Abstract
Experimental autoimmune encephalomyelitis (EAE) is a reliable model to study the pathogenesis of Multiple sclerosis (MS), which is a progressive autoimmune-mediated inflammation of the central nervous system (CNS). Tim-3 is one of the crucial immune checkpoints in immune tolerance. We investigated the impact of Tim3 in EAE by the anti-Tim3 antibody and detected the immune cell and inflammation through flow cytometry and ELISA. In this study we found that CD4 T cells express low levels of Tim-3 in EAE mice. Tim-3 suppression exacerbated the disease progression in EAE mice. Furthermore, the Galectin-9/Tim-3 pathway promoted the apoptosis of CD4 T cells and inhibited the differentiation of Th17 in EAE mice. Our study unravels the anti-inflammatory Galectin-9/Tim-3 pathway in EAE mice and provides a potential therapeutic target for EAE and MS treatment.
Introduction
Multiple sclerosis (MS) is an autoimmune-mediated central nervous system (CNS) inflammation affecting 2.3 million people worldwide. 1 MOG35-55 induced experimental autoimmune encephalomyelitis (EAE) is an animal model to study the pathogenesis of MS, which shares many features with MS. 2 Auto-reactive T cell is the central effector cell in the MOG35-55 induced EAE. 3 Most of the researches on MS and EAE have focused on the role of effector CD4 T cells, and accumulating data suggests that IL17A secreting T cells may play a critical role in the MS and EAE. 4
In recent years, the roles of immune checkpoint pathways have been uncovered in the control and eradication of infections and tumors.5,6 Antibodies against cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) and programmed cell death-ligand 1(PD-L1) were applied in immunotherapies that achieved the US Food and Drug Administration approval by mediating the inhibition of T cells.7,8 With the uncertain effectiveness in some patients, the overall effectiveness of immune checkpoint therapy remains unsatisfactory. Recent studies have identified several novel immune checkpoint molecules, including lymphocyte activation gene-3 (Lag-3), T cell immunoglobulin and mucin-domain containing-3 (Tim-3), T cell immunoglobulin and ITIM domain (TIGIT), V-domain Ig suppressor of T cell activation (VISTA). 9 Tim-3 is a type I transmembrane protein that marks IFN-γ-producing Th1 and induces autoimmune diseases. 10 Meanwhile, we are still in the early stages of understanding Tim-3. There are still some mechanical puzzles to be solved, like how Tim-3 and its ligand, Galectin-9, regulate autoimmune response, which may be a key to therapeutic potential. 11
Th17 cell is a subset of CD4 T cells secreting interleukin-17 (IL17), which induce several immune-related diseases, including inflammatory bowel disease, systemic lupus erythematosus, rheumatoid arthritis, and multiple sclerosis.12–14 Th17 cells mediate myelin-specific inflammation in the pathogenesis of the mouse model of multiple sclerosis, EAE. 15 In wild type mice, immunization with myelin antigen MOG35-55 promotes proliferation and polarization of Th17 cells. 16 Moreover, Th17 cells could be polarized from naïve CD4 T cells under non-pathogenic polarization conditions. 17 Thus, we were able to investigate the molecular mechanism of the Galectin-9/Tim-3 pathway in Th17 cells in vitro.
In this study, we evaluated the low expression of Tim-3 in mouse EAE models. Then, our data showed that the anti-Tim-3 treatment exacerbates the clinical severity of EAE. Finally, flow cytometry results indicated that the Galectin-9/Tim-3 pathway promoted the apoptosis of CD4 T cells and inhibited the differentiation of Th17 in vitro.
Material and method
EAE induction
Seven to eight-week-old C57/BL6 mice were housed under pathogen-free conditions and maintained in compliance with the guidelines of the Department of Laboratory Animals. EAE was induced in C57/BL6 mice using MOG35–55 (R&D Systems) emulsified in Complete Freund’s Adjuvant (InvivoGen). Mice were injected subcutaneously at two side using MOG35-55. Three hundred microgram/mouse anti-mouse Tim-3 treatment was initiated during five consecutive days from day 8 post-immunization.
