Abstract
An adult Bengal cat (Felis catus × Prionailurus bengalensis) with a prolonged history of partial anorexia, regurgitation, and weight loss and a clinical, radiographic, and ultrasonographic diagnosis of persistent megaesophagus and gastrointestinal ileus was submitted for necropsy. The intestinal tract was diffusely distended by gas and fluid with appreciable loss of muscle tone and an absence of luminal obstruction, consistent with the clinical history of chronic intestinal pseudo-obstruction. Histologically, the autonomic nervous system was intact, but the smooth muscle within the gastrointestinal wall exhibited a marked basophilia that was most pronounced in the jejunum. Immunohistochemistry for neurofilament, synaptophysin, CD117, and desmin demonstrated that the number of myenteric ganglia, number of interstitial cells, and leiomyocyte desmin content were similar when compared with the unaffected age- and species-matched control. Immunohistochemistry for smooth muscle α-actin demonstrated a striking loss of immunoreactivity, predominantly in the circular layer of the jejunum, that corresponded with the tinctorial change in leiomyocytes. Transmission electron microscopy revealed loss of myofibrils, loss of organelle polarity, and significantly larger central mitochondria (megamitochondria) in affected leiomyocytes, as well as nonspecific degenerative changes. Although the presence of a primary leiomyopathy and a causal relationship could not be confirmed in this case, leiomyopathies are considered a cause of chronic intestinal pseudo-obstruction in human medicine, and loss of smooth muscle α-actin immunoreactivity is one recognized marker for intestinal dysmotility.
Keywords
Intestinal pseudo-obstruction is a clinical syndrome defined by functional (clinical, radiographic, or manometric) evidence of intestinal dysmotility in the absence of mechanical obstruction of the lumen. 1,2,4,13 Intestinal pseudo-obstruction can be further dissected into acute and chronic syndromes. In human and veterinary medicine, acute intestinal pseudo-obstruction is referred to as adynamic ileus and is a relatively common, acquired, and frequently transient disorder. 1,2,4,10 Conditions associated with adynamic ileus, shared in common between human and veterinary medicine, include abdominal surgery (postoperative ileus), peritonitis, hypokalemia, spinal or pelvic trauma, viral enteritis, pancreatitis, intestinal infarction, and anticholinergic or opioid drug administration. 1,2,4,10
In contrast, chronic intestinal pseudo-obstruction (CIPO) is a rare clinical syndrome that is characterized by recurrent bouts of functional occlusion and can be a congenital or an acquired disease. In human medicine, CIPO can be caused by any one of a heterogeneous group of gastrointestinal neuromuscular diseases (GINMDs) that involve components of the neurointerstitial-muscular control of gastrointestinal contractility (Table 1). 1,2,10,11 In veterinary medicine, the differential list of GINMDs is significantly shorter and includes infiltrative intestinal neoplasia, 4 canine idiopathic sclerosing enteropathy/fibrosing gastrointestinal leiomyositis, 4,8 and dysautonomia. 5,9,17 Primary visceral neuropathies, interstitial cell diseases, and myopathies are presumed to occur based on the human correlate but have not been recognized previously. This report represents the first documented case of intestinal pseudo-obstruction associated with visceral smooth muscle abnormalities in a veterinary species and briefly reviews the current diagnostic algorithms and techniques for evaluating GINMD.
ICC, interstitial cells of Cajal; IND, intestinal neuronal dysplasia; MNGIE, mitochondrial neurogastrointestinal encephalomyopathy.
aIncludes but is not limited to the following diseases. Those listed, particularly the human correlates, are considered representative or the best described.
bUseful markers, in addition to hematoxylin and eosin, include the following: GFAP, glial fibrillary acid protein; NF, neurofilament; NSE, neuron-specific enolase; PAS, periodic acid–Schiff; SMA, smooth muscle α-actin; SYN, synaptophysin; TEM, transmission electron microscopy.
