Abstract
Within a 24-hour period, 7 out of 200 three- to four-week-old pastured Katahdin lambs died after showing clinical signs of hemoglobinuria, red-tinged feces, weakness, and recumbency. One of the lambs that was examined clinically before natural death also had abdominal pain, trembling, tachycardia, and severe anemia with a packed cell volume of 4%. Pathologic findings included icterus, hemoglobinuric nephrosis, dark red urine, pulmonary edema, hydrothorax, splenomegaly, and acute centrilobular to midzonal hepatocellular degeneration and necrosis with cholestasis. The differential diagnoses and diagnostic workup to achieve the diagnosis are briefly discussed.
History and Clinical Findings
Seven out of 200 three- to four-week-old pastured Katahdin lambs died within a 24-hour period after showing clinical signs of hemoglobinuria, red-tinged feces, weakness, and recumbency. One of these lambs that was examined clinically before death also had abdominal pain, trembling, tachycardia, and severe anemia with a packed cell volume of 4%. The lamb died naturally, and the carcass was submitted to the California Animal Health and Food Safety Laboratory in Davis for necropsy and diagnostic workup.
Pathologic Findings
On gross examination, the carcass was in good nutritional condition. The subcutaneous, peritoneal, and pericardial fat was diffusely and moderately icteric, and there was multifocal red-tinged subcutaneous edema. Approximately 100 ml of dark red serosanguineous fluid was free in the thoracic cavity. The lungs were diffusely edematous and congested, with expansion of the interlobular septae