Abstract
Six pregnant Standardbred mares aged between 6 and 14 years were gavaged with 50 g or 100 g of suspended emulsified whole Processionary caterpillars (Ochrogaster lunifer) for 5 days during 2 experiments undertaken to study the etiology of equine amnionitis and fetal loss (EAFL). The 6 treated mares and 1 untreated mare were between 128 and 252 days gestation. Mare 1 (untreated) was euthanized on day 5 of the treatment period, while the treated mares were euthanized on days 2, 4, 8, 10, 12, and 24 days from their first treatment. Caterpillar setae were not found in the untreated mare. Setal fragments were present in all regions of the gastrointestinal tracts in all treated mares, the uteri and mesenteric lymph nodes of 5 mares, and the liver of 4 mares. Acute gastroenteritis of varying severity was present in all treated mares, and 5 of 6 mares had acute colitis and endometritis. Focal hyperplastic serositis was found in the duodenum, cecum, dorsal colon, and uteri of various mares occasionally with embedded setal fragments. Setal invasion of the mucosa evoked a range of lesions including superficial erosion to deep ulceration. Inflammation in deeper tissues ranged from unapparent to neutrophilic (microabscesses), eosinophilic, or mononuclear (microgranulomas). The finding of setal fragments within the uterus of experimental mares suggests that direct migration of setal fragments acting as a bacterial vector is a likely mechanism for the bacterial abortions that characterize equine amnionitis and fetal loss.
A previously unrecognized form of equine abortion was reported in 2004 from the Hunter Valley region of New South Wales, one of the major areas of Thoroughbred breeding in Australia. The cases were related by specific histological and microbiological findings in the aborted fetuses. Equine amnionitis and fetal loss (EAFL) was the term adopted to describe this syndrome. 42 Systematic investigations were undertaken to determine a cause, and similarities with mare reproductive loss syndrome (MRLS) resulting from ingestion of the Eastern tent caterpillar (Malacosoma americanum) were noted. 33,47 Eastern tent caterpillars (ETC) are not found in Australia, but exposure to Processionary caterpillars (Ochrogaster lunifer), White cedar moth caterpillars (Leptocneria reducta), Browntail mistletoe moth caterpillars (Euproctis edwardsii), and possibly the plant, pennyroyal (Mentha pulegium) were identified as putative risk factors for EAFL. 32 It has now been shown that experimental ingestion of whole or exoskeleton of Processionary caterpillars (PC) by pregnant mares induces fetal loss. 9,10 It is postulated that inadvertent ingestion of caterpillar material by horses occurs during normal grazing of pastures contaminated with shed exoskeletons from aged nests. 10
Necropsy of field cases revealed 2 forms of EAFL, acute midterm abortions and late-term abortions with chronic changes. 42 Acute abortions generally have no gross or histological lesions, with only isolation of atypical bacteria, often environmental coryneforms. 42 Chronic lesions encompass the extraembryonic coelom of the allantochorion, allantois, amnion, and umbilical cord. Grossly these manifest as thickening of the allantoic surface of the allantochorion, amnion, and umbilical cord. Lesions on the amnion are often restricted to a 30-cm radius around the umbilical cord and amniotic vessel convergence and can include thickening, increased opacity, or more chronic nodular thickening, increased vascular tortuosity, and diphtheritic membrane extending from the amnion down the amniotic section of the umbilical cord. 42 Histologically, there can be acute amnionitis characterized by variable amounts of fibrin and neutrophils within the coelom, neutrophil infiltration of the allantoic surface, and vasculitis with thrombosis. Acute funisitis manifests as superficial infiltration of the umbilical cord by neutrophils, acute neutrophilic vasculitis of surface vessels, edema, fibrin, and inflammatory cells within the coelom. Acute changes within the allantois involve patchy neutrophil margination and migration within the allantoic vessels, variable fibrin, and neutrophil exudation within the extraembryonic coelom and/or with neutrophil infiltration of the allantoic surface. 42 Chorionic involvement depends on the degree of inflammation affecting the coelom and allantois. 42 Chronic inflammation can vary but generally includes the presence within the stroma of the amnion, umbilical cord, allantois, coelom and chorionic stroma of increased numbers of plasma cells, lymphocytes, and macrophages and lesser neutrophils. Some degree of capillary and fibroblast proliferation is present either multifocally or diffusely within the chorionic stroma and/or allantois. The case definition for EAFL stipulates that other causes of infectious abortion must be ruled out; atypical bacteria cultured from lung, stomach contents, and/or fetal membranes and characteristic histological lesions of amnionitis and funisitis must be present to be considered a confirmed case of EAFL. 42
Caterpillar-induced abortion in mares was first investigated in Kentucky in the USA in 2001 following large numbers of abortions with unusual histological lesions in the fetal membranes and the isolation of atypical bacteria from fetal tissues. 34 Mare reproductive loss syndrome is the name given to this syndrome and has been shown to encompass a range of presentations including early fetal loss, late fetal loss, fibrinous pericarditis, unilateral uveitis, meningoencephalitis, and perinatal death in weak full-term foals. 7,34 As with EAFL there is no single gross or histological lesion that characterizes MRLS, but there is frequent involvement of the allantoic portion of the allantochorion, umbilical cord, and amnion and some degree of fetal pneumonia. A wide range of bacteria were cultured from the aborted fetuses with Actinobacillus spp. and non-beta haemolytic Streptococcus spp. predominating. 34 Uveitis and pericarditis have not been reported to be associated with EAFL, and the bacteria most often associated with EAFL are environmental coryneforms. 41,42
The Eastern tent caterpillar, Malacosoma americanum of the Lepidopteron family Lasciocampidae, is found throughout the eastern United States and Canada, associated with trees of the Rosaceae family. 15,47 Contact with these caterpillars can cause a mild irritant dermatitis, and feeding dried ETC as a source of protein in rats caused mild toxicity. 47 The setae (hairs) of ETC measure approximately 20 µm in diameter with minute barbs along the shaft. 38 The processionary caterpillars, members of the subfamily Thaumetopoeidae, have several urticating species found worldwide, including in Australia. 15 Members have true setae, 100 to 500 µm long and 3 to 10 µm in width with barbs along the shaft and a hollow interior located on the ventral surface in setae fields termed mirrors. 2 There are approximately 650,000 to 1,000,000 setae per caterpillar. 2 With each larval molt the setae are shed with the old exoskeleton and remain in the environment within nest material and soil, allowing exposure long after the caterpillars are gone. 2 Research on Thaumetopoea sp. shows its setae are composed of a chitin skeleton with a protein matrix covered by tannin-bound lipoproteins, wax, and mucopolysaccharides. 2 Syndromes reported after contact with processionary caterpillars include erucism (dermal urticaria), ophthalmia nodosa, and lepidopterism (systemic illness with various manifestations). 15
Experimental investigation into the effects of the ETC on laboratory animals showed the caterpillar setae caused intestinal granulomas when fed to rats and pregnant sows. 34 However, it is still unclear how ingestion of caterpillars by mares causes abortion or the effect, if any, on the mare’s gastrointestinal tract (GIT) and other organs. The pathological effects of ETC in mares have not been reported except for limited observations in 1 mare. 34 Proposed mechanisms for abortion associated with MRLS include penetration of the gastrointestinal wall by caterpillar setae resulting in bacteraemia and localization in the fetus or a toxin in the caterpillar setae that is detrimental to the fetus or placenta. 34,38 Either mechanism may result in fetal infection and ultimately lead to fetal death and/or severe stress and subsequent abortion. These mechanisms could also apply to EAFL. The objective of this study was to further elucidate the pathogenesis of EAFL by describing the histopathology of mares during midgestation following gavage with PC and euthanasia on sequential days during and after treatment. The implications for the fetal-placental unit are described in the following article. 40
Materials and Methods
The Standardbred mares examined in this study participated in an experimental program that sought to establish the relationship between PC ingestion and the occurrence of EAFL. Two experiments were undertaken using different amounts of whole caterpillar emulsified and suspended in distilled water with a laboratory blender. Each mare was gavaged with either 500 ml distilled water (controls) or 500 ml of the emulsified caterpillar suspension into the stomach using a nasogastric tube. The mares had been mated with the same Australian Stockhorse stallion and were all exposed to PC in midgestation (128–252 days). These experiments were reported in part previously. 10,11
Briefly, in Experiment 1, 2 groups of 4 mares were gavaged with either distilled water (controls) or 100 g of emulsified whole PC daily for 5 days. Experiment 2 consisted of 3 groups of mares that were gavaged with distilled water (Group A control mares, n = 6) or 50 g of emulsified whole PC (Group B, n = 6 and Group C, n = 4), once daily for 5 days. All mares in both experiments were naïve without previous exposure to caterpillars with the exception of mares in Group C. In their previous pregnancy, the mares in Group C had been gavaged daily for 5 days with 500 ml of distilled water containing either 1 g or 2 g of suspended caterpillar exoskeleton. Following that pregnancy, these mares were rebred and were back in foal when recruited for Experiment 2. Prior to the trial the mares were dewormed with an ivermectin product according to the manufacturer’s instructions. During the trial the mares were monitored daily by clinical examination, hematology, and ultrasonography. 10 The University of Queensland Animal Ethics Committee approved these studies and additionally allowed 1 healthy pregnant untreated and 3 treated pregnant mares to be euthanized as a limited extension of the second experiment. The untreated mare was included to provide background information to the treated mares. All mares had been managed together in the same research herd for the preceding 12 months.