To evaluate the success of the EAE model, we detected the golden standard, 0–5 clinical level of the EAE mice, which show the success and treatment effect of EAE mice after MOG35-55 infusion.18,19 Mice were monitored every 2 days as follow: 0, no clinical signs; 1, limp tail; 2, limp tail and weakness of hind legs; 3, limp tail and complete paralysis of hind legs; 4, limp tail, complete hind leg, and partial front leg paralysis; 5, moribund and dead. All animal experiments were approved by the Institutional Animal Care and Use Committee of the Jilin University. To decrease the variation in the group, we applied 4–9 mice/group in our experiments.
Isolation of spinal cord lymphocytes and splenocytes
Spinal cords and spleens were isolated from EAE mice on day 28 after immunization. A single-cell suspension was achieved by pressing the spleen or spinal cord with 1 mL syringe piston and wash the tissue through a 70 µm nylon cell strainer (BD Biosciences). The single-cell suspension resuspended in red blood cell (RBC) lysis buffer to acquire splenocytes. Splenocytes were cultured in RPMI 1640 medium (Corning) supplemented with 10% fetal bovine serum (Gibco), penicillin, and streptomycin (Corning).
Enzyme-linked immunosorbent assay (ELISA)
To identify the concentration of Galectin-9 in mouse serum, we proceeded the ELISA with mouse Galectin-9 Quantikine ELISA kit (Cusabio) according to manufactures’ description. We incubate isolated splenocytes of EAE mice, 10 μg/mL MOG35-55 and 10 μg/mL anti-Tim-3 antibody or 1 μg/mL Galectin-9 for 7 days. The supernatant was collected for ELISA assay. The concentration of IL17A was detected by the Ready-Set-Go ELISA kit (Thermo) according to the manufacturer’s protocol.
Cell surface protein and IL17A detection
For cell surface protein detection, 1 × 106 suspended cells were incubated with the antibody for 30 min. Anti-mouse CD 3 (17A2), CD8 (53–6.7), CD4 (GK1.5), Tim-3 (B8.2C12) antibodies were purchased from BD Bioscience and Biolegend company. For intracellular IL17A detection, cells were permeabilized with Fixation Buffer and intracellular staining Perm Wash Buffer and stained with anti-mouse IL17A (TC11-18H10.1) antibody according to the manufacturer’s protocol. Cells were washed with PBS buffer and detected with FACS Fortessa (BD Bioscience).
Cell apoptosis assay
Cell apoptosis assay was evaluated by flow cytometry. Splenocytes were stained with flow cytometry antibodies as previously described. Splenocytes were resuspended in Binding Buffer (BD Bioscience) and incubated with PE—Annexin V and 7-AAD (Biolegend, San Diego, USA) for 15 min at room temperature. The cells were then analyzed by flow cytometer FACS Fortessa (BD) after incubation without washing.
Polarization of Th17 cell in vitro
Mouse CD4+CD25− T cells were acquired by flow cytometry as described above. To achieve the Th17 cells, naïve CD4+ CD25− T cells were stimulated with anti-CD3 (3 μg/mL), anti-CD28 (3 μg/mL) antibody, TGF-β (5 ng/mL), and IL-6 (20 ng/mL) in RPMI 1640 (Corning) containing 10% fetal bovine serum (Gibco), penicillin, and streptomycin (Corning). Anti-IFN-γ (10 μg/mL) and anti-IL4 (10 μg/mL) antibodies were added to inhibit unwanted deviation to Th1 or Th2 subsets. To investigate the effect of the Galectin-9/Tim-3 pathway, 10 μg/mL anti-Tim-3 antibody or 1 μg/mL Galectin-9 were added to block or stimulate the pathway for 7 days.
Statistical analysis
Levels of significance for comparison between two groups were determined by the Student
Results
Tim-3 and Galectin-9 expression decreased in EAE mice
To analyze the influence of Tim-3 on EAE pathology, we investigated the Tim-3 expression on the CD4 and CD8 T cells in EAE mice (Figure 1(a) and (b)). Flow cytometry showed that the proportion of Tim-3+ CD4 T cells but not Tim-3+ CD8 T cells was significantly decreased in EAE mice. Furthermore, we investigated the Galectin-9 concentrations in serum of EAE mice. The concentration of serum Galectin-9 was also remarkably decreased in EAE mice (Figure 1(c)). These data showed the significant changes of Galectin-9 and Tim-3 in EAE mice, which might play an important role in the pathology of EAE.