dHilbe et al. 6
eJohnson et al. 8
fKnowles et al,,10,11 Knowles and Martin, 12 Knowles et al, 13 O'Donnell et al, 15 and Smith et al. 19
gKnowles et al. 11
An adult, 6-year-old, male-neutered Bengal cat (Felis catus × Prionailurus bengalensis) presented to the Veterinary Medical Teaching Hospital with a 3-week history of anorexia, regurgitation, lethargy, and weight loss (1.4 kg). Additional history provided later revealed episodic vomiting and regurgitation at least once per week for the past year. On physical examination, the cat was thin (body condition score 3/9), was dehydrated, and had a distended cranial abdomen. Serial thoracic radiographs taken over a 2-week period revealed thin body condition, persistent megaesophagus, and progressive cardiomegaly. Abdominal radiographs revealed marked gastric gas distention, diffuse small intestinal dilation, and poor peritoneal and retroperitoneal serosal detail (Fig. 1). On abdominal ultrasound, jejunal loops were fluid filled and exhibited bidirectional flow. The walls of the jejunal loops were mildly thickened, 0.26 cm thick (0.25-cm upper limit of reference range). Serum chemistry profile and complete blood count were consistent with mild prerenal azotemia and moderate normocytic, normochromic, nonregenerative anemia, likely reflecting chronic disease or malnutrition. 