Study Population
The mares examined in the study are detailed in Table 1. The untreated mare No. 1 was randomly selected from Experiment 2–Group A controls. Mare Nos. 2, 3, and 5 participated in Experiment 2. Mare Nos. 2 and 3 were from Group B and mare No. 5 was from Group C. These mares were euthanized on sequential days as per the study protocol. Mare Nos. 4 and 6 were from Experiment 1 and mare No. 7 was from Experiment 2–Group C. These mares were euthanized for humane reasons as detailed in Table 1. The fetuses of mare Nos. 1, 2, and 4 were alive at the time of euthanasia. The fetus of mare No. 3 had no detectable heartbeat on ultrasound 2 days prior to euthanasia but had not yet been expelled. Mare Nos. 5, 6, and 7 aborted their fetuses prior to euthanasia. Mare No. 7 retained the fetal membranes for 12 hours post abortion. A number of the mares experienced a transient urticarial reaction following gavage with caterpillar slurry (Table 1).
Study Population and Treatment Outcomes of Pregnant Mares Euthanised for Histopathological Examination After Experimental Gavage With Processionary Caterpillars.a
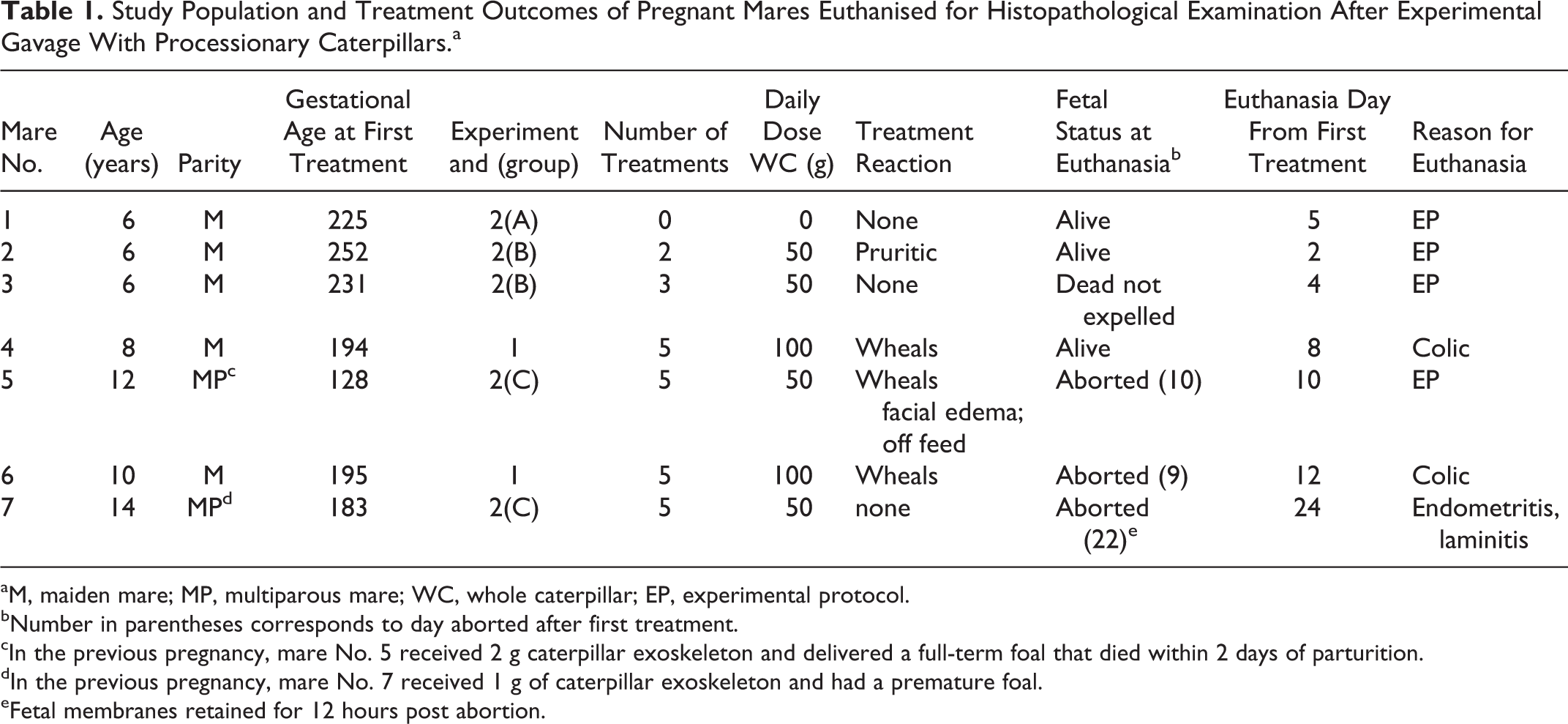
aM, maiden mare; MP, multiparous mare; WC, whole caterpillar; EP, experimental protocol.
bNumber in parentheses corresponds to day aborted after first treatment.
cIn the previous pregnancy, mare No. 5 received 2 g caterpillar exoskeleton and delivered a full-term foal that died within 2 days of parturition.
dIn the previous pregnancy, mare No. 7 received 1 g of caterpillar exoskeleton and had a premature foal.
eFetal membranes retained for 12 hours post abortion.
