Abstract
Immunohistochemistry (IHC) is a common adjunct in pathology for morphologic diagnosis, research pathology, and studying the pathogenesis of the disease. Proper technique and interpretation of an immunohistochemistry assay is of utmost importance. A variety of problems, including the presence of artifacts (nonspecific background or other staining problems) and the differentiation between nonspecific and specific staining, commonly occur. It is essential that antibody quality and IHC technique be optimized. We review the histologic patterns of specific and nonspecific staining after using IHC techniques, as well as basic troubleshooting procedures, and provide some examples of nonspecific staining and other artifacts especially in formalin-fixed, paraffin-embedded tissues (FFPE) of mice.
Immunohistochemistry (IHC) can be an excellent method for determining antigens (protein or gene) in formalin-fixed, paraffin-embedded (FFPE) cells and tissues, but IHC requires procedures that have many steps that can go wrong, even with appropriate antibodies and in experienced laboratories. There have been many review publications and books on IHC that can aid the veterinary pathologist and investigator. 2,5 –12,15 –19 We review some of our own methods for IHC interpretation and troubleshooting investigative procedures. Various procedures are used to confirm and optimize specific antigen localization in FFPE mouse tissues. These methods also apply for other fixatives and frozen sections and to all species of animals. Most of our figures are from mouse tissues, but the principles and procedures apply to all species.
Interpretation of the IHC Slide
After IHC staining of paraffin tissue sections with an antibody at a select dilution, the detection reagents and a chromogen at a select dilution, a color reaction occurs (eg, brown) when 3,3′-diaminobenzidine (DAB) is used as the chromogen. As the antibody dilution is increased, the color of the chromogen gradually disappears. There should be a dilution-related appearance of specific staining (ie, brown color with DAB). After use of proper controls and review of the slides with the various antibody dilutions, one concludes what constitutes specific staining and gives the best signal to noise for the antigen studied. Unfortunately, with DAB, there is always brown staining on the slide at some dilution. Investigators often want a positive result. Consequently, they often conclude that the antigen is found in a specific tissue or cell from the appearance of the brown color reaction in the tissue sections even without the proper use of controls. Many publications show “brown” color in cells and tissues that appears to be nonspecific background staining. 18 Often the publications do not have a pathologist as a coauthor, even though it does not take a pathologist to verify specific positive staining. That said, many investigators do not understand the importance of controls and critical detailed interpretation. Pathologists have fewer problems with accepting background staining as not being “positive” and specific. Any experienced technician or scientist can perform IHC and get positive results with the use of proper controls and enough knowledge of the tissue involved. It is also the experience of the authors that some scientists see brown color in some cells in the section and consider it “specific positivity.” The evaluation of the IHC slide requires interpretation of positive and negative tissue controls for all dilutions and pretreatments of the antibody used. This interpretative procedure is part of the training of pathologists and follows them throughout their careers. As a result of their training, pathologists know that “brown” is not always a positive result. 18 Interpretation of nonspecific (background) and specific staining requires thorough slide evaluation and will be discussed next.
Differentiation of Positive Staining From Nonspecific Background Staining
After performing a dilution of the primary antibody on the control or test tissues, one may often easily conclude what is specific and nonspecific staining. The specific and nonspecific staining should decrease as the dilution increases. If cells still stain at very high dilutions, such as 1:10 000, it is possible that the cells staining do not contain antigen, unless, of course, the primary antibody is very concentrated and specific. A comparison with a test tissue slide stained with all reagents except the antibody will usually provide evidence for specificity. This type of “secondary-only” control, however, will identify background caused by reagents from the development technique only and not from nonspecific binding of primary immunoglobulin. At the highest primary antibody concentrations, all cells and tissues usually stain the color of the chromogen indicative of nonspecific staining. Many publications show this nonspecific phenomenon as specific staining when in fact the figures are illustrating nonspecific immunoreactivity. Preimmune serum from the rabbit or other species in which the primary antibody was made can often replicate the nonspecific staining patterns at low antibody dilutions.
Patterns of Nonspecific and Specific Staining
Each antigen used in a specific protocol results in a specific pattern of staining in a cell and tissue of an organ and species. To differentiate nonspecific from specific staining, we must consider patterns of nonspecific background and specific staining.
Nonspecific staining
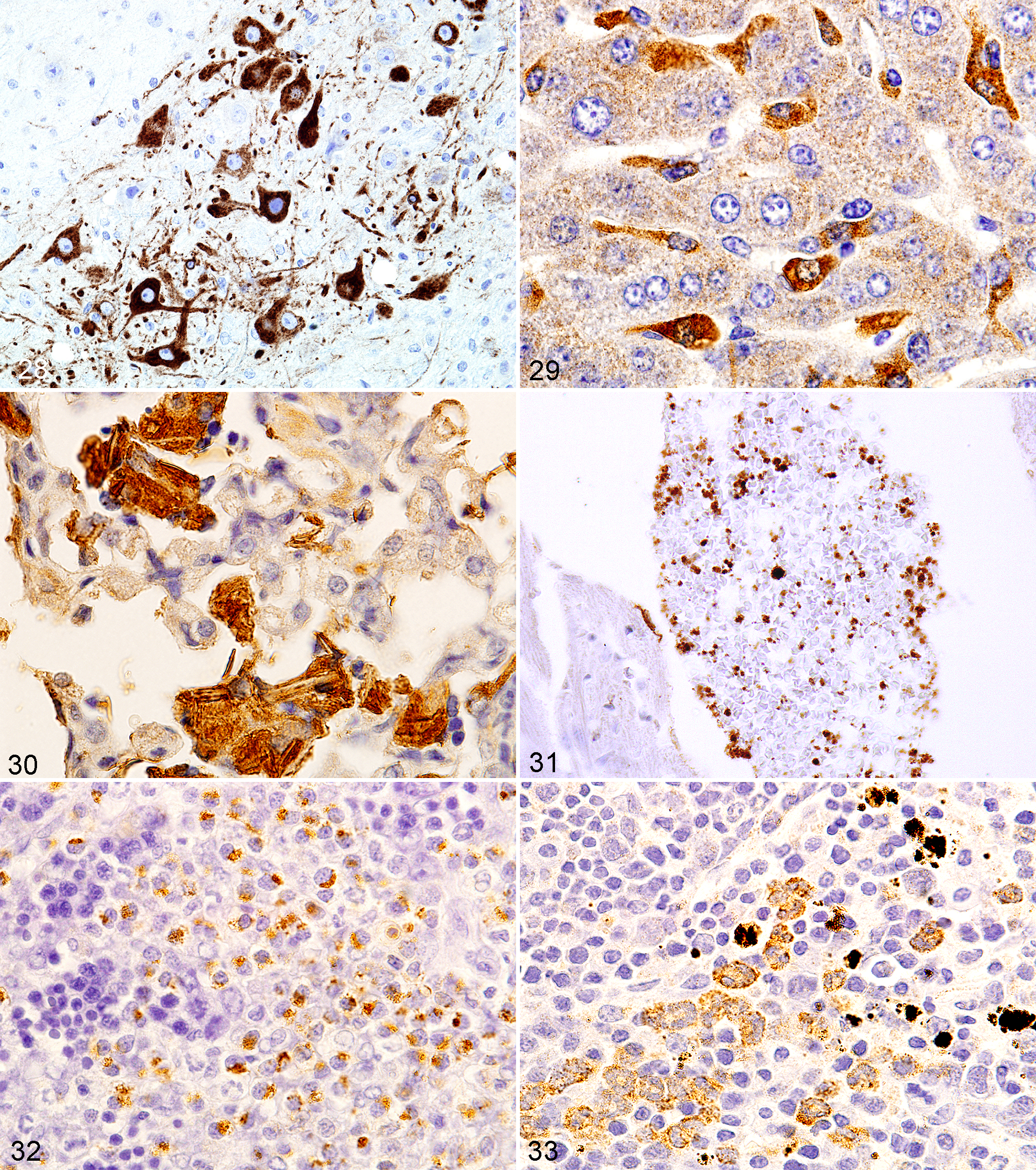
Dilutions of antibodies first used often reveal nonspecific background staining (NSBS) at the highest antibody concentrations. This can be replicated with nonspecific immunoglobulins and preimmune serum if made in an animal or normal serum from the species from which the antibody is made. Nonspecific immunoglobulin or preimmune serum must be concentration matched, however, to the primary antibody. Causes of this NSBS have been shown to be due to 1 or more of the following: hydrophobic interactions of protein; ionic and electrostatic interactions; endogenous peroxidase activity; hydrophobic interactions of protein, avidin, and biotin; Fc receptors; nonspecific antigen diffusion (sequestration); and pigments. 8,21,22 Some patterns of nonspecific staining are shown in Figs. 1 to 21. They can often mimic specific staining for various reasons.
