Abstract
Over a period of 6 months, dozens of moon jelly (Aurelia aurita) medusae from a single-species exhibit at the California Science Center (CSC) developed exumbrellar ulcers. Ulcers were progressive, causing umbrellar creases that expanded radially to the bell rim and occasional adoral erosions that extended into gastrovascular cavities. Husbandry interventions, including addition of ultraviolet light sterilizers, repopulation with fresh cultures, and enclosure disinfection, did not arrest the recurrence of lesions. Biopsies or whole specimens representing 17 medusae (15 affected and 2 grossly unaffected) from CSC and 2 control medusae from Aquarium of the Pacific were submitted to a private diagnostic laboratory and processed for light and electron microscopy. Microscopic lesions were present in all CSC medusae and were not observed or negligible in control medusae. Lesions included ulceration, necrosis, and hyperplasia in all umbrellar layers, with most severe lesions in the exumbrella and amoebocyte infiltration in the underlying mesoglea. Special stains, electron microscopy, and fungal culture did not associate microorganisms with the lesions. Bacterial cultures from the CSC population consistently grew Shewanella and Vibrio spp, both of which were considered commensal. Trauma and environmental stress are proposed as possible causes for the ulcers.
Jellies, which are commonly called “jellyfish,” are not fish but belong to the phylum Cnidaria, which represents approximately 9000 species of jellies, hydras, sea anemones, and corals. 29 They are referred to as coelenterates (Greek koilos, meaning “hollow,” plus enteron, meaning “intestine”) as their gastrovascular cavity functions in both circulation and digestion. The distinguishing feature of cnidarians is their cnidocytes, epithelial cells containing organelles capable of eversion. The most common cnidocyte organelles are nematocysts, the stinging structures used for capturing prey. 24
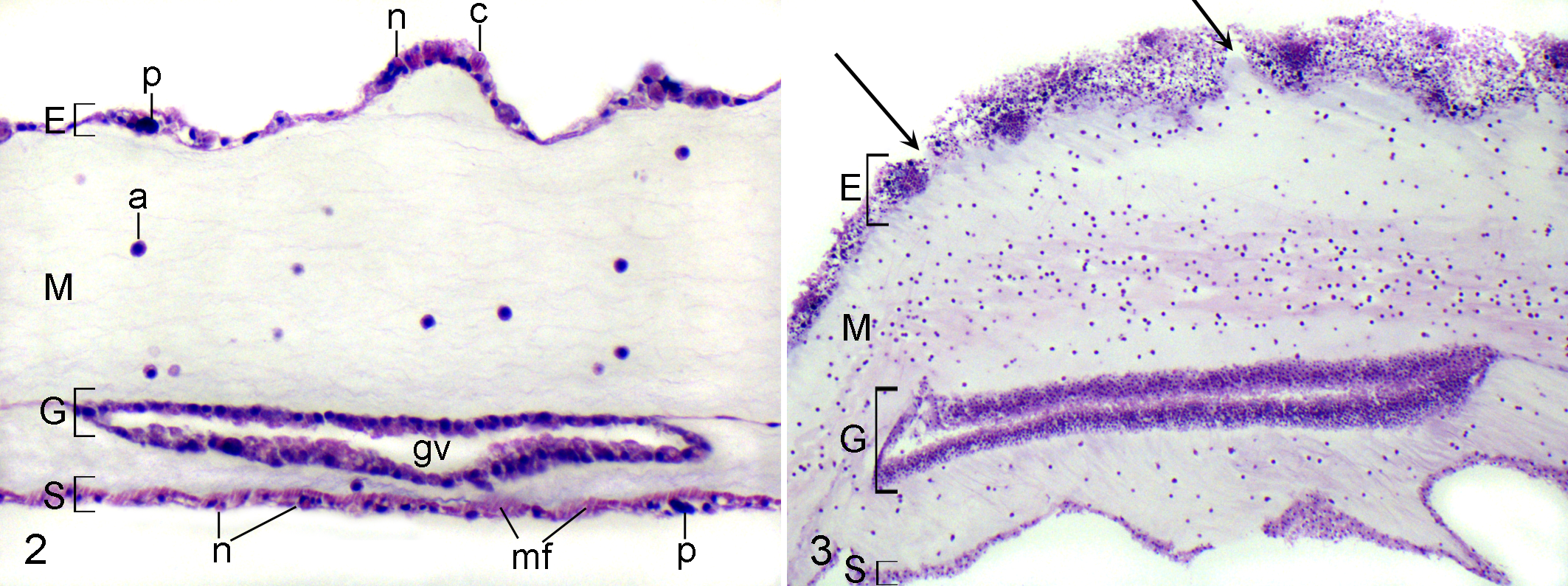
Jellies are in the cnidarian class Scyphozoa, which are characterized by a complex life cycle and 2 body forms: a smaller, asexual, benthic polyp and a larger, sexual, pelagic medusa resembling a “bell” or umbrella. 17,18,24,29 Cnidarians are usually considered diploblastic, consisting of only endodermal and ectodermal derivatives, although some evidence of mesodermal muscle has been proposed. 26 They have 3 body wall layers: epidermis (outer epithelium), gastrodermis (inner epithelium), and mesoglea (gelatinous layer between epithelial layers). 24,29 The epidermis is divided by location into the exumbrella, or aboral epithelium, and the subumbrella, or adoral myoepithelium. The gastrodermis lines gastrovascular cavities that radiate from the stomach throughout the mesoglea. 24
Medusae are a popular attraction at aquaria worldwide. 8,29,30 Captive jellies are also used as model organisms in comparative research, particularly in immunology and genetics. 2,18,29 Aurelia sp are prolific, being one of the most common jellies in captivity and widely dispersed jellies in the natural world. 1,5,30 Despite their ecologic importance, use in research, and popularity in zoos and aquaria, very little is known about the pathologic conditions of cnidarians. 1,7,8,29,31,32 The purpose of this study is to describe the clinical, microbiological, gross, and light and electron microscopic findings associated with an ulcerative condition of moon jelly (Aurelia aurita) medusae occurring at a public science center.