Postmortem Examination and Sample Collection
Postmortems were carried out in a systematic manner for each mare as per standard procedure. Samples required for other studies (fluids and tissues for culture and EHV-1 exclusion) were taken prior to any tissues for histopathology. Mares were euthanized with an overdose of pentobarbital sodium (Lundbec Inc) and laid in right lateral recumbency. Organ systems were removed to be sampled independently on a stainless steel surface. Labels were attached to each tissue corresponding to the preplanned areas of the gastrointestinal and reproductive tracts to be sampled. Approximately 1-cm pieces of tissue were taken from 6 areas of the stomach: the junction of the cardiac sphincter and the nonglandular stomach, the nonglandular stomach approximately 20 cm from the margo plicatus, the margo plicatus at the junction of the nonglandular and glandular stomach, glandular stomach at the curvature, glandular stomach approximately 20 cm from the pylorus, and junction of the glandular stomach and pylorus. Sequential areas of the gastrointestinal tract were taken in 3 areas from each section (proximal, middle, and distal) of the esophagus, duodenum, ileum, cecum, left and right dorsal colon, left and right ventral colon, and small colon. The jejunum was sampled in sequential 30- to 50-cm segments. Representative samples were also taken of organs (liver, spleen, lungs, kidney, heart,), lymph nodes (tonsil, mediastinal, mesenteric), and reproductive tract (ovary, uterus, cervix). Variable tissues were not available from treated mare Nos. 4 and 6 due to the unexpected outcome of colic and euthanasia without a necropsy protocol specific for these experiments in place. Total parasite burden was not quantified for the control or experimental mares. Necropsy and histopathology of the in utero or aborted fetuses and placental membranes are described in the companion article. 40
Histopathology
Collected tissue samples were fixed in phosphate-buffered 10% formalin for 48 hours or longer. Thin (3–4 mm) pieces of tissue were embedded in paraffin, sectioned at 6 µm, and stained with Wrights modification of the haematoxylin and eosin (HE). Selected sections were stained with Toluidine blue (TB). All organs were examined for inflammation, the presence of setae, and any reaction to the presence of setae. Setal fragment numbers were semiquantified as follows: no setae found (neg), 1 to 5 setal fragments (low), 6 to 10 setal fragments (moderate), or >10 setal fragments (high). Any other abnormalities were noted. Well-established descriptions in the literature were used as a comparison to assess normal changes and normal cellular infiltrates in tissues 1,12,17,21,23,27,31,35,37,45,48 along with tissues from the untreated mare. Additional specific sections were stained with TB for mast cell presence within the tissues or with Gram stain to confirm microorganism morphology if detected.
For comparison, a whole PC collected for use in the aforementioned experiments was placed in 10% formalin for 48 hours. The caterpillar was selected beginning at the head in sequential 3-mm sections and processed as described previously. Slides were stained using HE and TB and used as a standard for setae morphology and staining characteristics.
Results
Caterpillar Setae
Sections of the caterpillar showed large numbers of true setae present within multiple ventral depressions in the cuticle, suggestive of the described mirrors (Fig. 1). These setae were golden and refractile with sharp tapered ends measuring approximately 120 µm in length. Short angled barbs were present along the shaft, longer at the anterior end to barely present at the posterior end (Fig. 2). Setae diameter ranged from 3 to 10 µm with a hollow interior. Many setae were fragmented and lost during processing. Multiple locations on the outer cuticle had fractured shafts of larger hair-like structures up to 35 µm in diameter with golden parallel walls and central eosinophilic interior (not illustrated).
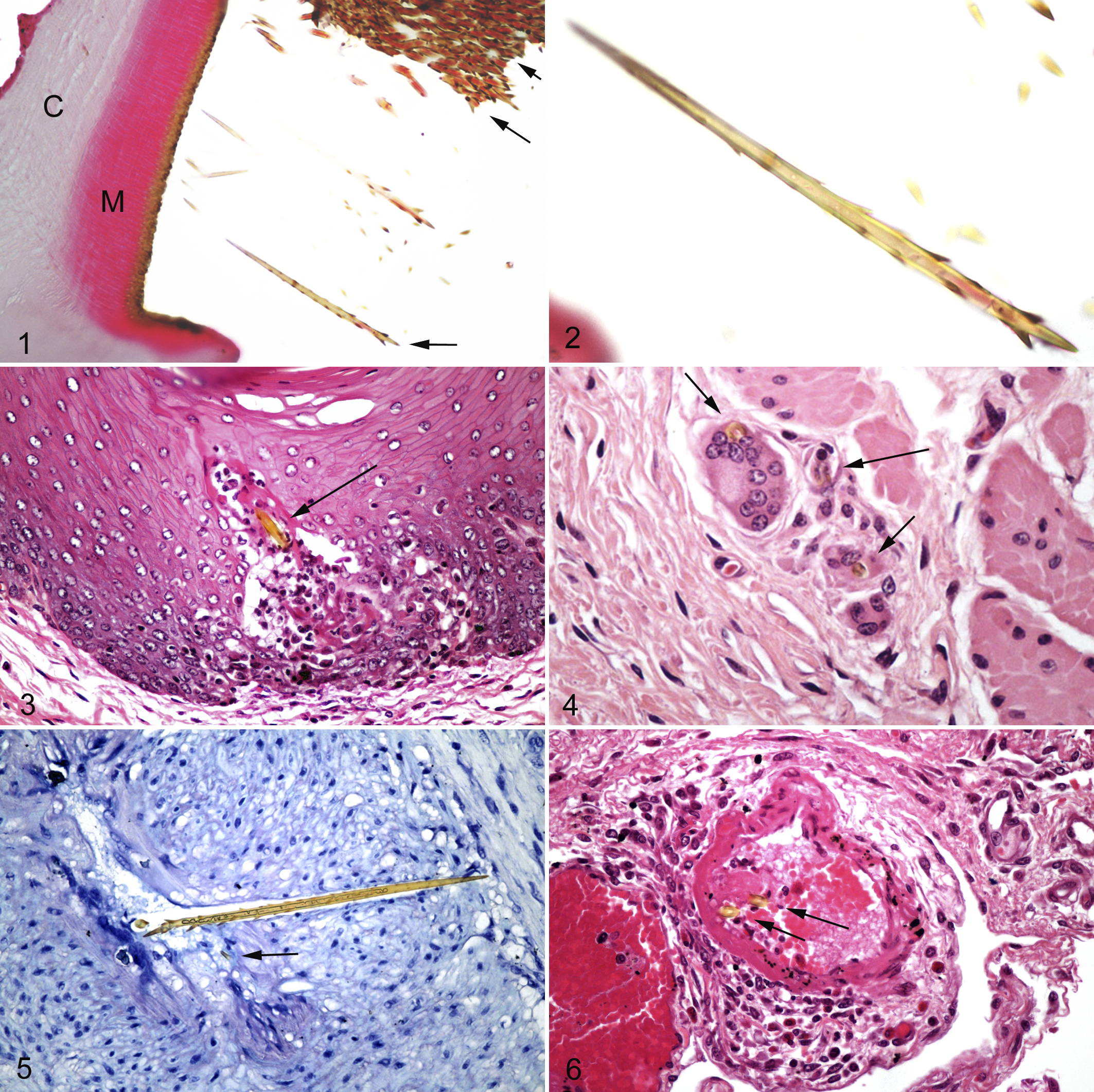
Setae were found throughout the tissues of exposed mares frequently accompanied by an inflammatory reaction. Detailed tissue histopathology of collected tissues is described in the following sections, and a summary of relative numbers for setal fragments found in tissues is shown in Table 2. Setal fragment numbers on average were highest in the large colon followed by the glandular stomach. The nonglandular stomach and cecum had moderate numbers while the small intestine, small colon, uterus, and lymph nodes had low numbers of fragments. Setal fragments were not found in the spleen or kidney of any mare where tissue was available. Mares dosed with 100 g of whole caterpillar had greater numbers of setal fragments in the gastrointestinal tract and uterus (mare No. 6). Setae fragment size ranged from the entire barb of greater than 100 µm in length to the smallest setal fragment of 7 µm in length with fragments in cross-section, transverse, and oblique orientation. Tissues around the anterior end of lengthy setal fragments frequently had a “pinched” appearance with occasional protruding barbs. Results for inflammation within selected tissues of the treated mares are summarized in Table 3.
