Abstract
Scleroderma is a skin disorder characterized by persistent fibrosis. Macrophage properties influencing cutaneous fibrogenesis remain to be fully elucidated. In this rat (F344 rats) model of scleroderma, at 1, 2, 3, and 4 weeks after initiation of daily subcutaneous injections of bleomycin (BLM; 100 μl of 1 mg/ml daily), skin samples were collected for histological and immunohistochemical evaluations. Immunohistochemically, the numbers of cells reacting to ED1 (anti-CD68; phagocytic activity) and ED2 (anti-CD163; inflammatory factor production) began to increase at week 1, peaked at week 2, and decreased thereafter. In contrast, the increased number of cells reacting to OX6 (anti–MHC class II molecules) was seen from week 2 and remained elevated until week 4. α–Smooth muscle actin–positive myofibroblasts were increased for 4 weeks. Double labeling revealed that galectin-3, a regulator of fibrogenic factor TGF-β1, was expressed in CD68+, CD163+, and MHC class II+ macrophages and myofibroblasts. mRNA expression of TGF-β1, as well as MCP-1 and CSF-1 (both macrophage function modulators), were significantly elevated at weeks 1 to 4. This study shows that the increased number of macrophages with heterogeneous immunophenotypes, which might be induced by MCP-1 and CSF-1, could participate in the sclerotic lesion formation, presumably through increased fibrogenic factors such as galectin-3 and TGF-β1; the data may provide useful information to understand the pathogenesis of the human scleroderma condition.
Scleroderma is regarded as a fibrotic lesion with excessive deposition of collagen and loss of skin apparatuses.2,33 The pathogenesis of scleroderma involves a complex interplay between vascular damage, inflammation, and fibrosis; however, the detailed mechanisms remain to be determined.2,25,44 It has been suggested that factors released from inflammatory cells in the early stages lead to the formation of myofibroblasts capable of producing collagens, resulting in fibrosis.15,17,21,27,45 In particular, macrophages are thought to act as important cells for the onset of scleroderma.13,22 Recruitment of macrophages into injured areas is mediated by chemokines such as monocyte chemoattractant protein–1 (MCP-1) and colony-stimulating factor–1 (CSF-1).1,4 Transforming growth factor–β1 (TGF-β1) is a major fibrogenic factor produced mainly by macrophages. 29 Galectin-3, a β-galactoside–binding animal lectin secreted by macrophages, has been considered a downstream regulator of TGF-β1 action. 11
Macrophage populations are heterogeneous.19,23,38 Generally, there are 3 subtypes of macrophages: exudative macrophages, resident macrophages, and antigen-presenting cells.16,28,40 In rat renal and hepatic lesions, these subtypes of macrophages can be identified immunohistochemically by an antibody (ED1, ED2, or OX6).45,46 ED1 (against CD68) labels exudative macrophages. 3 ED2 (against CD163) labels resident macrophages. 32 OX6 labels MHC class II molecules in antigen-presenting cells. 31 Besides these grouping, currently, macrophages appearing in lesions are classified as classically activated M1 (proinflammatory) and alternatively activated M2 (reparative) macrophages.23,38 Macrophage properties influencing scleroderma formation have not been fully clarified.
Bleomycin (BLM), an antibiotic used for cancer treatment in humans, has side effects including localized scleroderma at injected skin and lung fibrosis.18,25,30,42,43 Repeated injections of BLM into the subcutaneous tissue in experimental animals produce fibrotic lesions mimicking human scleroderma.6,18,25,42,43 In this study, we attempted to clarify possible macrophage properties in rat BLM-induced scleroderma through immunohistochemical methods.
Materials and Methods
Animals and Scleroderma
All the experimental protocols and animal housing conformed to the institutional guidelines of Osaka Prefecture University for animal care. Specific pathogen-free, 5-week-old male F344 rats were purchased from Charles River Japan (Hino, Shiga, Japan) and maintained at a controlled temperature of 21 ± 3°C and with a 12-hour light-dark cycle; they were provided a standard rat chow (MF; Oriental Yeast Co, Ltd, Tokyo, Japan) and tap water ad libitum. Bleomycin (5 mg/ml; Nippon Kayaku, Tokyo, Japan) was diluted to 1 mg/ml with sterile phosphate-buffered saline (PBS). Using a 27-gauge needle, 100 μl of BLM solution was injected subcutaneously into 2 sites on the shaved back of rats daily for 4 weeks. Skin samples from the BLM-treated rats were harvested at weeks 1, 2, 3, and 4. Controls were injected daily with an equivalent volume of sterile PBS and sacrificed at week 4. Five rats were examined at each examination week in the BLM-treated and control groups.
Histopathology and Immunohistochemistry
Animals were humanely euthanized, and skin samples from the injection sites were obtained and fresh frozen for RNA isolation and for cryosectioning or fixed in both 10% neutral buffered formalin and periodate-lysine-paraformaldehyde (PLP) solutions. The PLP-fixed samples were processed with the AMeX method (acetone–methyl benzoate–xylene) 39 and then embedded in paraffin. Fresh-frozen tissues were embedded in Tissue-Tek OCT compound (Sakura, Tokyo, Japan) and kept in –80°C. Formalin-fixed paraffin embedded tissue sections (5 μm in thickness) were routinely stained with hematoxylin-eosin (HE) for morphological evaluation and with the Azan-Mallory stain for collagens.
For immunohistochemical stainings, PLP-AMeX–processed or formalin-fixed tissue sections were used. PLP-AMex–processed tissue sections were used for antibodies to all macrophages and galectin-3. Immunohistochemical detection for myofibroblasts was performed using formalin-fixed tissue sections. Antigen retrieval was performed by heat pretreatment in 10 mM citrate buffer (pH 6.0) at 80°C for 6 hours. Sections were incubated overnight at 4°C with a primary antibody for macrophages. ED1 (Millipore, Bedford, Massachusetts; 1:500) labels CD68-expressing macrophages, 3 ED2 (AbD Serotec, Oxford, UK; 1:500) is used to identify CD163-expressing macrophages, 32 and OX6 (AbD Serotec; 1:500) recognizes MHC class II molecules in rats. 31 Myofibroblasts were identified using an antibody to α–smooth muscle actin (α-SMA) (clone IA4; DAKO, Carpinteria, California; 1:200). An antibody to galectin-3 (polyclonal; sc-20157; Santa Cruz Biotechnology, Santa Cruz, California; 1:500), which may be associated with the production of fibrogenic proteins by macrophages, was also applied. Then, slides were rinsed in PBS and treated with the horseradish peroxidase–conjugated polymer as secondary antibody (Histofine simple stain MAX PO; Nichirei, Tokyo, Japan) for 30 minutes at room temperature. After visualization of positive reactions using 3,3′-diaminobenzidine (DAB; Vector Laboratories, Burlingame, California), sections were counterstained lightly with hematoxylin. As negative controls, primary antibodies were replaced by nonimmunized mouse or rabbit IgG.