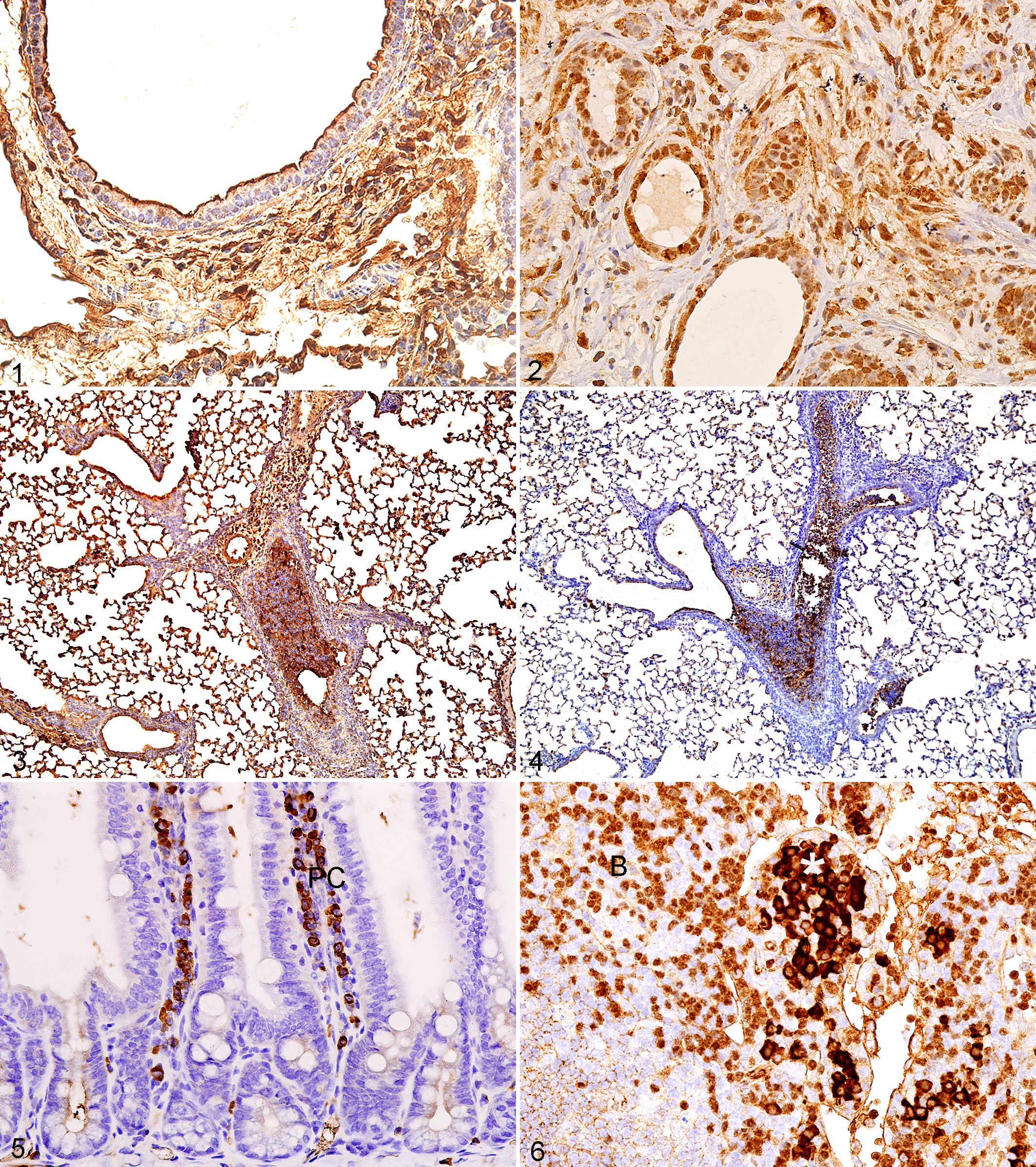
Lung; mouse. Diffuse, nonspecific background staining. Almost all cells and extracellular tissues are brown.
Preimmune and immune sera can be compared if the antibody was prepared by injecting the antigen into animals (rabbits, goats, guinea pigs, etc). The appearance of the NSBS at the highest primary antibody concentrations can mimic in color the staining seen with specific staining. At the lowest dilutions, all cells and tissues are brown with DAB as the chromogen (Fig. 1). Epithelial, mesenchymal, and neural cells and extracellular tissues are brown (Fig. 1). Even when the nonspecific background is very high, specific staining can be detected for some antigens such as the nuclear estrogen receptor (ESR2) (Fig. 2). As the dilution is increased, there is loss of brown from the various tissue components (Figs. 3, 4). At the lowest antibody concentrations tested, there should be no NSBS and only the specific staining of the antigen if the primary antibody immunoreacts in the tissue sections. Some cell types (lacking the antigen of interest) such as mast cells and plasma cells, as well as plasma (Figs. 5, 6), may appear brown at all dilutions depending on the antibody and the IHC technique used, but they will also be brown in the control slides that were not exposed to the primary antibody. With dilution of the antibody, the intensity of staining of these cells or plasma remains the same. The staining of immunoglobulins in plasma cells and blood vessels (Figs. 7, 8) is a particular problem with the use of mouse monoclonal antibodies on mouse tissues regardless of whether the mouse monoclonal antibody is to mouse antigens or antigens of other species. Detailed strategies for avoiding this type of background are discussed elsewhere. 6
Liver; mouse. Note the nonspecific staining of plasma (immunoglobulins) in the sinusoids. The primary antibody was not used in this case.