Materials and Methods
Cases
A. aurita medusae were obtained by the California Science Center (CSC; Los Angeles, CA) in 2009 from Santa Monica Pier Aquarium (Santa Monica, CA) and Cabrillo Aquarium (San Pedro, CA). Less than 10% of the population was supplemented with in-house culture. In October 2011, 24 A. aurita medusae were obtained from the Aquarium of the Pacific (AOP; Long Beach, CA) and moved to the CSC population.
At CSC, jellies were exhibited in a 643.52-liter pseudokreisel tank (ie, tank with curved edges to facilitate laminar flow and minimize wall trauma) with biofiltration and cultured in a 613.24-liter pseudokreisel tank with mechanical filtration. Jellies were treated as one population and shifted freely between these enclosures. Water quality parameters (pH: 8.1–8.3, salinity: 31–35 ppt, alkalinity: >2.5 meq/l, NH3: <0.04 mg/l, NO2: <0.1 mg/l, NO3: <25 mg/l) were checked twice weekly and were within normal limits, with the exception of 2 mildly aberrant readings (pH: 8.33, salinity: 35.5 ppt) that occurred at separate time points over several months in the inclusive study period. The temperature was maintained at 15.28°C–16.28°C. Daily exhibit maintenance included siphoning detritus and wiping down all internal exhibit surfaces. Once a month, jellies were removed from their exhibit to disinfect the tank. Disinfection was accomplished by scrubbing with 5% bleach (sodium hypochlorite) solution (Tough Guy Bleach®; Champion Packaging, Woodridge, IL), neutralizing with sodium thiosulfate (Wintersun Chemical Inc, Ontario, CA), and rinsing with loop water (ie, natural sea water; Catalina Water Company, Long Beach, CA). The culture tank was disinfected weekly with the same protocol. Jellies were fed enriched brine shrimp (Artemia nauplii; AquaFauna Bio-Marine Inc, Hawthorne, CA; enriched with Algamac 3000, AquaFauna Bio-Marine Inc, and Pavlova 1800 Instant Algae, Reed Mariculture, Campbell, CA) twice daily, and a mixture of krill (Euphausia pacifica and E. superba; State Fish Co, San Pedro, CA), mysid shrimp (Americamysis bahia; San Francisco Bay Brand Inc, Newark, CA), Cyclop-eeze (Argent Laboratories, Redmond, WA), blackworms (Lumbriculus variegates; Bayou Aquatics, Ontario, CA), and a multivitamin (Marine Cuisine; San Francisco Bay Brand Inc, Newark, CA) twice weekly.
Lesions were identified in CSC medusae 3 weeks after the addition of AOP medusae. Affected medusae were visually monitored, and no individual treatments were attempted. Aerobic bacterial cultures were performed on krill and biomedia. Aerobic bacterial cultures were also performed on 2 affected medusae, 10 days apart. A fungal culture was performed on the first medusa sampled. Ultraviolet (U/V) light sterilizers (Aqua Logic Inc, San Diego, CA) were added to both exhibit and culture systems (108 and 40 W, respectively) to inactivate microorganisms. The jelly culture tank and system were depopulated, deconstructed, and disinfected between case Nos. 1–10 and Nos. 11–17. Disinfection was performed as previously described. At the same time, polyp culture plates were placed in a freshwater dip for 5 minutes in attempt to eradicate suspected pathogenic microorganisms.
Medusae were euthanized when ulcers interfered with normal belling or eroded into gastrovascular cavities. Over a period of 6 months, samples from 17 affected medusae were collected by netting medusae and euthanizing them whole by placing them directly in 10% neutral buffered formalin or by excising 1-cm2 samples of representative areas and placing biopsies in 10% neutral buffered formalin. For comparative purposes, samples were also taken from 4 grossly normal A. aurita medusae: 2 from the same population at CSC and 2 from a different population at AOP. All samples were submitted to Northwest ZooPath for further examination.
Histopathology
During trimming, specimens were moved between formalin containers and cutting surfaces by balancing tissues on forceps’ handles or cupping specimens to avoid unnecessary trauma that may hinder histologic examination. Tissues were embedded in paraffin, sectioned at 5-μm-thick sections and stained with hematoxylin and eosin (HE). Select duplicate sections from affected and unaffected medusae were also stained with Giemsa, Gimenez, Warthin-Starry, periodic acid–Schiff, Gomori methenamine silver, and Brown and Brenn stains.