Double Immunohistochemical Labeling
Frozen sections (10 μm in thickness) were fixed in ice-cold acetone for 5 minutes, followed by blocking with 10% normal goat serum in PBS for 30 minutes. The slides were incubated overnight at 4°C with the anti–galectin-3. The sections were then incubated with anti-CD68, anti-CD163, anti–MHC class II, or anti–α-SMA antibody. After rinsing with PBS, sections were incubated with a secondary antibody of goat–anti-rabbit–Alexa 488 (Invitrogen, Carlsbad, California; 1:2000) or goat anti–mouse-Cy3 (Jackson Immunoresearch, West Grove, Pennsylvania; 1:2000) at room temperature for 30 minutes. All slides were mounted with the mounting medium, including 4′,6-diamino-2-phenylindole (DAPI; VECTASHIELD; Vector Laboratories). Images were taken and analyzed with a confocal laser scanning microscope (C1Si; Nikon, Tokyo, Japan).
To identify the macrophages coexpressing CD68/CD163, CD68/MHC class II, and CD163/MHC class II, we performed double immunohistochemistry according to the method described elsewhere. 10 First, the sections were immunolabeled with the anti-CD68 or anti–MHC class II antibody and then visualized with DAB (brown in color) as mentioned above. Second, sections used for CD163 were reacted with anti-CD68 or anti–MHC class II antibody, as well as sections for MHC class II with anti-CD68 antibody. The positive reactions at the second labeling were visualized red by the Fuchsin substrate-chromogen system (DAKO).
Terminal Deoxyribonucleotide Transferase (TdT)–Mediated Deoxyuridine Triphosphate Nick End Labeling (TUNEL) for Apoptotic Cells
A standard in situ terminal deoxyribonucleotide transferase (TdT)–mediated deoxyuridine triphosphate nick end labeling (TUNEL; ApopTag Peroxidase In Situ Apoptosis Detection Kit; Millipore) method was used for detection of DNA fragmentation for apoptotic cells according to the manufacturer’s instructions. Negative control sections were incubated with distilled water instead of TdT enzyme, to confirm the specificity.
Quantitative Real-Time Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from control skin and BLM-injected skin samples using an SV Total RNA isolation system (Promega, Madison, Wisconsin), and the final concentration of RNA was adjusted to 0.5 μg/μl. Briefly, first-strand cDNA was synthesized by using the SuperScript first-strand synthesis system (Invitrogen). Quantitative PCR was performed with a SYBR green real-time PCR master mix (Toyobo, Osaka, Japan) by a LineGene system detector (BioFlux, Tokyo, Japan). The primers were as follows: for TGF-β1, 5′-TGTCTACTGAACTTCGGGGTG-3′ (forward) and 5′-GAGGCTGACTTTCTCCTGGTA -3′ (reverse); for CSF-1, 5′-ACAGGTGGAACTGCCGCCAGTGTAGAA-3′ (forward) and 5′-GGTGGACGTTGCCATAATGTCTC-3′ (reverse); for MCP-1, 5′-CTATGCAGGTCTCTGTCACGCTTC-3′ (forward) and 5′-CAGCCGACTCATTGGGATCA-3′ (reverse); and for β-actin, 5′-TAAAGACCTCTATGCCAACAC-3′ (forward) and 5′-CTCCTGCTTGCTGATCCACAT-3′ (reverse). Data were calculated using the comparative Ct method. 37 Relative quantitation of mRNA level was normalized by that of β-actin as an internal control gene.
Cell Count and Statistical Evaluation
Distinct immunopositive cells for anti-CD68, anti-CD163, anti–MHC class II, anti–α-SMA, or anti–galectin-3 antibody were quantified in 5 randomly selected areas (each 0.2 mm2). The percentage of double-labeled macrophages (CD68+/CD163+, CD68+/MHC class II+, and CD163+/MHC class II+ per 0.2 mm2) was also calculated.
Significant testing was assessed by the Student’s t-test or Tukey’s multiple comparison test. Spearman’s correlation coefficient test was used to determine the correlation between galectin-3+ cell number and CD68+, CD163+, or MHC class II+ cell number. A value of P < .05 was considered significant.
Results
Histologically, no noticeable lesions were observed in PBS-treated control rats (Figs. 1 and 3). In BLM-treated rats at weeks 1 and 2, cell infiltrate began to be seen in the lower part of dermis, with atrophy of hair follicles (Fig. 2); in the atrophying hair follicles, apoptotic cells were detected by the TUNEL method. The Azan-Mallory staining showed that subcutaneous fat tissue in normal skin (Fig. 3) was replaced by collagens (Fig. 4). At weeks 3 and 4, the increased collagen deposition was characterized by hyalinization, particularly around the atrophying hair follicles (Fig. 4, inset), with notable thickening of the dermis. The skin lesion at BLM injection sites was diagnosed as dermal sclerosis (scleroderma).