Since cells of interest may stain brown at some dilution, the tendency is to conclude that any cells that stain brown are positive for the antigen. Edge artifact is common in tumors (Fig. 9), but surprisingly, some publications appear to show edge artifact as positive staining. Careful use of appropriate controls should allow one to determine what is nonspecific background staining and what is specific antigen staining. 8,9
TUNEL
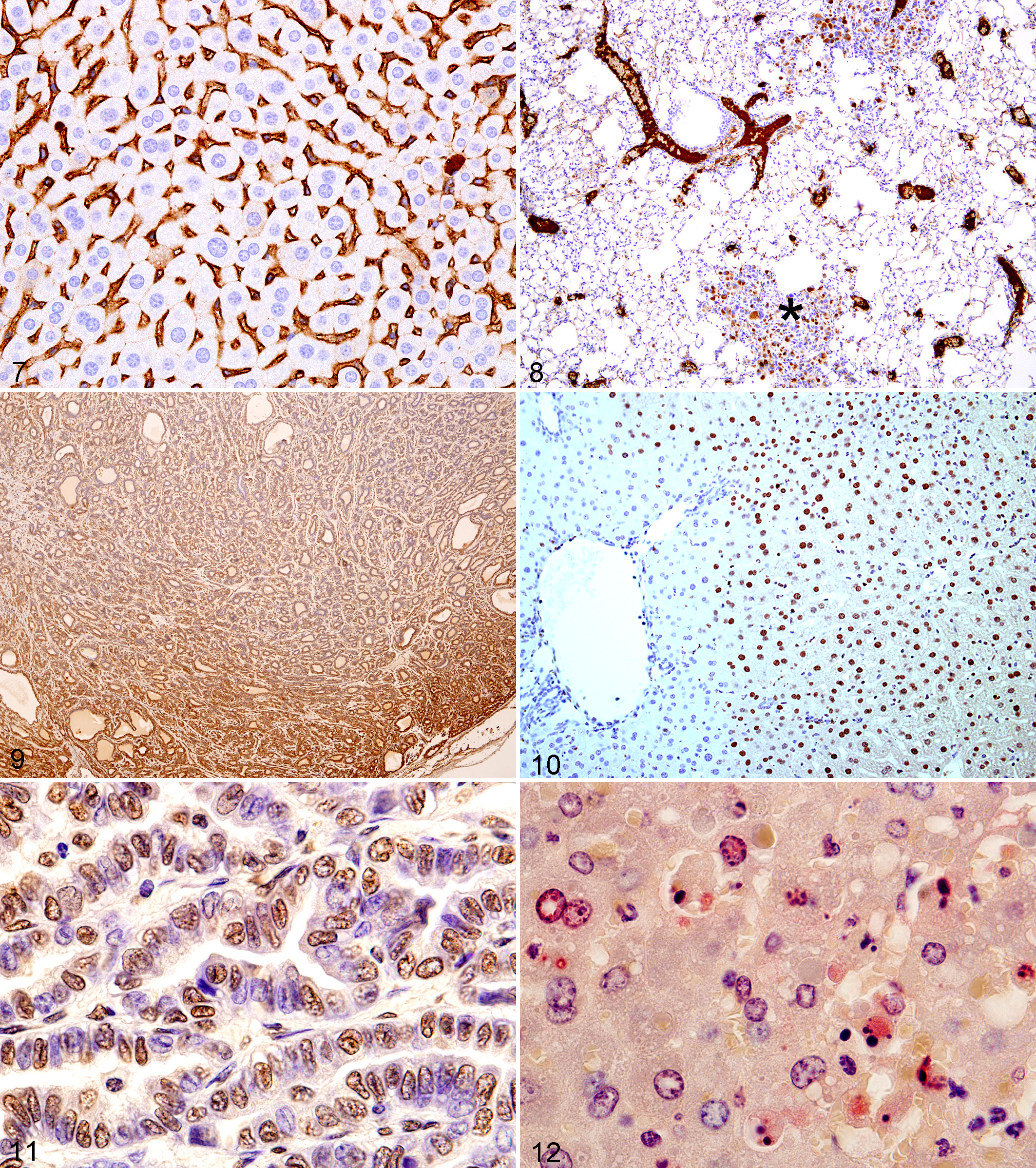
A particular specific example of 2 problems with NSBS is the use of the terminal deoxynucleotidyl transferase (TdT) dUTP nick end labeling (TUNEL) method, often with commercial kits. 4 The TUNEL assay attempts to identify apoptosis in tissue sections by detecting DNA fragmentation in situ, by end labeling of DNA strand breaks using TdT. However, it also labels cells dying by other cellular mechanisms in addition to other nonspecific results. 4 After many years of experience with these kits, most pathologists will not diagnose apoptosis unless the cytomorphology of apoptotic bodies is seen with positive staining. The problem is that investigators who are not pathologists do not understand this problem and often publish any positive staining as apoptosis without morphological evidence of apoptosis. Morphologically, normal cells in a tissue without obvious apoptosis or focal necrosis can be observed to stain the color of the chromogen using the TUNEL kits (Figs. 10, 11). This occurrence may be due to a focal staining artifact and other factors. The use of antibodies to a specific caspase (cleaved caspase 3 for some tissues of mice and others species) 3 for IHC should be a more accurate measure of viable cells going into apoptotic cell death (which may be morphologically normal and/or not morphologically apoptotic) and the cell structures (degenerating cells and apoptotic bodies) representing apoptosis itself (Fig. 12). It has been stated in some publications by pathologists that apoptosis should not be diagnosed unless morphological evidence of apoptosis is seen with or without TUNEL+ cells or apoptotic bodies. 4 We agree. Some investigators use other methods as well to demonstrate apoptosis such as electron microscopy and biochemical and molecular assays.