16 After stabilization, the diagnostic plan was to include endoscopy with possible exploratory laparotomy and biopsy. Supportive care was instituted, but replacement fluid therapy induced left-sided heart failure with development of respiratory distress. Cardiac insufficiency and pulmonary edema were confirmed on echocardiography and follow-up thoracic radiographs, respectively, and were responsive to clinical management; however, euthanasia was elected due to the poor prognosis associated with the persistent gastrointestinal dysmotility. The cat was submitted for necropsy with a clinical diagnosis of partial proximal intestinal outflow obstruction and decompensated cardiomyopathy.
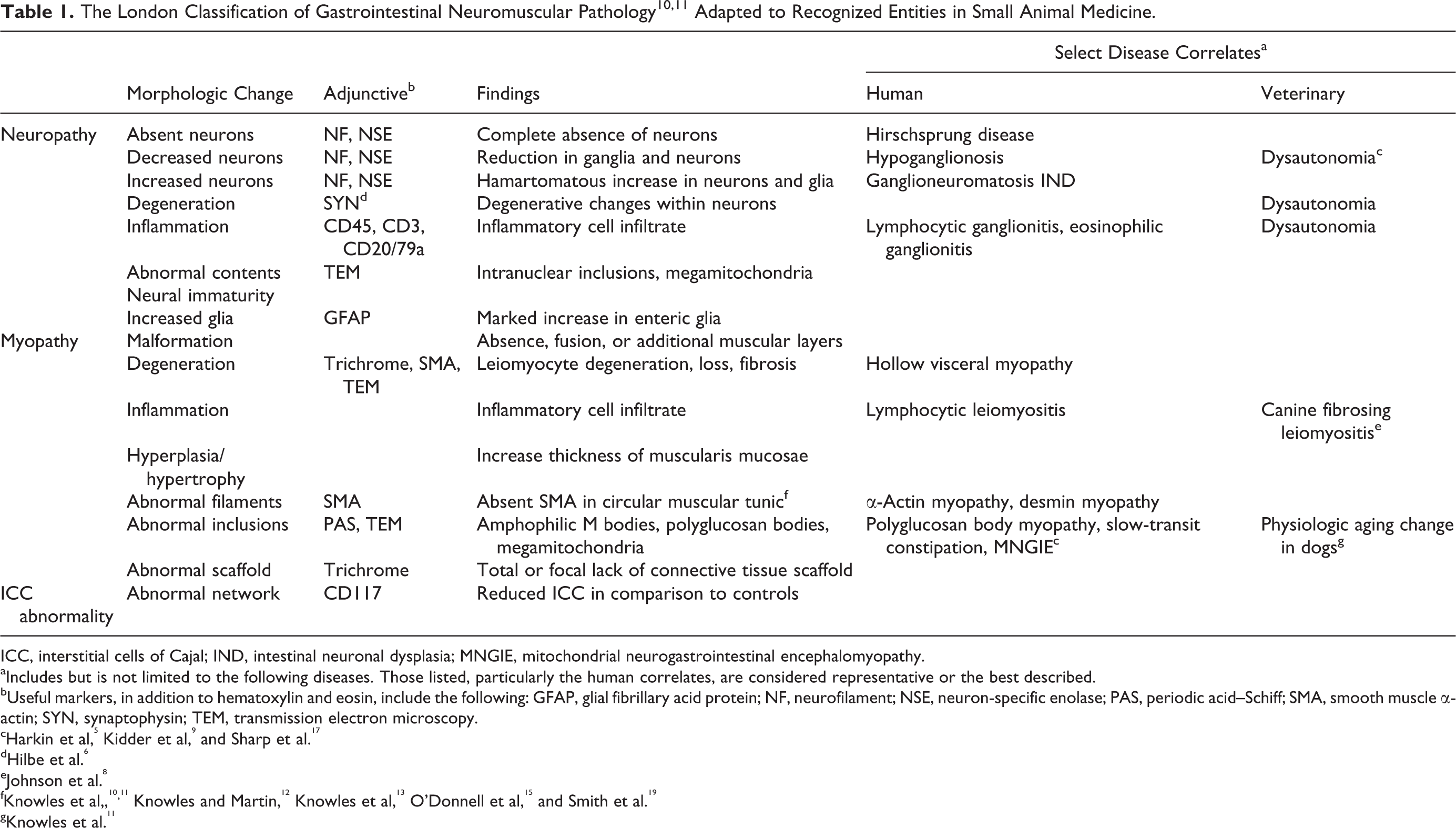
Abdomen; cat. Abdominal radiographs revealed marked gas distention of the stomach and diffuse dilation of the small intestines with loss of peritoneal and retroperitoneal serosal detail.
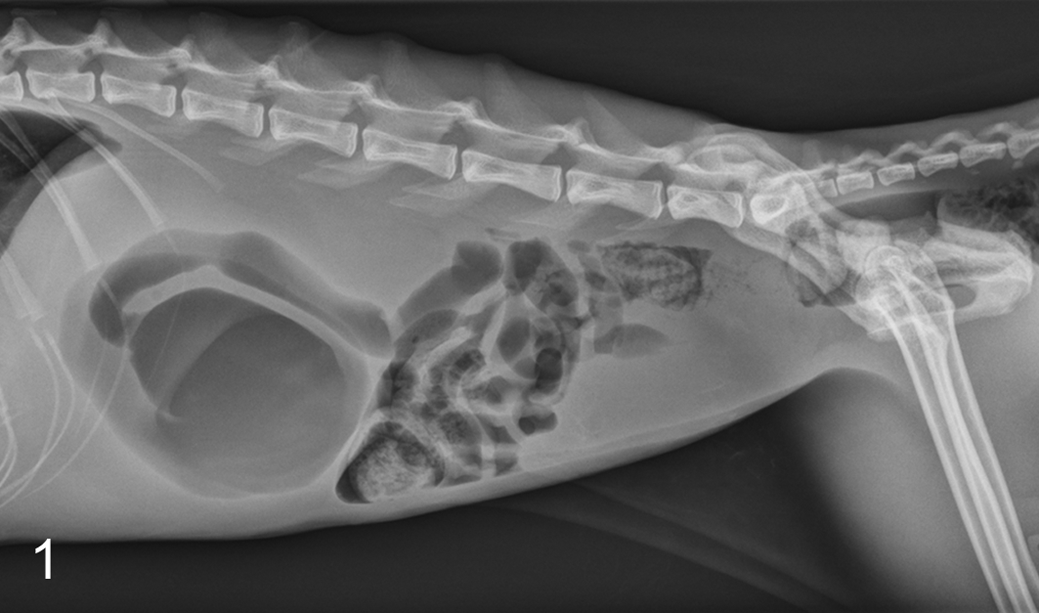
Gross necropsy confirmed that the cat was in thin body condition with diffuse gas and fluid dilation of the intestinal tract, from caudal esophagus to colon, and loss of intestinal smooth muscle tone (Fig. 2). The mucosa of the dilated caudal segment of the esophagus was eroded to ulcerated. Also confirmed was cardiomegaly with left ventricular hypertrophy and pulmonary edema.