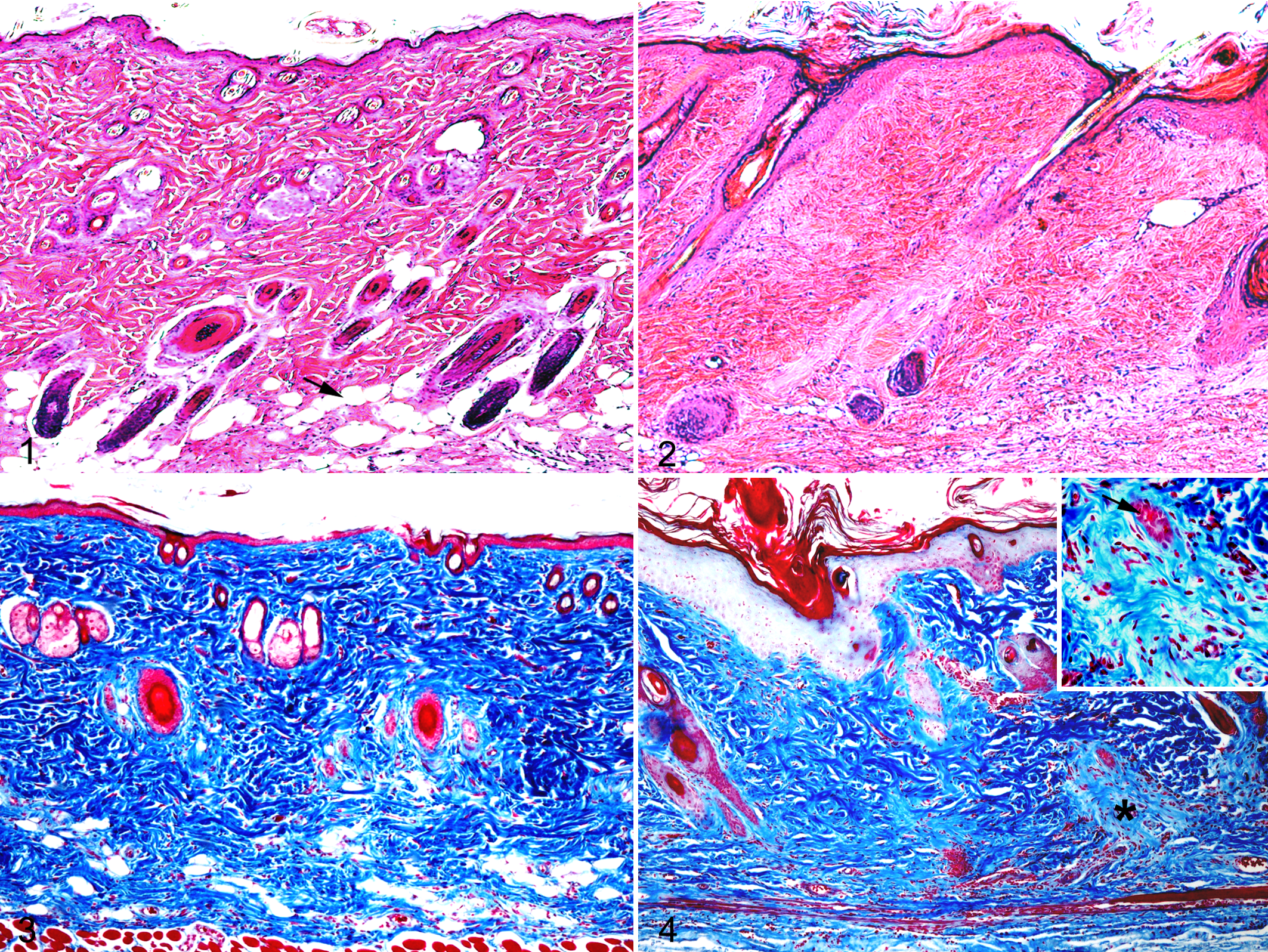
Macrophage Immunophenotypes
In the dermis of control rats, there were a small number of CD68+, CD163+, and MHC class II+ cells.
Following subcutaneous BLM injection, a significant increase of CD68+ cells was observed in the dermis at weeks 1 to 4; the number peaked at week 2 and thereafter tended to decrease up to week 4 (Fig. 5). Similar to the kinetics of CD68+ cells, the CD163+ cell number at weeks 1 to 4 showed a consistently significant (P < .05) increase with the highest number at week 2 (Fig. 6). Although the MHC class II+ cell number was similar to that in controls at week 1, the number began to significantly (P < .05) increase at week 2 and thereafter remained consistently increased up to week 4 (Fig. 7). When compared with CD68+, CD163+, and MHC class II+ cell numbers, the CD68+ and CD163+ cell numbers were greater than the MHC class II+ cell number at week 1; at weeks 2, 3 and 4, the MHC class II cell number showed an equivalent density to the CD68+ and CD163+ cell number.
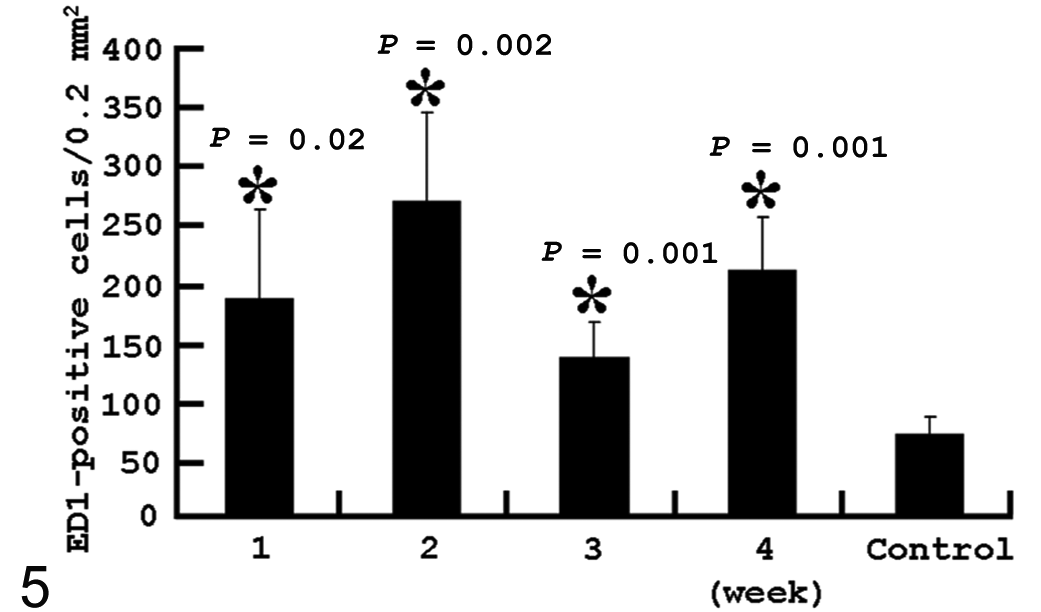
Kinetics of CD68+ (ED1) macrophages in bleomycin-treated skin. *Significantly different (P < .05) from control. Error bars indicate standard deviation (SD).
