Abstract
Fatty liver hemorrhagic syndrome, characterized by sudden death in overconditioned hens due to hepatic rupture and hemorrhage, is one of the leading noninfectious idiopathic causes of mortality in backyard chickens. Nutritional, genetic, environmental, and hormonal factors, or combinations of these, have been proposed yet not proven as the underlying cause. In an attempt to characterize the hepatic changes leading to the syndrome, this retrospective case study examined 76 backyard chickens that were diagnosed with fatty liver hemorrhagic syndrome between January 2007 and September 2012 and presented for necropsy to the diagnostic laboratory of the California Animal Health and Food Safety Laboratory System. A majority of the birds were female (99%), obese (97.5%), and in active lay (69.7%). Livers were examined histologically, and the degree of hepatocellular vacuolation (lipidosis), the reticular stromal architecture, the presence of collagenous connective tissue, and vascular wall changes were evaluated and graded using hematoxylin and eosin, Gomori’s reticulin, oil red O, Masson’s trichrome, and Verhoeff-Van Gieson stains. Interestingly, there was no correlation between lipidosis and reticulin grades; hepatocellular lipidosis was absent in 22% of the cases and mild in 26% of the cases. Additionally, there was evidence of repeated bouts of intraparenchymal hemorrhage before the acute “bleed-out” in 35.5% of the cases. These data are not supportive of the previously proposed causes and provide a framework for future studies to elucidate the pathogenesis of this condition. Furthermore, the data shown in this study support hemorrhagic liver syndrome as a more accurate name, as hepatic lipidosis is absent in a significant proportion of ruptured livers.
Keywords
Fatty liver hemorrhagic syndrome (FLHS) is the most common noninfectious cause of mortality in laying backyard chickens in Northern California. 17 First reported in commercial layers by Couch in 1956, 7 FLHS presents clinically as sudden death in nutritionally overconditioned laying hens. Pathologic examination reveals abundant coelomic fat reserves and an enlarged, pale, tan to yellow, friable liver with subcapsular and parenchymal hematomas and hemorrhages. 11,26 There is often a capsular rupture with associated peracute intracoelomic suffusive hemorrhage (hemoabdomen), with a large blood clot overlaying the liver capsule, and the carcass is noticeably pale due to hypovolemic anemia. Histologically, there is often hepatic lipidosis (HL) with hemorrhage and often disruption of the reticulin framework of the liver, described as reticulolysis. 29 HL, although present in most cases, is not a constant finding in overweight chickens with hepatic rupture and hemorrhage, contrary to the name FLHS.
The cause of FLHS has yet to be fully elucidated but is likely a combination of nutritional, genetic, environmental, and hormonal factors. 11,26 It was noted early in the characterization of the syndrome that hens that died due to FLHS were in good to obese body condition, suggesting that diet may play a role in the creation of the HL. 7 One plausible hypothesis for the pathogenesis of FLHS is that the vacuolar swelling of hepatocytes disrupts the reticulin structure of the hepatic plates due to mechanical stress, leading to hemorrhaging from the sinusoids. 11,29 In addition to the mechanical stress to the liver architecture, some have suggested that the excessive lipid metabolism taking place in overfed laying hens may facilitate oxygen radical-mediated destruction of the reticulin fibers. 25,26 Spurlock and Savage demonstrated that feeding a diet rich in antioxidants reduces the incidence of FLHS; 25 however, other studies have not shown the same associations. 23 Genetics may play an integral role in susceptibility of FLHS; in fact, Squires and others have extensively studied a strain genetically predisposed to FLHS, although a specific mechanism has not been elucidated. 8,28,32 Clearly, further characterization of the syndrome is warranted.
At the California Animal Health and Food Safety Laboratory System (CAHFS), Davis, FLHS is diagnosed commonly in backyard chickens submitted for necropsy. 17 In commercial flocks in California, poultry veterinarians report a decrease in incidence presumably related to a carefully calculated diet (personal communication with Dr. B. Charlton, CAHFS, Turlock, California), although FLHS has been reported as a major cause of death in commercial layers, reaching up to 5% mortality during a laying cycle, and also as a reason for sudden drop in egg production in affected flocks. 14,22 If predisposing factors can be elucidated and the pathogenesis more fully understood, it may be possible to significantly reduce the incidence of FLHS in backyard flocks. The goal of this study was to characterize the changes in hepatic architecture and clarify the distribution of the pathologic lesions in spontaneous FLHS cases, in particular whether or not there is a clear association between this syndrome and HL, in an attempt to elucidate the mechanism underlying the lethal liver rupture and associated bleed-out. The spectrum of hepatic vacuolar changes was evaluated and compared with the integrity of hepatic reticulin, collagen, and vascular elastin within a population of chickens diagnosed with naturally-occurring FLHS.
Materials and Methods
Case Selection
A retrospective analysis of backyard chicken cases that presented to CAHFS, Davis, for pathologic examination and diagnostic testing was performed. Cases were selected from the CAHFS database (STARLIMS 10.5.67) with the search criteria of “fatty liver” and/or “lipidosis” in the diagnosis or comments fields between January 1, 2007, and August 31, 2012. These search criteria retrieved all cases of backyard chickens that were diagnosed with a ruptured liver, regardless of fatty change, as the primary cause of death or morbidity of the bird, as hepatic rupture and hemorrhage cases have long been diagnosed as FLHS. The submissions were from backyard chicken flocks, defined as flocks with less than 1000 birds in Northern California. In addition to histopathologic assessment, the following data from each case was collected when given on the submission form or from the comments in the report of the pathologist: age, breed, sex, laying status, date of presentation for necropsy, body condition, gross description of liver changes, and concurrent diagnoses. We also noted when copper, selenium, and/or vitamin E concentrations in liver were determined. The overall numbers of case submissions per month from backyard chicken flocks to CAHFS, Davis (from January 1, 2007, to August 31, 2011) were collected to be able to compare the seasonal distribution of FLHS cases to all backyard chicken cases.