Relative Setal Fragment Numbers Within Tissues of Pregnant Mares After Experimental Gavage With Processionary Caterpillars.a

aWC, whole caterpillar; NG, nonglandular; GL, glandular; NA, not available; 0, none; +, 1 to 5 setal fragments in the tissues; ++, 6 to 10 setal fragments in tissue; +++, >10 setal fragments in the tissues.
Distribution of Inflammation and Setal Fragment Locations Within the Tissues of Pregnant Mares After Experimental Gavage With Processionary Caterpillars.a

aWC, whole caterpillar; GIT, gastrointestinal tract from the stomach to the small colon; Inflam, inflammation; NA, tissue not available; St, stomach including nonglandular and glandular regions; SI, small intestine; LI, large intestine including cecum, dorsal and ventral colon, and small colon; Lv, liver; LN, lymph node; Ut, uterus; acute, presence of neutrophils in varying numbers within the epithelium or mucosa; Neg, negative.
b+, scattered neutrophils between the microcaruncle crypts; ++, diffuse neutrophils in the stratum compactum and between the microcaruncle crypts; +++, diffuse large numbers of neutrophils in the stratum compactum with extension into the stratum spongiosum to the level of the deep endometrial glands.
cControl mare—not treated.
dMare euthanized due to severe colic while still pregnant.
eMare euthanized due to severe colic after aborting 9 days after first treatment with Processionary caterpillar.
fMare euthanized due to endometritis and laminitis after aborting 22 days after first treatment with Processionary caterpillar.
Untreated Mare
Setae were not found in mare No. 1. Cell populations of the intestinal tract were similar to normal equine tissues without presence of neutrophils in the mucosa or submucosa. 23,31 Draschia megastoma was identified within the lumen of the stomach, and ciliates were present within the intestinal lumen. 20 Individual small strongyle larvae were present in the deep mucosa of the duodenum and left dorsal colon and another on the mucosal surface of the cecum with mild mucosal disruption. Eosinophil numbers varied from 1 to 5/hpf in the deep mucosa of the small intestine to >30/hpf in the large colon. Submucosal eosinophils varied from 5 to 20 in the small intestine to 30 to 50/hpf in the large colon. Eosinophils were generally confined to submucosal areas directly adjacent to the muscularis mucosae. Mare No. 1 was 260 days pregnant at the time of euthanasia, and microcotyledon development was equivalent to descriptions of mares at this stage of pregnancy. 1 Neutrophils were not a feature of the uterine tissue. Mast cells were present in low numbers within the duodenum (1 per 10–400× fields). Within the left dorsal colon however, mast cells were within the mucosa at a rate of approximately 20/400× field and in the submucosa at >10/hpf. The uterus of the control mare exhibited rare mast cells primarily located within the deepest areas of the stratum spongiosum adjacent to vessels.
Treated Mares
Gross Lesions
At necropsy, mare No. 5 had linear erosions of the distal esophagus as well as multifocal to coalescing ulcerations of the nonglandular stomach focusing around the cardiac sphincter. Mare Nos. 4 and 6 exhibited mild to moderate hyperemia of the small and large intestine.
Esophagus
Esophageal tissue was not collected from mare Nos. 4 and 6. Mare No. 3 was the only mare with setal fragments embedded in the epithelium with reactions ranging from unapparent to focal necrosis to microabscess formation around the setae (Fig. 3). Lesions in the remaining mares included no obvious abnormalities (mare No. 2), multifocal areas of bulla formation and ballooning degeneration of the superficial epithelium (mare No. 7), and multifocal extensive ulceration with complete loss of the epithelium to the level of the lamina propria with neutrophilic inflammation and bacterial colonization of the deeper layers (mare No. 5).
Nonglandular Stomach
Severe erosions involving incomplete superficial epithelial loss and deep ulcerations to the level of the lamina propria with neutrophilic inflammation were present in 3 mares (mare Nos. 4, 5, 6) with setal fragments in all tissue layers. Mare No. 4 had multifocal microabscesses in the muscularis externa. Bacterial colonization of the superficial epithelium without evidence of inflammation was present in 2 mares (mare Nos. 2 and 3) of which 1 (mare No. 2) had multifocal superficial erosions with setae present in both the epithelium and underlying lamina propria. The remaining mare (mare No. 7) had a mixed population of lymphocytes and plasma cells with rare neutrophils within the lamina propria without setal fragments.
Glandular Stomach
Setae fragments and variable inflammation were present in all mares. Setae were located in all layers of the glandular stomach from the mucosa to the serosa with partial vascular penetration to intraluminal presence. Reaction to setae was frequently mononuclear with fibrin, lymphocytes, and macrophages as well as occasional multinucleated giant cells (mare Nos. 4 and 7) (Fig. 4). There were low numbers of neutrophils around the deeper pits (mare Nos. 2, 3, 6, 7) and mild epithelial necrosis involving low numbers of neutrophils (mare No. 5). Severe mucosal changes were present in mare No. 4, with extensive necrosis, ulceration, and diffuse neutrophilic infiltrate effacing the mucosa. Neutrophil presence extended to the submucosa.
Duodenum
All mares had rare mucosal and submucosal setal fragments often with only focal necrosis around the fragment. There were low to moderate numbers of neutrophils in the villous tips and deeper crypts in all mares as well as mild mucosal vascular engorgement with occasional crypt dilation. Two mares (mare Nos. 4, 5) had areas of focal proliferation of serosal mesothelium in irregular fronds or nodules with low to moderate numbers of lymphocytes, plasma cells, or macrophages within the underlying adventia of the serosa or within the hyperplastic mesothelium. One area exhibited necrosis and hemorrhage but without obvious setal fragments.
Jejunum, Ileum
The ileum was not available for mare No. 5. All mares had rare mucosal and submucosal setal fragments in the jejunum and ileum with surrounding reactions ranging from unapparent to granulomas composed of lymphocytes and macrophages. Mare No. 4 had severe diffuse necrotizing inflammation within the jejunum and ileum with extensive deep mucosal necrosis, multifocal loss of mucosa to the level of the muscularis mucosae, intense neutrophil infiltration, perivascular edema, and lymphatic dilation. An intact setal shaft was present traversing a large submucosal artery (Fig. 5). The remaining mares had mucosal inflammation similar to the duodenum.
Cecum
Setae were present in moderate to heavy numbers in all mares from the mucosa to the serosa. Setae fragments were present within arterial and lymphatic lumens, arterial walls, veins, submucosa, and villi. Reactions ranged from unapparent (mare No. 2) to focal necrosis (all mares) to granulomas (mare No. 3). Setae found within a vessel in mare No. 3 had early microthrombus formation around the fragment with segmental necrosis of the media (Fig. 6). All mares had low numbers of neutrophils within the superficial mucosa and crypts. Lymphocytes were prominent within the lamina propria of the mucosa and submucosa of 3 mares (mare Nos. 2, 6, 7). Eosinophils numbers were comparable to the control mare. Mare No. 5 had multifocal sites of serosal hyperplastic mesothelium associated with embedded setal fragments. There were mixed inflammatory cells in these areas, including neutrophils, lymphocytes, plasma cells, and multinucleated giant cells.
