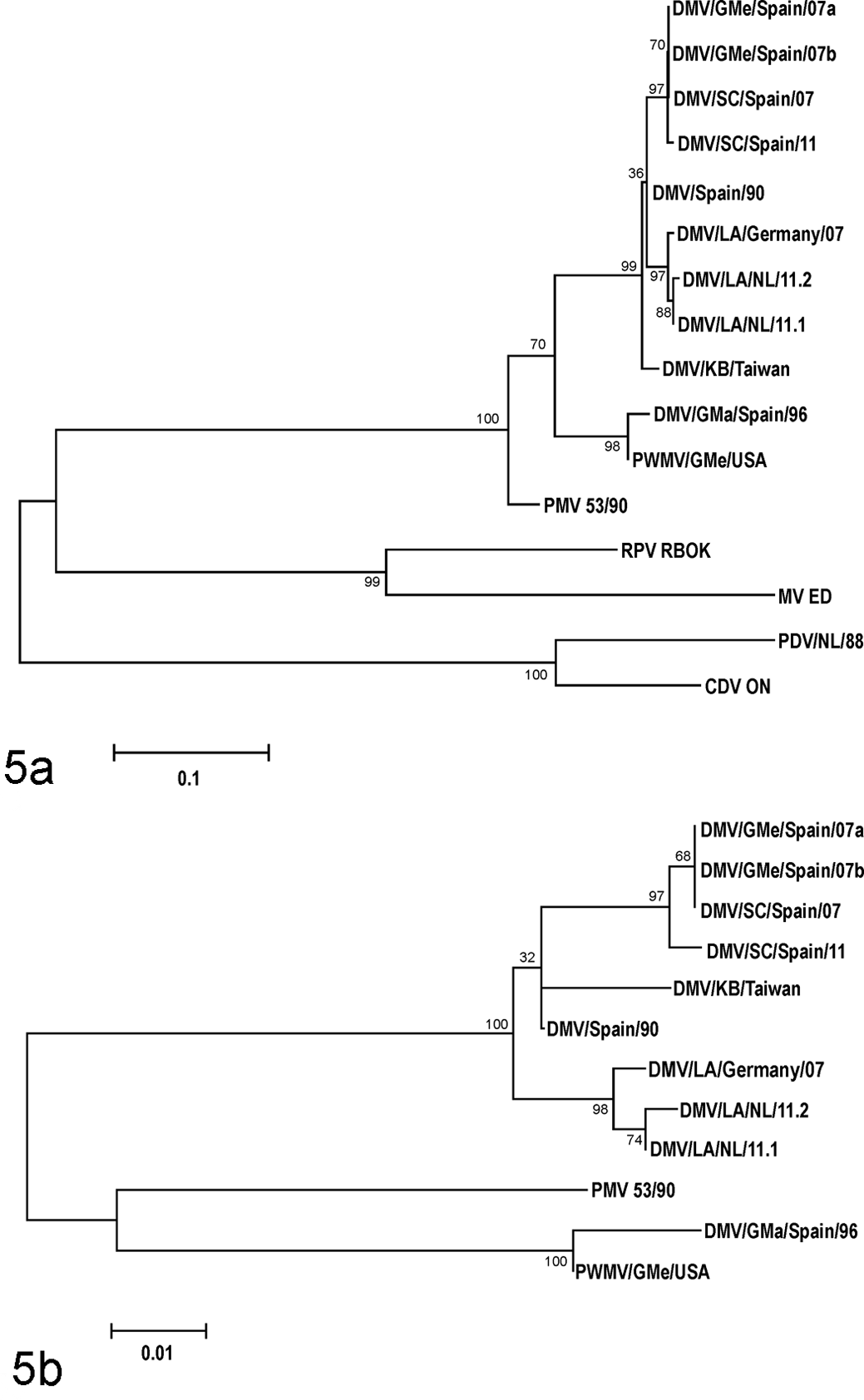
Maximum likelihood phylogram of a phosphoprotein (P) gene fragment of morbilliviruses (A) and cetacean morbilliviruses (B). Bootstrapping was performed with 1000 replicates using MEGA 5. 31 Genbank accession numbers are given in parentheses: MV ED, measles virus Edmonston strain (K01711); RPV RBOK, Rinderpest virus RBOK strain (X68311); CDV ON, canine distemper virus Onderstepoort strain (AF305419); PDV/NL/88, phocine distemper virus, the Netherlands, 1988 (AF525289); PMV 53/90, porpoise morbillivirus, isolate 53/90 (KF650727); DMV/GMe/Spain/07a, dolphin morbillivirus, long-finned pilot whale (Globicephala melas), Spain, 2007 (HQ829972); DMV/GMe/Spain/07b, dolphin morbillivirus, long-finned pilot whale, Spain, 2007 (EU039963); DMV/SC/Spain/07: dolphin morbillivirus, striped dolphin (Stenella coeruleoalba), Spain, 2007 (HQ829973); DMV/SC/Spain/11, dolphin morbillivirus, striped dolphin, Spain, 2011 (JN210891); DMV/SC/Spain/90, dolphin morbillivirus, striped dolphin, Spain, 1990 (Z47758); DMV/LA/Germany/07, dolphin morbillivirus, white-beaked dolphin (Lagenorhynchus albirostris), Germany, 2007 (EF451565); DMV/KB/Taiwan, dolphin morbillivirus, pygmy sperm whale (Kogia breviceps), Taiwan (AF333347); DMV/GMa/Spain/96, dolphin morbillivirus, short-finned pilot whale (Globicephala macrorhynchus), Canary Islands, 1996 (FJ842381); PWMV/GMe/USA, dolphin morbillivirus, long-finned pilot whale, New Jersey, USA (AF200817); DMV/LA/NL/11.1, dolphin morbillivirus, white-beaked dolphin case No. 1, the Netherlands, 2011 (KC888945); DMV/LA/NL/11.2, dolphin morbillivirus, white-beaked dolphin case No. 2, the Netherlands, 2011 (KC888946).