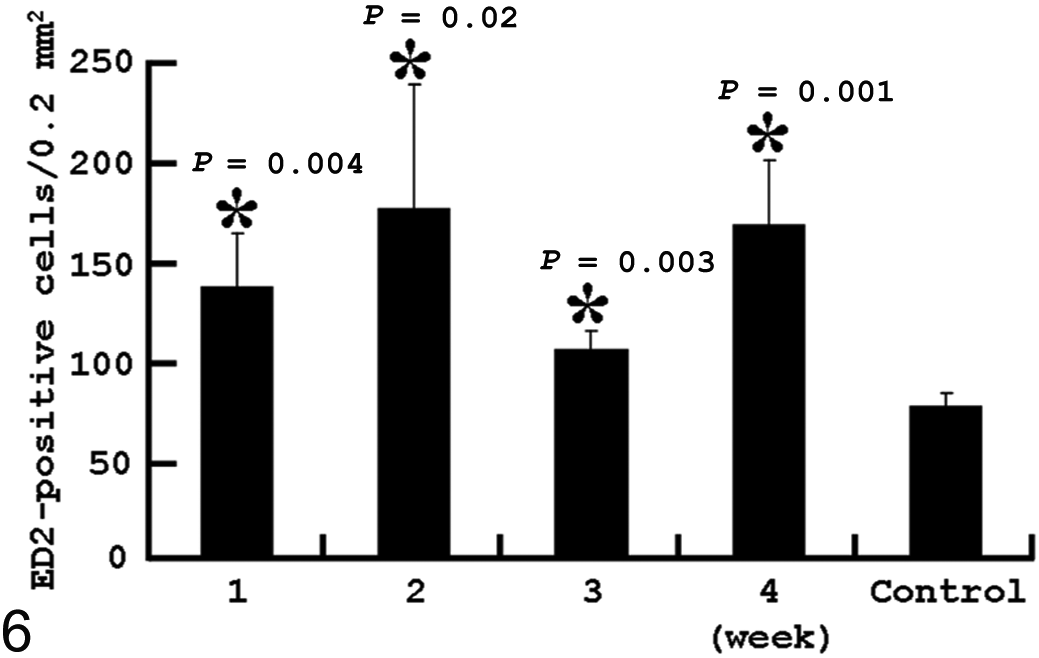
Kinetics of macrophages reacting to CD163 (ED2) in bleomycin-treated skin. *Significantly different (P < .05) from control. Error bars indicate SD.
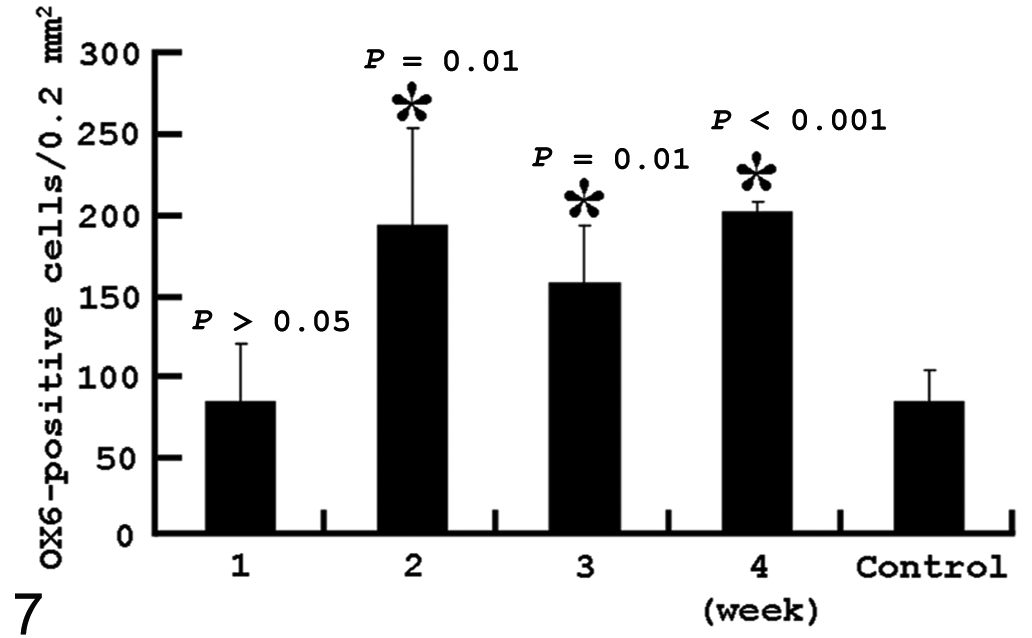
Kinetics of macrophages labeled with MHC class II antibody (OX6) in bleomycin-treated skin. *Significantly different (P < .05) from control. Error bars indicate SD.
In BLM-injected skin, CD68+, CD163+, and MHC class II+ cells were diffusely observed in the dermis, particularly in the lower dermis with progressing fibrosis. CD68+ cells were round, oval, or elongated, with distinct granular reactions in the cytoplasm (Fig. 8). CD163+ cells exhibited spindle-shaped or round configuration (Fig. 9). Besides round and elongated cells, the MHC class II+ cells had a dendriform shape with projecting cytoplasmic processes (Fig. 10).
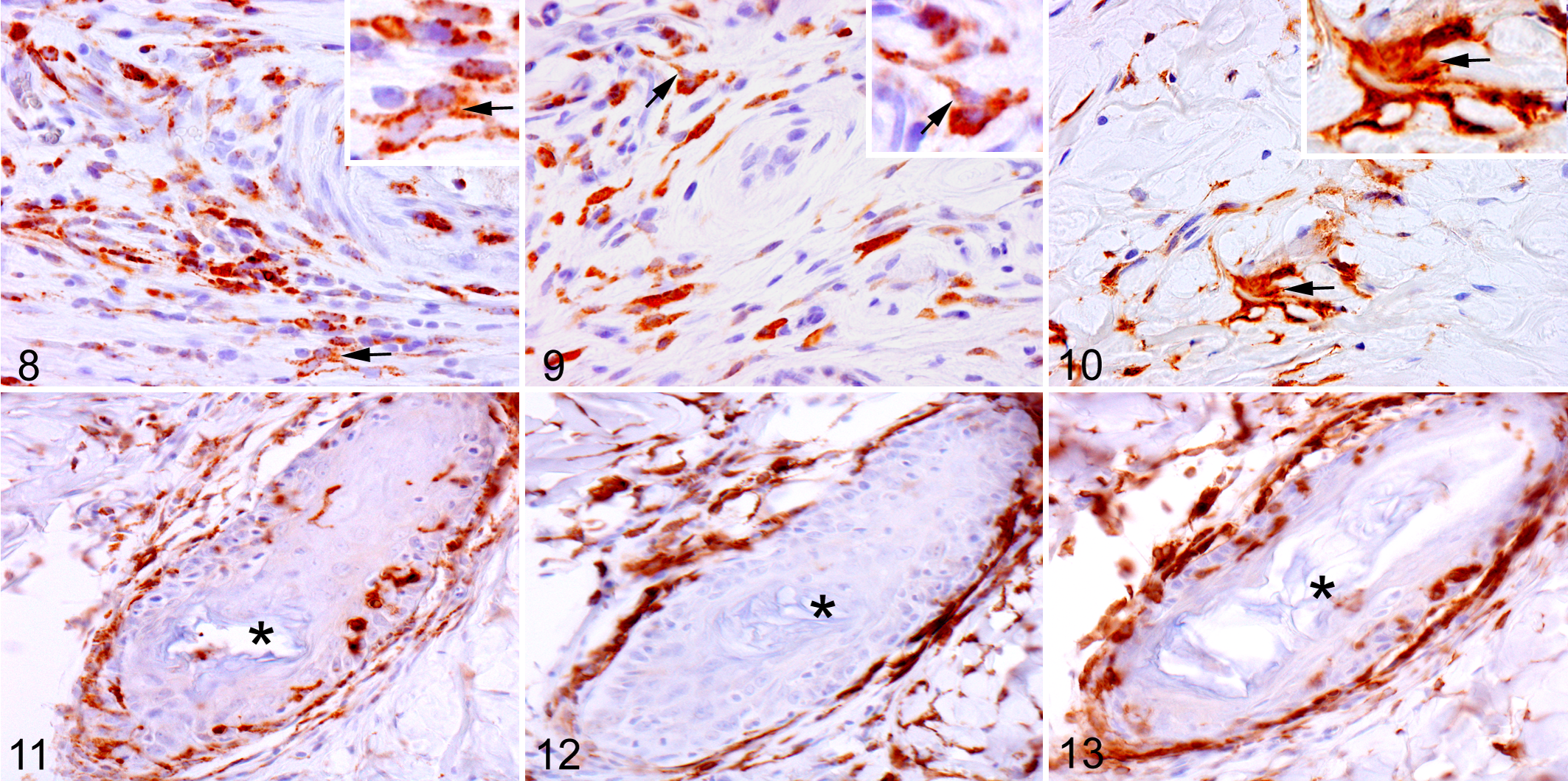
The double labeling between CD68+, CD163+, and MHC class II+ cells in BLM-treated skin revealed that there were cells reacting to both CD68 and CD163 or to both CD163 and MHC class II, ranging from 9% to 14% at weeks 2 and 4 (Table 1). Cells reacting to both CD68 and MHC class II (28% and 30% at weeks 2 and 4, respectively) showed a greater number than CD68+/CD163+ or CD163+/MHC class II+ cells (Table 1).
Percentage of Double-Labeled Cells in Skin Treated With Bleomycin.
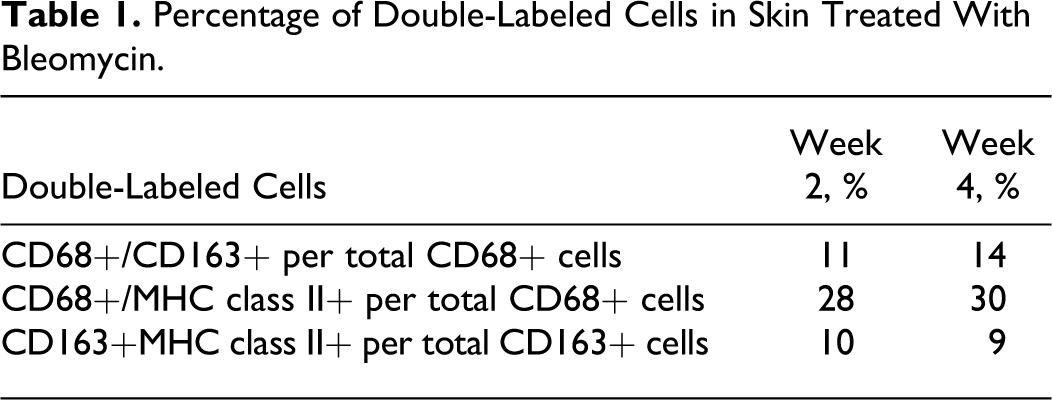