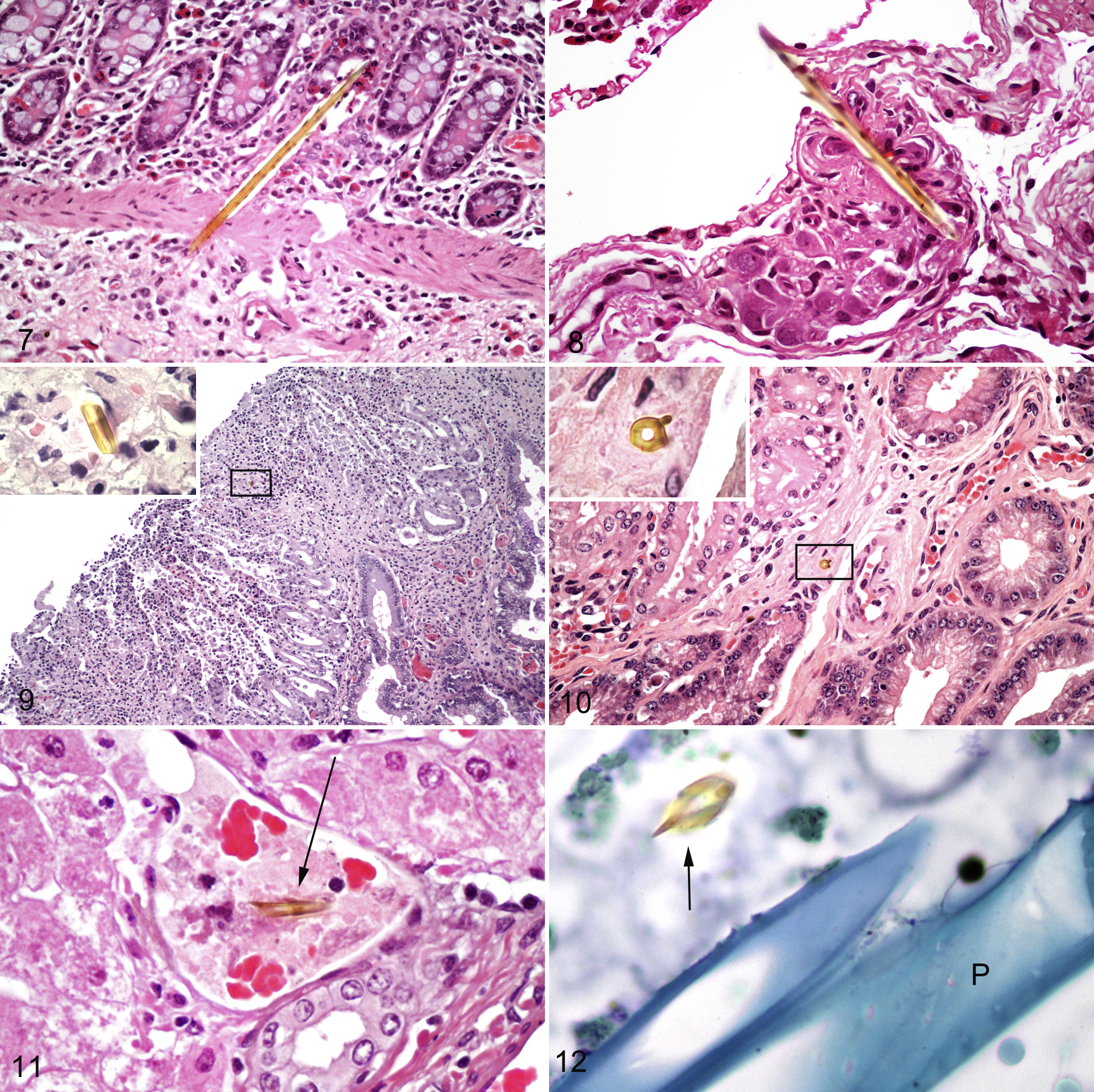
Ventral Colon
Samples from the ventral colon were not available from mare Nos. 4 and 6. All remaining mares had large numbers of variably sized setal fragments from the mucosa to the serosa (Fig. 7). The inflammatory reaction varied and included unapparent, focal necrosis, microabscesses, lymphocytic, and multinucleated giant cells (serosa of mare Nos. 5 and 7). Within the submucosa, fragments were found adjacent to and within vessels, lymphatics, and nerve bundles (mare No. 5) (Fig. 8) often with a granulomatous reaction. All other mares had low numbers of neutrophils within the superficial mucosa. Mare 7 had areas of mixed population of eosinophils and neutrophils within the lamina propria of the mucosa and foci of lymphocytes and plasma cells in the submucosa. Small strongyle larvae were found in the intestinal lumen in 2 mares (Nos. 5, 7) with eosinophil numbers similar to the control mare in the mucosa and submucosa.
Dorsal Colon
Tissue was not available from the dorsal colon of mare No. 4. Large numbers of setal fragments were found within the dorsal colon of all remaining mares with a range of reactions from unapparent, focal necrosis, lymphoplasmacytic, and microabscess formation. A longitudinal cross-section of a large artery within the dorsal colon of mare No. 6 had 18 setal fragments embedded from the lumen to the tunica adventitia. Fragment sizes ranged from 10 to 30 µm and were frequently obliquely fractured. Mare Nos. 5 and 7 had multifocal areas of hyperplastic serosal mesothelium similar to the cecum with embedded setal fragments. The reaction around the fragments was mononuclear, including lymphocytes, plasma cells, macrophages, and multinucleated giant cells. All other mares had low numbers of neutrophils in the superficial mucosa and rare crypt microabscesses. Two mares (mare Nos. 3, 7) had a prominent mixed population of lymphocytes, plasma cells, and macrophages in the lamina propria of the mucosa and submucosa. A single encysted small strongyle larva was embedded in the deep mucosa of mare No. 5 with scattered peripheral lymphocytes and rare eosinophils in the immediate vicinity.
Small Colon
Tissue from the small colon was not available from mare No. 4. Setae numbers varied between mares ranging from rare (mare Nos. 3, 7) to numerous (mare No. 6) (Table 2). Reactions included severe focal necrosis to granulomas (mare No. 7). Mare No. 6 had multifocal crypt dilation and mucosal necrosis associated with local setae penetration. This mare had a dense population of mixed mononuclear cells including lymphocytes, plasma cells, macrophages, and occasional multinucleated giant cells in the submucosa. Mare Nos. 3, 5, and 6 had low to moderate numbers of neutrophils within the mucosa. Eosinophil numbers within the submucosa in 2 mares (No.s 2, 5) were slightly increased compared to the control mare. Mare No. 5 had a single small strongyle larva within the lumen.