Bacteriology
In case No. 1, few Escherichia coli were recovered in the lung and the pulmonary lymph node, plus a mixed culture (few bacteria) in the prescapular lymph node. In case No. 2, a moderate number of E. coli and Enterococcus sp were recovered in the lung, a single E. coli in the liver, a few E. coli and a mixed culture (few bacteria) in the spleen, and a few coagulase-negative Staphylococcus sp in the pulmonary lymph node. Further identification of the mixed cultures with few bacteria was not pursued, because the colonies present were typical of organisms that most likely represented postmortem contamination.
Serology
Case No. 1 had an antibody titer against morbillivirus of 15 in samples obtained on June 13 and 14 and December 12, 2011. Case No. 2 had a titer of 40 on December 4 and 30 on December 5, 2011.
Primary Diagnoses
The primary diagnosis in case No. 1 was anaphylactic shock, based on temporal correlation of its sudden death with intramuscular midazolam and cefovecin injection, pulmonary edema, mediastinal edema, and acute hemorrhages in the stomach and thymus. The primary diagnosis in case No. 2 was aspiration pneumonia, based on observed problems with force feeding 1 day before euthanasia, fibrinosuppurative pneumonia associated with fish remains in airways, and mixed culture of E. coli and Enterococcus sp from the lung.
Discussion
In this study, we diagnosed DMV infection in 2 white-beaked dolphins, 1 subacute case and 1 chronic case. The different stages of pathogenesis were reflected by the differences in tissue tropism and associated lesions. We also ascertained that the DMV identified from these white-beaked dolphins was distinct from those from other odontocete species.
In case No. 1, DMV infection was diagnosed in the cerebrum and not in the cerebellum or brainstem, which corresponds with chronic cases of DMV infection in the central nervous system (CNS) of striped dolphins 4 years after the 1990 DMV epidemic in the Mediterranean Sea. 9 Surprisingly, despite the extensive DMV-associated polioencephalitis in case No. 1, behavior appeared completely normal (Fig. 1). Possibly the tremor of the dorsal fin noted at the start of its rehabilitation and during his period of illness from July 22 until August 8 was due to cerebral infection. If this animal had been autopsied without knowledge of its clinical history, the effects of the observed encephalitis would have been judged to be far graver. Throughout its entire stay in the rehabilitation center, continuous medication with antibiotics was necessary. Each attempt to stop resulted in overt clinical signs. It might well be that long-term immunosuppression by DMV infection was responsible for this dependence on antibiotics. Immunosuppression due to CDV infection in dogs may last well after virus clearance. 2 In macaques, measles virus targets memory T cells and follicular B cells, which causes temporary immunologic amnesia and explains the long-term immunosuppression. 6 In case No. 1, the antibody titer against morbillivirus remained relatively low, although a chronic CNS infection was present. This is contrary to what is found in other species, for example, in rats and humans.20,29 DMV antigen was observed in glial cells, which means the virus did not escape contact with the host’s immune system. A possible explanation is compromised immune response from morbillivirus infection, as has been suggested for phocine distemper virus infection in harbor seals, where out of 22 harbor seals diagnosed with phocine distemper virus infection by PCR and/or immunohistochemistry on tissues, only 8 had the IgG antibody to morbillivirus. This includes 6 seals that were positive in the brain, of which only 1 had IgG antibody to morbillivirus. 28 Case No. 1 was 6 months in the facility before it died and had close contact after initial quarantine with harbor porpoises. This raises the question if DMV infection might have occurred after admittance. Several observations make this an unlikely event. Neither DMV nor porpoise morbillivirus (PMV) have been observed in the 45-year history of the rehabilitation center. Porpoise morbillivirus is highly different from the virus strains found in white-beaked dolphins (Fig. 5). The only morbillivirus known to occur in harbor porpoises is PMV, which is distinct from the DMV isolates found in white-beaked dolphins (Fig. 5). Five harbor porpoises that died in the period when case No. 1 was present at the facility tested negative for morbillivirus by PCR on samples of lung, kidney, brain, and urinary bladder, while the remaining harbor porpoises tested negative for serum antibody to morbillivirus by virus neutralization (unpublished data). Based on the chronicity of its lesions, case No. 2 was in the facility too short to have acquired DMV infection there.