The “lipidosis” search criterion retrieved 24 birds without hepatic rupture or hemorrhage in which “hepatic lipidosis” was a part of the reported diagnosis in birds that died of various causes. The liver from 3 of these birds, also obese, without evidence of concurrent coelomic inflammation or severe autolysis were randomly chosen as lipidotic controls (henceforth, HL). Liver from an adult female laying chicken without histopathologic lesions was consistently used as the baseline control throughout the study. All 4 control livers underwent the same procedures of special stains and grading. Additionally, liver sections from three 4-week-old clinically healthy broiler chickens in good but nonobese nutritional body condition from a commercial facility were used as controls for comparison of our vacuolar grading system, as described below.
Hepatic Histopathology and Scoring System
Each liver was fixed in 10% buffered formalin, embedded in paraffin, and sectioned at 4 μm. Tissues were stained with hematoxylin and eosin (HE), Gomori’s Reticulin (GR), Masson’s Trichrome (MT), and Verhoeff-Van Gieson (VG). The degree of hepatocellular vacuolization characterized by single or multiple clear, well-demarcated cytoplasmic vacuoles was graded on the following criteria as viewed on HE: (0) no to very rare vacuolization, (1) less than 50% of hepatocytes containing vacuoles of any size, (2) 50% or greater hepatocytes containing variably sized vacuoles or diffuse vacuolization with small vacuoles, and (3) diffuse vacuolization with large or medium to large vacuoles. For all grading schemes to be described, when multiple liver sections were available for a case, all sections were viewed. If there was a marked difference among the sections for a particular case, those sections with evidence of hemorrhage were used for grading.
After scoring all cases for degree of vacuolization on HE, 5 cases from each vacuolar grade category were randomly selected (n = 20), and formalin-fixed liver tissue from these cases was embedded in Tissue-Tek optimal cutting temperature compound (Sakura, Torrance, California), sectioned at 8 μm, and stained with oil red O (ORO) for comparison of vacuolization score to true lipid content. ORO staining was performed only on cases from 2010 through 2012, as stored formalin-fixed tissues were unavailable from earlier cases. Formalin-fixed liver from the 3 HL control cases and the 3 broiler control cases were similarly processed and stained with ORO. The broiler control cases were used to assess the normal levels of hepatic vacuolization in liver from healthy chickens.
The amount of reticulin fibers present was graded on the following criteria as viewed on GR: (0) reticulin fibers present and continuous in sinusoids (no loss), (1) reticulin fibers present in 50% or greater of sinusoids (mild loss), (2) reticulin fibers present in less than 50% of sinusoids consistently across the section (moderate loss), and (3) reticulin fibers absent to rare in sinusoids in the section (complete loss).
Sections stained with MT were examined for the presence of intraparenchymal and sinusoidal fibrosis as well as adequate collagen surrounding portal vessels compared to control liver sections. Evidence of prior hemorrhage in the liver parenchyma was evaluated by the presence of hemosiderosis and/or hematomas surrounded by fibrosis, as viewed on HE and MT stains. The integrity of hepatic vasculature was assessed by the presence or absence of elastin and elastic laminae in both medium and large vessels viewed on sections stained with VG.
Statistical Analysis
Statistical analyses were performed with Prism 5 for Mac (GraphPad Software Inc., San Diego, California). Submissions by calendar month of backyard birds with and without FLHS were compared using chi-square or Fisher exact tests to assess whether FLHS submissions were significantly associated with calendar month, whether degree of hepatic vacuolization was associated with hepatic rupture, whether reticulin loss was associated with prior hemorrhage, and whether degree of vacuolization was associated with prior hemorrhage. Correlation between reticulin grade and vacuolar grade was tested using Spearman rank correlation coefficient. For all analyses, a P value <.05 was considered significant.
Results
The search criteria retrieved 76 backyard chicken cases of FLHS in the study period. Birds ranged in age from 7 months to 7 years. The majority were female (n = 74 of 75 reporting sex). Breeds represented include Rhode Island Red (n = 8), Wyandotte (n = 7), Ameraucana (n = 6), Orpington (n = 6), Brahma (n = 3), Silkie (n = 2), Delaware (n = 1), Barred Plymouth Rock (n = 1), and Leghorn (n = 1); breed was not reported for 41 birds. Birds presented for necropsy throughout the year. Although not statistically significant (P = .08), FLHS incidence tended to be seasonal, compared to total chicken submissions, with FLHS submissions most heavily concentrated in spring and summer (Fig. 1).