In the perifollicular area of controls, there were a few CD68+, CD163+, and MHC class II+ cells. In BLM-injected skin, a large number of CD68+, CD163+, and MHC class II+ cells appeared in the perifollicular area with progressing fibrosis (Figs. 11, 12, and 13, respectively). Interestingly, CD68+ and MHC class II+ cells were present within hair follicles as infiltrating cells (Figs. 11 and 13, respectively), whereas no CD163+ cells were seen within hair follicles. CD163+ cells were limited to perifollicular areas (Fig. 12).
α-SMA+ Myofibroblasts
In skin with fibrotic lesions, spindle-shaped cells reacting to α-SMA are regarded as myofibroblasts. 33 In control skin, no myofibroblasts were found in the dermis, although vascular smooth muscles and arrector pilli muscles reacted to α-SMA. In BLM-injected skin, α-SMA+ myofibroblasts began to be seen in the dermis as early as week 1, and the number gradually increased at weeks 2, 3, and 4 with advancing fibrosis (Fig. 14). Myofibroblasts were spindle shaped and appeared exclusively in the lower dermis within the fibrotic lesion (Fig. 15).
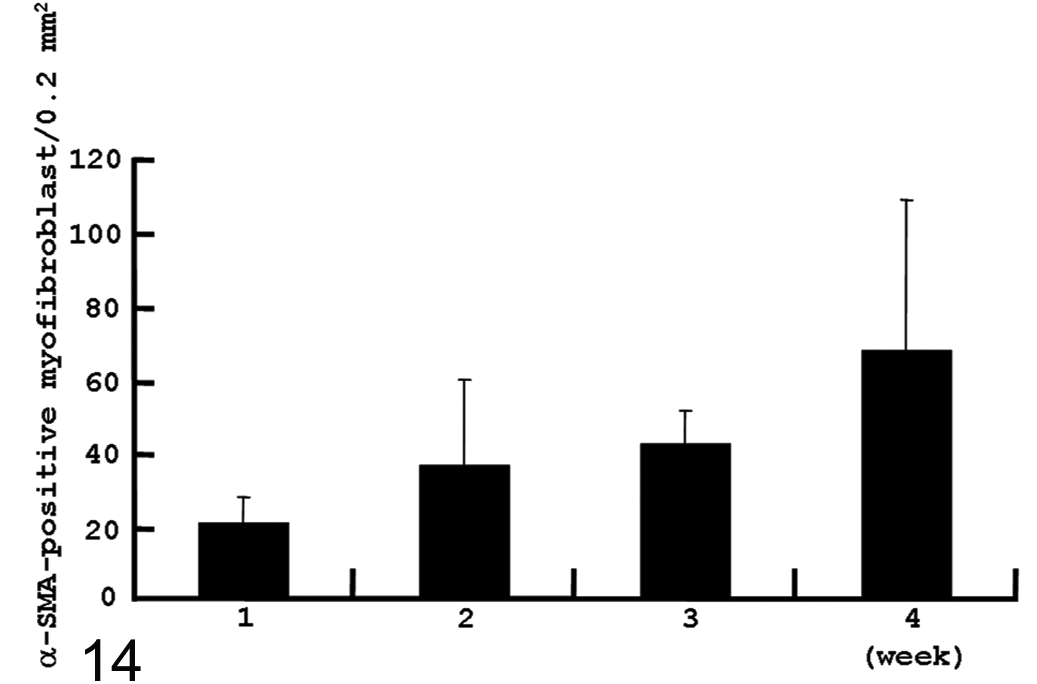
Kinetics of myofibroblasts reacting to α–smooth muscle actin (α-SMA) in bleomycin-treated skin from weeks 1 to 4. Because no α-SMA+ myofibroblasts were found in control skin, statistical evaluation was not performed. Error bars indicate SD.