Microscopic lesions were categorized by location and type of lesion, then graded by severity. The exumbrella, subumbrella, and gastrodermis were graded on epithelial ulceration, necrosis, and hyperplasia. The subumbrella was also graded on hypertrophy of the myofibrillar component of myoepithelial cells. The mesoglea was graded on increased density of amoebocytes. Denuded areas were excluded if not accompanied by hyperplasia, necrosis, or inflammation to rule out artifactual epithelial separation. Hyperplasia was diagnosed only in appropriately sectioned areas and when accompanied by mesogleal inflammation or other lesions to rule out sectioning artifact. Grades for each category included not observed (histologically normal), mild (rare), moderate (occasional), marked (frequent), and severe (minimal to no normal tissue was observed histologically). Multiple sections were examined and graded for each medusa. The median grade was applied, except when categories had both not observed and mild grades, in which case a mild grade was applied to document the presence of a lesion.
Electron Microscopic Analysis
Formalin-fixed biopsies from 1 affected medusa (case No. 12) were retrimmed and placed into modified (50% strength) Karnovsky’s fixative. After immersion in Karnovsky’s fixative, the tissues were postfixed in 1% osmium tetroxide. After osmification, the tissue was rinsed in 0.1M sodium cacodylate, dehydrated through a graded ethanol series, transitioned through propylene oxide, and infiltrated and embedded in Eponate-12 epoxy formulation (Eponate-12; Ted Pella Inc., Redding, CA). Thick sections were cut, mounted on glass slides, stained with toluidine blue O, and examined by light microscopy. Thin sections were mounted on 300-mesh copper grids and stained in 4% uranyl acetate in 75% ethanol, followed by post staining in lead citrate. The grids were examined in a Zeiss 906E transmission electron microscope at 100 kv accelerating voltage (Carl Zeiss SMT, Peabody, MA).
Results
Gross Findings
Lesions occurred in medusae with bell diameters of ≥ 4 cm. Ulcers, 1–2 mm in diameter, were first noted on the exumbrella. Over approximately 6 weeks, ulcers enlarged up to 6 mm in diameter, at which point the affected area of the bell collapsed and developed a crease that extended radially from the ulcer to the bell rim (Fig. 1) and interfered with normal belling. Occasionally, ulcers eroded adorally into gastrovascular cavities. All lesions were progressive and necessitated humane euthanasia. Surgical debridement was not attempted, as the lesions were extensive with areas of bell collapse and treatment was unlikely to be therapeutic or result in exhibit quality animals.
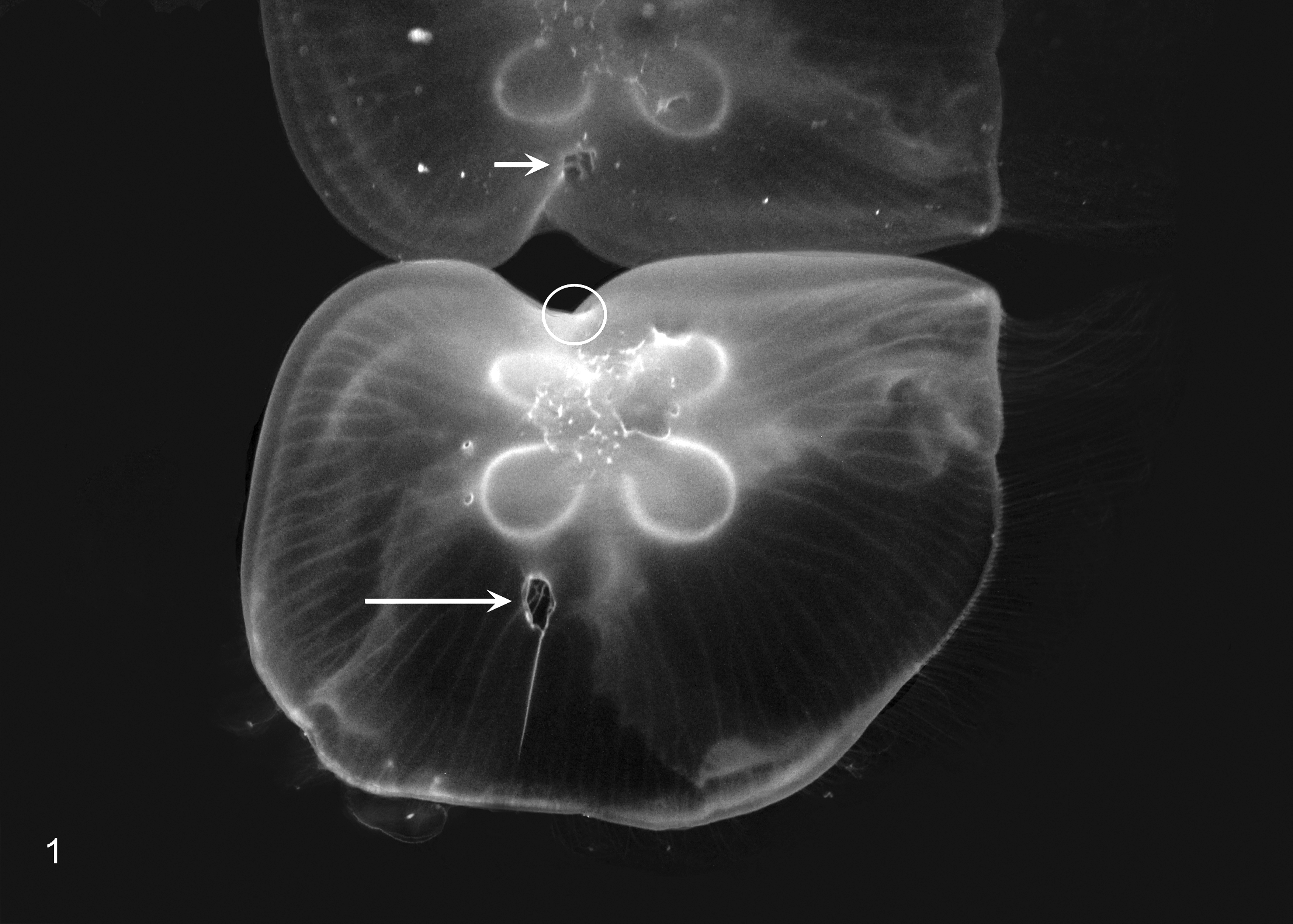
Medusa; umbrellar ulcers. The exumbrella has 2 large ulcers, each with an associated bell crease that extends radially toward the bell rim. One large ulcer is visible on the exumbrella (long arrow) with the crease extending ventrally in the image. The other large ulcer is visible only in the reflection on the water’s surface (short arrow) with the crease extending ventrally in the image and represents an area on the distal surface of the exumbrella (circle). Multiple small ulcers without creases are present adjacent to the horseshoe-shaped gonads.
