Abstract
Necrotizing meningoencephalitis (NME), necrotizing leukoencephalitis (NLE), and granulomatous meningoencephalomyelitis (GME) are idiopathic inflammatory diseases in the central nervous system (CNS) of dogs. In our previous study, the proportion of inflammatory cells, except for CD3-positive T cells, were not different in parenchymal and perivascular lesions in the brain. However, breed specificities, clinical courses, and specific lesions were distinct among these diseases. Thus, similarities and differences in the pathologies of these diseases have been implied. In this study, the messenger RNA (mRNA) and/or protein expression levels of cytokines and chemokine receptors were investigated in NME (n = 2), NLE (n = 4), and GME (n = 2) cases, and their relationship in the formation of specific lesions was discussed. The mRNA and protein expression levels of interferon (IFN)–γ and interleukin (IL)–17 were marked in NME and GME, respectively. The mRNA expression levels of CXCR3 and CCR2 were also marked in NME and GME, respectively. The results of double-labeling immunofluorescence, used to identify cells producing IL-17 in these lesions, showed that most CD163-positive macrophages/microglia but fewer CD3-positive T cells were IL-17 positive in GME. These results indicate that IFN-γ plays a key role in NME lesions and that the macrophages/microglia that infiltrate brain lesions producing IL-17 are more important in GME than T cells.
Necrotizing encephalitis (NE) and granulomatous meningoencephalomyelitis (GME) are idiopathic inflammatory diseases in the central nervous system (CNS) of dogs. NE is characterized by malacic changes and is subdivided into necrotizing meningoencephalitis (NME, the gray matter type) and necrotizing leukoencephalitis (NLE, the white matter type) according to the location of lesions. NME appears to be breed specific and affects the Pug, 6,16 Shi-tzu, 28 Papillon, 28 Maltese, 8,27 Chihuahua, 9 Yorkshire Terrier, 14,30 and French Bulldog. 26 Conversely, GME is characterized by granulomatous lesions in the cerebral white matter and cerebellum and is not breed specific. Although the cause of these diseases still remains obscure, CD3-positive T cells have been reported to play significant roles in these diseases. 11,28,29 In our previous report, 20 CD3-positive T cells were also the main inflammatory cells observed in these diseases, especially in GME lesions. The number of CD163-positive macrophages was higher in GME than in NME and NLE, although this was not significant. The proportion of IgG-, CD20-, CD79acy-, and myeloperoxidase-positive inflammatory cells was not different among these diseases. Overall, the proportion of inflammatory cells in parenchymal and perivascular lesions in the brain did not vary much among these diseases, although marked differences in the histopathology of these lesions and their location have been observed. Thus, we concluded that these diseases may share a common pathogenesis.
Th1 cells have been shown to mediate human autoimmune diseases, such as multiple sclerosis (MS) 15,25,32 and rheumatoid arthritis (RA). 5,22 Similar phenomena have also been observed in murine experimental autoimmune encephalitis (EAE), a disease model of MS. 1,32 However, treatment with an antagonist of Th1 cells did not prevent and/or ameliorate the symptoms of MS and EAE. 1,2,15 Humoral immunity, especially B cells, may also mediate these diseases. 13 Th17 cells were shown to play a key role in MS and EAE, 1 and self-specific Th17 cells were reported to be highly pathogenic in autoimmune diseases such as RA, 10,23 autoimmune uveitis, 33 and Crohn disease. 3,12 Th17 cells have been suggested to be important at the initiation of such diseases, and Th1 cells are also important for the maintenance and/or deterioration of these diseases. 1,21
Chemokine receptors, such as chemokine (C-X-C motif) receptor 3 (CXCR3), C-C chemokine receptor type 2 (CCR2), and C-C chemokine receptor type 4 (CCR4), have also been associated with MS and EAE. 1 CXCR3 is expressed primarily on activated T cells, such as Th1 cells. 4 Th1 cells that express CXCR3 produce a large amount of interferon (IFN)–γ. 1,4 T cells expressing CCR2 have been shown to produce a large amount of interleukin (IL)–17. 1 CCR4 is expressed on leukocytes. 1,4 T cells that express CCR4 produce IL-4 or IL-17 according to the coexpression of CRTh2 or CCR6. 1,4
In the present study, we compared the levels of inflammatory cytokines and chemokine receptors in the brain lesions of NME, NLE, and GME cases and discussed the relationship of these cytokines in the formation of specific lesions.
Materials and Methods
Brain Tissues
Frozen brain tissues from NME (n = 2), NLE (n = 4), GME (n = 2), and control (n = 3) cases were used in this study (Table 1). NME, NLE, and GME were diagnosed by histopathologic examination. Potential infectious causes were excluded by both histopathologic examination and immunohistochemistry against canine distemper virus (CDV).
