Abstract
In 2010, Coxiella burnetii was identified in 75% of northern fur seal placentas from a single rookery in Alaska, but nothing was known about the significance of this organism in the population. Although many infectious organisms cause increased cell death, C. burnetii has been shown to suppress apoptosis of the host macrophages as an intracellular survival mechanism. To determine if infection induces a similar functional change in the placenta, immunohistochemistry for antibodies to cleaved caspase-3 (activated caspase-3) and the (TDT)-mediated dUTP-digoxigenin nick end labeling (TUNEL) technique were used to compare the amount of placental apoptosis in infected and noninfected placentas. There was a statistically significant difference in the frequency of apoptotic cells between infected and uninfected placentas, with more apoptosis identified in the uninfected placentas. This finding suggests that the survival mechanism of C. burnetii in host macrophages to reduce apoptosis may also be utilized in trophoblasts. The significance of decreased trophoblastic apoptosis for the northern fur seal fetus requires further investigation.
Keywords
Coxiella burnetii, an intracellular bacterium found worldwide in numerous animal reservoirs, is the causative agent of Q fever in humans and causes reproductive failure in many species of mammals. 3 The decline of the northern fur seal (NFS; Callorhinus ursinus) population on St Paul Island, Alaska, has been well documented but is not easily explained; in a 20-year study evaluating pup mortality, the most common cause of death was trauma and nutrition, with infectious disease only infrequently identified. 8 In 2010, 75% of opportunistically collected NFS placentas from a single rookery were positive for C. burnetii by polymerase chain reaction (PCR). 4 Histologic lesions were infrequent; bacteria was visualized in 5 (3%) of the samples, and only a single placenta had arteritis. 4 As the placentas were assumed to be collected from live, healthy births, this result was surprising, and the significance of the high prevalence of infection in this declining population was unclear.
In all mammals, apoptosis of trophoblasts is a normal part of placental growth and separation during parturition. 5,8 In a normal placenta, the rate of apoptosis increases throughout gestation; however, apoptosis can also increase in placentas during disease states, such as preeclampsia, intrauterine growth restriction, 6 and placental infections such as Toxoplasma 1 or Listeria. 5 In contrast, C. burnetii has been shown to inhibit apoptosis in host macrophages as a survival strategy. 7,10 Like macrophages, trophoblasts have a phagocytic function, 2 suggesting the mechanism of bacterial survival within the cell may be similar and that cell death is likewise inhibited by infection. The objective of this study was to compare the frequency of apoptotic cells in NFS placentas infected with C. burnetii to noninfected placentas in attempt to determine if the organism elicits a functional change in the tissue.
A subset of placentas collected in 2010 from a single NFS rookery on St Paul Island 4 were organized by infection status into 3 groups; group A, those infected with histologically identifiable organism, confirmed by both immunohistochemistry (IHC) and PCR (n = 5); group B, C. burnetii–positive placentas (PCR) without organism seen on hematoxylin and eosin (n = 5); and group C, negative placentas as determined by hematoxylin and eosin and PCR (n = 5). The group A positives were the only placentas of the original 146 samples collected where organism was seen histologically. 4 Group B positive samples were selected per the highest number of genomic equivalents per gram of tissue, and the group C negative samples were randomly selected from the samples that were PCR and histologically negative. All formalin-fixed, paraffin-embedded tissues were cut at 4 μm, deparaffinized, then rehydrated with descending alcohol concentrations to buffer. Apoptosis was evaluated using the IHC marker cleaved caspase-3 and (TDT)-mediated dUTP-digoxigenin nick end labeling (TUNEL) with NFS lymph node used as a positive control. caspase-3 staining was performed using standard immunohistochemical technique on an automated stainer (Discovery System, Ventana Medical Systems, Tucson, Arizona). Reagents were purchased from Ventana Medical Systems. Antigen retrieval was accomplished with heat-induced epitope retrieval with EDTA buffer (pH 9.0) for 1 minute at 125°C. Sections were incubated for 8 hours at room temperature in anti-cleaved caspase-3 primary antibody (AF835, R&D Systems, Minneapolis, Minnesota) at a 1:500 dilution. A prediluted, universal biotinylated secondary antibody and a DAB MAP detection kit (Ventana Medical Systems) were utilized to detect the immunoreactive complexes, and slides were counterstained with Mayer’s hematoxylin. TUNEL staining was performed with In situ Cell Detection Kit POD (Roche Applied Science, No. 11684817910, Indianapolis, Indiana) following the manufacturer’s instructions. Heat-induced epitope retrieval with citrate buffer (pH 6.0) for 1 minute at 110°C was followed by endogenous peroxidase blocking with 3% hydrogen peroxide in methanol for 10 minutes. TUNEL reaction solution was applied for 1 hour at 37°C in a humidity chamber, followed by nonspecific protein blocking for 10 minutes with BackgroundSniper (Biocare LLC, No. BS966H, Concord, California). Converter solution was applied for 30 minutes at 37°C in a humidity chamber, followed by counterstaining with Mayer’s hematoxylin.