After introducing U/V light sterilization and culling approximately one-half of the population due to extensive lesions, the incidence of ulcers decreased dramatically, from > 80% affected to < 20%. After depopulation, deconstruction, and disinfection of the culture tank and freshwater treatment of the polyp culture, the new crop of medusae grew to ≥ 4 cm in bell diameter, were added to the remaining exhibit population, and subsequently developed ulcers similar to those seen prior to husbandry interventions. Affected medusae were continually culled until the exhibit population decreased to approximately 20–30 jellies. Ulcers did not recur at this population size, and the population was maintained at 20–30 jellies without incidence.
Cultures
All aerobic bacterial cultures grew both Vibrio and Shewanella spp. Medusa cultures had moderate growth of Vibrio and light to moderate growth of Shewanella sp. The first medusa culture also grew one colony of methicillin-resistant, coagulase-negative Staphylococcus sp. The biomedia culture had moderate growth of Vibrio sp, Shewanella putrefaciens, and Pseudomonas aeruginosa. The krill culture had abundant growth of Vibrio sp and S. putrefaciens and grew a few colonies of nonpathogenic Coryneform bacteria. The medusa fungal culture had no growth.
Histopathology
All medusae from the CSC population (case Nos. 1–17), including grossly unaffected specimens, had microscopic lesions in all umbrellar layers (Table 1). In CSC medusae, the most severe lesions occurred in the exumbrella and underlying mesoglea, and the epithelium of all layers had variable hyperplasia, necrosis, and ulceration (Figs. 2, 3). Necrotic epithelial cells had vacuolar degeneration, swelling, and lysis (Fig. 4). Necrosis preferentially affected epithelial cells as opposed to cnidocytes. Hyperplasia was characterized by increased numbers of cells and nuclei in affected epithelium. Mitotic figures and anisokaryosis were not observed. In affected areas of the bell, the exumbrellar epithelial cells had multifocal to coalescing, moderate to marked necrosis with hyperplasia that was occasionally associated with microscopic ulceration (Figs. 4, 5); the gastrodermal epithelial cells had diffuse, moderate to marked hyperplasia that was occasionally associated with sloughing of necrotic cells into the gastrovascular lumen, excessive mucus production, and rare ulceration; the subumbrellar myoepithelium was thickened due to hyperplasia and hypertrophy of myoepithelial cells and was multifocally necrotic with rare ulceration; and the mesoglea had moderately to severely increased density of amoebocytes (inflammation) in varying stages of apoptosis.
Histologic Features of Umbrellar Ulcers in a Captive Population of Moon Jelly (Aurelia aurita) Medusae.