Tim-3 and Galectin-9 expressions in EAE mice: (a) Tim-3 expression in splenic CD4 T cells of EAE mice was detected by flow cytometry, (b) Tim-3 expression in splenic CD8 T cells of EAE mice was detected by flow cytometry, and (c) the serum Galectin-9 concentration of EAE mice was detected by ELISA. Data are presented as the mean ± SEM; n = 4 mice/group. Levels of significance for comparison between two groups were determined by the Student
Tim-3 pathway blocking accelerated the clinical symptoms in EAE mice
The Tim-3 pathway plays a crucial role in T cell activation in vivo. We injected 200 µg/mL anti-mouse Tim-3 antibody into EAE mice intravenously every 3 days from day 8 after MOG vaccination. Over the 20 days after immunization, clinical assessment of EAE demonstrated that anti-mouse Tim-3 treatment had increased mean disease severity scores compared with the control group (Figure 2(a)). This result suggests that the Tim-3 pathway may inhibit the disease progression in EAE mice. To investigate the effect of Tim-3 blocking, we detected the CD4 T cell differentiation of spinal lymphocytes on day 20. The proportion of CD4 T cells increased remarkably after Tim-3 blocking (Figure 2(b)). Similarly, the IL17A secreting of CD4 T cells increased significantly in Tim-3 blocking mice (Figure 2(c)).

Anti-Tim-3 antibody treatment accelerated the EAE in vivo: (a) mean clinical scores ± SEM of isotype antibody or anti-Tim-3 treated mice immunized with myelin peptide MOG35-55 and CFA on day 20. Mononuclear cells were enriched from the spinal cords and counted, and the proportions of (b) CD4 T and (c) IL17 secreting CD4 T cells of EAE mouse spinal cord were detected with flow cytometry. Data are presented as the mean ± SEM; n = 8–9 mice/group. Levels of significance for comparison between two groups were determined by the Student
Galectin-9/Tim-3 pathway selectively induced CD4 T cell apoptosis
To unveil the impact of the Galectin-9/Tim-3 pathway in lymphocytes of EAE mice. We isolated splenic cells from EAE mice sacrificed on day 28 after immunization. Anti-Tim-3 antibody (4 μg/mL), Galectin-9 (1 μg/mL) or related isotype control antibody were added to the supernatant for 3 days. Apoptotic splenic lymphocytes were labeled with 7-AAD and Annexin V and detected by flow cytometry (Figure 3). Data showed that the proportion of apoptotic splenic lymphocytes and CD4+ T cells decreased after Tim-3 blocking (Figure 3(a) and (b)). Furthermore, with the stimulation of Galectin-9, the proportion of apoptotic lymphocytes and CD4+ T cells accelerated significantly (Figure 3(a) and (b)). While we did not observe significant effects of Tim-3 blocking or Galectin-9 stimulation in CD8+ T cells (Figure 3(c)).