Uterus
Tissue from the uterus of mare No. 4 was not available. Setal fragments were found within the tissue of all remaining mares including the serosa (mare Nos. 2, 5, 7), the myometrium (mare No. 5), and endometrium (mare Nos. 2, 3, 5, 6, 7) (Figs. 9, 10). Mare No. 6 had 11 setal fragments throughout the uterus located from the outer myometrium to the endometrium with a mononuclear reaction (lymphocytes, macrophages) around a few fragments in the myometrium and a neutrophilic reaction to a setal fragment in the endometrium. Mare Nos. 2 and 3 were still pregnant at the time of euthanasia with microcotyledon development equivalent to descriptions for their gestational stage. 1 Mare No. 2 had patchy areas of neutrophil infiltration between the microcaruncle crypts with rare lymphocytes in the stratum compactum. Mare No. 3 had large numbers of neutrophils within the superficial stratum compactum and between the crypts of the microcaruncles. Moderate numbers of lymphocytes were also present in these areas as well as scattered plasma cells and macrophages. Mare Nos. 5, 6, and 7 had aborted 12 hours, 72 hours, and 48 hours, respectively, prior to euthanasia. Mare No. 5 had diffuse infiltration of low to moderate numbers of neutrophils within the superficial stratum compactum and between the crypts of the microcaruncles. Moderate numbers of lymphocytes, plasma cells, and lesser numbers of macrophages were present at the junction between the stratum compactum and spongiosum extending into the superficial stratum spongiosum. The endometrium of mare No. 6 had moderate to heavy numbers of lymphocytes and plasma cells diffusely within the stratum compactum with low numbers of neutrophils. These cells extended into periglandular areas with low numbers of scattered macrophages. Mare No. 7 had extensive severe necrosis (Fig. 9) of the endometrial surface with diffuse infiltration of the stratum compactum by intact and degenerate neutrophils extending and effacing the endometrial glands in the stratum spongiosum. Bacilli were present within a small venule of the endometrium of mare No. 7 as well as mixed Gram-negative bacilli and Gram-positive cocci within the necrotic debris on the surface of the endometrium. Three mares (mare Nos. 2, 5, 7) had multifocal areas of hyperplastic serosal mesothelium similar to the duodenum with a setal fragment acting as a focus in mare No. 5.
Mesenteric Lymph Node
Low numbers of setal fragments were present within the mesenteric lymph nodes of 5 mares (Table 2) within sinuses and cortical areas. There was focal necrosis with a neutrophilic infiltrate surrounding a setal fragment in mare No. 2 and granulomatous reaction involving lymphocytes and macrophages surrounding a setal fragment in mare No. 7. Unapparent or focal necrosis was present around the setal fragments found in mare Nos. 4, 5, and 6 including a long setal shaft. Setae were not evident within the lymph nodes of mare No. 3.
Liver
Setal fragments were found in the parenchyma of mare Nos. 4, 6, and 7 with multiple fragments present in mare No. 6. Inflammation around 1 parenchymal fragment in mare No. 6 consisted of mild focal necrosis and small numbers of neutrophils and lymphocytes. Mare Nos. 4 and 6 had a fragment within a portal vein (Fig. 11). Inflammation was not evident within the livers of mare Nos. 5 and 7. There was patchy portal infiltration of neutrophils and rare lymphocytes and plasma cells in mare Nos. 2, 4, and 6. Mare No. 3 had a degenerate parasite fragment in 1 area with focal periportal eosinophils and focal biliary hyperplasia.
Spleen
There were no obvious abnormalities in the spleen of any mare.
Mast Cells
Setal fragments did not take up the toluidine blue stain in tissues from the treated mares, retaining the golden refractory color while other plant material and debris stained the homogenous blue of the background tissue (Figs. 5, 12). Setal fragments embedded in tissues without any obvious cellular reaction did not have mast cells around or in the immediate vicinity within the gastrointestinal tract of mare Nos. 2, 3, 5, 6, and 7 and in the uterus of mare Nos. 2, 6, and 7. Around setal fragments in the gastrointestinal tract where there was a mononuclear reaction (mare Nos. 2, 4, 6), only mare No. 2 had mast cells inconsistently present around the periphery of the reactions. Numbers ranged from 1 to 3 cells when present.
Bacteria
Microorganisms were present on the mucosal surface of the gastrointestinal tract of all mares. Mixed Gram-negative bacilli and Gram-positive cocci were present within the endometrium of mare No. 7 as previously described. Gram-stained sections of tissues other than the gastrointestinal tract excluding mare No. 7 failed to highlight bacteria intimately associated with setal fragments or elsewhere.
Discussion
Setae were found throughout every tissue layer of the gastrointestinal tract including the serosa, within the liver (both parenchyma and vasculature), lymph nodes (parenchyma and sinuses), mesentery, and uterus, indicating extensive setal fragment dissemination from the GIT. The streamlined shape of the setae with sharply pointed ends and angled barbs would facilitate mechanical penetration through tissues as the means of setae dissemination. Movement of setae through tissues by mechanical transfer has been well documented in cases of ocular exposure in people, with migration through the conjunctiva and subconjunctiva into the cornea, iris, and ultimately retinal penetration. 8 The ocular migration was postulated to be caused by the movements of the globe, respiration, and pulse along with the constant movement of the iris propelling the setae forward with the angled barbs preventing backward movement. 8 In horses, the peristaltic movements of the gastrointestinal tract and myometrial contractions of the gravid uterus are likely to facilitate penetration of these tissues. Movement into tissues without muscle layers like the liver and lymph nodes may slow or halt the migration.
Vascular penetration by setal fragments found in many tissues indicates that the circulatory system may also facilitate tissue dissemination. Penetration into the portal circulation may explain the presence of a setal fragment within the portal vein of mare Nos. 4 and 6. Similarly, lymphatic drainage into the local lymph node may also account for some of the small fragments found in the mesenteric lymph nodes in 5 of 6 treated mares. It is possible that large setae could be disseminated via the circulatory or lymphatic system, but the long length of the setal fragments might prevent travel in any but the largest vessels. The presence of setal fragments in the gastric serosa of 3 of 6 treated mares supports the suggestion of continued direct setal migration into the liver. Hepatic parenchymal, uterine, and mesenteric lymph node penetration is likely via direct mechanical penetration from the GIT. The migration of caterpillar setae is akin to the migration of other foreign bodies in tissues. Grass awns, porcupine quills, toothpicks, and sewing needles, all of similar shape to setae, have been found in aberrant locations including the brain, joints, bladder, and uterus of animals, generally far distant from the point of entry. 3,5,14,24,26
Relative setal numbers (Table 2) within the tissues were highest in the stomach and large colon where tissues were available with low numbers or none in the small intestine and other organs. Gastrointestinal transit time and dosage of whole caterpillar may play a role in setal fragment density within the tissues. Ingesta (and consumed setae) spends a small amount of time in the esophagus (seconds) and small intestine (travelling approximately 30 cm/min) as opposed to longer times in the stomach (2–6 hours) and large colon/cecum (>10 hours depending on diet). 43 Mare Nos. 4 and 6, both receiving 100 g of whole caterpillar, had the highest number of setal fragments in the glandular stomach of all the mares, and mare No. 6 had 11 setal fragments in various locations in her uterus. Large numbers of setal fragments in small uterine tissue sections may indicate that massive numbers of setal fragments are migrating in a virtual shower through the uterus. The presence of high numbers of setal fragments in the uterus of mare No. 6 is likely due to the higher dose of caterpillar setae as well as the time of euthanasia (12 days from the first treatment). The increased frequency of finding setal fragments in the GIT, liver, and uterus of higher dosed mares suggests the dose of whole caterpillar does play a role in the numbers of setae migrating.
The different locations of setal fragments within the layers of the GIT as well as the uterine serosa, myometrium, and endometrium of various mares suggest fragments migrate at different rates. Setal fragment migration into tissues and the uterus can be very rapid, with fragments present in the liver, lymph nodes, uterine serosa, and endometrium of mare No. 2 within 2 days after the first treatment. However, setal fragments continued to be found in tissues up to 24 days from the first treatment. Variables affecting the rate and ultimate location of setae in the uterus may include the size and fragmentation of the setae, the exit point from the gastrointestinal tract (small intestine, large colon, or small colon), stage of pregnancy (contact points with the GIT and thickness of the myometrium), as well as the strength and frequency of myometrial contractions. The rate of setal migration into the uterus is most likely affected by close and continual anatomical contact with the GIT, which would be dependent on the location and stage of gestation.