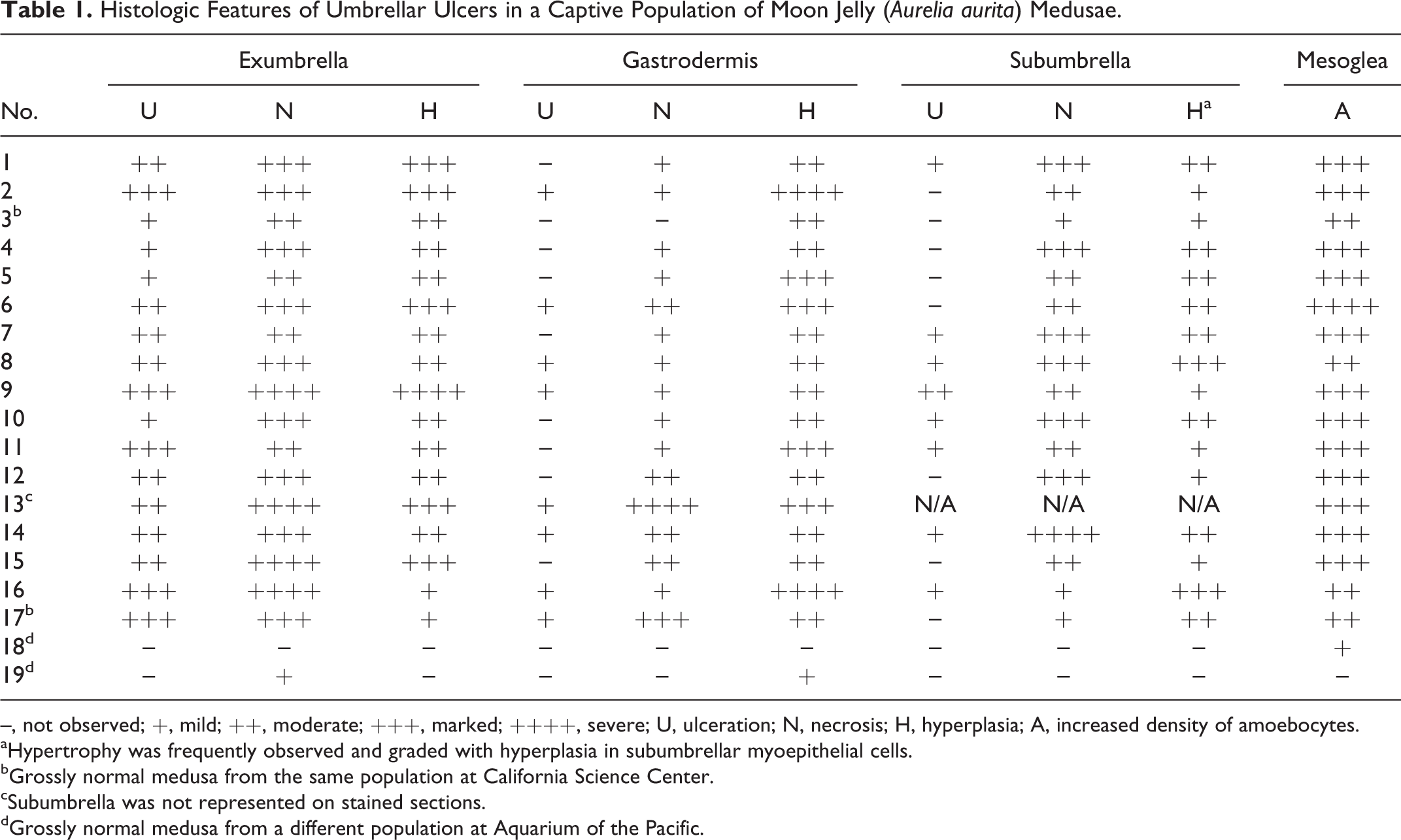
–, not observed; +, mild; ++, moderate; +++, marked; ++++, severe; U, ulceration; N, necrosis; H, hyperplasia; A, increased density of amoebocytes.
aHypertrophy was frequently observed and graded with hyperplasia in subumbrellar myoepithelial cells.
bGrossly normal medusa from the same population at California Science Center.
cSubumbrella was not represented on stained sections.
dGrossly normal medusa from a different population at Aquarium of the Pacific.
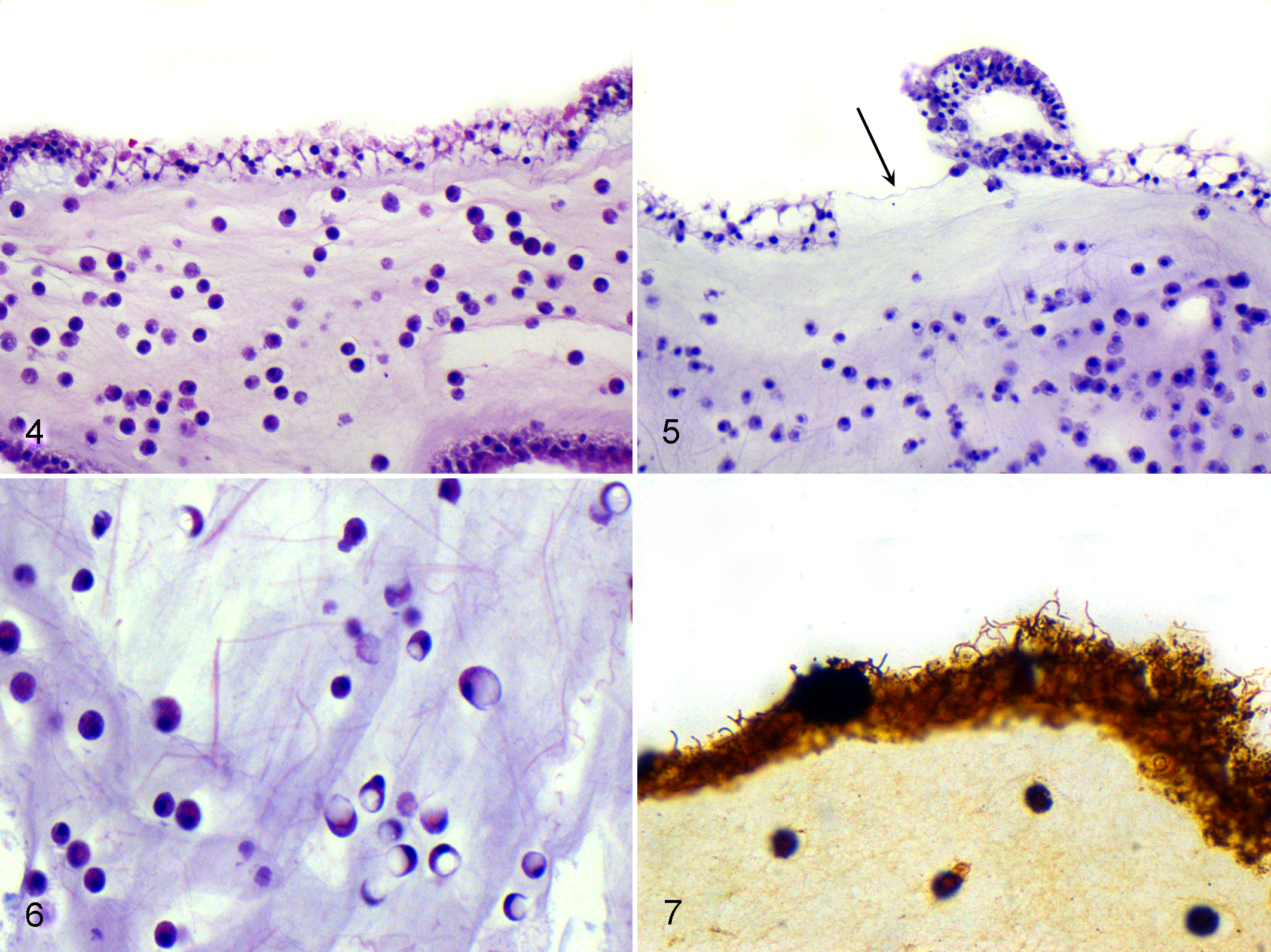
In 2 grossly ulcerated medusae (case Nos. 1, 4), amoebocytes had large, clear, spherical cytoplasmic vacuoles that caused cellular swelling and peripheral displacement of nuclei (Fig. 6) that was interpreted as phagocytosis of edema. On HE-stained sections, low densities of pleomorphic bacteria were observed in the mesoglea of 4 medusae (case Nos. 12–15) and associated with ulcers in 2 medusae (case Nos. 2, 11). These bacteria were not observed with special stains. In both CSC and AOP medusae, Warthin-Starry staining demonstrated spirochetes that were associated with all layers of epithelial cells and were particularly abundant in gastrovascular canals (Fig. 7). These organisms were not associated with lesions.
Medusae from AOP had few histologic lesions (Table 1). One medusa (case No. 18) had mild inflammation. The other (case No. 19) had multifocal, mild exumbrellar epithelial necrosis and focal, mild gastrodermal hyperplasia. Amoebocytes in all medusae (case Nos. 1–19) were present in varying stages of apoptosis, ranging from normal to degenerate to apoptotic. The proportion of amoebocytes in each apoptotic phase was equivalent in all cases and independent of the presence of lesions. Amoebocyte density, however, was apparently associated with lesions and was moderately to severely increased in areas with epithelial necrosis, hyperplasia, and ulceration.
Electron Microscopy
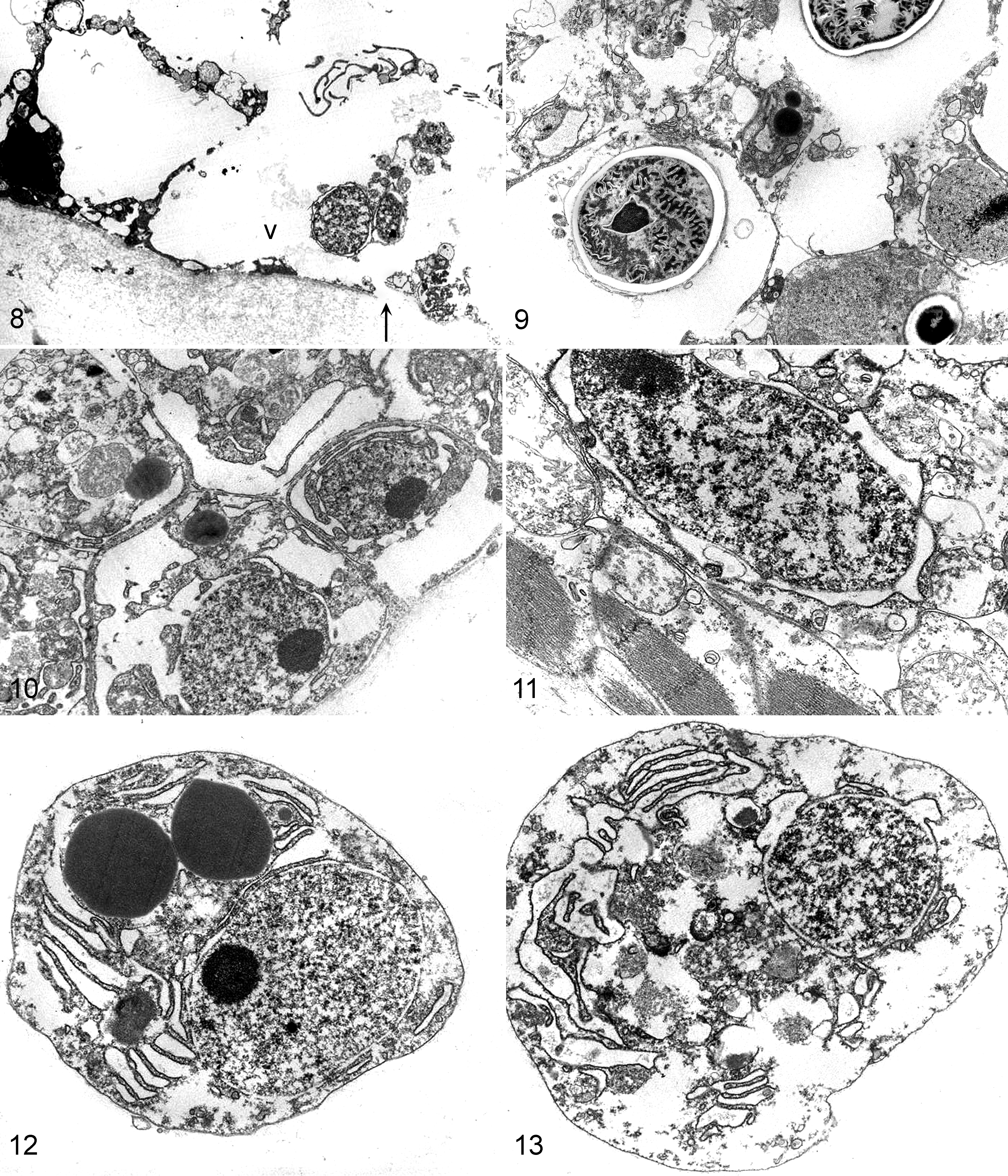
Ultrastructurally, medusa case No. 12 had lesions that corresponded with the light microscopic findings. The exumbrellar epithelial cells were severely vacuolated, and organelles were lysed or absent. Cell membranes were often lysed, and nuclei were pyknotic (Fig. 8). The exumbrellar epithelial cells near cnidocytes were less vacuolated compared to areas void of cnidocytes, and in general, cnidocytes were less severely affected than epithelial cells (Fig. 9). Hyperplasia of the gastrodermis was characterized by stratification of the epithelial layer (Fig. 10). The swollen subumbrellar myoepithelial cells had intracellular edema and markedly swollen mitochondria (Fig. 11). The amoebocytes were in various stages of degeneration (Figs. 12, 13). No microorganisms were identified ultrastructurally.