by dark red fluid and noticeable rib imprints on the pleural surfaces (Fig. 1). Pink stable froth filled the lumen of the trachea and lower airways. There was moderate, diffuse, dark red discoloration of the cortex and medulla of both kidneys (Fig. 2), and the urinary bladder was distended with abundant dark red urine. The spleen was moderately enlarged with rounded edges. The brain was mildly icteric. The skeletal muscles were diffusely pale. The liver was pale brown and friable. No other significant gross findings were seen in the rest of the carcass.
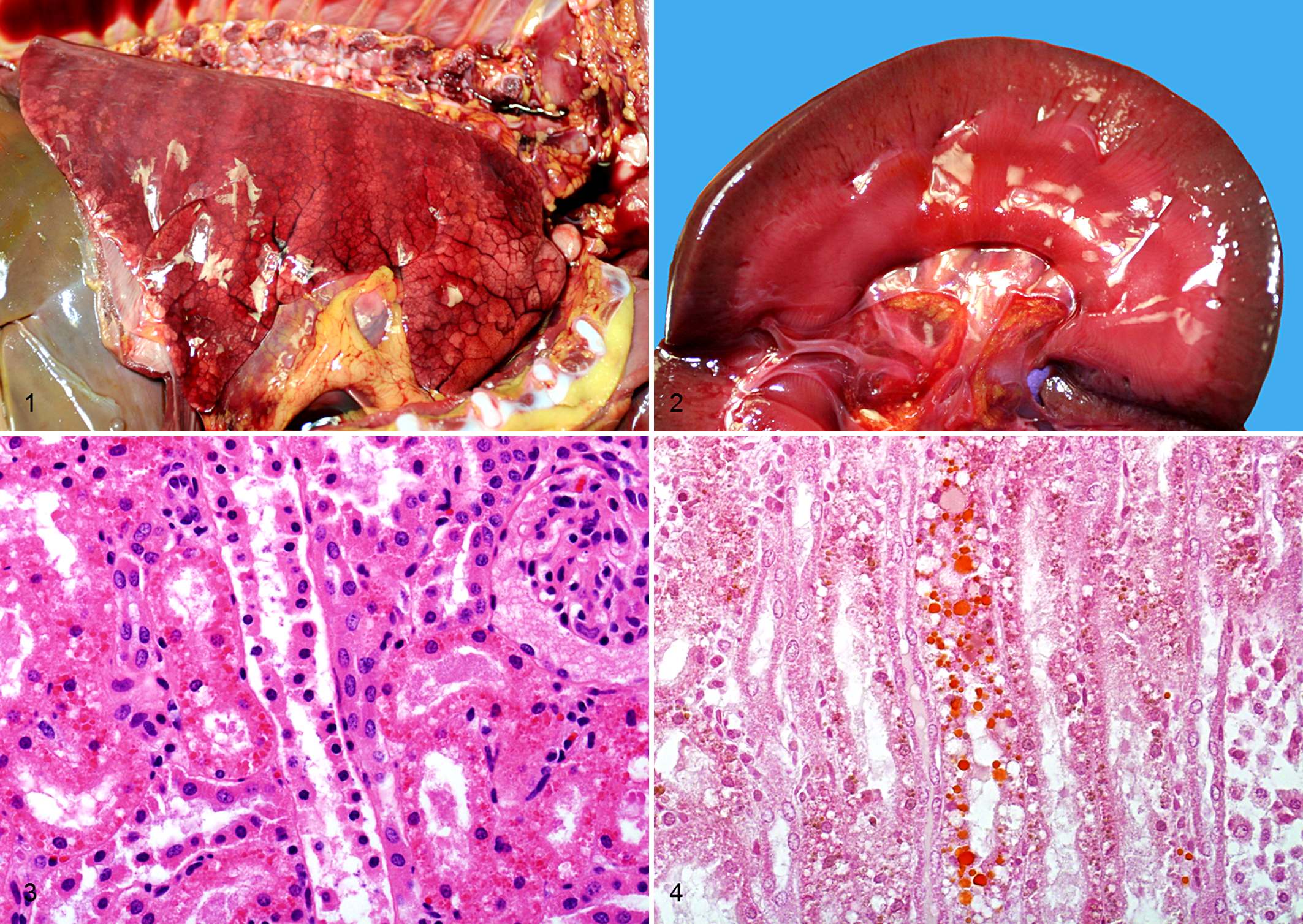
On histology, the epithelial cells of the renal proximal and distal convoluted tubules had numerous, multifocal, deeply eosinophilic granular to globular intracytoplasmic hyaline droplets that stained orange brown with Okajima stain for hemoglobin (Figs. 3, 4). There was also mild multifocal lymphoplasmacytic cortical interstitial nephritis. The lungs had extensive areas of proteinaceous alveolar and interstitial edema, as well as fibrin exudation into alveoli. The liver had multiple areas of centrilobular to midzonal hepatocellular degeneration and necrosis without hemorrhage, and bile canaliculi were frequently plugged with bile (cholestasis). No significant microscopic lesions were seen in the brain (cerebral cortex, hippocampus, corpus striatum, thalamus, medulla oblongata, and cerebellum), spleen, heart, trachea, esophagus, thymus, diaphragm, skeletal muscle, sciatic nerve, skin, adrenal gland, mesenteric lymph node, testicle, urinary bladder, abomasum, rumen, omasum, and small and large intestines.