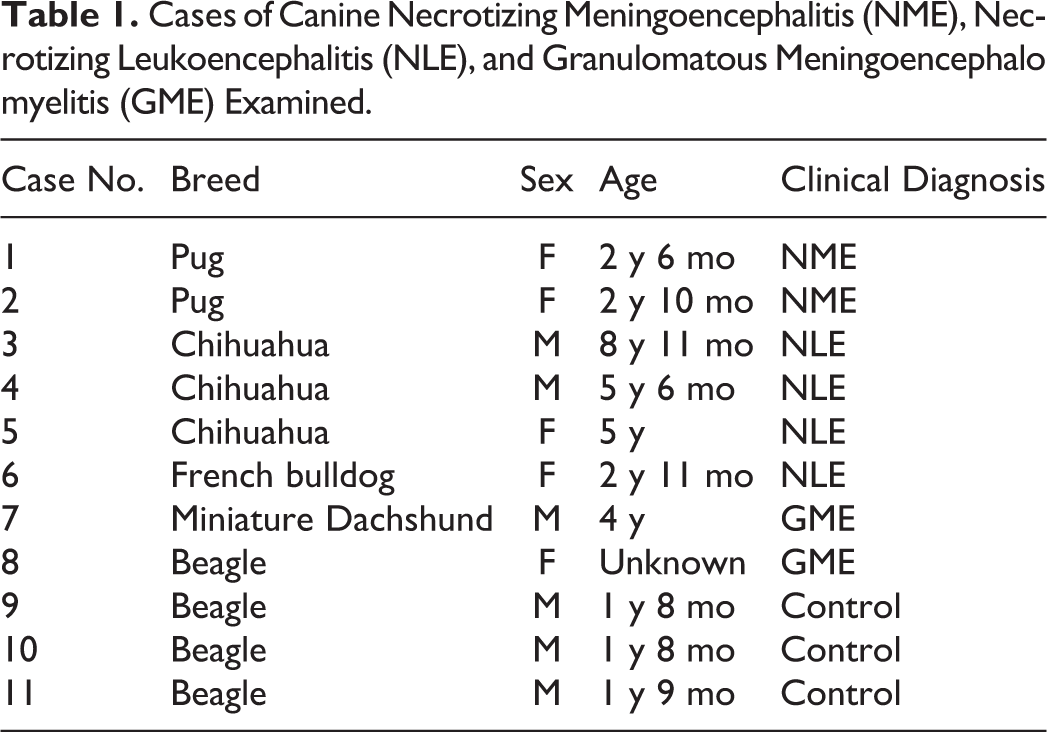
Cases of Canine Necrotizing Meningoencephalitis (NME), Nec-rotizing Leukoencephalitis (NLE), and Granulomatous Meningoencephalomyelitis (GME) Examined.
Immunohistochemistry
Deparaffinized sections were autoclaved at 121°C for 15 minutes for antigen retrieval. Endogenous peroxidase activity was blocked by incubating the sections with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were treated with skimmed milk at 37°C for 40 minutes for tissue blocking. These sections were then incubated at 4°C overnight with CDV nucleoprotein (NP) antibody (VMRD, Pullman, WA). These sections were then incubated with the EnVision polymer reagent (DAKO Japan, Kyoto, Japan) at room temperature for 40 minutes. Visualization was performed with 3,3′-diaminobenzidine tetrahydrochloride (Wako, Osaka, Japan), and counterstaining was performed with hematoxylin.
RNA Extraction and cDNA Synthesis
Total RNA was prepared by the guanidinium thiocyanate-phenol-chloroform method (ISOGEN; Wako) from brain tissues and reverse-transcribed with a PrimeScript RT-PCR kit (Takara, Otsu, Japan).
Real-time PCR Quantification Using SYBR Green I
Real-time polymerase chain reaction (PCR) on an ABI PRISM (Applied Biosystems, Foster City, CA) was performed in a total volume of 25 μl SYBR Green Realtime PCR Master Mix (TOYOBO, Osaka, Japan) and primers for CCR2, CCR4, CXCR3, GAPDH, IFN-γ, IL-12p35, IL-12p40, TBX21, IL-4, IL-10, GATA3, IL-17, IL-21, or RORγt (Table 2). The PCR reaction was performed on a MicroAmp Optical 96-well Reaction Plate (Applied Biosystems) with an initial degeneration step at 54°C for 30 seconds and 95°C for 3 minutes, followed by 10 cycles at 95°C for 30 seconds and an annealing step at 68°C for 1 minute, 72°C for 30 seconds, and 30 cycles at 95°C for 30 seconds. At the end of each cycle, the intensity of fluorescence emitted from SYBR Green was measured. After the completion of this process, samples were subjected to a dissociation curve analysis (from 60°C above annealing temperature to 95°C). Samples were then quantified automatically (7900HT version 2.3 Sequence Detection Systems; Applied Biosystems). A single narrow peak was observed by the dissociation curve analysis at the specific melting temperature, and a single band of the predicted size was observed by 2% agarose gel electrophoresis. Each case was compared with a control group by the ▵▵Ct method and relative quantification was performed.