A complete set of tissue samples was immersion-fixed in 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. Formalin-fixed, paraffin-embedded tissues were sectioned 4 to 5 μm thick and stained with hematoxylin and eosin (HE). Select sections of intestinal tract, including esophagus, stomach, and small intestine, were evaluated using Masson’s trichrome, periodic acid–Schiff (PAS), and Von Kossa stains. Similar sections were processed for indirect immunohistochemistry. Sections were deparaffinized, rehydrated, and incubated in citrate buffer (pH 6) for 20 minutes using the Lab Vision PT Module (Lab Vision Corporation, Fremont, CA) for antigen retrieval. Endogenous peroxidases were blocked by incubation in 3% hydrogen peroxide for 10 minutes. Nonspecific immunoreactivity was blocked by incubation in Ultra V Block (Lab Vision Corporation) for 5 minutes. Primary antibodies and dilution factors used included the following: mouse monoclonal anti–smooth muscle α-actin (1:1000, clone 1A4; DAKO, Glostrup, Denmark), mouse monoclonal anti-neurofilament (1:200, clone 2F11; DAKO), mouse monoclonal anti-synaptophysin (1:200, clone SY38; DAKO), rabbit polyclonal anti-CD117 (1:150; DAKO), and mouse monoclonal anti-desmin (1:400; DAKO). Immunoreactivity was detected using a horseradish peroxidase–conjugated anti–mouse or rabbit secondary antibody, developed using the chromogen, 3,3′-diaminobenzidine, and counterstained with hematoxylin. Tissue sections processed simultaneously, with omission of the respective primary antibody, were used as negative controls. Archival sections of intestinal tract from a 3-year old female-spayed Bengal cat that was submitted for necropsy with unrelated disease were used as a positive control for the smooth muscle α-actin, synaptophysin, CD117, and desmin immunohistochemistry.
For transmission electron microscopy, selected formalin-fixed intestinal samples were postfixed in 2.5% glutaraldehyde (pH 7.2) for 12 hours, followed by 1% OsO4 in 0.1 M cacodylate buffer for 60 minutes. The samples were subsequently dehydrated through an ethanol series, rinsed in propylene oxide, and embedded in epoxy resin (Durcupan; ACM Fluka, Sigma-Aldrich, St Louis, MO). Toluidine blue–stained 1-μm sections were examined under a light microscope, and ultra-thin sections (70 nm) from selected areas were prepared and stained with uranyl acetate and lead citrate for contrast. Electron micrographs were acquired using a JEOL 100CX electron microscope (JEOL Ltd., Tokyo, Japan). Nuclear dimensions from affected and unaffected leiomyocytes were measured using ImageJ software (National Institutes of Health, Bethesda, MD). Statistical comparisons were performed using independent samples t test (Prism version 5; GraphPad Software, La Jolla, CA).