Differential Diagnoses
The clinical signs (hemoglobinuria), clinical findings (packed cell volume of 4%), and anatomic pathologic findings in this lamb (icterus, splenomegaly, hepatic necrosis, hemoglobinuric nephrosis, and hemoglobinuria), as well as the clinical signs observed in the other lambs involved in this outbreak, were highly suggestive of an intravascular hemolytic process. The differential diagnoses for intravascular hemolysis in sheep include toxic conditions (copper toxicosis, 8 plant toxicosis such as that caused by onion [Allicum cepa] and Brassica spp 1 ), infectious conditions (leptospirosis, 9 Clostridium perfringens type A enterotoxemia [yellow lamb disease], 6 bacillary hemoglobinuria [Clostridium haemolyticum hepatitis], 1 eperythrozoonosis [Mycoplasma ovis]), 1 and parasitic conditions (Babesia ovis, Babesia motasi). 1
Ancillary Test Results
A set of diagnostic tests was selected to further investigate the case. Small intestinal contents and tissue were processed for bacterial anaerobic culture, and lung and liver swabs were cultured aerobically. A pooled sample of colon content and bile was processed for the detection of Salmonella spp by polymerase chain reaction (PCR). Smears of kidney were analyzed by a direct fluorescent antibody test (FAT) for the detection of Leptospira spp, and a PCR that detects L. interrogans, L. kirschneri, L. borgpetersenii, L. santarosai, L. weilii, and L. pomona was performed in urine. Formalin-fixed paraffin-embedded sections of kidney were processed for immunohistochemistry (IHC) using a rabbit multivalent anti-Leptospira primary antibody (LEP-FAC, National Veterinary Services Laboratories, Ames, IA), horseradish peroxidase–labeled anti-rabbit polymer (EnVision+, K4002, Dako, Carpinteria, CA), and AEC substrate chromogen (K3464, Dako). Additionally, serum samples were processed for detection of antibodies to several L. interrogans serovars, including L. icterohemorrhagiae, L. canicola, L. grippotyphosa, L. hardjo, and L. pomona by microscopic agglutination test, as previously described. 2 Samples of intestinal contents were tested for C. perfringens alpha toxin (CPA), beta toxin (CPB), epsilon toxin (ETX), and structural antigen using a commercially available capture-qualitative ELISA kit (Bio-X Diagnostics SPRL, Jemelle, Belgium) following the instructions of the manufacturer. Finally, heavy metal analyses for the detection of lead, manganese, cadmium, copper, iron, zinc, molybdenum, arsenic, mercury, and selenium were performed by spectrometry (FISONS, Accuris Model, Thermo Optek Corp., Franklin, MA) on fresh liver and kidney.
C. perfringens was isolated in large numbers from small intestinal contents and tissue on anaerobic culture, and the isolate was genotyped as C. perfringens type D negative for enterotoxin (CPE) and beta2-toxin (CPB2) genes by a previously described multiplex PCR technique designed to amplify segments specific for the genes encoding toxins CPA, CPB, ETX, iota toxin (ITX), CPE, and CPB2 of C. perfringens. 3 C. perfringens structural antigen and its major toxins CPA and ETX were detected in small intestinal contents by ELISA, while CPB was not.
Small and rare numbers of Escherichia coli were isolated on aerobic cultures from liver and lung, respectively. Salmonella spp PCR was negative. Serum antibody titers of 1:200 against L. icterohemorrhagiae were detected by microscopic agglutination test, while titers against the other serovars were all negative at a dilution of 1:100. Leptospira spp IHC and FAT in kidney and PCR in urine yielded negative results. All evaluated liver and kidney metal concentrations were within acceptable ranges for sheep, with the exception that the liver and kidney copper concentrations were slightly below the lower end of the reference range (liver: 18 mg/kg wet weight [ww], reference range: 25–100 mg/kg ww; kidney: 2.8 mg/kg ww, reference range: 4–5.5 mg/kg ww).
Diagnosis and Discussion
In this case, several causes of intravascular hemolysis were ruled out by specific testing (copper toxicosis and leptospirosis), lack of typical gross lesions (bacillary hemoglobinuria), absence of hemoparasites in tissue sections, or lack of history of exposure to hemolytic toxic plants (onions or Brassica spp). Although low, the serum antibody titers to L. icterohemorrhagiae were suggestive of previous exposure to this agent; however, this was not considered to be clinically relevant, as leptospirosis was ruled out by 3 techniques (IHC, FAT, and PCR).
All the pathologic changes in this lamb—including icterus, splenomegaly, hemoglobinuric nephrosis, hemoglobinuria, centrilobular hepatic necrosis, and pulmonary edema—have been described in cases of C. perfringens type A enterotoxemia in lambs, 6 and this condition was strongly suspected at necropsy in this case. For this reason, anaerobic bacterial cultures and C. perfringens toxin ELISA were performed in intestinal contents/tissue, and these tests allowed the confirmation of enterotoxemia, as discussed below.