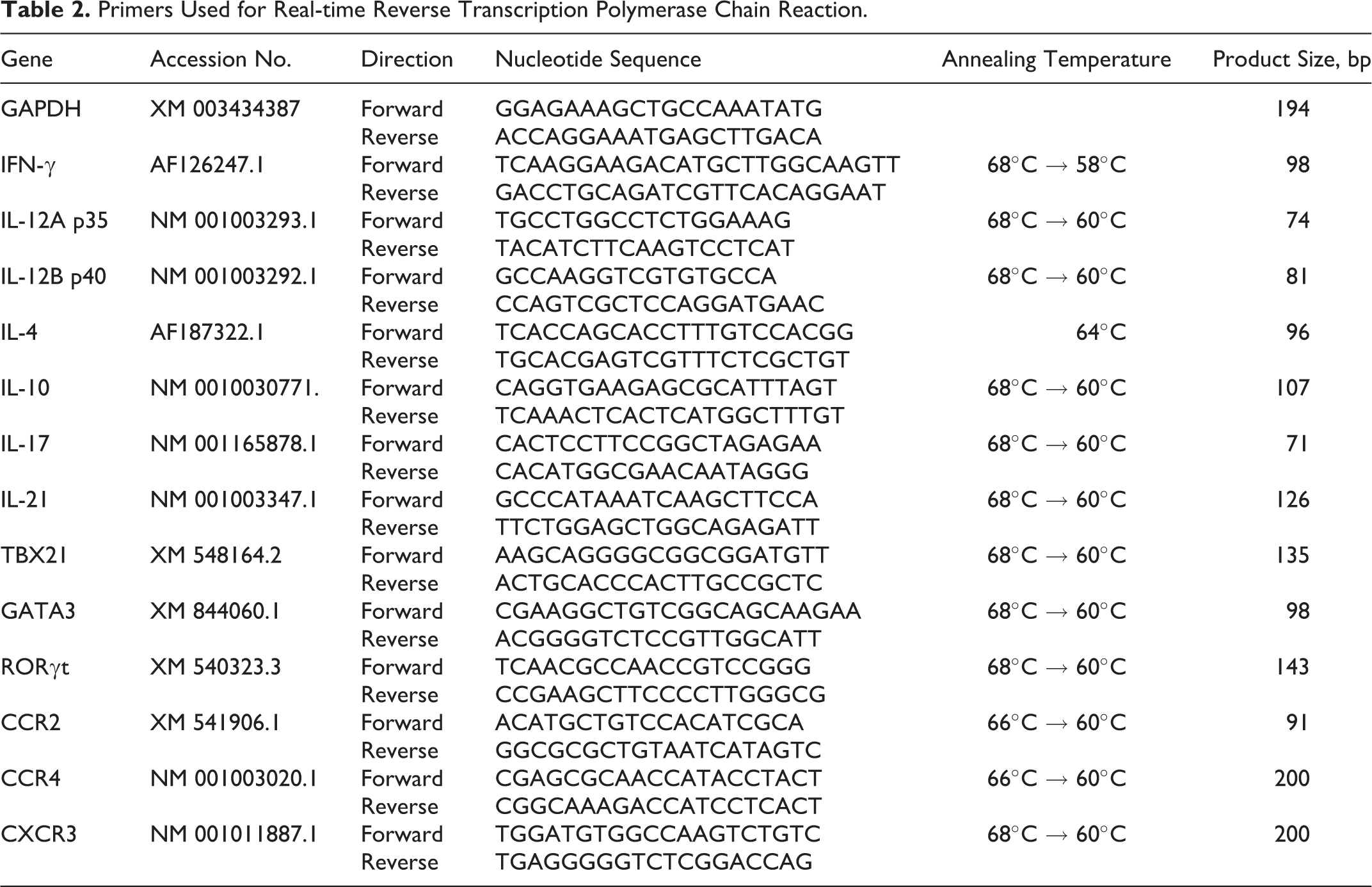
Primers Used for Real-time Reverse Transcription Polymerase Chain Reaction.
Immunoblot
Brain tissues from NME, NLE, and GME cases were homogenized with a TissueRuptor (QIAGEN, Homebrechtikon, Switzerland) and TissueRuptor disposable probes (QIAGEN) in a buffer containing 10 mM Tris-HCl (pH 7.8), 1% NP-40, 0.1% sodium dodecyl sulfate (SDS), 150 mM NaCl, 1 mM EDTA (pH 8.0), Proteinase inhibitor cocktail, 2 mM Na3VO4, 10 mM NaF, and distillated water (DW). Lysates were incubated on ice for 30 minutes and centrifuged at 12 000 × g at 4°C for 20 minutes. Supernatants were collected. Proteins were separated by SDS–polyacrylamide gel electrophoresis (PAGE) and transferred to Immun-Blot PVDF Membranes (Bio-Rad, Hercules, CA). Membranes were blocked with 150 mM NaCl, 10 mM Tris-Base, and 40 mM Tris-HCl buffer (pH 7.8) containing 5% skim milk and 0.05% Tween-20 and were incubated with one of the primary antibodies at 4°C overnight and then with appropriate secondary antibodies at room temperature for 1 hour. The primary antibodies used are listed in Table 3, and the secondary antibodies were donkey anti–goat IgG horseradish peroxidase (HRP) conjugated (1:5000; Santa Cruz Biotechnology, Santa Cruz, CA) and donkey anti–rabbit IgG HRP conjugated (1:5000; GE Healthcare, Buckinghamshire, UK). Membranes were visualized with the Amersham ECL Prime Western blotting detection reagent (GE Healthcare, Uppsala, Sweden) using a Molecular Imager ChemiDox XRS+ with Image Lab software (Bio-Rad, Tokyo, Japan). Positive bands were quantified relative to a β-actin band using Image Lab software (Bio-Rad).
Primary Antibodies Used for Immunoblot Analysis and Double-Labeling Immunofluorescence.
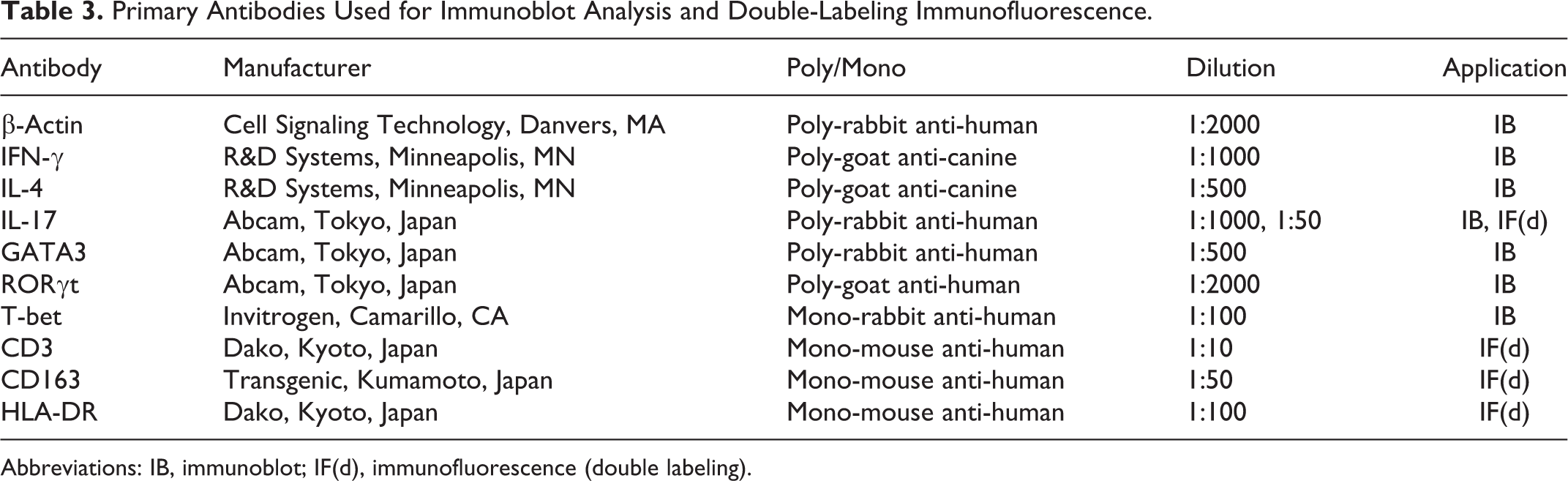
Abbreviations: IB, immunoblot; IF(d), immunofluorescence (double labeling).