In case No. 2, DMV antigen and/or RNA was found in lung, intestine, spleen, and lymph nodes, which corresponds with the subacute systemic phase of morbillivirus infection in terrestrial species and in marine mammals.15,17 The marked lymphoid depletion in spleen and lymph nodes is well known as the primary effect of morbillivirus infection, resulting in immunosuppression and allowing facultative pathogens such as Bordetella bronchiseptica to cause severe disease. 27 Surprisingly, case No. 2 had no pathological evidence of disease from such facultative pathogens. It is noteworthy that the infection was over its peak. This was suggested by the hyperplasia of lymphocytes in the prescapular lymph node and limited virus antigen expression in combination with lymphoid depletion. Again it is noteworthy that observed antibody titers were low despite the suspected duration of infection of several weeks based on the above observations. In seals, a maximum antibody titer is developed 3 weeks after infection despite lymphoid depletion. 16
The potential relationship between DMV infection and live stranding in these 2 white-beaked dolphins is speculative. In case No. 1, the CNS infection may have affected feeding behavior (resulting in emaciation) and/or ability to navigate and orientate. In case No. 2, the systemic infection may have caused malaise, resulting in separation from the mother and the rest of the social group, and subsequently emaciation and disorientation. In both cases, these effects may have led to stranding.
These 2 cases, as well as previous cases of DMV infection in white-beaked dolphins, raise questions about the role of white-beaked dolphins in the epidemiology of DMV and about the virulence of DMV for white-beaked dolphins. Regarding epidemiology, our data, together with the previously reported individual strandings of white-beaked dolphins infected with DMV in the Netherlands in 1990 24 and on the North Sea coast of Germany in 2007, 34 indicate that DMV is recurrently present in the UK–North Sea population of white-beaked dolphins. Furthermore, phylogenetic analysis (Fig. 5) shows that the 3 DMV isolates from white-beaked dolphins in 2007 and 2011 had a common source, since they were more closely related to each other than to DMV isolates from other odontocetes, including striped dolphins in 2007 and 2011. Together, these findings suggest that white-beaked dolphins could be acting as a reservoir for this DMV. DMV observed in white-beaked dolphins in 2007 and 2011 is closely related to DMV observed in striped dolphins in 1990. Morbilliviruses are highly contagious, cause infections of short duration, and induce long-lasting immunity in survivors. Therefore, reservoir populations for morbilliviruses need to be large. 28 Potentially, the North Norway-Barents Sea population, with up to 100 000 individuals, could fulfill this role. From there, DMV might spread recurrently to the smaller UK–North Sea population of 22 000 individuals. We observed a high incidence of morbillivirus infection in a small sample from this population. Regular infection of the UK–North Sea population may induce immune protection and prevent large-scale epidemics.
Regarding virulence of DMV for white-beaked dolphins, there is no evidence that these 2 DMV cases were representatives of a major DMV epidemic in white-beaked dolphins. First, there were 6 months between the dates of stranding of case No. 1, which had a chronic infection, and case No. 2, which had a subacute infection. Second, 2 other white-beaked dolphins that stranded on the coasts of the Netherlands and Belgium in winter 2011–2012 (case Nos. 3 and 4) were not infected by DMV. Third, there was no reported increase of white-beaked dolphin strandings on the coasts of the Netherlands, Belgium, and Schleswig-Holstein, Germany, in 2011 (n = 5) or 2012 (n = 3)4,33 (U. Siebert, personal communication, 2013). Furthermore, DMV infection was not diagnosed as the proximate cause of death in either of the 2 white-beaked dolphin cases reported here. Together, these findings suggest that DMV is less virulent for white-beaked dolphins than, for example, for striped dolphins, in which major die-offs from DMV occurred in 1990 and 2007. 8,30 Species variation in susceptibility to morbillivirus infections is well known. For example, CDV is known to be more virulent for ferrets than for domestic dogs, 7 and phocine distemper virus is known to be more virulent for harbor seals than for gray seals. 27
In conclusion, we have demonstrated 2 forms of DMV infection in white-beaked dolphins: subacute systemic and persistent neural. The combined data on individual strandings of DMV-infected white-beaked dolphins on the coasts of the Netherlands and Germany indicate that the UK–North Sea population is recurrently infected with DMV without undergoing a major die-off and suggest that DMV is not highly virulent for white-beaked dolphins. There is a need for further research on DMV in this species to further clarify its epidemiology and virulence. Rehabilitation centers should be aware of the risk of introducing morbillivirus into their center when admitting white-beaked dolphins. Upon autopsy of stranded white-beaked dolphins, special attention should be paid to analysis for morbillivirus infection in multiple organs, including the CNS. The relationship between cause of death and postmortem evidence of a morbillivirus infection in incidental mortalities of odontocetes should be considered cautiously.
Footnotes
Acknowledgements
We thank the Dolfinarium in Harderwijk for their financial support of this research and all benefactors and volunteers of the association SOS Dolfijn for their support of the rehabilitation of stranded cetaceans.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