Role of temperature causing nonspecific staining and optimizing specific staining
Heat-induced epitope retrieval (HIER) is 1 method that can be used, and several commercial devices can be used to produce heat for epitope retrieval. Three common commercial devices for HIER are a vegetable steamer, microwave oven, and pressure cooker. As a rule, as the temperature is increased, the incubation time is decreased. The optimal temperature, antigen retrieval buffer reagent pH, and incubation time may vary depending on the epitope (antigen) of interest and the heat device used (Figs. 13–16). Some antibodies and antigens can be temperature sensitive and the immunoreactivity may be decreased or lost at higher temperature (Figs. 13, 14, 17, 18). For example, a 2- to 3-minute variation in pressure cooking time may have a substantial impact on the end result, including the presence of nonspecific staining (Fig. 15). Therefore, it is recommended that each laboratory determine its own optimal temperature and incubation time for the heating device used. It is also important to keep the cool-down time consistent, since variation in the cool-down time will cause variation in the results. Figures 13 to 14 and 17 to 18 illustrate that immunoreactivity of an antibody for an antigen may be decreased with higher temperature or longer incubation times.
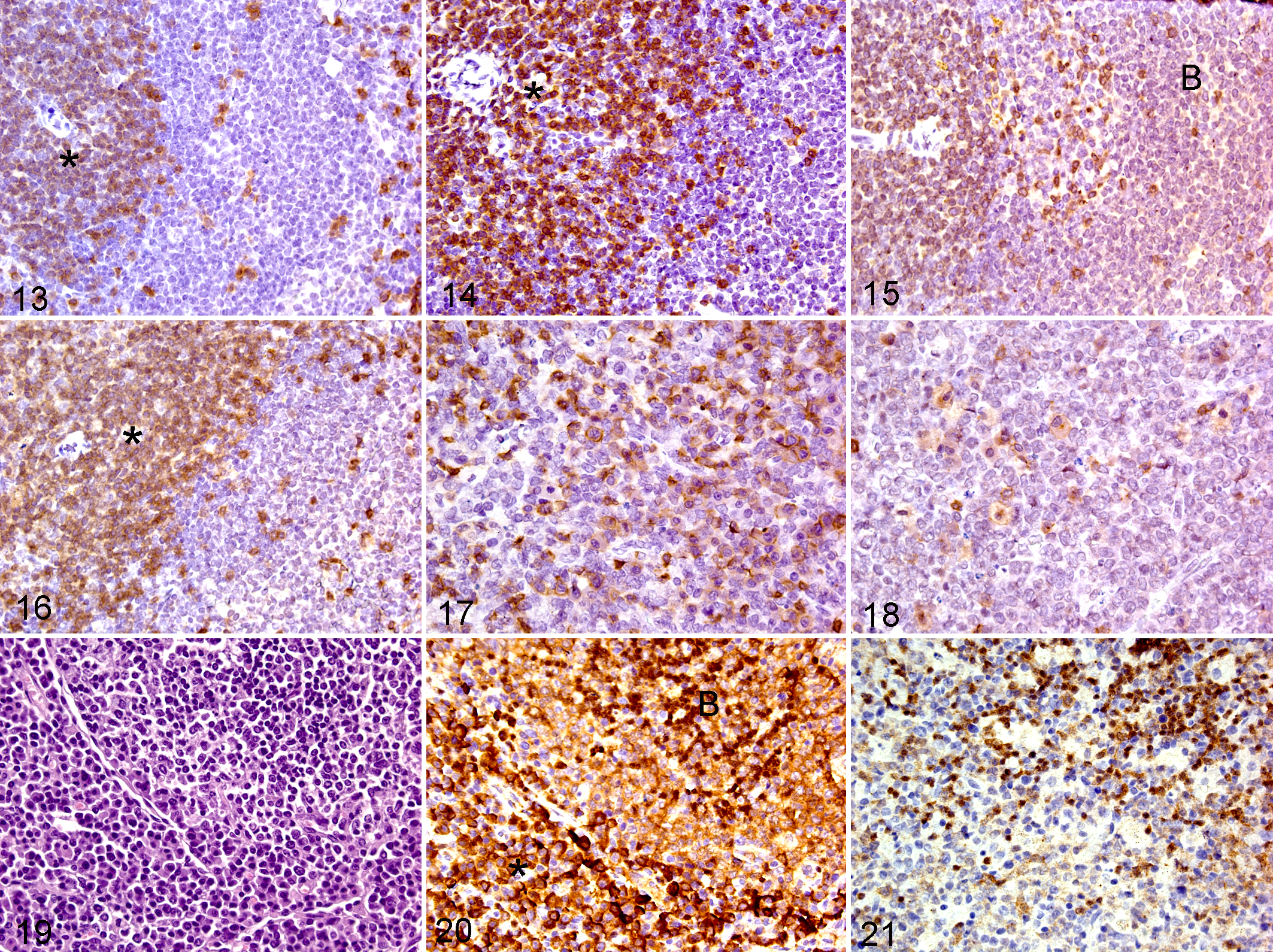
CD3 (CD3e); spleen; mouse. Heat-induced epitope retrieval (HIER) of 30 seconds at 125°C. There is less intense T-cell staining in the periarterial lymphocyte sheaths (PALS) (asterisk) than in Fig. 14.
Role of secondary antibody causing nonspecific staining
There are several types of link antibodies available. The optimal secondary reagent must be made in another animal species rather than in the species of the target antigen, and the immunoglobulin (Ig) isotype (IgG, IgM, etc) should match the type of the primary antibody. As is the case with primary antibodies, the quality of link antibodies can substantially have a major effect on the quality of the immunohistochemical assay and the interpretation of the tissue changes, as shown in Figs. 19 to 21. Nonspecific staining of plasma cells was caused by the use of a secondary antibody (Fig. 20). The use of the optimal secondary antibody resulted only in specific staining of B lymphocytes (Fig. 21).
HRAS
An excellent example of IHC nonspecific staining published initially as specific staining is the case of HRAS1 (Hras1, Harvey rat sarcoma virus oncogene 1) in FFPE tissue. Antibodies to HRAS were claimed to stain cytoplasm but not cell membranes of colon adenocarcinomas in humans.