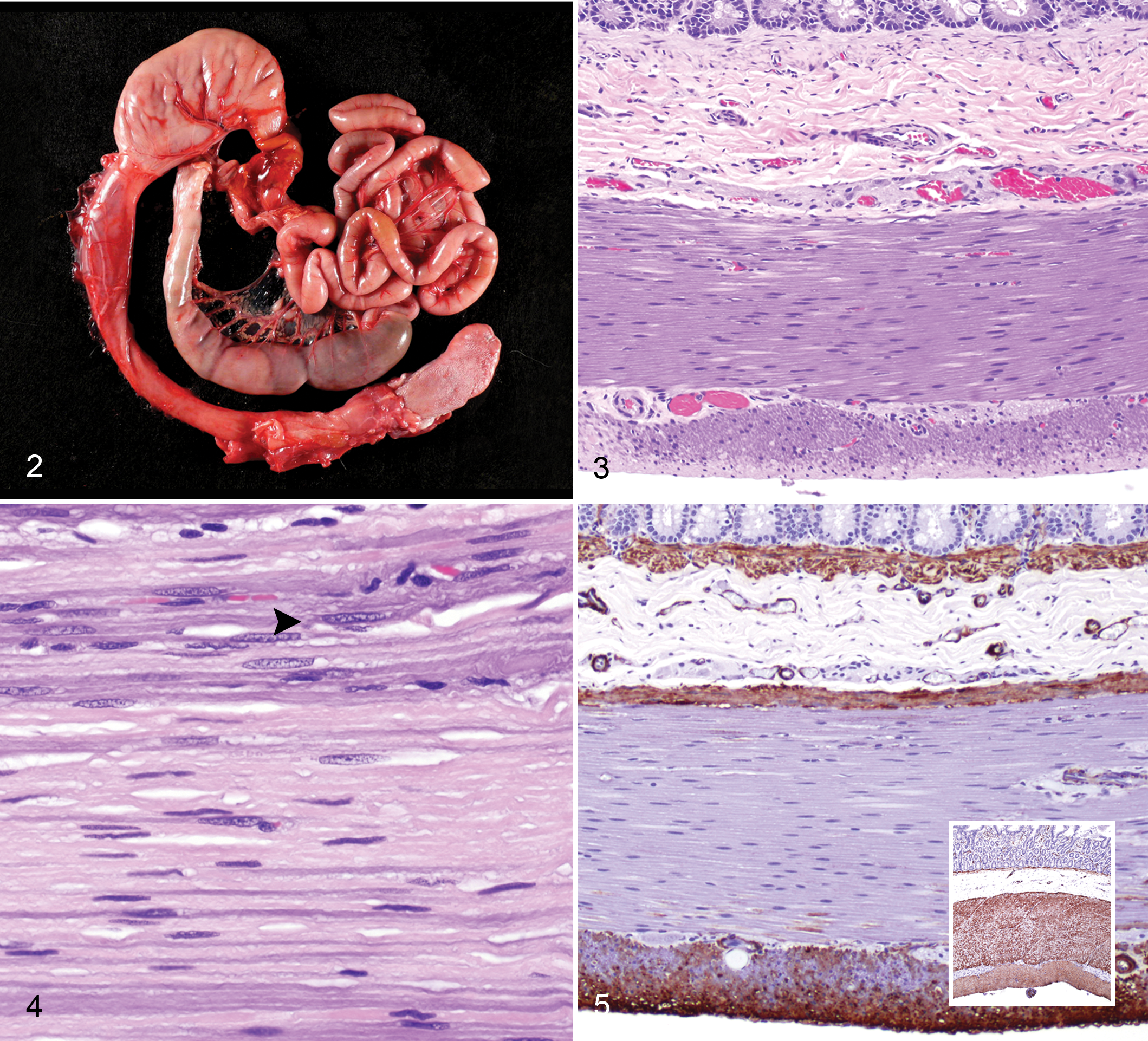
Histopathologic findings revealed plentiful and intact ganglion cells within myenteric plexi and a lack of mucosal inflammation, except for erosion and ulceration of the caudal esophageal mucosa. Within 1 Meissner’s plexus, a single shrunken, hypereosinophilic ganglion cell with a pyknotic nucleus was identified. A distinct tinctorial change, characterized by marked basophilia, was appreciated in leiomyocytes at all levels of the intestinal tract. The leiomyocyte basophilia was most prominent in the jejunum, primarily in the inner circular layer but in patches within the outer circular layer (Fig. 3). A similar basophilic tinctorial change was present in leiomyocytes within the walls of blood vessels and the urinary bladder (not shown). Affected basophilic leiomyocytes often contained larger nuclei with a more open to coarsely granular chromatin pattern compared with the smaller, more condensed nuclei of unaffected leiomyocytes (Fig. 4). The esophageal skeletal muscle fibers were spared from exhibiting a similar basophilic tinctorial change and subjectively were hypertrophied. Esophageal submucosal fibrosis and multifocal fibrosis of the outer muscular layer of the stomach were observed. Other histopathologic lesions included serous atrophy of fat, myocardial hypertrophy, pulmonary edema, centrilobular hepatocellular lipidosis, chronic interstitial nephritis, and adrenocortical hyperplasia.