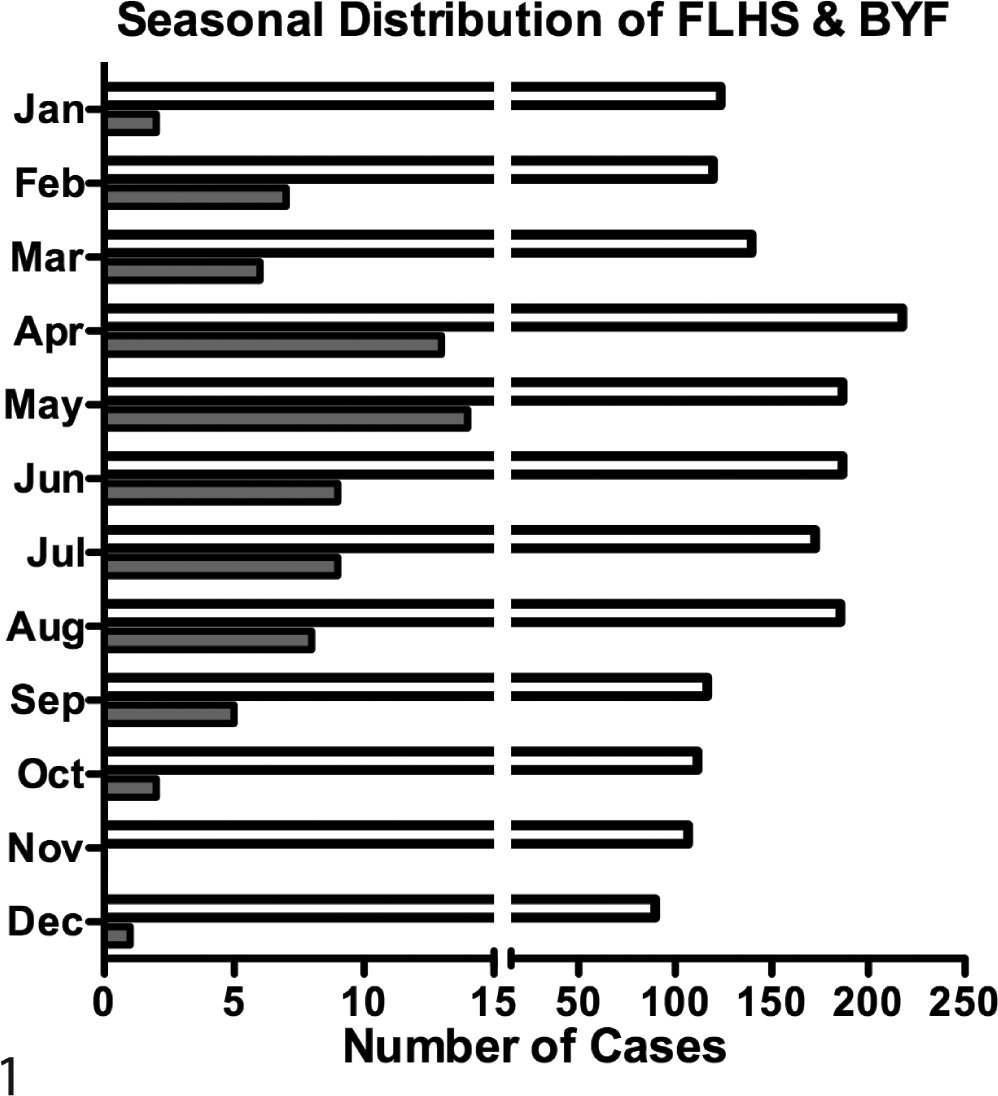
Seasonal distribution of fatty liver hemorrhagic syndrome (FLHS) and backyard flock (BYF). Number of cases of FLHS (gray bar) and BYF (white bar) that presented to the California Animal Health and Food Safety Laboratory, Davis, between January 1, 2007, and August 31, 2012, stratified by month.
Of the 76 FLHS-affected birds, 53 (69.7%) were in active lay, defined as having follicles greater than or equal to 2 cm, and 6 (7.9%) were inactive; ovary size was not reported for 16 (21.1%), and 1 (1.3%) was a male. The majority (n = 74, 97.4%) of FLHS-affected birds had abundant to excessive subcutaneous and intracoelomic fat stores and were classified as overweight to obese; 1 (1.3%) bird was emaciated, and body condition was not reported for 1 bird.
FLHS cases presented with a history of sudden death or peracute nonspecific clinical signs, followed by euthanasia or natural death. FLHS was determined to be the cause of death or reason for euthanasia in all but 1 case—namely, a chicken euthanized for neurologic signs and found to have fulminant Marek disease and concurrent liver rupture and intracoelomic hemorrhage. Among the 76 FLHS cases, additional diagnoses were as follows: low liver copper (16 of 32 tested); yolk peritonitis (n = 9); respiratory lesions, including pneumonia, bronchitis, and bronchopneumonia (n = 6), latent Marek disease lesions (n = 5); endoparasite infestation (n = 4); low vitamin E (n = 3 of 5 tested); leiomyoma of the mesosalpinx (n = 2); visceral gout (n = 2); septicemia (n = 1); and fulminant Marek disease (n = 1). There was no evidence of selenium deficiency in any of the 26 birds tested.
As the majority of FLHS cases had similar signalment, nutritional body condition, and history, specific case details are not discussed. However, brief details are given for the 2 cases that were outliers. The sole case of FLHS in a male chicken was in a nutritionally overconditioned adult found dead after signs of lethargy and anorexia for less than 24 hours. He was diagnosed with Escherichia coli septicemia, vitamin E deficiency, and FLHS with severe fatty change. The only case of FLHS in an emaciated chicken occurred in a 1-year-old female of unknown laying status with a clinical history of anorexia, weakness, and weight loss. In addition to FLHS, severe diffuse histiocytosis was described in multiple organs, including the liver, intestinal mucosa, ovarian stroma, spleen, and lung. No intralesional acid-fast bacteria were evident upon additional staining with Ziehl-Neelsen, and a chronic inflammatory process was suspected as the likely cause of the emaciation. Severe inflammatory changes in addition to the vitamin E deficiency in the first bird likely played a role in the development of hepatic rupture and bleeding for both chickens.
In addition to FLHS cases, our search criteria retrieved 24 cases of moderate to severe hepatic vacuolar change diagnosed as HL. These birds had no intracoelomic or hepatic hemorrhage and died or were euthanized for diagnoses different from that of hepatopathy. Three of these HL cases were randomly selected to serve as controls as previously described. All 3 were obese adult female chickens with inactive ovaries. They presented to CAHFS for necropsy in August (n = 1) and December (n = 2) and, in addition to HL, were diagnosed with encephalitis (n = 1) and cervical trauma (n = 1) or had an undetermined cause of death (n = 1).