The presence of setae barbs and fragments within tissues induced variable focal tissue reactions ranging from focal necrosis to neutrophilic, lymphocytic, or granulomatous inflammation with multinucleated giant cells. It is unknown how these focal reactions could affect the gastrointestinal tract on a larger scale. Vascular necrosis as seen in Fig. 6 may result in focal gastrointestinal ischaemia. Penetration through nerve bundles could cause acute abdominal pain (colic) or segmental gut stasis. In many instances, the setae were found in all tissues without an accompanying focal tissue reaction. The variation in the reactions may be due in part to differences in the duration of setae within tissues and therefore the speed of migration. It might be expected that setal migration through tissues would cause tissue disruption, cell injury, and focal necrosis, as was seen in many tissues. The lack of reaction may be that the setae were found midmigration before tissue reactions occurred or that the reaction occurred behind the setae in a different tissue plane and therefore was not visible. Rapid setal migration as occurred within mare No. 2 could result in detection of more setae without an accompanying reaction. In addition, setae may be able to exist in tissues for variable periods of time without inciting an inflammatory reaction, as has been reported with intraocular setae in the human eye. 25
The variations in the cellular inflammatory response (neutrophilic, lymphocytic, or granulomatous) to setae in the mares were similar to the suppurative and/or granulomatous inflammatory reactions reported in human intraocular tissue. 22,46 Granulomatous reactions were described as an initial lymphocytic infiltration followed by macrophages and multinucleated giant cells. 46 Experimental exposure to ETC setae resulted in submucosal granulomas in a nonpregnant mare, rats, pigs, and goats. 30 Dermal reactions in humans and mice to caterpillar setae were also predominantly mononuclear. 13 Caterpillars have setae composed of chitin, which is degraded in mammalian tissues by chitotriosidase, a chitinase-like protein present in macrophages. 2,16 Chitin and its breakdown products stimulate a pro-inflammatory response in humans with subsequent macrophage activation. 2 The reactions within human ocular tissue were thought to occur once the setal fragment had become relatively fixed, but it is unknown if the reaction was to the setae itself or a possible toxin within the setae. 22 The fixation of setal fragments within the tissue for any reason such as denser tissue or setal blunting makes it more likely that a cellular reaction can be stimulated by the chitinous skeleton of caterpillar setae. This response in the form of macrophage stimulation may be responsible for the mononuclear and granulomatous reactions seen around some setal fragments penetrating vasculature (Fig. 8), within the GIT, serosal lesions, and uterus of the experimental mares. 2 The neutrophilic response as demonstrated by microabscesses surrounding setae within the crypts, submucosa, and muscularis externa of the GIT in some mares may indicate the presence of other pro-inflammatory components such as bacteria that could be chemotactic for neutrophils. This may be the underlying cause of the focal mucoid placentitis lesions induced by gavaging mares with caterpillar exoskeleton in the pre- and early placentation stages of pregnancy. 11
Bacteria were found intimately associated with setal fragments on the mucosal surface of the GIT but not elsewhere. Bacilli were found within the endometrium and vasculature of mare No. 7 but not intimately associated with the setae also present. Intravascular bacteria that might indicate bacteraemia were not seen in other tissues. While there is a likely association between the presence of a setal fragment and acute inflammation, bacteria could not be confirmed in association with the migrating setae. The lack of microorganisms intimately associated with migrating setal fragments may be due to a number of factors. Bacteria may not actually be associated with setal fragments except where they are in contact in the GIT lumen and mucosa. However, this would make the process of setal fragment migration and acute bacterial abortion involving a wide variety of enteric and environmental bacteria highly coincidental. It is more likely that routine light microscopic histology is an insensitive method of detecting small numbers of bacteria that may be attached to the setae. Bacteria that have located in the inner hollow core or around barbules may not be apparent in a single tissue cross-section, nor can the whole setal fragment be examined in limited thin tissue sections with varying orientation in different tissue planes. The mares’ defense systems in most tissues are able to remove small numbers of introduced bacteria. In cases where the bacteria are deposited in tissues or fluid-filled spaces where rapid multiplication can occur ahead of an immune response, as in the pregnant uterus and the fetal membranes, progression and spread to the fetus can occur as outlined in the companion article, leading to EAFL and MRLS. 40 A similar mechanism could explain the pericarditis and uveitis seen in MRLS cases.
In the gastrointestinal tract, setal penetration evoked diffuse mucosal inflammation resulting in acute gastritis, duodenitis, jejunitis, ileitis, typhlitis, and colitis characterized by variable numbers of neutrophils in the superficial mucosa. Mechanisms responsible for this development may be multifactoral. Neutrophils are not part of the cell population in the mucosa of normal equine small or large intestine, as demonstrated by the control mare and other studies on the cell population in the equine gastrointestinal tract. 23,31 In addition, neutrophil presence in naturally acquired small strongyle infections are not described unless fenbendazole is used, which was not the case in these experiments. 37 The influx of neutrophils into the mucosal surfaces is most likely caused by disruption of the mucosal barrier from diffuse and repeated mechanical penetration of setal fragments. Mechanical disruption of the mucosal barrier in addition to focal necrosis in the path of the setal fragments would allow increased exposure of the mucosa to gut microflora, stimulation of inflammatory mediators, and influx of neutrophils in an acute inflammatory response. The gastroenteritis in mare No. 4 was severe enough to cause colic and result in euthanasia. Similarly, oral associated contact with caterpillar setae in dogs resulted in tongue edema, necrosis, and oral mucosal ulceration. 6
A hypersensitivity reaction may also be involved in the gastrointestinal inflammation in some mares as 4 mares showed some form of urticarial reaction after oral dosing with caterpillars (Table 1). 9,10 Humans allergic to Thaumetopoea pityocampa, another Processionary caterpillar, have IgE antibodies that recognize the proteins thaumetopoein, a 28 kDa protein, and Thap1, a 15 kDa protein isolated from T pityocampa suggesting an IgE-mediated allergic reaction after caterpillar exposure, although as yet these proteins or toxins have not been demonstrated in O. lunifer. 29,44 An IgE–mediated Type 1 hypersensitivity could induce inflammatory cell infiltration into the mucosal surfaces after repeated challenges with caterpillar setae from the gut lumen over 5 days. 36 In addition, experimentally induced anaphylaxis and increased histamine levels were shown to cause inflammation, edema, and congestion in the large colon and cecum of the horse and to cause uterine contractions, placental separation, and fetal death in cats. 4,18 Any histamine-related or toxin-induced changes in the cecum, colon, and uterus of the mares would be difficult to distinguish from pathology associated with mechanical setal penetration as seen in this study, and therefore the role of a hypersensitivity reaction from any possible setal toxin remains unclear.