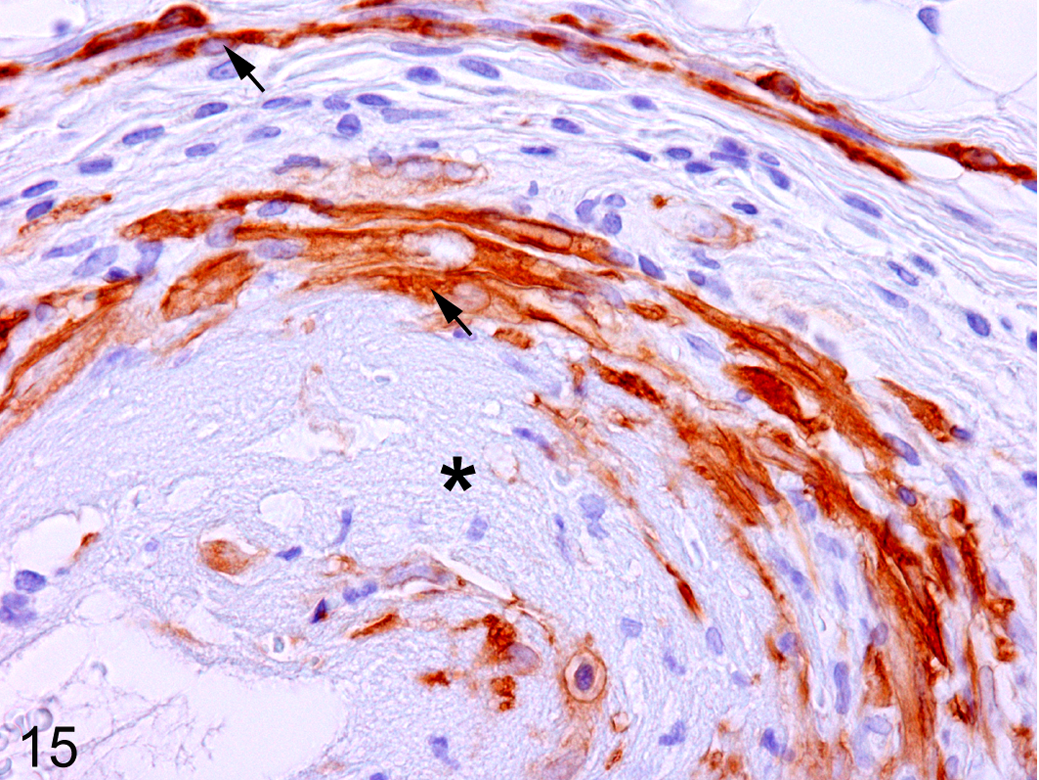
Skin; bleomycin-treated rat at week 2. Spindle-shaped α–smooth muscle actin (SMA)–positive myofibroblasts (arrows) are present in the sclerotic lesion (*, hyalinization area). Immunohistochemistry, counterstained with hematoxylin.
Galectin-3 Immunoexpression
In BLM-injected skin, the galectin-3–expressing cell number began to be significantly increased as early as week 1, and the significant elevation was maintained up to week 4 (Fig. 16). Cells positive for galectin-3 were round, oval, or spindle in shape (see Suppl. Fig. S1 online at http://vet.sagepub.com/supplemental).
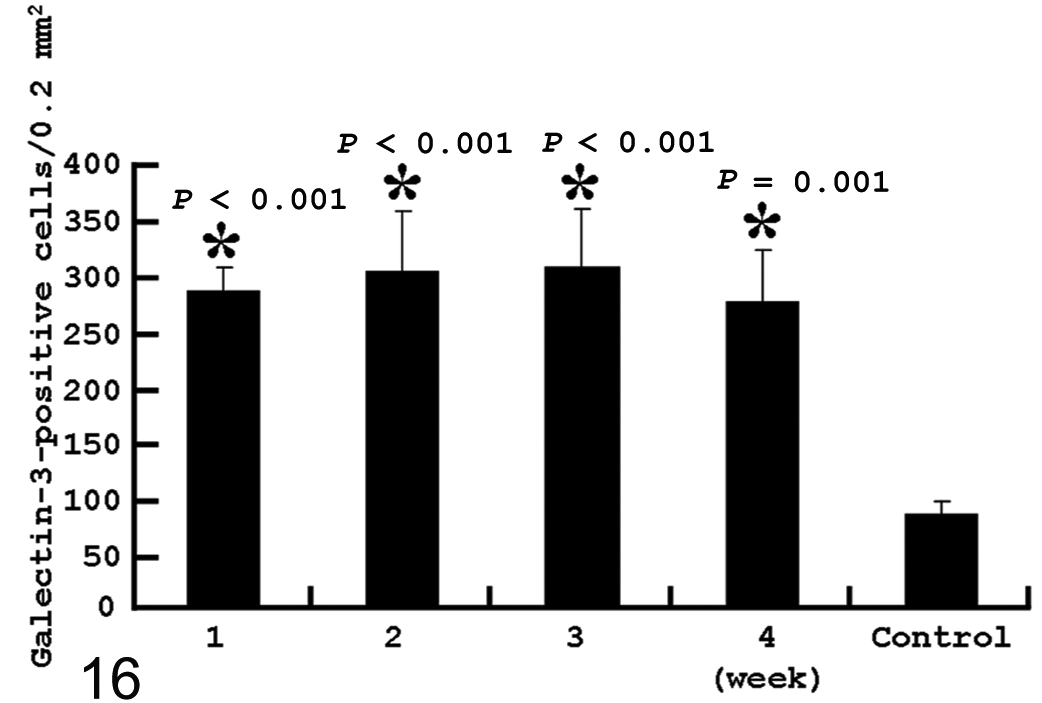
Kinetics of galectin-3+ cells in control and bleomycin-treated rats. *Significantly different (P < .05) from control. Error bars indicate SD.