Grossly, livers from FLHS-affected birds were reported as large, friable, and soft and varied in color from tan to yellow to orange. Subcapsular hematomas and pinpoint hemorrhages were visible on the liver capsule, and intraparenchymal hemorrhages and hematomas were often visible upon cut section. Large intracoelomic blood clots were adhered to the left or right liver lobe, often overlying and continuous with a grossly visible capsular rupture (Fig. 2). Livers from HL-affected birds were also enlarged, soft, friable, and pale tan to yellow but had no hemorrhage.
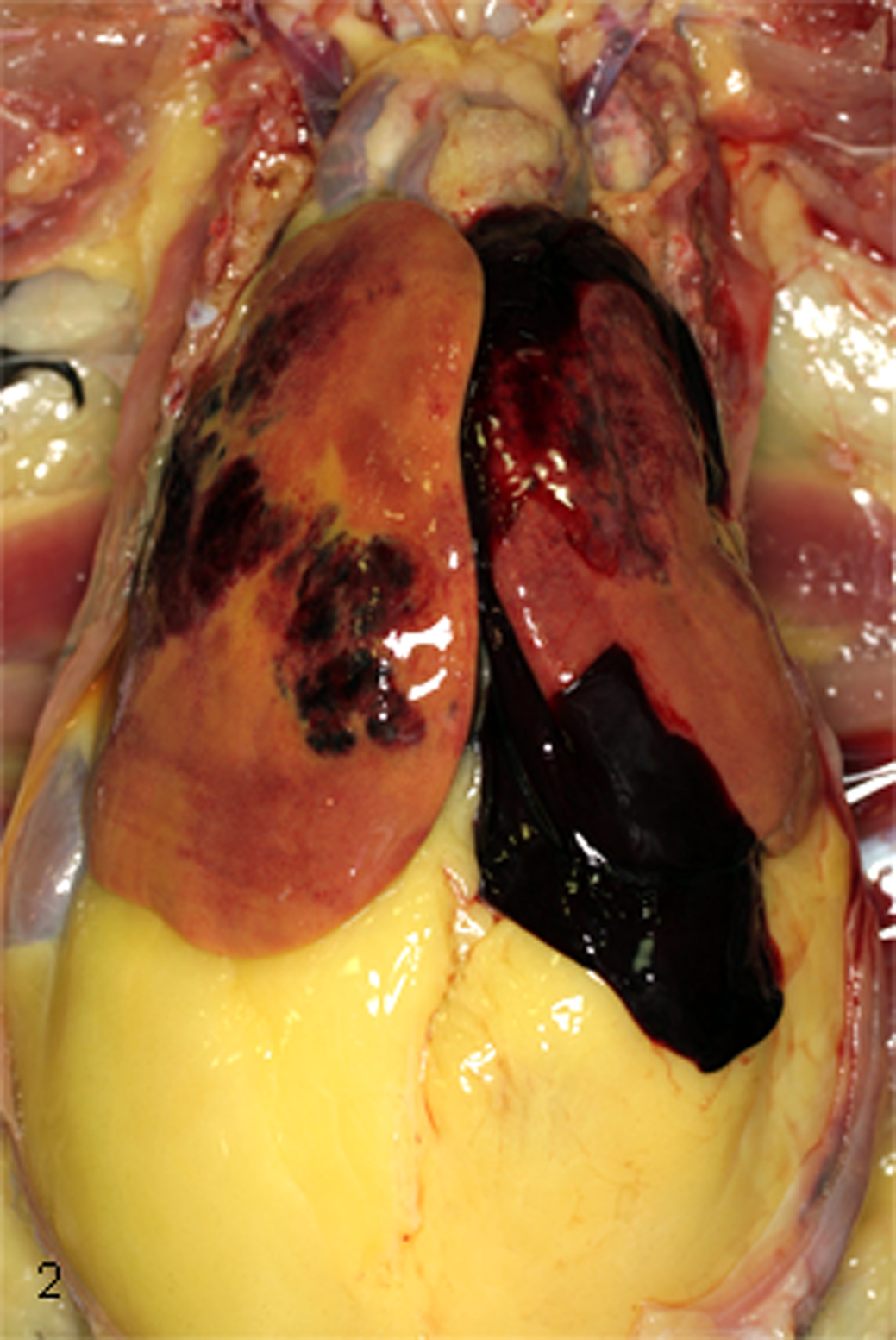
Coelomic cavity; chicken. Multifocal to coalescing subcapsular hepatic hemorrhages with diffuse hepatic pallor, intracoelomic blood clots, and abundant intracoelomic fat.
Histologic changes of FLHS-affected livers spanned a broad range of severity. Degree of vacuolar change was highly variable, from rare evidence of mild hepatocyte vacuolization to diffuse severe involvement. Vacuolar change was often patchy and more heavily focused in the centrilobular areas with centrilobular bridging. Vacuole size and number ranged from numerous small pinpoint vacuoles to large single vacuoles markedly distending the hepatocyte cytoplasm. Hepatocytes in HL-affected control livers diffusely contained medium to large vacuoles. Subcapsular and/or intraparenchymal hematomas, smaller unorganized sinusoidal hemorrhages, and occasionally blood clots adhered to the capsule were present in FLHS-affected livers. Many liver sections had evidence of previous and ongoing hemorrhage as characterized by the presence of hemosiderin-laden macrophages and fibrosis surrounding organizing hematomas. Additional findings included sinusoidal fibrosis, portal bridging fibrosis, nodular regeneration, portal inflammatory infiltrates, and extramedullary hematopoiesis.