20
HRAS is primarily expressed on the cell membrane, and thus, antigen should be found on cell membranes by IHC. By using detailed controls, including absorption of antisera with the antigen, we showed that the antibody used by the investigators who claimed it only stained cytoplasm did not stain cell membranes where
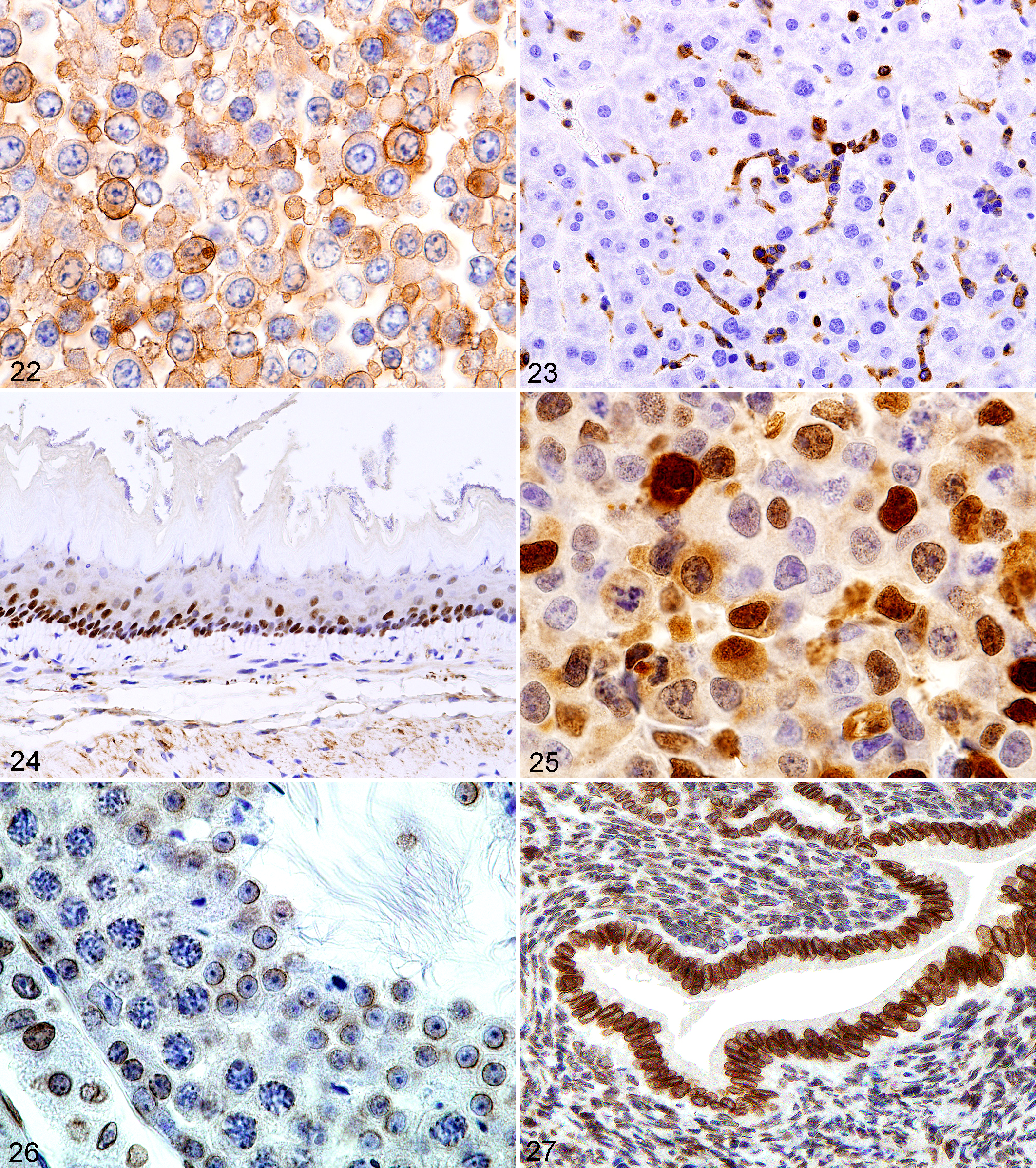
Diffuse cell membrane staining of HRAS on cells in a formalin-fixed, paraffin-embedded (FFPE) pellet prepared from a mouse cell line with an
Primary antibody specificity problems—the case of CD3
Monoclonal antibody clones prepared to the same antigen may not produce the same IHC result. They may not react with the same epitope and not be useful in FFPE sections. We noticed this phenomenon for CD34 in mouse megakaryocytes. 10 One clone stained megakaryocytes while another clone did not. We found that a new lot of CD3 from the same supplier we had used for years suddenly started staining Kupffer cells in normal mouse liver (Fig. 23). It is possible the 1 antibody stains both T cells and T/NK cells, whereas the other antibody only stains T cells (authors’ experience).
In summary, proper controls need to substantiate the colored reaction in an immunohistochemical assay.
Specific staining
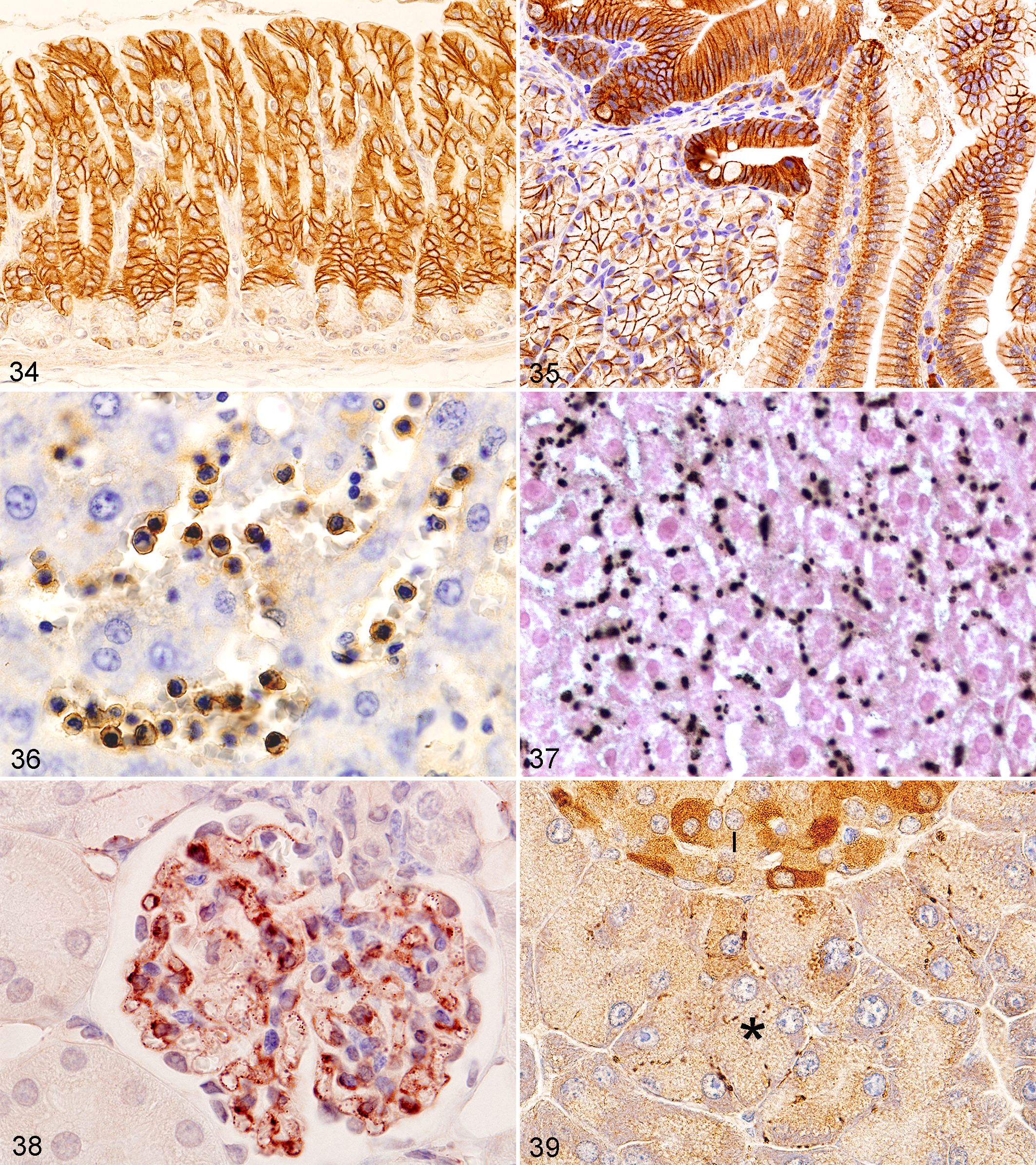
Antigens (usually proteins) may be present in various organelles and anatomical locations within or outside cells. The patterns of antigen distribution can often be seen in 4- to 6-μM sections of FFPE tissue sections stained by an IHC technique. A human protein atlas provides some information on antigens and their staining patterns in human tissues that may be of help in veterinary pathology (http://www.proteinatlas.org). 14 The cell anatomical locations include intranuclear (Figs. 24, 25), intracytoplasmic (Figs. 28–33), membranous (nuclear or cytoplasmic) (Figs. 22, 26, 27, 34–39), and extracellular. In the nucleus, viral inclusions, abnormal inclusions of known or unknown etiology, nucleoli, chromatin, the nucleoplasm, and the nuclear membrane may be IHC positive. Inside the cell’s cytoplasm, mitochondria, lysosomes, rough and smooth endoplasmic reticulum, lipid droplets, peroxisomes, abnormal proteins, pigment granules, and other organelles or other structures can reveal antigen. Often, an antigen is found in several anatomical locations, in several organelles, in the nucleus and cytoplasm, and in the cell membrane and cytoplasm. Cell surface antigens include diffuse membrane antigens (Figs. 34–36), antigens in gap junctions (Fig. 37), and receptors (Fig. 38). Also, a specific cellular anatomical location of an antigen may not have been detectable. Outside the cell, connective tissue, tissue matrices, extracellular abnormal fluids, necrotic debris, blood components (Fig. 8), and other substances both normal and abnormal can be found to be immunoreactive. Antigens may be found in specific anatomical locations in normal cells but in other locations in tumors cells or reactive cells. Also, they may be found in specific cell anatomical locations in some cell types and other anatomical locations in other cell types even within the same tissue (Fig. 39). Fixative may influence the anatomical location of cellular antigens. IHC techniques used can also affect staining results, causing artifacts.