All 5 of the examined treated mares had caterpillar setal fragments in their uteri as well as some degree of uterine inflammation. This involved mild to heavy neutrophil infiltration of the subepithelial areas and between the crypts of the microcaruncle. Neutrophil presence in the stratum compactum of uterine biopsies of nonpregnant mares in estrus is an indicator of acute inflammation. 35 However, normal postfoaling involution of the uterus in mares without a history of pathological problems involves an intense influx of neutrophils and macrophages into the areas of the microcaruncles with orderly microcaruncle degeneration over a period of 10 days. 21 The pregnant untreated mare did not have neutrophils within the endometrial tissue sampled, and comparison with the 2 mares still pregnant at the time of euthanasia (mare Nos. 2, 3) suggests the neutrophilic inflammation in both treated mares was due to the effects of caterpillar setae migration rather than part of the normal cell population at that stage of gestation. However, mare Nos. 5, 6, and 7 aborted 12 hours, 72 hours, and 48 hours, respectively, prior to euthanasia. Comparison of these treated postabortion mares to normal involution is more difficult due to the normal intense inflammatory process in the stratum compactum, which occurs immediately postpartum in normal mares. 21,27 The effect of any setal migration through the endometrium in mare Nos. 5 and 6 is difficult to discern from the normal progression of uterine involution. Acute neutrophilic inflammation was present in mare No. 5 at 12 hours post abortion, which under normal circumstances may not have developed until 24 hours post foaling. 21 In mare No. 6, 72 hours would have been adequate time to develop the predominance of mononuclear cells masking any underlying inflammation. The changes in this mare suggested involution was proceeding as normal. The remaining mare had extreme changes that involved severe and extensive necrosis of the endometrium down to the deeper endometrial glands, which would not have developed in 48 hours in a normal foaling. 21 The severe changes in this mare were most likely complicated by the retention of the fetal membranes, and any underlying cellular changes caused by caterpillar setal migration would have been masked by time.
Causes of the variable inflammation in the uterus of the pregnant mares may be multifactoral but is most likely related to the migration of setal fragments. Direct inoculation of antigens (mammalian tuberculin, ascarid antigen, and bovine serum albumin) into the endometrium of nonpregnant mares elicited transient acute inflammation as diffuse hyperemia and edema with foci of necrosis and predominant neutrophils. 28 In the same study, mechanical disruption by endometrial biopsy in control mares elicited a similar cellular reaction. 28 The mechanical disruption of setae migration through the uterus, the chitin within the setae, any toxin associated with the setae, or any bacteria translocating with the setae must be considered as a potential cause of any endometrial inflammation. While the presence of abnormal inflammation in the aborted mares is difficult to discern from normal involution, it is important to note that any inflammation elicited by the migration of the setae during pregnancy may be cleared by the inflammation of normal uterine involution after abortion as in mare Nos. 5 and 6 unless there are postpartum complications, as in mare No. 7.
The hyperplastic serosal lesions found multifocally on the GIT and uteri in treated mares in contrast to the untreated mare are likely a response to setal penetration. The reaction was variable and included focal hyperplasia of mesothelial cells as multilayered cells or small papillary fronds with variable inflammation (unapparent, neutrophilic, or mononuclear) similar to the inflammatory response to setae seen in other tissues. Inflammation and chemical irritation have been found to cause a hyperplastic response in mesothelial cells, and both mechanisms may be evoked by setal migration through the tissues. 49 The presence of setal fragments in some serosal lesions confirms the lesions as an indicator of setal migration.
Migration of setae in tissues did not evoke a significant mast cell response, with only mare No. 2 having a few mast cells at the periphery of mononuclear reactions around setae in the GIT. It has been suggested that formalin fixation may interfere with the staining of equine mast cells with TB, and therefore other staining techniques may have to be utilized to fully investigate the involvement of mast cells in any cellular reactions after caterpillar exposure. 17 Previous studies in horses of different ages have correlated the presence of mast cells and eosinophils in the large intestine of horses with cyathostome worm burdens. 12 Intestinal parasites were detected in some experimental mares, and although a total worm burden was not determined, the incidence was low, creating difficulty when deciding if the presence of mast cells in the GIT was due to parasite infestation or secondary to setal fragment presence. Mast cells were not part of the cellular reactions within the uterus of the mares in which tissues were stained with TB as cell numbers were low and comparable with the control mare and previously documented studies. 45,48 While initially TB stain was used to determine if mast cells were a component of any cellular reactions to setal fragments in the experimental mares, it became apparent the stain highlighted the presence of the golden refractory setal fragments in tissues and allowed rapid differentiation from contaminating plant material, which stained blue (Fig. 12). As such, it may be a useful stain to aid detection of setae in tissue from field cases of equine abortions.
Mare Nos. 5 and 7 had exposure to caterpillar exoskeleton in their previous pregnancy (Table 1), delivered compromised foals, and were rebred successfully to participate in the current study. The presence of setal fragments within multinucleated giant cells in the serosa of the glandular stomach of mare No. 7 and large colon of both mares could be due to the previous exposure to exoskeleton. Case reports on ocular setae indicate that endogenous removal of setae may take 8 weeks to 6 months or setae can persist for over a year after initial penetration with very little visible degradation. 8,19 However, mare No. 4, a naïve mare, also had inflammation with multinucleated giant cells within the mucosa and serosa of the glandular stomach after 8 days from the first treatment. Mare Nos. 5 and 7 were euthanized 10 and 24 days from the first treatment, which would allow a mononuclear inflammatory response to develop. Both of these previously exposed mares aborted when reexposed following gavage with 50 g of whole caterpillar with a moderate to severe acute endometritis (Table 3). The acute inflammatory reaction coupled with the presence of setae in the uterus suggests that reexposure and new setal migration rather than past exposure was the cause of the histopathological findings in the uterus of these mares.
Conclusion
The histological findings of setal fragments throughout the GIT, mesenteric lymph nodes, liver, and uterus in this study identify more than one possible mechanism by which caterpillar setae can facilitate bacterial translocation from the GIT to the reproductive tract. The frequent presence of setal fragments adjacent or within the lumen of vasculature in all areas of the gastrointestinal tract allow for the possibility of bacteraemia secondary to setal vascular penetration. However, the finding of setal fragments within the uterus of experimental mares suggests that direct migration of setal fragments acting as a bacterial vector is the more likely possibility for the bacterial abortions that characterize exposure to caterpillars. This is strongly supported by the rapid setal migration into the uterus within 2 days of exposure despite the lack of absolute direct association of bacteria with migrating setal fragments. It should also be noted that in the companion article, 40 the presence of setal fragments within the allantochorion of fetuses from the experimental mares in the current study are detailed, and more recently setal fragments have been identified in the allantochorion of EAFL clinical cases. 39 Both observations further support the importance of a setal vector for bacterial translocation in EAFL.
If toxins are present in O lunifer setae, the possible effects could not be differentiated from any mechanical effects of setal penetration in these experiments with nonspecific diffuse acute inflammation affecting the GIT of all experimental mares. Uterine inflammation ranged from mild to severe but appeared to be cleared by normal uterine involution, which supports practitioners’ claims and experimental evidence that mares aborting from EAFL have few problems getting back in foal (A. J. Cawdell-Smith, 2012, personal communication). Caterpillar setal migration likely plays an important role in the pathogenesis of EAFL and highlights the need for further research to delineate the manner in which setae fragments facilitate the transfer of enteric or environmental bacteria into an environment where their presence initiates infection and subsequent fetal loss.
Footnotes
Authors’ Note
Parts of these data were presented as a short abstract at the 10th International Symposium on Equine Reproduction in Lexington, Kentucky, in July 2010.
Acknowledgements
The authors thank the staff of the Equine Research Unit for their technical support during the study.
Declaration of Conflicting Interests
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Horse Program of the Rural Industries Research and Development Corporation, Canberra, Australia.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Horse Program of the Rural Industries Research and Development, Canberra, Australia.