All slides were reviewed by 4 authors (EM, EE, BC, CD) for quality and pattern of staining. Quantitation of staining was conducted by a manual count of the number of positively staining trophoblasts per 400× field. Three regions of the placenta were reviewed: maternal interface, fetal interface, and a middle area between maternal and fetal portion of the placenta. Five 400× fields were counted in each region of the placenta, evenly spaced along the length of the tissue. Descriptive and comparative statistics were performed using commercially available software (SPSS 20.0). The nonparametric Kruskal–Wallis test was used to compare the frequency of positive cells between infection groups and between areas in the placenta. When only 2 groups were compared, the Mann–Whitney U test was used.
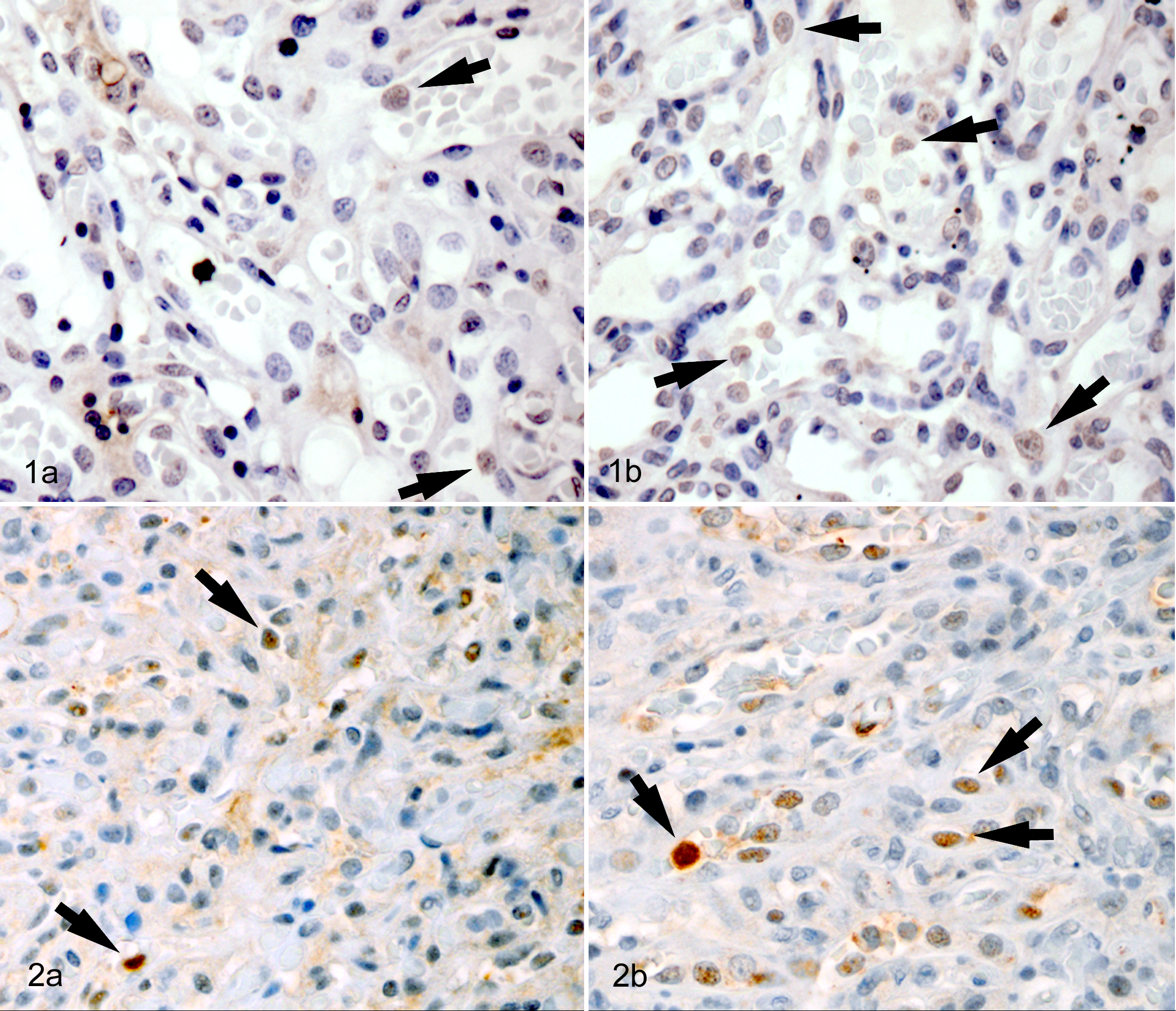
Immunohistochemical staining pattern and the frequency of positively stained cells are presented in Figures 1 and 2 and Table 1, respectively. Identification of specific cell types was challenging on IHC; most positive stained cells were interpreted as trophoblasts. caspase-3 staining was primarily intranuclear, but cytoplasmic staining was occasionally present, while TUNEL staining was exclusively nuclear. In all groups, more positively stained cells were identified in the caspase-3 samples compared to TUNEL. The frequency of apoptotic cells varied significantly between all 3 (A, B, C) groups (TUNEL, P = .012; caspase-3, P = .023); however, there was no significant difference in the frequency of stained cells between the 2 infected groups (A and B; TUNEL, P = .937; caspase-3, P = .211), so these 2 groups were merged into a single “infected” variable for subsequent analysis. Given that abundant bacteria is shed during parturition in domestic animals, 3 environmental contamination with C. burnetii was proposed as a potential cause for the high number of PCR-positive NFS placentas in which no organisms were seen histologically. 4 As there was no difference in the frequency of apoptosis between samples where C. burnetii was seen in tissue section compared to placentas positive on PCR alone, it is more likely that the 2010 NFS placentas were truly infected but that the organism is not distributed uniformly throughout the tissue and multiple regions of the placenta need to be evaluated to increase the sensitivity of histopathology to identify infection.