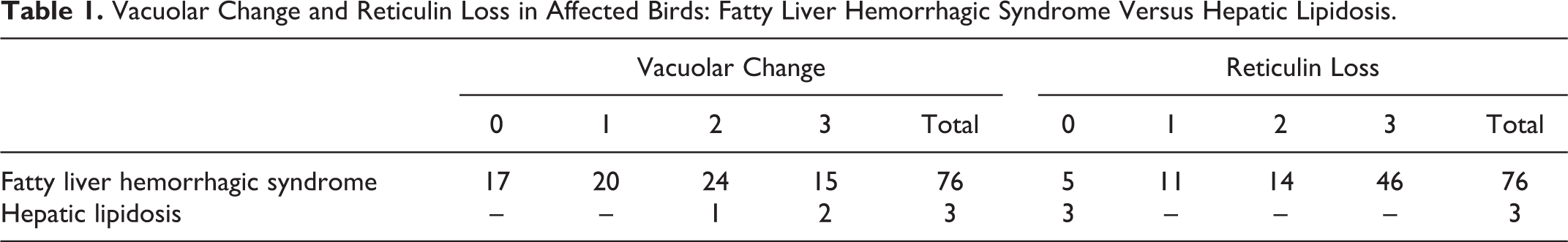
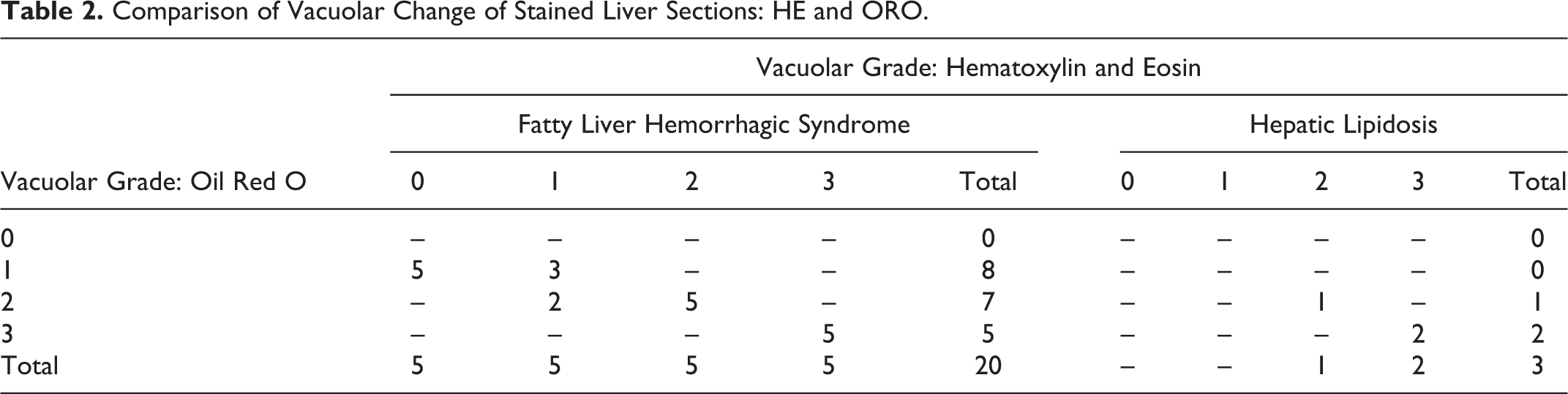
The grades of hepatic vacuolization of FLHS cases as assessed on HE sections were distributed relatively evenly among the categories: grade 0 (n = 17), grade 1 (n = 20), grade 2 (n = 24), and grade 3 (n = 15) (Fig. 3a–c, Table 1). The cases of HL with no accompanying hemorrhage were graded 2 (n = 1) and 3 (n = 2). There was good correlation between HE and ORO for grade 2 and 3 livers, but ORO staining revealed positive lipid staining in small pinpoint vacuoles that were not evident on HE in grade 0 and 1 livers (Fig. 4a–c, Table 2). Additionally, livers from the 3 nonobese healthy commercial broiler chickens showed rare to occasional small vacuoles on HE, grade 0 (n = 2) and grade 1 (n = 1), while small pinpoint vacuoles were evident diffusely throughout the parenchyma on ORO-stained sections.