Discussion
Aquatic invertebrates have no acquired immunity and rely on an innate immune system. 2,22 In cnidarians, the lack of a true mesoderm precludes development of circulating immune cells, such as hemocytes, which are present in triploblastic coelomic invertebrates. 11,27 The principal defense cells of cnidarians are phagocytic cells in the mesoglea called amoebocytes, which were first described over 100 years ago with the exposition of phagocytosis. 4,11,19,24,27 Despite this long-standing history, histologic features of inflammation in jellies have not been well documented. In this report, inflammation in jellies is characterized by an influx of amoebocytes in the mesoglea, which is consistent with the working definition of inflammation in phylogenetically linked corals. 31,32
In all CSC medusae, mesogleal inflammation was consistently associated with epithelial lesions. In soft corals, mesogleal edema has been reported as a component of inflammation and is demonstrated by dilation of mesogleal connective tissue spaces. 31 This change could not be distinguished from separation artifact in most medusae but was appreciated in 2 cases due to phagocytosis of edema by amoebocytes, suggesting this component of inflammation may be conserved among cnidarians.
Histologically, the most severe lesions frequently occurred in the exumbrella, which was the nucleus for gross ulceration. Exumbrellar epithelial necrosis was the most common histologic lesion. Although characterized by several features of necrosis, such as cellular swelling, pyknosis, and lysis, epithelial cells lacked some histologic changes expected with necrosis, including karryorhexis and hypereosinophilia. 14 It is not clear if cnidarian epithelial cells follow a typical pattern of necrosis as observed in their mammalian counterparts, and for this reason, these epithelial changes may be more accurately described as degenerative, though the term necrotic is used throughout this work for descriptive consistency. Necrosis occurred preferentially in epithelial cells and rarely affected cnidocytes. The mechanism of this cnidocyte-sparing effect is unknown but may be related to the robust nematocyst capsule. 16 Epithelial hyperplasia was consistently observed concomitant to necrosis and inflammation, suggesting a regenerative response.