The preservation of ganglion cells in the myenteric plexi was further confirmed using neurofilament and synaptophysin immunohistochemistry (not shown). Interstitial cells of Cajal, represented by CD117-immunoreactive cells within the muscular tunics, were present and in similar numbers within affected and control tissues (not shown). The cause of the leiomyocyte basophilia was not identified histochemically. Mineral deposition was excluded based on the Von Kossa stain. No leiomyocyte cytoplasmic inclusions or lipofuscin granules were observed in the PAS-stained section. Masson’s trichrome only confirmed segmental fibrosis in the esophagus and stomach. However, smooth muscle α-actin immunohistochemistry revealed a striking absence of immunoreactivity that colocalized with the leiomyocyte basophilia. Loss of immunostaining was multifocal to diffuse, predominantly affecting the proximal to mid-intestinal tract and most prominent in the inner circular layer of the muscular wall. Complete loss of immunostaining was most striking in the jejunum (Fig. 5). The age and hybrid species-matched control exhibited diffuse and strong smooth muscle α-actin immunoreactivity in all layers (Fig. 5, inset). Comparison between the pattern of α-actin staining in case and control sections demonstrated mild hypertrophy/hyperplasia of the muscularis mucosae in the case (Fig. 5).
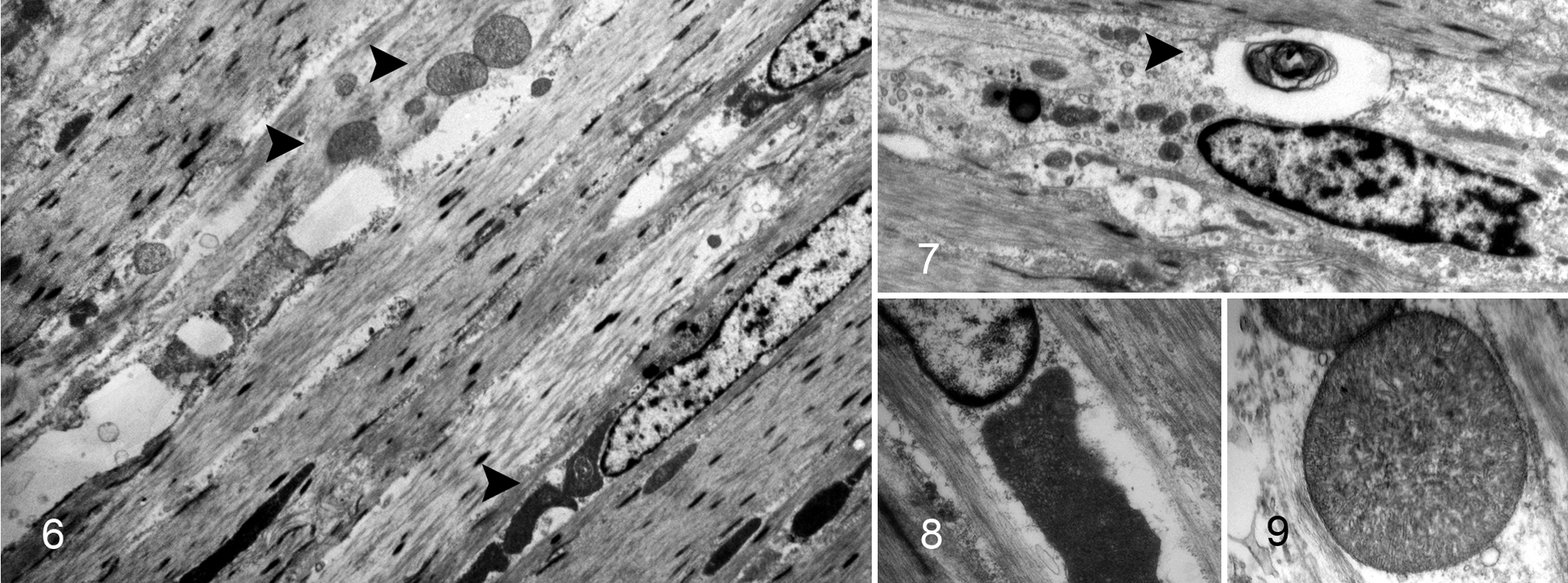
Transmission electron microscopy of basophilic leiomyocytes demonstrated loss of myofibrils, loss of organelle polarity, large central mitochondria (megamitochondria) (Figs. 6, 8, 9), and nonspecific degenerative changes characterized by accumulation of electron-dense intralysosomal and free debris and lamellar myelin figures (Fig. 7). Mean