Double-Labeling Immunofluorescence Quantification
The 10-μm-thick OCT compound–embedded frozen brain tissue sections were first treated with skimmed milk at 37°C for 40 minutes. Sections were then incubated at 4°C overnight with a first primary antibody and then with a second primary antibody at 37°C for 1 hour. The primary antibodies used are listed in Table 3. A secondary antibody cocktail was employed at room temperature for 1 hour. The cocktail included fluorescein isothiocyanate-labeled horse anti–mouse IgG (1:200; Vector Laboratories, Burlingame, CA) and Alexa Fluor 594–labeled donkey anti–rabbit IgG (1:200; Invitrogen, Eugene, OR). Sections were finally mounted with Vectashield HardSet with DAPI (Vector Laboratories) and observed using a Leica DMI3000 B fluorescence microscope (Leica microsystems, Wetzlar, Germany). The number of positive cells was counted under 400× magnification in 5 to 10 selected fields, perivascular cuffs, or neuroparenchymal infiltrates of each case, and the mean percentage was calculated.
Results
Pathologic Findings
Based on pathologic findings, the 8 cases examined were divided into 3 disease entities: NME (case Nos. 1, 2), NLE (case Nos. 3–6), and GME (case Nos. 7, 8). In NME cases, malacic lesions and inflammatory lesions, such as perivascular cuffs and the infiltration of inflammatory cells in the neuroparenchyma and leptomeninges, were marked in the cerebral cortex, hippocampus, and thalamus. In NLE cases, malacic lesions and inflammatory lesions were marked in the cerebral white matter, subcortical region, cerebellum, and spinal cord. However, malacic changes were mild, and inflammatory lesions were marked in the cerebral white matter, thalamus, midbrain, and cerebellum in 1 NLE case (case No. 5). In GME cases, nodular granulomatous lesions, composed of macrophages/microglia, lymphocytes, plasma cells, and a few neutrophils, and inflammatory lesions were marked in the cerebral white matter, subcortical region, midbrain, cerebellum, and spinal cord. These lesions and their distribution in the 3 diseases were similar to those of previous studies.
Messenger RNA Levels of Cytokines and Transcription Factors
Among the cytokines for the Th1 immune response, IFN-γ messenger RNA (mRNA) levels in the 2 NME cases were markedly higher than those in the NLE and GME cases. IL-12B mRNA levels were high in the 2 GME cases and moderately high in 1 NLE case (Fig. 1, upper). However, the expression levels of the Th1 cytokine, IL-12A, and transcription factor, TBX21, were not different between the diseases (Fig. 1, upper). Among cytokines for the Th2 immune response, IL-4 mRNA levels were high in NLE and GME cases (Fig. 1, middle). However, although IL-10 levels were not different between the diseases, they were moderately high in 1 NME case (Fig. 1, middle). GATA3 levels, a transcription factor of Th2 cells, were high in all NME and GME cases, as well as in 1 NLE case, but not in the other 3 NLE cases (Fig. 1, middle). In cytokines of the Th17 immune response, IL-17 and IL-21 mRNA levels were the highest in GME cases and were also marked in 1 NLE case (Fig. 1, bottom). RORγt levels, a transcription factor of Th17 cells, were moderate and similar between the diseases (Fig. 1, bottom). IL-12B is a subunit of IL-23, a stimulating factor for the secretion of IL-17 and IL-12. Thus, the high expression level of IL-12B in GME cases signified that the Th17 immune response was more connected than the Th1 immune response. In this study, IFN-γ was prominent in NME, and IL-17 and IL-21 were marked in GME.