In the cases presented herein, an infectious etiologic agent was initially suspected, as the appearance of ulcers closely coincided with the addition of medusae from another population. Bacteria were not consistently associated with lesions, which obviated the need for additional bacterial culture attempts, including anaerobic cultures. Additionally, no other types of infectious agents, such as viruses, fungi, or algae, were demonstrated by electron microscopy or fungal culture, which precluded sequencing by polymerase chain reaction. All bacterial cultures grew Vibrio and Shewanella spp. Both genera represent members of normal bacterial flora in cnidarians yet have a wide array of potentially pathogenic toxins, including hemolytic, septicemic, and necrotic compounds. 25 The symbiotic flora of marine invertebrates are highly species specific and in most cases, such as A. aurita, have not been wholly described. 2,25 Without defining the normal flora of A. aurita, it cannot be definitively determined if these bacteria represent a shift toward opportunistic invaders or are part of the symbiont. Warthin-Starry staining revealed spirochetes in both control and affected medusae that were not associated with lesions, suggesting that these organisms are likely commensal. The krill culture and 1 medusa culture grew Coryneform and Staphyloccocus spp, respectively. Colony growth was very low in both cases, and these organisms were likely culture contaminants. Only the biomedia culture grew P. aeruginosa, which is a nearly ubiquitous bacteria that lives in a diversity of environments, including streams, shorelines, and open oceans. 13 Though P. aeruginosa is a common opportunistic pathogen, it is unlikely to have caused lesions in the cases presented herein, as it was not cultured from medusae and is prolific in aquatic environments. An association between epithelial insults and susceptibility to pathogens has been demonstrated both clinically and experimentally in cnidarians. 2,6 Epithelial disruption was a consistent finding among CSC medusae, and the ability to defend against pathogen invasion may have been conferred by areas of intact epithelium adjacent to lesions and/or underlying amoebocyte infiltration.