ultrastructural dimensions (sum of width × length ± SEM) of megamitochondria within basophilic leiomyocytes (25.09 ± 2.60 μM) were statistically significantly greater (P = .0003) than the dimensions of mitochondria in unaffected nonbasophilic leiomyocytes (12.00 ± 1.35 μM).
The leiomyocyte basophilia, with corresponding loss of smooth muscle α-actin immunoreactivity and ultrastructural intraleiomyocyte megamitochondria, was interpreted as an acquired morphologic and biochemical abnormality associated with chronic intestinal pseudo-obstruction in this adult Bengal cat. The clinical history (3-week duration of clinical signs in a 6-year-old feline) was most consistent with an acquired condition. The clinical progression was interpreted as proximal intestinal pseudo-obstruction resulting in chronic regurgitation, vomiting, and caudal segmental esophagitis. Megaesophagus can occur secondary to esophagitis, but the other alternate possibility is a direct association with the leiomyocyte abnormality as the feline esophagus transitions from skeletal muscle to smooth muscle in the caudal segment. 7 The leiomyocyte abnormality was focused on after exclusion of other possible causes of CIPO. The most significant adjunctive diagnostic finding was loss of smooth muscle α-actin immunoreactivity. Other possible causes of the leiomyocyte basophilia, including mineralization and lipofuscinosis, were excluded histochemically. Disuse or neurogenic atrophy was not considered the underlying process for the leiomyocyte basophilia since functional but disorganized motility was present ultrasonographically, loss of smooth muscle mass or individual leiomyocytes was not appreciated, and expression of other muscle protein (desmin) was comparable to the control.