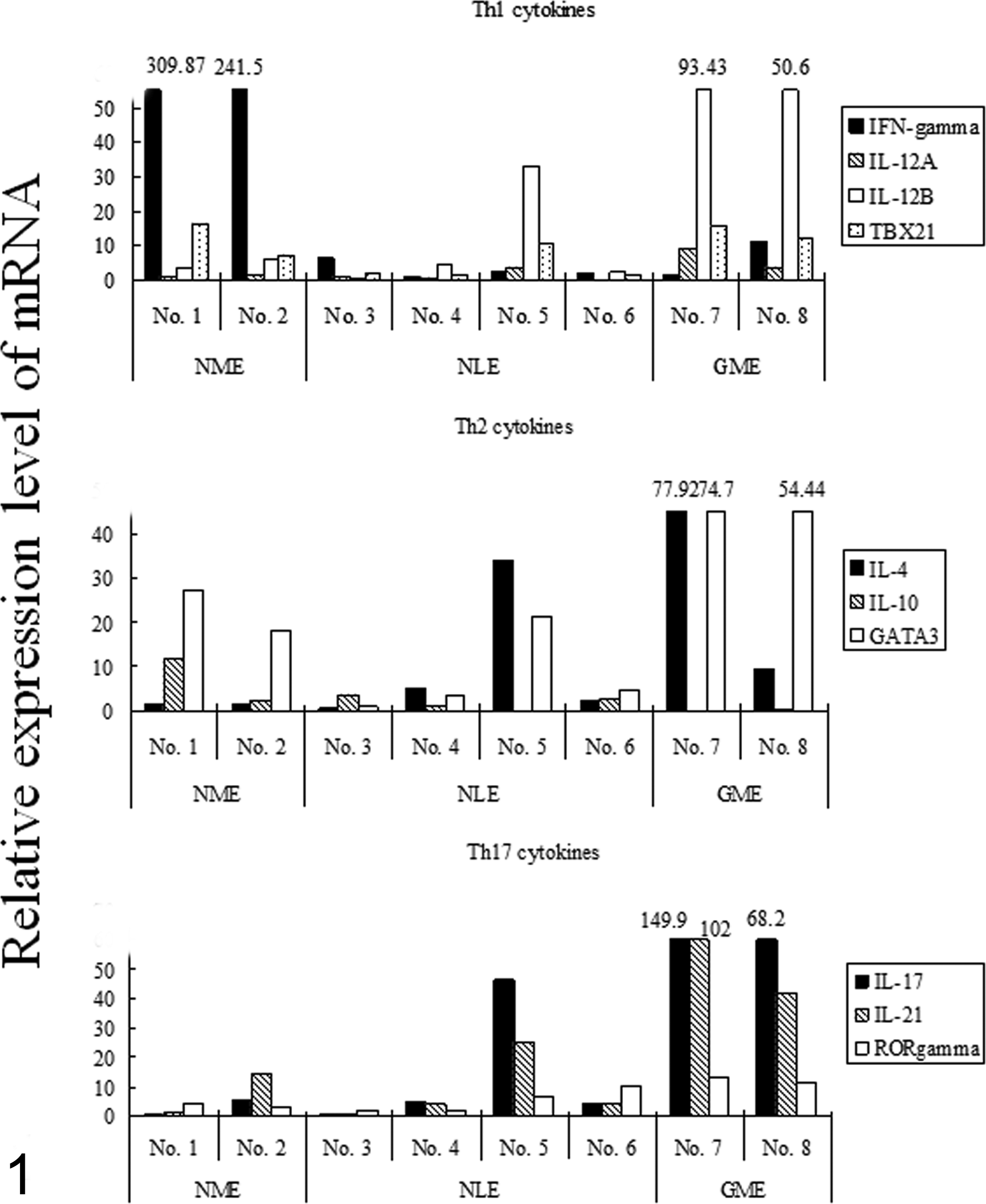
Messenger RNA (mRNA) levels of cytokines by real-time quantitative reverse transcription polymerase chain reaction. The relative mRNA level of each cytokine relative to the controls was measured and calculated in necrotizing meningoencephalitis (NME; case Nos. 1, 2), necrotizing leukoencephalitis (NLE; case Nos. 3–6), and granulomatous meningoencephalomyelitis (GME; case Nos. 7, 8). (Upper) Th1 cytokines, interferon (IFN)–γ, interleukin (IL)–12A, and IL-12B, and the transcription factor, TBX21. (Middle) Th2 cytokines, IL-4 and IL-10, and the transcription factor, GATA3. (Bottom) Th17 cytokines, IL-17 and IL-21, and the transcription factor, RORγt.
mRNA Levels of Chemokine Receptors
CXCR3, CCR2, and CCR4 mRNA levels were examined among the chemokine receptors. In NME and NLE cases, CXCR3 mRNA levels were the highest among the 3 chemokine receptors (Fig. 2). CCR2 and CCR4 mRNA levels were similar in NME and NLE cases (Fig. 2). In GME cases, CCR2 mRNA levels were the highest among the 3 chemokine receptors, although the levels in all 3 were similar (Fig. 2). CCR2 mRNA levels in GME cases were also higher than those in NME and NLE cases (Fig. 2). These results imply that cells expressing CXCR3, such as Th1 cells and macrophages/microglia, infiltrated NME or NLE cases, and the infiltration of cells expressing CCR2, such as Th17 cells and macrophages/microglia, was marked in GME cases.
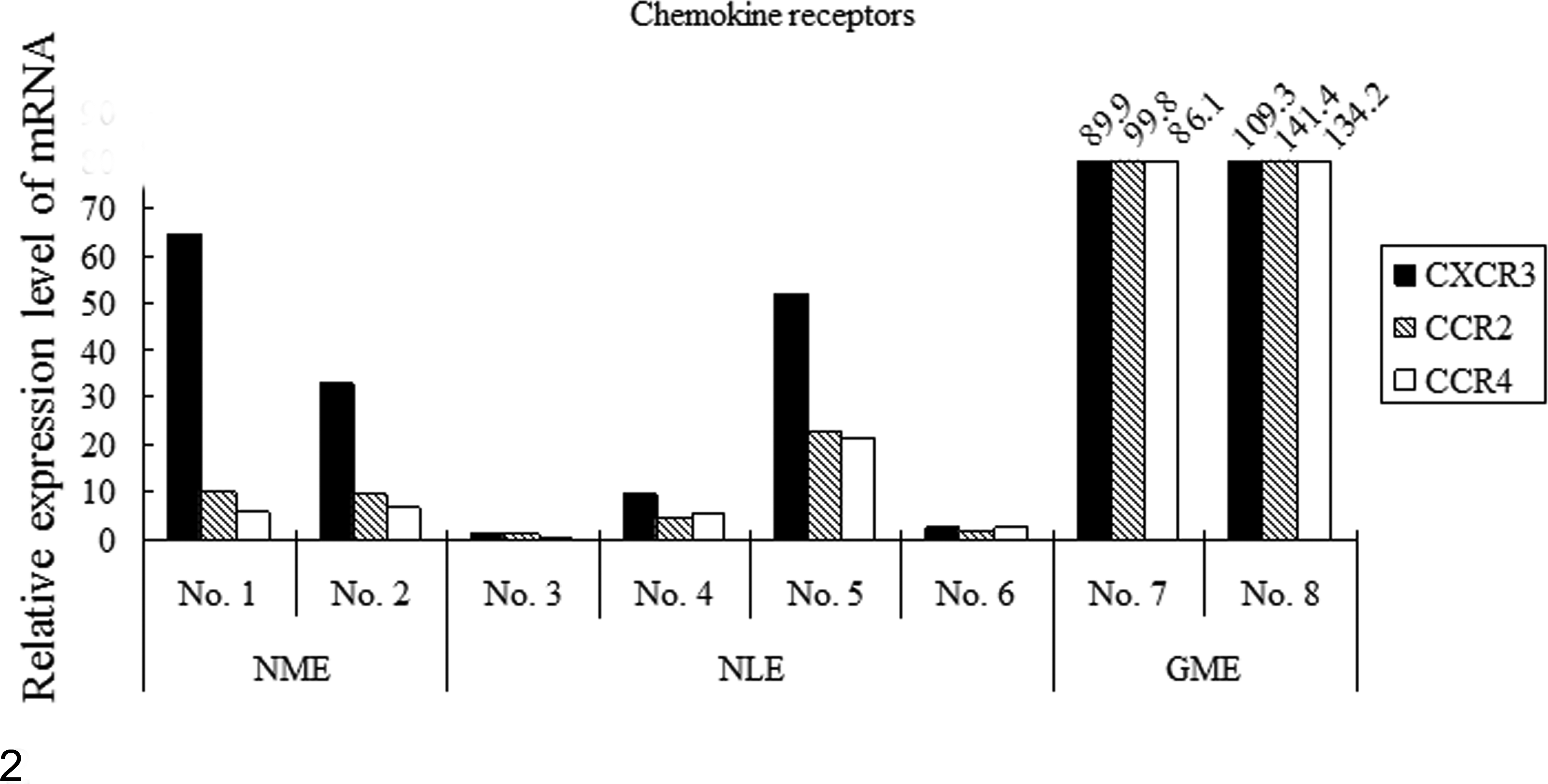