C. perfringens is classified into 5 types (A–E) according to the production of 4 so-called major toxins—namely, CPA, CPB, ETX, and ITX. While all types produce CPA, types B and C produce CPB; types B and D produce ETX; and type E produces ITX. 10 The most common form of enterotoxemia in sheep is that produced by C. perfringens type D, also known as overeating or pulpy kidney disease; most clinical and pathologic changes of the disease are thought to be produced by ETX. 10 The disease is mostly characterized by neurologic and respiratory signs and lesions. 10 A rare form of enterotoxemia known as yellow lamb disease has typically been associated with strains of C. perfringens type A that produce unusually high amount of CPA, 6,10 a hemolytic lecithinase (phospholipase). 7 This form of enterotoxemia is characterized by severe hemolysis and jaundice. 6 Even though all types of C. perfringens can produce CPA, genotypes B, C, D, and E have not been associated with this syndrome.
Surprising, in our case the isolated strain was genotyped as type D. The ELISA assay for detection of C. perfringens CPA, CPB, and ETX—which allows for the differentiation of C. perfringens types A, B, C, and D—was done on small intestinal contents and was positive for CPA and ETX, which is a toxin profile consistent with C. perfringens type D. Thus, the main diagnostic criterion for C. perfringens type D enterotoxemia in sheep (detection of ETX in intestinal content) was fulfilled in this case. 10
CPA is a highly hemolytic lecithinase produced by all the aforementioned types of C. perfringens; 7,10 therefore, CPA-induced hemolysis could theoretically be caused by any type of C. perfringens. However to our knowledge, hemolysis in lambs has been associated with only C. perfringens type A enterotoxemia, not other types. To further investigate the amount of CPA produced by this strain of C. perfringens type D, the supernatant of the anaerobic 5-hour culture was analyzed by the same ELISA method but slightly modified to semiquantify the amount of CPA and ETX present in the sample, as previously described. 5 The amount of CPA and ETX was estimated by mathematic regression using samples with known concentrations of these toxins. The toxin concentrations produced by the field strain were compared with 4 reference strains isolated from other cases of C. perfringens type D enterotoxemia (strains 1–4) incubated under the same conditions. The field strain produced higher amounts of CPA (1500 ng/ml) than did the 4 reference strains (strain 1: 500 ng/ml, strains 2–4: 1 ng/ml), though the amount of ETX produced by this strain was not significantly different (García and Uzal, data not shown). Because the field strain produced a high amount of CPA, we speculate that this toxin was responsible for the hemolytic crisis in this lamb.
While all genotypes and strains of C. perfringens carry the gene that encodes for alpha toxin, not all strains produce this toxin; of those strains that do produce alpha toxin, the majority produce it in a small amount. Furthermore, a small percentage of C. perfringens type A strains produce significant amounts of this toxin, which is why we think that the rare cases of yellow lamb disease diagnosed before were associated with C. perfringens type A strains. In our study, we found hemolytic disease in a lamb infected with an unusually high alpha toxin producing type D strain of C. perfringens.
ETX has been shown to increase vascular permeability, which is believed to be the pathogenic mechanism of the pulmonary edema, hydrothorax, and perivascular brain edema with secondary cerebellar coning in typical acute cases of C. perfringens type D enterotoxemia and multifocal symmetric encephalomalacia in subacute to chronic cases. 4,10 The pulmonary edema and mild hydrothorax described in this lamb were likely caused by ETX toxemia because this toxin was also detected in the intestinal contents. This finding suggests a combined CPA and ETX action in this case. However, no perivascular brain edema, cerebellar coning, or multifocal symmetric encephalomalacia was noted. This does not preclude a diagnosis of type D enterotoxemia, as perivascular edema of the brain may not be present in a small proportion of cases. 10 Cerebellar coning and multifocal symmetric encephalomalacia are infrequently observed in cases of type D enterotoxemia, and it is hence not surprising that they were not seen in this lamb.
This report illustrates an unusual presentation of a case of enterotoxemia caused by a high CPA-producing strain of C. perfringens type D in a lamb in which combined effects of CPA (intravascular hemolysis) and ETX (pulmonary edema and hydrothorax) were found clinically and pathologically.
Footnotes
Acknowledgements
We thank Karen Sverlow and all California Animal Health and Food Safety bacteriology and pathology technicians for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