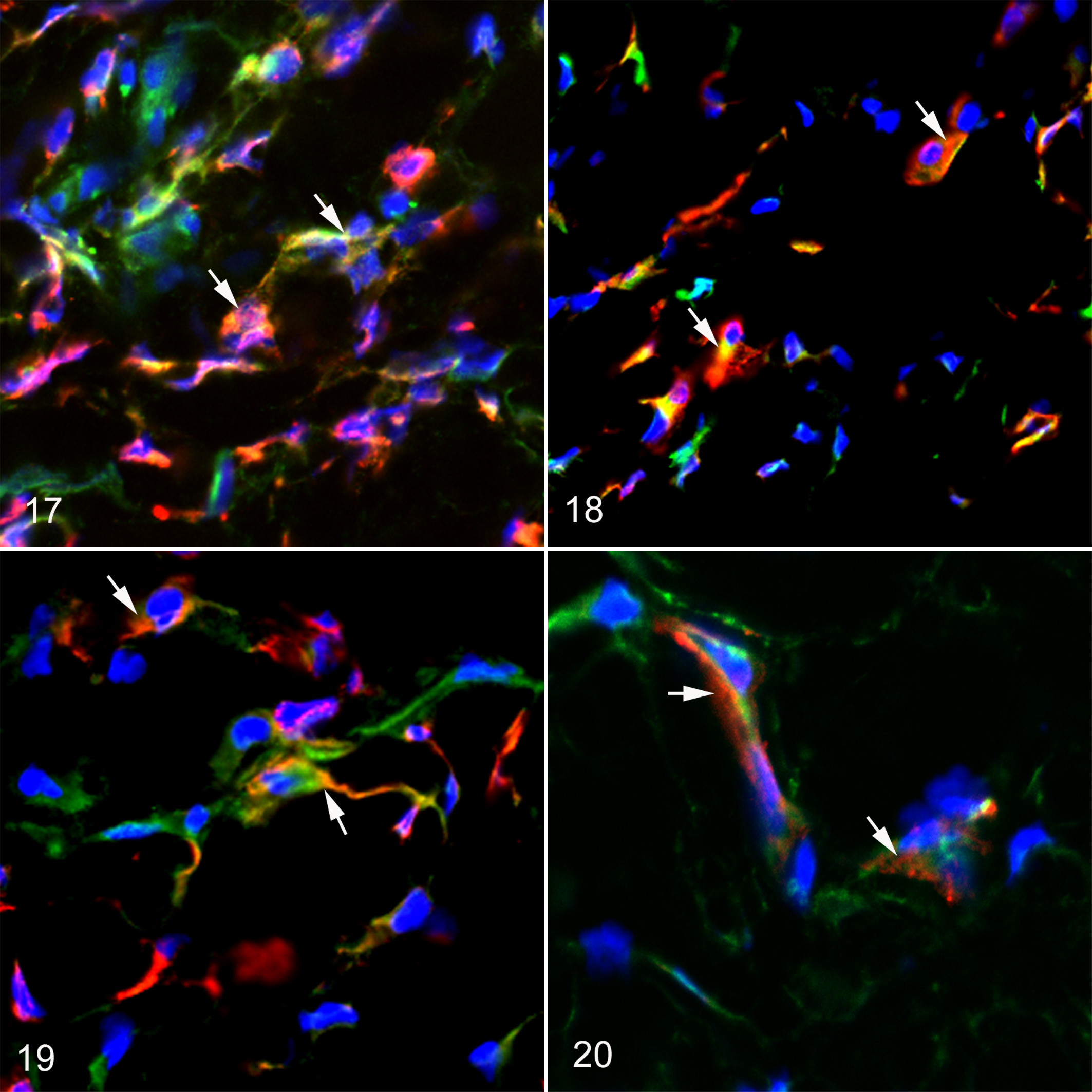
To identify galectin-3+ cells, we performed the double immunofluorescence labelings. Galectin-3+ cells also reacted to CD68 (Fig. 17), CD163 (Fig. 18), MHC class II+ (Fig. 19), or α-SMA (Fig. 20). To further investigate the relationship between the appearance of galectin-3–expressing cells and macrophages or myofibroblasts, we conducted correlation analyses. The analyses showed that there was a significant positive correlation (P < .05) between the galectin-3+ cell number and the CD68+ (r = 0.5786, P = .03), CD163+ (r = 0.6536, P = .01), or MHC class II+ (r = 0.6286, P = .02) cell number. In addition, the galectin-3+ cell number correlated positively with the α-SMA+ myofibroblast number (r = 0.7714, P = .004).
mRNA Expressions of TGF-β1, MCP-1, and CSF-1
Compared with that in controls, TGF-β1 mRNA expression was significantly increased at weeks 1 to 4 after BLM injection, being 3- to 9-fold greater. Expression level of MCP-1 mRNA was 12- to 28-fold greater in rats receiving BLM at weeks 1 to 4, with a significant increase. Although CSF-1 mRNA level did not show significant change at week 3, that at weeks 1, 2, and 4 was about 2.5-fold higher, showing a significant (P < .05) change.