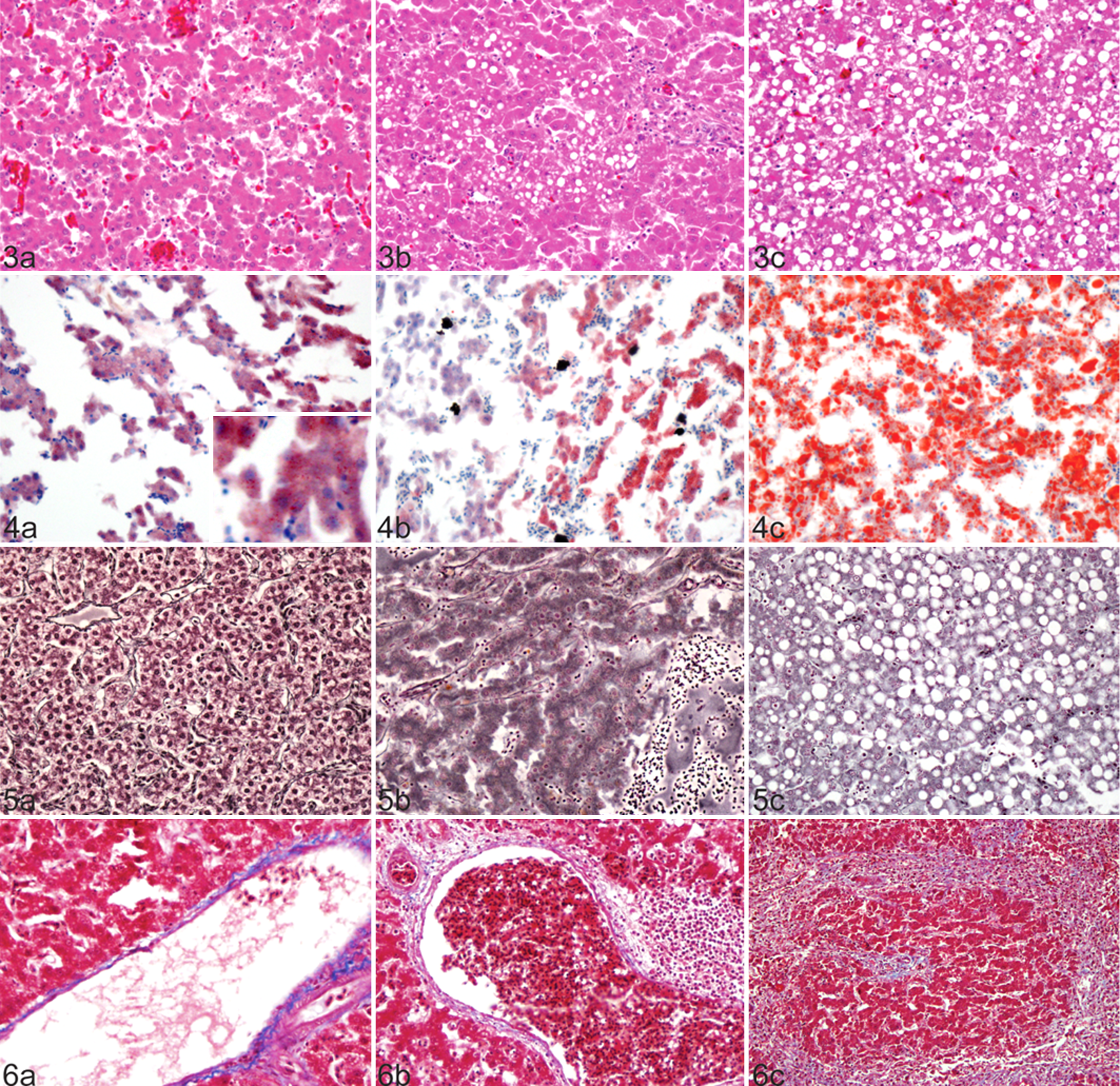
Liver; chicken. (a) Nonvacuolated hepatocytes, vacuolar grade 0, are present amid intraparenchymal hemorrhage. (b) Small to medium vacuoles fill approximately 60% of hepatocytes, vacuolar grade 2. (c) Medium to large vacuoles distend hepatocytes diffusely throughout the tissue, vacuolar grade 3. Hematoxylin and eosin.
Vacuolar Change and Reticulin Loss in Affected Birds: Fatty Liver Hemorrhagic Syndrome Versus Hepatic Lipidosis.
Comparison of Vacuolar Change of Stained Liver Sections: HE and ORO.
To assess the level of reticulin loss in our set of cases, we assessed the reticulin fibers in FLHS- and HL-affected livers with GR. Reticulin staining on control tissues was strongly positive surrounding vessels and ducts in both portal and centrilobular regions and streaming continuously along sinusoids of hepatic plates 2 hepatocytes thick (Fig. 5a). Conversely, FLHS-affected livers illustrated varying degrees of reticulin absence from sinusoids (Fig. 5b, c). The majority (79%) of livers from FLHS-affected birds showed marked loss of sinusoidal reticulin; 60 of 76 cases had greater than 50% loss of reticulin fibers (grade 2 or 3), while another 11 had some detectable loss, and only 5 were normal (Table 1). All liver sections from HL-affected birds had normal reticulin. There was no correlation (P = .39) between severe reticulin scores and high-grade vacuolar change, or vice versa, in our population (Table 3). There was also no association (P = .9086) between reticulin loss and prior hemorrhage.
Comparison of Vacuolization and Reticulin Loss.a
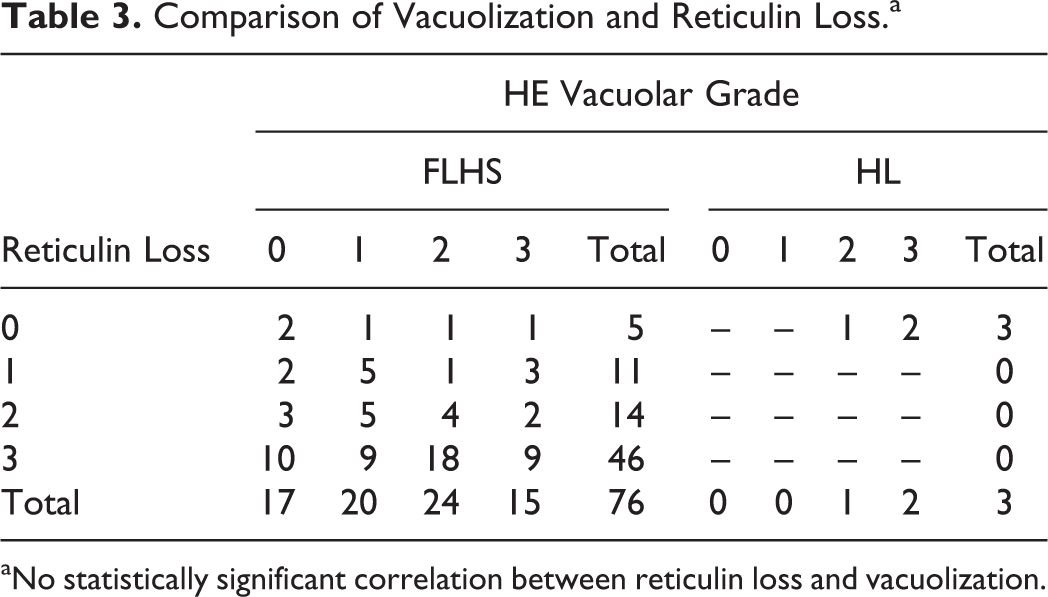
aNo statistically significant correlation between reticulin loss and vacuolization.