Messenger RNA (mRNA) levels of chemokine receptors by real-time quantitative reverse transcription polymerase chain reaction. The relative mRNA level of each chemokine receptor relative to the controls was measured and calculated in necrotizing meningoencephalitis (NME; case Nos. 1, 2), necrotizing leukoencephalitis (NLE; case Nos. 3–6), and granulomatous meningoencephalomyelitis (GME; case Nos. 7, 8).
Protein Levels of Cytokines
To confirm whether the protein expression of cytokines in brain lesions was similar to that of mRNA, we measured expression levels by immunoblot analysis. The expression of the IFN-γ protein was high in NME cases, which reflected the mRNA expression results (Fig. 3, upper). IL-4 expression was marked in 1 NLE case (case No. 5; Fig. 3, middle). IL-17 expression was marked in 2 GME cases (Fig. 3, bottom). Transcription factors were expressed similarly in all cases and controls, except for the expression of the RORγt protein, which was slightly higher in NME and GME (data not shown). In total, predominant cytokines were distinct in the diseased tissues of these diseases; IFN-γ and IL-17 expression was marked in NME and GME, respectively.
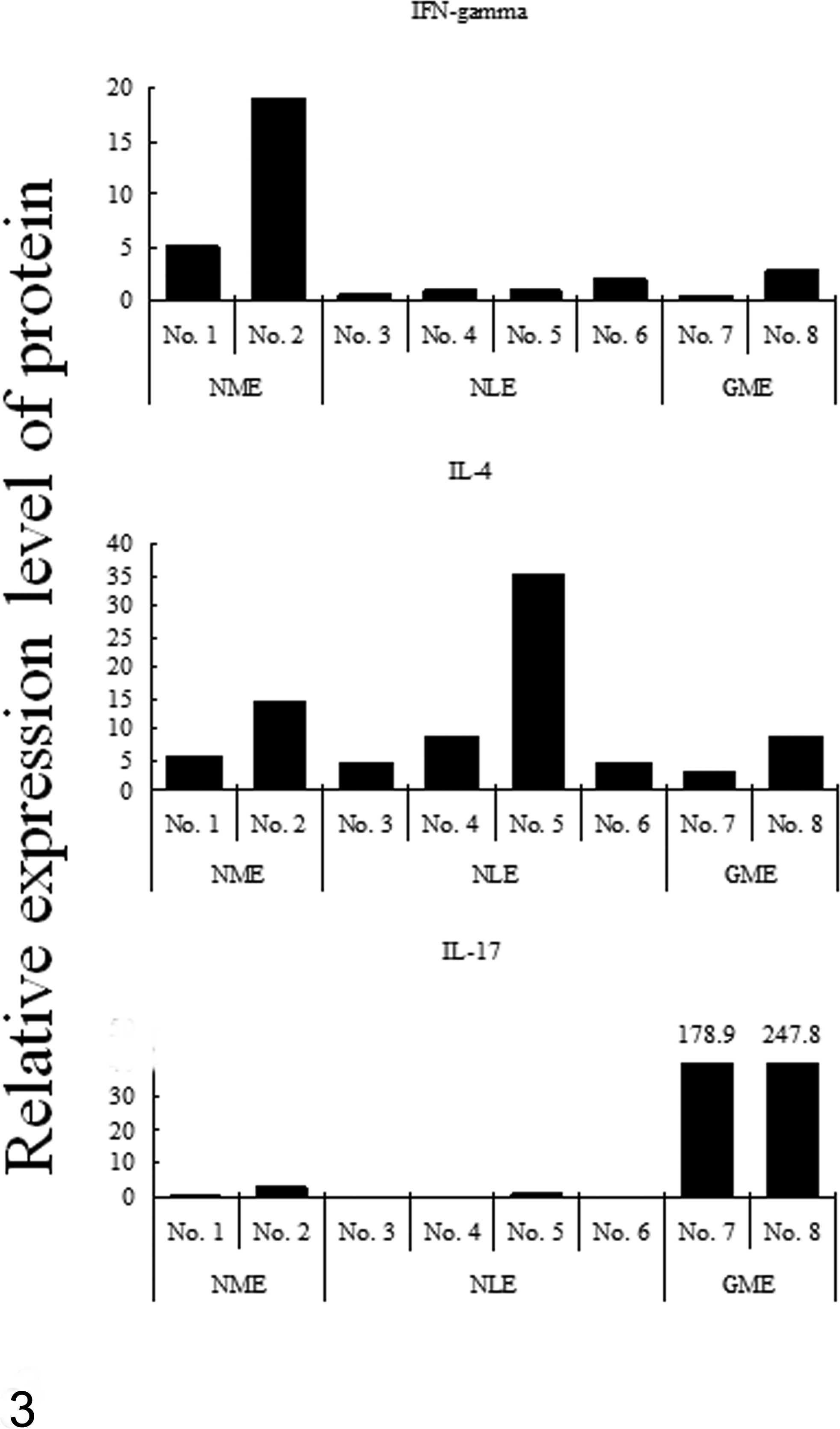
Protein levels of cytokines by immunoblot analysis. The relative level of each cytokine protein to that of β-actin was measured and calculated in necrotizing meningoencephalitis (NME; case Nos. 1, 2), necrotizing leukoencephalitis (NLE; case Nos. 3–6), and granulomatous meningoencephalomyelitis (GME; case Nos. 7, 8) (Upper) Interferon (IFN)–γ. (Middle) Interleukin (IL)–4. (Bottom) IL-17.