Discussion
In the present study, the development of sclerotic lesions following BLM injection was evident particularly in the lower dermis, of which findings correlated with those of sclerotic lesions in human cases.30,33,42 Thus, the present model represents a good tool to study scleroderma pathogenesis.6,18,25,42,43
The present study showed that macrophages exhibiting different immunophenotypes appear in fibrotic lesions following BLM injection. These macrophages were identified with anti-CD68, anti-CD163, or anti–MHC class II antibody. Rat anti-CD68 is used to identify exudative macrophages in pathological settings17,45,46; the antigen is present mainly on the membrane of phagolysosomes, and the degree of CD68 expression reflects phagocytic activity of macrophages. 3 In BLM-injected skin, CD68+ macrophages began to rise at week 1 and retained a significantly increased number until week 4. At week 1, CD68+ macrophages were the most predominant among macrophage populations, indicating their involvement in early inflammation as a phagocyte for cell debris. The beginning of an increase in CD68+ cell number at week 1 correlated with an increased number of myofibroblasts and TGF-β1 mRNA expression. It has been reported that macrophages are capable of producing large amounts of TGF-β1 at wound sites and fibrotic areas.8,45,46 It is well known that myofibroblasts, which are contractile cells, are induced by TGF-β1.15,17,21 These findings indicated that CD68+ cells might be responsible for myofibroblast induction through increased production of TGF-β1. 42 Consequently, the α-SMA+ myofibroblasts were gradually increased throughout the observation period in BLM-injected skin. Myofibroblasts are capable of producing collagens, resulting in fibrosis.15,17,21,45 Similar to human cases, α-SMA+ myofibroblasts were found in sclerotic dermis.15,30,33
In addition to exudative macrophages, macrophages reacting to CD163 were increased in BLM-injected skin, showing similar kinetics to that of CD68+ cells. The percentage of double-immunopositive cells to both CD68 and CD163 was small (11% to 14%), indicating that CD163+ macrophages differed greatly from CD68+ macrophages. Rat CD163 molecule has been used to label with resident macrophages such as Kupffer cells and histiocytes.16,29,40,41 The increased expression of CD163 is associated with the enhanced production of inflammatory factors (such as tumor necrosis factor [TNF]–α and interleukin [IL]–6) and phagocytosis.32,35,45 CD163+ macrophages have been seen in chemically induced renal and hepatic lesions as major macrophages.45,46 Like CD68+ cells, CD163+ macrophages might contribute to the development of myofibroblasts through the release of fibrogenic factors such as TGF-β1, although the source of TGF-β1 should be investigated more. Similarly, CD68+ and CD163+ macrophages have been found in human scleroderma. 13 These macrophages might be associated with the production of fibrogenic mediators such as TGF-β1.13,14 TGF-β1 might be produced by CD68+ and CD163+ cells, but this should be investigated in further studies. Definite roles of these macrophages in human scleroderma remained to be established. 13
Anti–MHC class II antibody labels with antigen-presenting cells such as dendritic cells and activated macrophages in normal skin and pathological lesions.5,20,31 In the present study, the MHC class II+ cell number showed a significant increase at weeks 2 to 4, of which the appearance was later than CD68+ and CD163+ cells. Interestingly, in the double labeling, the respective percentages of cells reacting to both CD68 and MHC class II or to both CD163 and MHC class II were 28% to 30% or 9% to 10%; these findings suggested that MHC class II+ cells were different from CD68+ or CD163+ cells, although some MHC class II+ cells might be generated from CD68+ and CD163+ cells. 27 In BLM-induced mouse skin fibrosis, CD4+ T lymphocyte activity was reported. 18 The activation of CD4+ cells might be mediated through antigen presentation in MHC class II+ cells.36,41 MHC class II+ cells have been suggested to modulate cytokine production from macrophages and collagen depositions by myofibroblasts. 36 Collectively, MHC class II+ cells seen in the present study might be responsible for the development of scleroderma, particularly at later stages.
Based on the activation pathway and phenotypes, macrophages in lesions have recently been divided into M1 (proinflammatory) (classically activated by Th1 lymphocyte–related responses) and M2 (reparative) (alternatively activated by Th2 lymphocyte–driven responses) types.13,14,23,38 The former are attributable to phagocytosis of cell debris in early inflammation, and the latter are responsible for tissue repair.23,24,38 It was difficult to determine the relation of M1 and M2 macrophages with macrophage phenotypes examined in this study because the appearance of CD68+, CD163+, and MHC class II+ macrophages almost overlapped each other; M1 and M2 macrophage concepts are mainly based on in vitro studies and have been controversial.24,38 Although it has been reported that CD68 expression is associated with the M1 phenotype, and CD163 is highly expressed in M2 macrophages, more detailed studies are needed to know the functions of macrophages with different immunophenotypes.13,14,23
Bleomycin injection has been reported to induce apoptosis of hair follicles, resulting in alopecia.43,44 In this study, apoptotic bodies were often seen in the hair follicles. Follicular cell apoptosis may further promote the recruitment and phagocytic activity of macrophages. 26 The increased number of CD68+, CD163+, and MHC class II+ cells in the perifollicular area might be related to ingestion and removal of apoptotic cells. Interestingly, CD163+ macrophages were not detected within hair follicles, although the CD68+ and MHC class II+ cells were seen; there might be differences in macrophage properties. 32 Because the greater sclerotic lesions (hyalinization) were seen exclusively around the hair follicles undergoing apoptosis, it would be interesting to pursue the relationship between degenerating hair follicles and cutaneous fibrosis or macrophage functions.
It is well known that MCP-1 and CSF-1 are chemoattractants for macrophage recruitment.1,4,7 MCP-1 and CSF-1 mRNAs were elevated in rats treated with BLM, being paralleled by the increased numbers of macrophages and TGF-β1 mRNA. The upregulation of TGF-β1 gene expression can be induced in cultured macrophages under MCP-1 or CSF-1 treatment.1,9,27 It was also reported that MCP-1 addition to a rat macrophage cell line enhanced expression of CD68 and CD163 molecules. 27 Moreover, the blockade of MCP-1 functions in BLM-treated mouse skin resulted in reduced macrophage recruitment and decreased fibrosis. 7 Macrophage chemoattractants such as MCP-1 and CSF-1 might be very important factors to recruit and retain macrophages for maintaining the active fibrogenesis.
Galectin-3 is a kind of animal lectin that may be expressed and secreted by activated macrophages.11,12,34 Galectin-3 expression is related to increased fibrosis in liver and kidneys.11,12 This study showed that the galectin-3+ cell number was significantly increased in BLM-injected skin. The double labeling revealed that galectin-3 was expressed in CD68+, CD163+, and MHC class II+ cells with positive correlations, indicating that macrophages were the major source of galectin-3 in sclerotic skin. In addition, α-SMA+ myofibroblasts expressed galectin-3 in the double labeling. Since the increased galectin-3 expression is a prerequisite for TGF-β1–mediated myofibroblast activation and collagen production, 11 the expression of galectin-3 by macrophages and myofibroblasts themselves is likely to be important for collagen deposition by myofibroblasts, leading to progression of scleroderma. Galectin-3 expression patterns in sclerotic lesions have not been reported previously.
In conclusion, the present study shows that macrophages exhibiting heterogeneous immunophenotypes participate in the BLM-induced rat scleroderma. The macrophages also appeared in and around the hair follicles undergoing apoptosis. It is considered that upregulated MCP-1 and CSF-1 levels could be related to recruitment of macrophages in an injured area and might influence macrophage properties such as phagocytosis, antigen presentation, and production of fibrogenic factors (particularly TGF-β1 and galectin-3); these factors might be responsible for induction of myofibroblasts capable of releasing collagens. The expressions of these macrophage properties should depend on microenvironmental conditions induced by cell-to-cell and cell-to-matrix interactions.8,17,23,28,38,40 More detailed studies including M1 and M2 macrophage dichotomy are necessary to clarify functions of divergent macrophages and factors affecting the functions in scleroderma.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Grant-in-Aids for Scientific Research (B) (Nos. 18380188 and 22380173 to J. Yamate), Japanese Society for the Promotion of Science.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