Brain; mouse. Diffuse cytoplasmic staining of a flavivirus antigen in neuron body, dendrites, and axons.
Stomach; mouse. CLDN18 (claudin 18) on cell membranes of the gastric epithelium. Minimal reactivity in the crypt epithelium.
Unexpected staining of cells
This is a relatively common observation when using tumor biomarkers. Keep in mind that antibodies often recognize small peptides (6–10 amino acids) that can be present in other antigens (molecular mimicry) in other cells. Moreover, the same antigen may be present in multiple cell types. 6 If staining produces results that indicate cells or tissues with an antigen not previously known to show antigen present, it may be possible that these cells may be truly positive. Many antibodies stain cells and tissues that are not the main cell of concern for use of the antibody in IHC. For example, many tumor antigens are often found in normal cells and tissues not related to the tumor origin. The tissues may not have been tested previously or observed closely for antigen. Molecular mimicry (especially cross-reactions of infectious agent antigens with normal tissue antigens) may also occur. 13 When investigating a new antibody, it may be useful to run a panel of normal tissues to more fully characterize the assay.
Changes in normal patterns of specific staining in tumors and other conditions
The staining pattern of an antigen in a tumor cell can be identical to that seen in its normal cell-of-origin counterpart (Fig. 40). Genetic changes in genes (eg, mutations) can result in antigen location shift: a cell membrane or cytoplasmic antigen being found by IHC in the nucleus (eg, β-catenin, CTNNB1), which by itself would suggest a mutation has occurred. Also, due to the low levels of an antigen in normal cells or its normal chemical form being unstable especially after tissue fixation, an antigen may not be able to be easily found by IHC but visible readily by IHC after mutant protein of the gene. An example is TRP53 (Fig. 41), where the mutant protein is more stable than the wild-type protein and immunoreacts in the tissue sections. Other tumor antigens may also have IHC patterns different from that found in normal cells. Tumor cells are often less differentiated than normal cells. The location of a tumor cell and stage of differentiation in the tumor may also reveal differences in staining patterns. An example is the histiocytic sarcoma tumor cells in the blood in the hepatic vein with abundant F4/80 cell membrane expression, while the tumor cells in the hepatic parenchyma show less antigen expression (Fig. 42).
Lymphomas often show expression of cell-specific antigens, but their appearance in IHC may be different from normal cells. The antigens may be found in tumor cells at higher or lower levels than in normal cells (Figs. 43, 44). CD45R is a B-lymphocyte antigen found diffusely present on cell membranes of normal B lymphocytes, but in B-cell lymphoma cells, it can be found in a stippled membrane pattern (Fig. 44) at much lower staining intensity than in normal B cells. CTNNB1 (catenin [cadherin-associated protein], β1) is expressed on cell membranes of normal cells but is found in the nucleus in benign tumor cells (adenomas) of the mouse intestine in some mouse models (Fig. 45). Expression, however, may be decreased in the nucleus in invasive adenocarcinomas, perhaps due to phosphorylation of CTNNB1 in cell transformation.