Deficiency in smooth muscle α-actin has been associated with chronic intestinal pseudo-obstruction in human cases of GINMD. 13 –15,18,19 The pattern of smooth muscle α-actin deficiency is strikingly similar to this feline case with loss of immunoreactivity in the circular layer of the jejunum. 13 –15,19 In 1 large multi-institutional study of idiopathic CIPO, partial to complete absence of jejunal smooth muscle α-actin immunoreactivity was relatively prevalent, identified in 24% (28/115) of patients. 13 In many human CIPO cases, including cases from the aforementioned study, loss of smooth muscle α-actin immunoreactivity was the sole finding and was observed in both congenital and acquired disease states. 13,15,19 Some authors argued that the smooth muscle α-actin abnormality could be the etiological condition altering motility. 15,19 Others consider alterations in smooth muscle α-actin expression to be an associated morphologic change and a diagnostically valuable but nonspecific biomarker for intestinal dysmotility. 13,14,18 Despite this, the consensus statement from the International Working Group does recognize a filament protein abnormality subgroup of gastrointestinal myopathies, defined by absent smooth muscle actin, in the London Classification of GINMD (Table 1). 10
Smooth muscle α-actin was the only myocyte contractile protein (eg, smooth muscle γ-actin, myosin, tropomyosin, caldesmon, filamin) evaluated in this case, due to the limitation in available antibodies. Having stated this, smooth muscle α-actin is currently the only diagnostically relevant contractile protein in the evaluation of GINMD. 10 –12 The reports of adult and pediatric smooth muscle α-actin deficiency postulated that the absence of immunoreactivity reflected absent expression. 13,15,19 Alternate possibilities include loss of the smooth muscle α-actin epitope due to conformational change or structural interference. To determine the nature of the absent immunoreactivity, further investigation into isoactin gene expression levels, protein content, and protein sequence/conformation would be required. Lack of immunoreactivity in specific anatomic sites can also represent a normal physiologic variation. Partial to complete loss of smooth muscle α-actin immunostaining in the inner circular layer of the ileum is consistently observed in normal human adult controls, and differential expression in the ileum has been reported during fetal development. 13 In this case and in the context of increased cytoplasmic basophilia, loss of smooth muscle α-actin immunoreactivity was considered to reflect loss of expression. 20
Megamitochondria are also features of certain human GINMDs, included in the subgroup of abnormal content myopathies in the London Classification (Table 1). 10 The ultrastructural identification of megamitochondria or increased numbers of mitochondria in visceral leiomyocytes is seen in mitochondrial disorders, such as mitochondrial neurogastrointestinal encephalomyopathy (MNGIE). 3 These mitochondrial disorders manifest disease in energy-dependent tissues, such as muscle and brain, and can be associated with CIPO. However, there are many other circumstances in which megamitochondria can be identified, including subsequent to starvation or malnutrition. 21 Thus, in the broadest sense, formation of megamitochondria can be considered a nonspecific response to an unfavorable subcellular environment. In this case, the megamitochondria were considered a feature of the leiomyopathy but not considered primarily causal because of the absence of other lesions attributable to a systemic mitochondrial disorder (ie, the absence of the leukoencephalopathic component of MNGIE).
Given the clinical presentation (pseudo-obstructive ileus with megaesophagus in a cat) and geographic locale (Midwest), dysautonomia was considered. 9 Based on the absence of neuronal degeneration (central chromatolysis), neuronal loss, gliosis, and/or ultrastructural changes in myenteric plexi and in the central nervous system as well as absent extravisceral abnormalities (eg, mydriasis, prolapse of nictitans) and consultation with multiple veterinary and human neuropathologists, the available case material was not consistent with dysautonomia. 9,17 Unfortunately, only the intramural autonomic ganglia were available for evaluation. The cranial cervical, stellate, and abdominal ganglia (most commonly used for diagnosis) 5 and spinal cord were not sampled for histopathology. Therefore, it remains possible that an autonomic neuropathy could be at least contributory, if not causal.
A recently compiled comprehensive consensus statement created by an international working group defined diagnostic guidelines and a classification scheme for evaluation of human GINMDs and could be useful in standardizing the evaluation of veterinary cases. 10,11 The case documented here exhibits smooth muscle abnormalities that required immunohistochemistry and electron microscopy to characterize, emphasizing the importance of adjunctive diagnostics, including histochemical, immunohistochemical, and ultrastructural techniques (Table 1). The underlying pathogenic mechanism is yet to be determined. Loss of α-actin immunoreactivity may or may not reflect the primary pathogenesis of chronic intestinal pseudo-obstruction in this case but, at minimum, is a valuable diagnostic marker for dysmotility. 12,13
Footnotes
Acknowledgements
The authors are grateful to Sandy Cheasty, Barb Reese, Laura Calvo, and Jim Langteig for technical support and to Drs Robert Higgins, Shariar Salamat, and Richard Dubielzig for their expertise and advice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.