Interestingly, 27 of the total 76 (35.5%) FLHS livers showed evidence of previous bouts of intraparenchymal hemorrhage before the acute bleed-out, as evident by hemosiderosis (n = 16) and/or fibrosis around the hematomas (n = 12). Statistically significant association was found between a low degree of vacuolization (grades 0 and 1) and increased incidence of prior hemorrhage: 18 of 37 (49%) of cases with rare to less severe vacuolization (grades 0 and 1) had previous hemorrhages, while only 9 of 39 (23%) livers with moderate to severe vacuolization (grades 2 and 3) had previous hemorrhage (P < .05).
To assess the level of fibrotic change present in FLHS-affected livers, sections were stained with MT. Of the 76 FLHS-affected sections, 17 (22.3%) had decreased bright blue mature collagen around smaller portal vessels compared to similarly sized vessels from control tissues (Fig. 6a, b). Rather, the vessels were surrounded by scant amounts of pale blue wispy collagen. Changes in portal vessel collagen were not seen in HL-affected livers. As described previously on HE, fibrosis bridging portal regions (n = 4) (Fig. 6c), perisinusoidal fibrosis (n = 2), and perihematoma fibrosis (n = 12) were also evident in FLHS-affected livers and more elegantly defined with MT. There was no difference in the elastin and elastic laminae, as viewed by VG across or between vacuolar grade groups in the study population.
Discussion
To the our knowledge, this is the first retrospective study of naturally occurring FLHS in backyard chickens. This study demonstrates that nutritionally overconditioned laying birds are at risk for developing FLHS, particularly in the spring and summer months. Additionally, we have confirmed that, contrary to the name of the condition, a significant proportion (22.4%) of FLHS cases have little to no lipid vacuolization in the liver. In our opinion, the term hemorrhagic liver syndrome is a more accurate name of the disease in cases in which HL is absent. Furthermore, there is no correlation between the degree of reticulin loss and vacuolar change, suggesting that the previously proposed hypothesis for mechanical reticulolysis in FLHS may be incomplete. 11,29
There was a profound variation in the degree of hepatocellular vacuolization within the FLHS case population. In contrast to mammals, the liver in birds is responsible for the majority of lipogenesis, and dietary fat in birds is directly transported to the liver by the portal vein. Therefore, birds are expected to normally have a higher hepatic lipid concentration and microvacuolization. 6,15 While there are many schemes for grading lipid vacuolization reported in both human and veterinary medicine, they generally span 0 (no change), 1 (mild change), 2 (moderate change), and 3 (severe change). 5,12,24,29 Based on the range of lipid accumulation that we observed in our population, we chose to classify livers with less than 50% vacuolization as mild and greater than 50% as moderate and severe. Because small vacuoles coalesce into larger vacuoles as disease progresses, 21 we chose to classify diffuse small vacuolization as moderate change (grade 2) and reserve the classification of severe change (grade 3) for diffuse medium-sized to large vacuoles. Liver sections described as grade 0 on HE in our study had varying degrees of microvacuolization as detected with ORO staining. The 3 healthy commercial broilers used as controls also showed this vacuolar change. Thus, 22% (n = 17) of the FLHS cases had no excess hepatic lipid accumulation compared to controls, and another 26% (n = 20) had only mild vacuolar change despite being overweight or obese. Additionally, we observed that moderate to severe HL without parenchymal or subcapsular hemorrhage/hematomas is not uncommonly seen in backyard chickens that presented to CAHFS. While some studies have suggested that hepatocyte swelling via fatty change may be a factor in hepatic rupture, 5 –7,11,29,30 our results show that there is no correlation between degree of fatty change and hepatic rupture; therefore, vacuolar change is not a good prognostic factor for FLHS. The cases with no to mild vacuolization were significantly more likely to have had prior hemorrhage before the acute bleed-out compared to cases with moderate to severe vacuolar grade. Perhaps the retained hepatocellular integrity of mildly vacuolated livers is protective against microhemorrhages leading to capsular rupture and massive bleed-outs in moderately to severely vacuolated livers.
Surprisingly, there was no correlation between the degree of vacuolization and reticulin loss, as was also observed in a 1975 study of naturally occurring FLHS in Australia. 18 Reticulin fibers, made of mainly collagen III with attached fibronectin and glycoproteins, line the sinusoids and help form the structural integrity of the liver. 9 Loss of reticulin fibers, reticulolysis, has been associated with FLHS, but the mechanism is not understood. 10,18 Some have proposed that grossly distended vacuoles may mechanically stress reticulin fibers; in fact, there are positive correlations between steatosis and reticulolysis in humans. 11,24 –26,29 We did occasionally note regional correlations between vacuolization and reticulin loss, but this was not common. If hepatocyte swelling induces mechanical stress upon the reticulin meshwork causing reticulolysis, then the diffusely vacuolated livers should also be those with severe loss of reticulin, which was not consistently observed in our case population. There were many sections of liver from FLHS-affected birds with rare vacuolization and absent reticulin and, conversely, heavily vacuolated livers from HL-affected birds had normal reticulin. Clearly, there are additional factors contributing to the reticulin loss in FLHS. However, it is clear that in our population, reticulin loss is a stronger predictor of FLHS than vacuolar change.