Frequency of Apoptotic Cells in Coxiella burnetii–Infected and Uninfected Northern Fur Seal Placentas Using 2 IHC Markersa
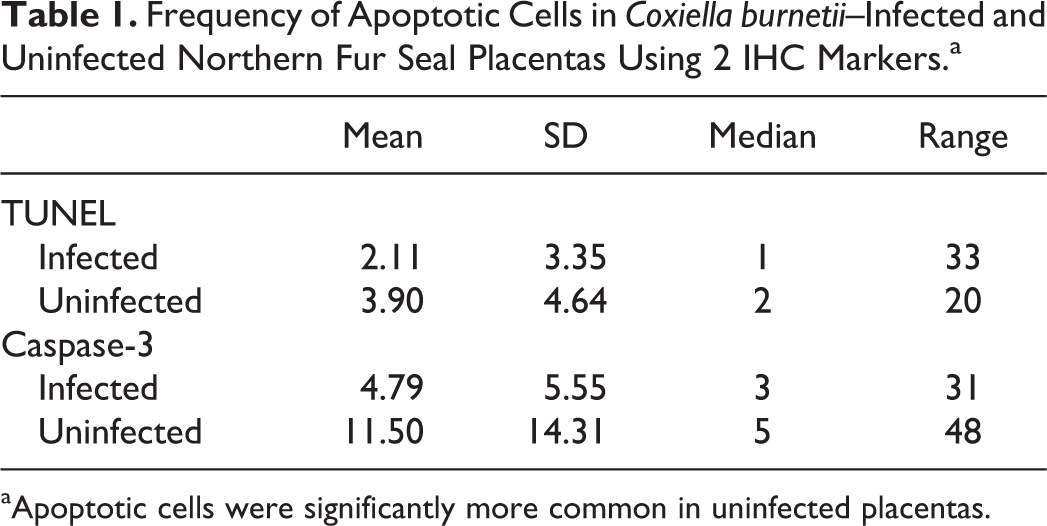
aApoptotic cells were significantly more common in uninfected placentas.
In noninfected placentas, there was no difference in the frequency of apoptosis between locations (fetal, middle, maternal) within the placentas for either TUNEL (P = .593) or caspase-3 (P = .191). In contrast, the infected placentas differed in the number of TUNEL-identified apoptotic cells that were identified within fetal, middle, maternal, and infected areas (P = .002) but not caspase-3 (P = .070). Subjectively, less staining was observed within or adjacent to C. burnetii–infected trophoblasts. The statistically different number of apoptotic cells in varying locations of the placenta by 1 IHC marker was interpreted to be due to proximity to infected trophoblasts and not a function of location within the placenta itself. Bacteria are not uniformly distributed throughout the placenta, but within areas of infection, cells contain a large number of organisms. It is possible that trophoblasts closer to identifiably infected cells have a higher probability of infection themselves compared to those farther away and small numbers of intracellular bacteria that may not be identifiable histologically could still prolong trophoblastic survival. Additionally, cell survival may be modulated by proteins secreted by regional, infected trophoblasts, through a paracrine signaling.
Overall there were significantly fewer stained cells in infected (A and B) placentas relative to the noninfected (C) placentas by both TUNEL (P = .003) and caspase-3 (P = .013). Decreased apoptosis in C. burnetii–infected placentas is consistent with the observations regarding apoptotic rates of macrophages when infected with this bacterium. 10 The mechanism for survival includes both altered expression of genes involved in apoptosis as well as increasing and blocking production of anti- and proapoptotic proteins respectively. 7,9,10 Increased apoptosis during disease states has been shown to be detrimental to the fetal viability, 6 but the significance of decreased apoptosis for NFS reproductive health and pup mortality is less clear. While placentas were collected from presumably successful live pup births, 4 nothing is known about the survival of the pup beyond placental detachment. Given that this intracellular bacterium proliferates and distorts the cell, it is likely that it impedes some cellular functions, which may hinder fetal nutrition, oxygenation, and waste removal; trophoblast survival alone does not equate to appropriate function. Given the decline of the population at this heavily infected site, further investigation into effect of C. burnetii infection on both individual animals and the population is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
E. Myers was supported by the Colorado State University, CVMBS Veterinary Summer Research Program. Partial funding for this project was provided by the National Marine Fisheries Service; all tissue samples were collected under authority of US Marine Mammal Permit No. 782-1708, issued to the National Marine Mammal Lab, Seattle, Washington.