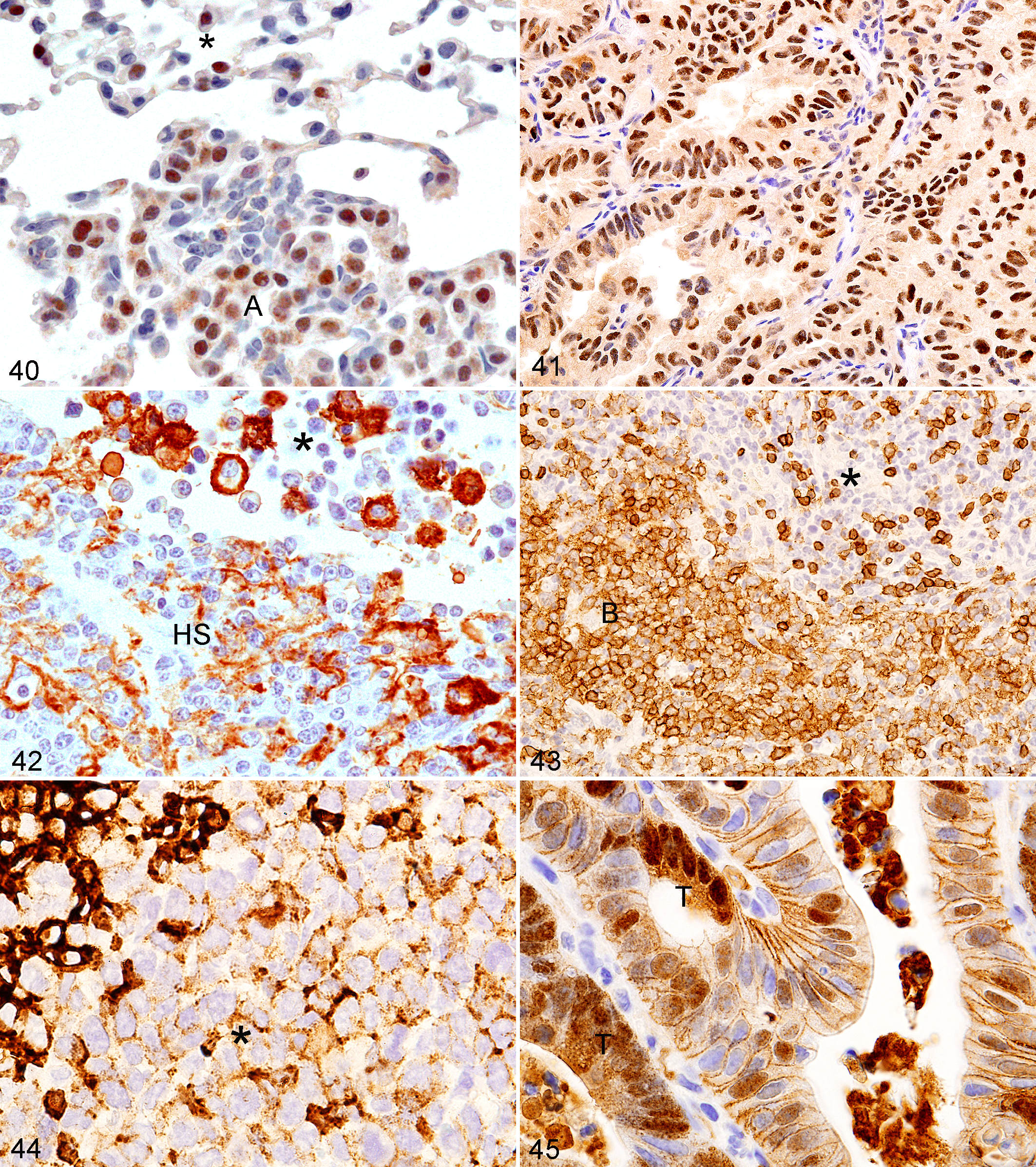
Lung; mouse. TTF1 in normal alveolar type II cells (asterisk) and edge of an adenoma (A).
In inflammatory cells, antigens normally found in a specific cellular distribution may be in the same cellular and tissue locations, in variable amounts or numbers of organelles. Cells not normally expressing an antigen, or cells with such low antigen levels that the antigen is barely detected with IHC, often have marked antigen expression upon activation of the cell due to various insults. Upregulation of genes due to inflammation and infectious processes can result in increased protein (antigen) in specific cells and tissues for a variety of conditions. An example is CHI3L3 (Ym1) in pulmonary macrophages (Fig. 30) in various pulmonary inflammatory conditions and induced in Kupffer cells (Fig. 29). Much less antigen is present in normal alveolar macrophages and none in normal Kupffer cells.
Of course, other methodologies such as in situ hybridization, measurement of mRNA, protein and gene expression methods, ultrastructure, and histochemistry should confirm such variations in protein levels and gene expression.
Troubleshooting
If the predicted or desired result is not found, troubleshooting methods should be attempted. The simple 6-step approach outlined in Table 1 should be the first consideration when troubleshooting IHC antibody problems.
Inadequate Staining With an Antibody: What to Do.

Verify the antibody is effective in the species tested, verify the antibody works in FFPE tissues, find and develop true-positive and true-negative controls including cell pellets with high or low antigen expression (high or low gene or protein expression), and perform literature searches on the use of IHC with the same or related antibodies. The antibody manufacturer can also be contacted for more product information. If all these approaches fail to help determine whether the staining observed is specific staining, use step-by-step troubleshooting protocol procedures as outlined by others. 1,8,9 In our experience, the most common IHC problem is whether the antibody has any utility for IHC, especially in FFPE sections. In research environments, many new antibodies are used and have not been shown to be useful for IHC, even with antigen retrieval and other staining enhancement methods.
How to Find a Good Antibody
Nonspecific staining can often be prevented by using a “good” antibody. Today, there are often numerous antibodies available for many antigens. So which ones to buy and use? Antibodies to specific antigens can be found by searches of websites of companies selling antibodies, publications, and the use of databases on the Internet. Such databases include Linscott’s Directory of Immunological & Biological Reagents (www.linscottsdirectory.com), www.antibodyresource.com, www.antibodydirectory.com, and www.antibodypedia.com. We also use an National Cancer Institute (NCI) database website, which provides very accurate information from its own experiences (http://ncifrederick.cancer.gov/rtp/lasp/phl/immuno/) and some publications on mouse IHC. 5,10,11,12,16 These websites and databases provide many sources for antibodies to the same antigen. You will often have to do more work to find what you may need for the tissues associated with a specific project. How does an individual go about choosing 1 or more antibodies to test? Solely because a company sells an antibody to the antigen of interest does not mean that it will work in FFPE tissue sections. The use of antibodies based on the experience of others, especially experienced colleagues, is a good approach. Publications and the company datasheet often provide valuable information. The figures in the publications, however, often provide equivocal and questionable evidence of the antibodies’ usefulness, and a company’s datasheet information is often incomplete or completely useless (Table 2). The best datasheets will describe how the antibody was made and to what specific antigen (immunogen), Ig isotype, protein concentration, species it works in, applications for which it is optimal (IHC, immunofluorescence, fluorescence-activated cell sorting), if it works in FFPE sections, antigen retrieval required, dilutions to start at, figures of specific staining, and some references on its previous use. What is the evidence that the antibody reacts with the antigen that it was made against? Is there a Western blot showing a single band at a defined molecular weight or convincing figures from a good positive control?
Antibody Datasheet Information Requirements.

FFPE, formalin fixed, paraffin embedded; IF, immunofluorescence; WB, Western blot; IHC, immunohistochemistry.