FLHS is most commonly observed in laying hens but can be recapitulated in male or female chickens treated with exogenous estradiol. 20,27 Most of the FLHS cases (97.3%) in our study were female, and the majority (69.7%) had active ovaries, suggesting that they were under the influence of high estradiol levels. It should be noted here that we did not perform a cross-sectional analysis to determine the percentages of sex, age, nutritional condition, and laying status of the total backyard chicken submissions to CAHFS, Davis, so whether the FLHS data are representative of the general population cannot be interpreted. Although the cases were a minority, we also observed FLHS in hens with inactive ovaries and in 1 male; however, all of these birds had excess fat stores, suggesting that obesity alone can promote FLHS. The male had multiple additional significant pathologies that may have contributed to the development of the HL and rupture or the death. The development of spontaneous FLHS in roosters cannot be evaluated due to the very low number of birds. The only case of FLHS observed in an emaciated chicken was in the presence of a chronic multisystemic histiocytic inflammatory condition of unknown etiology that likely contributed to the emaciation. This particular chicken had rare vacuolar change and no reticulin loss but did have evidence of perisinusoidal fibrosis, making the inflammation and subsequent fibrotic change the likely cause of the rupture, although the presence of additional contributory factors cannot be ruled out.
There appears to be a seasonality to the incidence of FLHS, with rates peaking in mid- to late spring and summer following the egg-laying pattern, as has been reported. 13,19,22 The individual husbandry practices of each premises is not known, but communication with clients by A.M. revealed that most are likely to allow the natural course of molting and undergoing of reproductive senescence during winter months. Thus, it is likely that with the onset of egg production in the spring, circulating levels of estradiol, food intake, and hepatic fat content/stores will increase, predisposing the birds to FLHS. Additionally, environmental stressors such as high heat have been shown to increase the incidence of FLHS, 7,11,22,26 which could also help to explain the high incidence during summer in Northern California. The potential role of trauma-induced hepatic rupture also cannot be totally ruled out in some of the cases.
Oxidative damage due to rancification of feed, increased lipid peroxidation, and/or antioxidant deficiencies may also contribute to reticulin loss in FLHS. 25 Indeed, there were 3 cases of vitamin E deficiency detected in our study, but only 5 birds had vitamin E testing performed, so broad conclusions cannot be made. There was no evidence of selenium deficiency among those tested in our case population.
In almost one quarter of FLHS cases, we observed a profound decrease of mature collagen surrounding portal venules and hepatic arterioles compared to similarly sized vessels from control livers. The decreased collagen may be an artifact of autolysis or due to periportal inflammation, as many of the affected sections also had portal inflammatory infiltrates. However, all birds including the controls had similar, minimal to moderate portal infiltrates, which is considered as a common feature of ground-dwelling birds. Nevertheless, the collagenization of vessels decreases in the periphery of liver, and the locations of the liver sections were not known for our cases. It has been reported that the hemorrhage associated with FLHS may be due to rupture of larger intrahepatic vessels. 22 However, we observed no difference in the integrity of vascular elastin or elastic laminae in FLHS-affected livers compared to controls as observed on VG-stained sections. This further supports that the hemorrhage is sinusoidal in origin and may reflect a loss of reticular integrity.
As this is a retrospective case study of privately owned birds, specific details regarding nutrition and husbandry are not known and cannot be ruled out as contributing factors to the development of FLHS. Multiple agents have been implicated in causing hepatic hemorrhage including aflatoxin, rapeseed oil, hepatitis E virus (HEV), and chelated minerals. 4,16,22,31 HEV, the causative agent of diseases such as hepatitis–splenomegaly syndrome, big liver and spleen disease, and hepatitis–liver hemorrhage syndrome, in particular, has been suggested as a causative agent of hepatic hemorrhage without histologic evidence of fatty change. 22 Histologically, HEV infection is characterized by lymphocytic phlebitis, lymphocytic and heterophilic periphlebitis, and/or fibrinoid necrosis in the liver with only occasional cases developing subcapsular hemorrhages. 2,3 Additionally, amyloid deposition, hepatomegaly, and splenomegaly are associated with infection with HEV and were not observed in the FLHS case population presented here. 1 However, as we did not check for the presence of HEV or seroconversion, we cannot rule out this viral infection. We noted that 50% (n = 16) of 32 tested cases of FLHS had copper levels below the reference range. Copper is essential for the production and cross-linking of collagen and elastin fibers. Copper deficiency has been associated with aortic rupture in poultry. 22 However, in our series, there appeared to be no correlation between cases with low copper and those with decreased collagen maturation around vessels.
With the significant increase in popularity of keeping backyard chickens and the high frequency of mortality due to FLHS in these flocks, it is necessary that proper veterinary and nutritional recommendations be made to owners to prevent FLHS. While multifactorial, it is clear that the cause of FLHS has a large nutritional component, as 97% of the affected birds had large fat depots or were obese. Studies clearly illustrate the role of dietary components in the development of FLHS. 6,29,33 Generally, lower-energy, high-protein rations with sufficient vitamin E, selenium, choline, and vitamin B12, as well as increased exercise opportunities, decrease obesity. 7,15,26 Educating the owners of backyard chickens regarding these nutritional and environmental recommendations should aid in decreasing the incidence of FLHS.
Footnotes
Acknowledgements
We are thankful to the histology team, Rosa Manalac, Alisha Curtis, James Koobs, and Karen Sverlow.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