IL-17– and IL-17–Producing and/or Secreting Inflammatory Cells
We performed double-labeling immunofluorescence to identify cells that produced IL-17 in brain lesions. CD163-positive macrophages/microglia were more positive for IL-17 than CD3-positive T cells. Indeed, CD163-positive macrophages/microglia infiltration was marked in GME, NME, and NLE brains in that order. The percentage of IL-17–positive cells in CD163-positive macrophages/microglia was the highest in GME cases: 46% (case No. 6) and 73% (case No. 7) (Fig. 4, upper). CD3-positive T cells infiltrated GME and NME cases, as well as NLE cases to a lesser extent (Fig. 4, middle). The percentage of IL-17–positive cells in CD3-positive T cells was higher in GME cases: 20% (case No. 6) and 28% (case No. 7). HLA-DR–positive antigen-presenting cell (APC) infiltration was marked in NME and GME brains, with 35% (case No. 6) and 32% (case No. 7) being IL-17 positive in GME brains (Fig. 4, bottom). We then performed double-labeling immunofluorescence against HLA-DR for APCs such as macrophages, B cells, and dendritic cells. Since the number of CD20-positive B cells was not marked among these diseases and these cells were negative for IL-17, the results of double-labeling immunofluorescence against HLA-DR and IL-17 reinforced that against CD163 and IL-17. Hence, these results showed that the macrophages/microglia that infiltrated brain lesions produced IL-17 more than T cells and that this phenomenon was more prominent in GME cases than in NME and NLE cases.
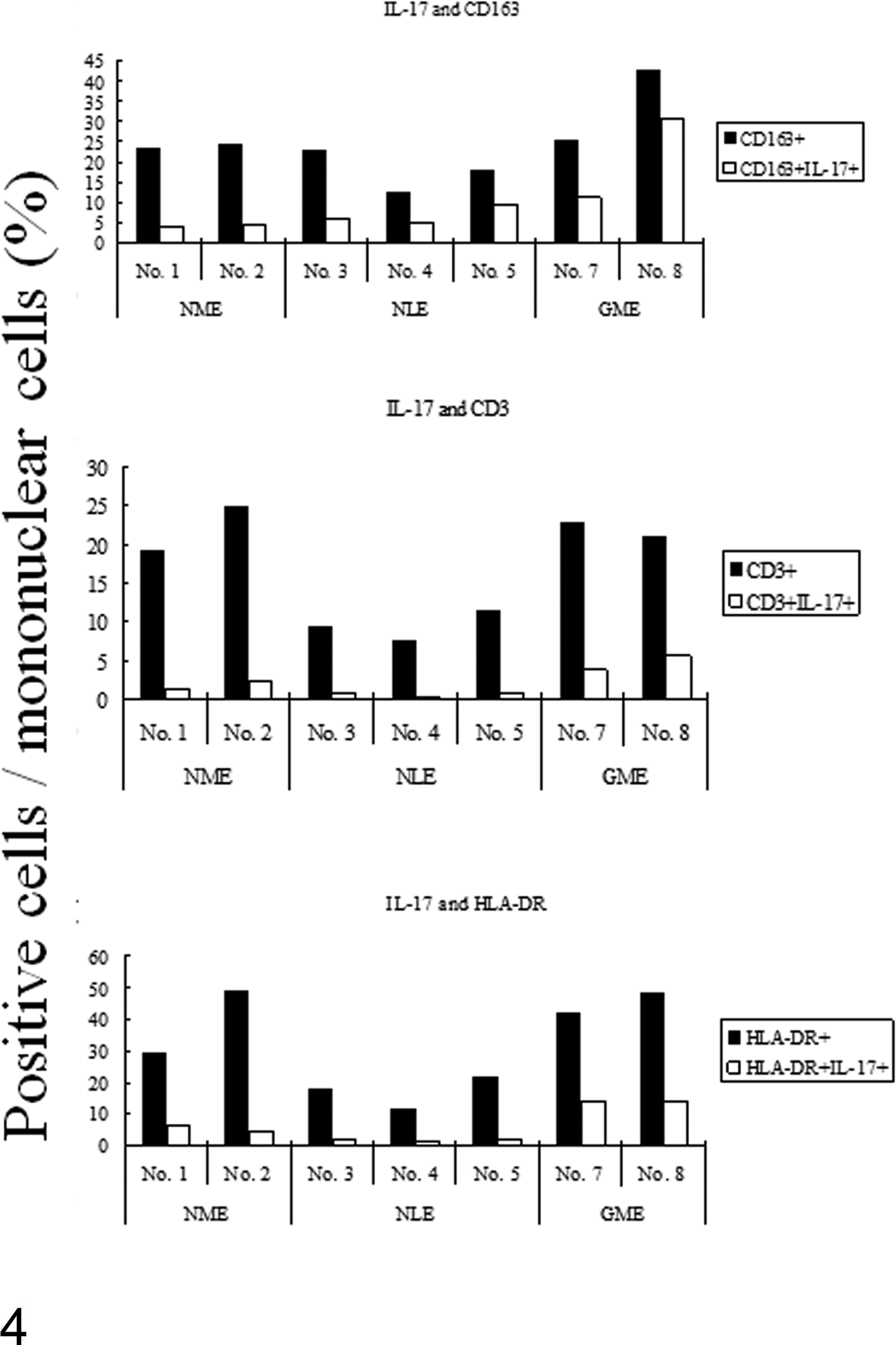
Percentage of positive cells by double-labeling immunofluorescence. Results are shown as the ratio of single-positive (black bar) and double-positive (open bar) cells in all nucleated cells. (Upper) Interleukin-17 (IL-17)–positive cells and IL-17 and CD163 double-positive cells. (Middle) IL-17–positive cells and IL-17 and CD3 double-positive cells. (Bottom) IL-17–positive cells and IL-17 and HLA-DR double-positive cells.