A good starting point is to use time-tested antibodies that many others have used and are commercially available. On the other hand, a new antibody just made available commercially or made by yourself, a colleague, or collaborator can bring problems. An antibody previously shown to work for IHC in FFPE tissues often has specific positive control-tested tissues by others, often on the same slide as the test tissue or lesion. Also, all other controls will work easily for this well-tested antibody in known positive control cells and are often shown in publications. For antibodies used in our laboratory for the first time and perhaps by anyone for the first time in IHC, we often try 3 dilutions of 1:100, 500, and 1000 or what, if any, dilutions are recommended in the company datasheet or by the investigator preparing the new antibody.
Sometimes specific methods are suggested by the company datasheet or the research laboratory supplying the antibody. The positive and negative control may be a simple one—a tissue that should contain known or hypothesized positive and negative cells. Often that is all you will need. If you are lucky, the cell pellet technique using high- and low-expressing cells from specific cell lines will definitely help. After staining with these 3 initial dilutions, search for the dilution that appears to not stain cells and tissues nonspecifically and stains specific cells differentially. If all dilutions produce a very diffusely brown (with DAB as the chromogen) tissue section, then try 3 incrementally higher dilutions (eg, 1:1000, 2000, 4000). If dilutions eventually show no definitive specific cellular staining, then the conclusion may be the antibody does not work on postulated cells thought to contain the antigen. The most important controls must be used. 6,9 There are many antibodies made in laboratories and sold commercially that will not provide evidence of antigen in FFPE tissues despite the company’s claims. Contact the company, colleagues, and authors after you search the literature for optimal figures of tissues showing positive immunoreactivity for the antigen and antibody of interest. If these simple steps do not work and you are convinced that the antibody should work (based on positive results in other laboratories, especially by trusted colleagues), go to the detailed troubleshooting references given below.
Inadequate Staining With a Tested Antibody
If, after using an antibody that should perform well for your tissues and with antibody dilutions and methods suggested by the product datasheet, publications, or colleagues, the staining does not appear specific, or cells show too little or too much staining (ie, brown with DAB as the chromogen), verify that the antibody cross-reacts with mouse antigen. If the product sheet does not indicate that the antibody works on mouse tissue, that situation becomes a problem. Many investigators assume that an antibody will be useful for all species. This assumption is often not true. No matter how the procedure is modified, it may never work. Under these circumstances, the best controls are needed (ie, tissues or cell pellets highly expressing antigen). Test species cross-reaction by running tissue from the antibody target side by side with the mouse tissues.
If the product sheet indicates that the antibody does react with the mouse antigen, then there may be another problem. Check if the fixative is important, or antigen retrieval may have to be modified. Check the step-by-step protocol procedures. 2,8,9
For monoclonal antibodies, various clones may have been made to the same antigen, but the antibodies may react with different epitopes, some of which may not be preserved in FFPE tissue. Thus, all monoclonals to an antigen may not react in FFPE to the same antigen under the same protocol conditions. One example is CD34. 10
Inadequate Staining With an Untested Antibody
New untested antibodies represent a problem for use and proper interpretation. 8,9 Controls become of greatest importance. 6,8,9 Proper controls include cells and tissues well known to show antigen in FFPE tissue sections. We will mention some of these procedures again because they are very important. In vitro cell lines of cells showing high or low antigen levels are the best simple controls to use. Cell pellets can be prepared from cell lines by scraping off cell monolayers with a rubber policeman from the flask, centrifuging it at low speed for 5 to 10 minutes, and then layering the fixative gently over the cell pellet in the centrifuge tube and fixing it overnight. Gently lifting the fixed pellet from the tube and embedding it in paraffin completes the task. A tissue processing gel can be used to more closely mimic normal solid tissue processing. Fixatives can play a major role for providing optimal results. 5 –7 Tissue macroarrays or microarrays are most helpful provided they contain positive and negative control tissues. Macroarrays are made easily by hand embedding small portions of normal or abnormal tissues into 1 block. Microarrays are prepared with the use of manual or automated tissue arrayer equipment, and many hundreds of small plugs of tissue can be embedded in 1 block. Microarrays are especially useful for studying many tumors. Many slides of tissue arrays are also commercially available but at high cost per slide (www.biomax.us/tissue-arrays; www.tissue-array.net; www.pantomics.com/tissuearrays.aspx).
If the new antibody is used on the controls noted above and the controls include cells or tissues with known high and low expression, the entire process of determining the presence of antigen in FFPE sections is made much easier. Often the new antibodies either stain nothing specifically at the lowest dilution or create nonspecific background, which in some instances may be erroneously interpreted as specific immunoreactivity by inexperienced investigators. The use of the above controls must confirm that the new antibody demonstrates that specific antigen in FFPE tissues.
Troubleshooting: Advanced Protocol and Reagent Considerations
Advanced troubleshooting may involve detailed stepwise procedures. The step-by-step procedures should be checked, including when there is weak or no staining of the positive control and weak or no staining of test slides, inadequate or no staining of the test slide and adequate staining of the positive control slide, and when excessive background staining is observed. The detailed methods for determining problems step by step have been well described for 24 items (www.dako.com/us/index/knowledgecenter/kc_publications/kc_publications_edu/immunohistochemical_staining_methods.htm) and in veterinary publications to determine the reason for the failure of specific staining for an antigen, especially in cases where it has worked previously in your laboratory or in other laboratories. 1,6 –9 These include problems with chemical components of specific steps, pH of solutions, temperature of reagents, quality of chemicals used, and preparation of solutions.
Summary and Conclusions
IHC is a technique that requires care in each step of the procedure, including tissue fixation, antibodies used, and details in the IHC protocol. In our experience, the most common problem is the quality of the primary antibody used in IHC on FFPE sections. Any deviation from an optimal protocol can result in nonspecific background staining, less than optimal specific staining, or no staining at all. Careful evaluation of all components involved in each step of the IHC technique and correction of deficiencies will likely result in optimal staining.
Footnotes
Acknowledgements
We are grateful for the laboratory work performed by our professional staff and other colleagues over the years. Dr Ward appreciates the important assistance over the past 30 years of Fred Argilan, Cindy Harris, Keith Rogers, Barbara Kasprzak, Donna Butcher, Larry Faucette, Dr Miriam Anver, Dr Diana Haines, Dr Alfredo Molinolo, Elizabeth Williams, and Dr Shioko Kimura. We thank Dr Ya-hui Chi for the SUN1 IHC and Dr Maria Lucia Dagli for GJB1 IHC. Dr Rehg is grateful for assistance from the St Jude Veterinary Pathology core staff and Dorothy Bush for her dedication to immunohistochemistry. We thank Dr Debbie Gillette for the plating of figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest involving the research, authorship and/or publication of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This publication was supported, in part, by grant P30C CA 21765 from the National Cancer Institute and by the American Lebanese Syrian Associated Charities (ALSAC). Its contents are solely the responsibility of the authors and do not necessary represent the official view of the National Cancer Institute.