In captive jellies, inappropriate currents and tank design have been implicated as sources of trauma and ulceration. 29,30 The gross and histologic morphology of ulcers in the cases presented herein is similar to that from a preliminary report in which umbrellar ulcers were attributed to traumatic collisions and/or inappropriate currents. The cases from both the previous and present reports had exumbrellar ulcers that caused bell creases and occasionally eroded into gastrovascular cavities, as well as epidermal necrosis with mesogleal inflammation. In the previous report, medusa collisions with tank walls were observed, and almost all ulcers were infiltrated with either monomorphic bacteria or algal-like organisms. 28 In the cases presented herein, neither collisions nor intralesional infiltration by the aforementioned organisms were observed, and pleomorphic bacteria rarely infiltrated ulcers. In the present cases, the occurrence of ulcers coincided with an increased density of animals and resolved when the density returned to pre-AOP levels. Though collisions were not observed, it is likely that the increased density of jellies exacerbated inappropriate currents and/or caused intraspecific collisions that led to primary ulceration. Specific density thresholds for said trauma are difficult to establish across tanks whereby flow pattern, flow rate, and tank design are independently adjusted. Instead, density thresholds for individual tanks must be established in light of the tank’s design.
In wild corals, a number of syndromes have been described that produce epithelial tissue loss and necrosis, yet it is exceedingly rare that the pathogenesis is known or that Koch’s postulates can be fulfilled. Regardless of etiology, tissue loss in corals is accepted as a nonspecific reaction to a wide variety of stressors. 29 A similar response has been demonstrated in fish that develop cutaneous ulcers in response to stress, which was induced by confinement or epinephrine injection. 21 The authors propose that the stress response seen in corals may be conserved among other cnidarians, specifically scyphozoans, whereby “tissue loss,” or ulceration, occurs in response to stress. In the cases presented herein, medusa ulceration may have been caused by a nonspecific environmental stressor, which compromised the epithelium, leading to the micro- and macroscopic lesions described. Senescence in invertebrates causes functional changes in the immune system, such as a reduced ability to clear infections. 15 This was not considered a contributing factor for the lesions in the cases presented herein, as jellies did not exhibit changes consistent with senescence, such as curved bells, tentacle loss, or alterations in oral arm length, nor had they undergone spawning, an almost ubiquitous prequel to senescence. 10
A common manifestation of disease in cnidarians is “bleaching,” or disruption of the symbiotic relationship with zooxanthellae. Hosts are dependent on this symbiotic algae for energy in the form of photosynthate and for recycling of nitrogenous wastes. 9 In anemones and corals, bleaching may be caused by a variety of stressors and is correlated to an increased susceptibility to disease. 3,12,23 Although many scyphozoans harbor symbiotic algae, A. aurita is possibly an azooxanthellate species, and their dearth of zooxanthellae may be normal. 20 The diversity of symbionts among scyphozoans underscores the importance of evaluating lesions in light of species-specific symbiosis.
There are multiple challenges to preparing medusae for histologic examination. Despite delicate collection techniques and immediate fixation, tentacles, gonads, oral arms, and portions of the umbrella often became separated from umbrellas within 2 days of being in formalin. Shipping containers included air-filled cells to minimize damage during transportation. During trimming, specimens were moved between formalin containers and cutting surfaces using atraumatic techniques, as any grasping caused dissecting erosion of the epithelium. Additionally, the jelly-like nature of the specimens frequently caused posttrimming realignment within cassettes, making oblique sections difficult to avoid.
Widmer provided an overview of signs, possible causes, and solutions to common jelly disorders. Over 20 gross signs of disease, including many bell alterations, were described. No descriptions were consistent with the ulcerative and erosive bell lesions observed in this study. 30 Lesions described herein are similar in gross and histologic morphology to those from a preliminary report in 2003. 28 To the authors’ knowledge, there are no other reports of lesions with semblance to those reported herein.
Footnotes
Acknowledgements
We are grateful to Jim Wellehan for preliminary PCR assays and to Bob Nordhausen for helpful advice regarding preparation of samples for electron microscopy. We also thank Roy Brown of Histology Consulting Service for slide preparation, Liz Post and Sue Hutton of Northwest ZooPath for slide and data retrieval, and Christie Buie of Northwest ZooPath for image layout. We would like to acknowledge Chelsea O’Toole and Cora Webber of California Science Center for helping to identify, track, and manage this condition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Northwest ZooPath.