Discussion
In the present study, the cytokine profiles of brain lesions were characteristic of each lesion in NME, NLE, and GME. The expression levels of IFN-γ and IL-17 were marked in NME and GME cases, respectively. The expression levels of CXCR3 and CCR2 were the highest in NME and GME cases, respectively. Given that T cells and/or macrophages/microglia expressing CXCR3 or CCR2 have been reported to produce a large amount of IFN-γ or IL-17, respectively, these results were consistent with the results obtained for the cytokines. 1,4
It is evident that NME and NLE could be associated with autoimmunity against glial fibrillary acidic protein. 17,20,31 Autoreactive T cells, B cells, and autoantibodies against astrocytes were identified in these diseases. 20 CD3-positive T cells, CD20-positive B cells, IgG-positive plasma cells, and HLA-DR–positive or CD163-positive macrophages/microglia accumulated in these lesions. 20 In this report, lesions such as perivascular cuffs and the infiltration of inflammatory cells in the neuroparenchyma were also markedly observed among the 3 diseases studied. It has been reported that IFN-γ predominated in the perivascular cuffs of active MS lesions. 19,32 IFN-γ has cytotoxic characteristics, 19,32 and malacic changes were marked in the NME cases of this report. Therefore, the high expression levels of IFN-γ may be associated with the inflammatory lesions in NME cases. A previous study has suggested that astrocytes stimulate the secretion of IFN-γ and IL-17 from T cells and macrophages through the secretion of IL-23. 18 The possibility that the low expression of IL-17 in NME and NLE in the present results may be due to astrocyte damage cannot be excluded. However, since the CXCR3 mRNA expression levels involved in the secretion of IFN-γ were marked in NME cases, IFN-γ has been suggested to play an important role in NME.
The mRNA expression profile of 1 NLE case was similar to those of GME and NME cases, which showed high IL-4, IL-17, and IL-21 cytokine levels similar to those of GME cases, as well as high CXCR3 expression levels similar to those of NME cases. However, only IL-4 was marked at the protein expression level. Since the other 3 NLE cases in this report included more lesions of the end phase, differences in the cytokine repertoire between NLE cases may be accounted for. We observed a few cases that had general inflammatory lesions, but not distinct lesions, or showed intermixed distinct lesions and distributions among the 3 diseases. Thus, differential diagnoses had to be considered. This 1 NLE case could be one of those cases and was not a typical NLE case. In fact, this case showed distinct malacic changes in the cerebral white matter, and the distribution was similar to that of GME. As a result, although the IL-4 expression level in this 1 NLE case was distinct, the importance of IL-4 in NLE cases is not clear.
The mRNA expression levels of IL-17, CCR2, or CCR4, which are involved in the secretion of IL-17, 1,4 and protein expression levels of IL-17 were higher in GME cases than in NME and NLE cases. We then performed double staining to confirm which inflammatory cells produced IL-17. Most CD3-positive cells were expected to produce IL-17 since the infiltration of these cells was the highest in GME lesions. 20 However, a large number of CD163-positive cells were positive for IL-17 and were the highest in the present GME cases. The number of CD3 and IL-17 double-positive cells was modest. Some HLA-DR–positive cells were IL-17 positive in GME.
IL-17 has been reported to be involved in autoimmune diseases, allergic reactions, and antitumor immunities, and infiltrating Th17 cells produce IL-17 in some autoimmune diseases, such as RA, 10,23 Crohn disease, 3,12 MS, and EAE. 1,21 Neutrophils, eosinophils, mast cells, and macrophages also produce IL-17 in the lesions of allergic reactions. 19,24 GME may be related to delayed-type hypersensitivity. 11,29 Mast cells may also participate in the acute-phase lesions of GME and stimulate the development of GME lesions by increasing the permeability of the blood-brain barrier. 7 Neutrophils included in the granulomatous lesions of GME as well as mast cells may be involved in IL-17 production directly or indirectly through macrophages. 7 Taken together, IL-17 produced by macrophages/microglia may be an important factor in the formation of GME lesions.
The present study was the first to analyze the expression levels of cytokines in canine NME, NLE, and GME. The results indicate that the pattern of mRNA and protein expression of cytokines and chemokine receptors may be distinctly different in diseased tissues and inflammatory cells in the necrotizing and granulomatous forms of immune brain disease in small breeds: IFN-γ and CXCR3 in NME, as well as IL-17 and CCR2 in GME. This may be associated with the pathologic characteristics of each disease, despite the limitation of the small sample of fresh tissue available. Furthermore, macrophages within inflamed brain tissue may be disproportionately involved with IL-17 production relative to CD3-positive T cells in GME.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