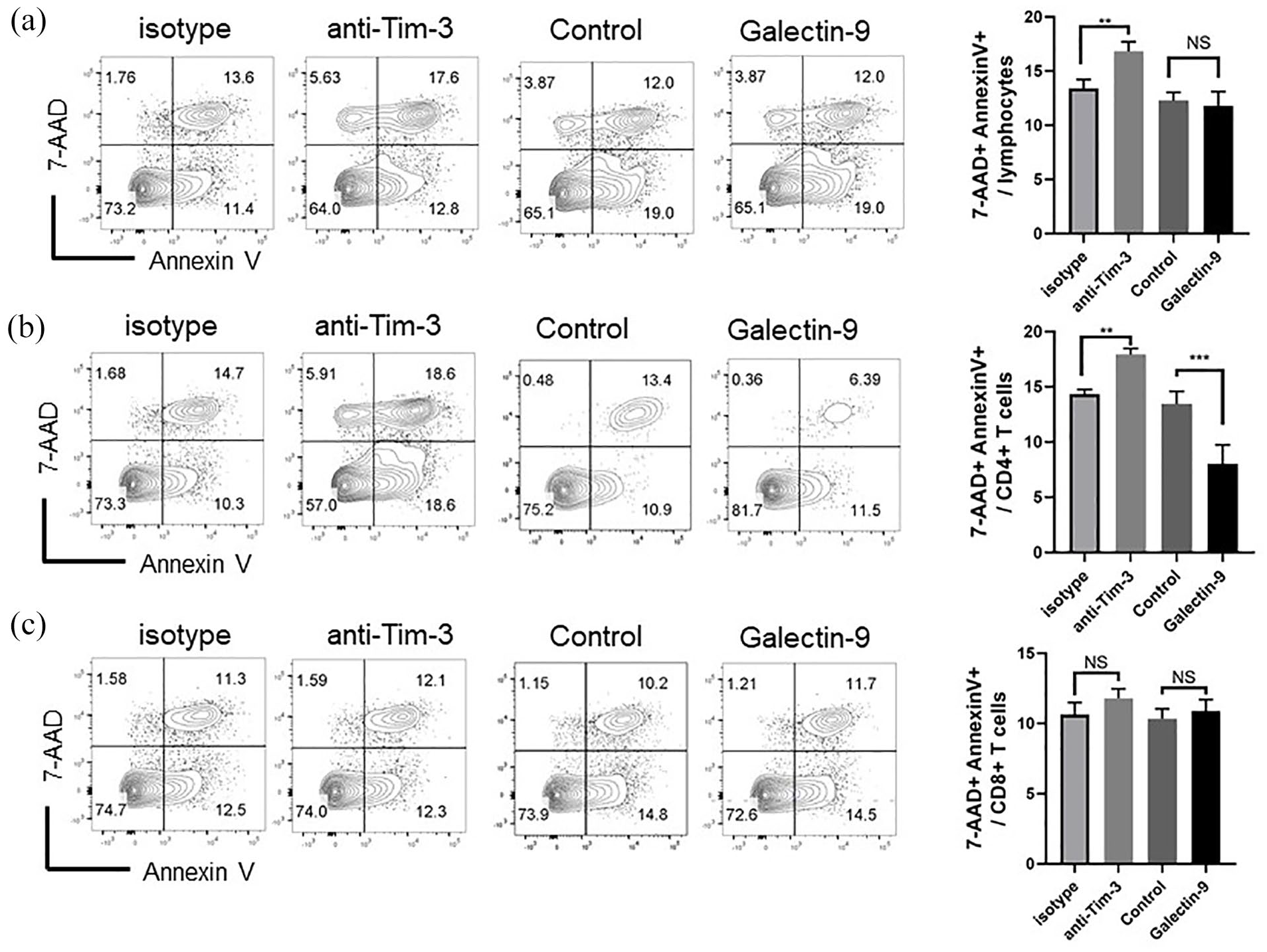
The Galectin-9/Tim-3 pathway inhibits the apoptosis of splenic CD4 T cells in EAE mice. Splenic cells were isolated from EAE mice sacrificed on day 28 after immunization. Anti-Tim-3 antibody, Galectin-9, or related isotype control antibody were added to the supernatant for 3 days. 7-AAD+ Annexin V+ apoptotic splenic lymphocytes (a), CD4+ T cells (b) and CD8 T cells (c) were detected by flow cytometry. Data are presented as the mean ± SEM; n = 4 mice/group. Levels of significance for comparison between two groups were determined by the Student
The Galectin-9/Tim-3 pathway inhibited the differentiation and function of Th17 cells
We observed the decreased production of IL17A in CD4 T cells from EAE mice after Tim-3 blocking. To clarify the impact of the Galectin-9/Tim-3 pathway on Th17 differentiation and function, we processed the Th17 polarizing experiment in vitro. Naïve CD4 T cells obtained from B6 mice were stimulated by TGF-β, IL-6, anti-IFN-γ, and anti-IL4 antibodies for 7 days. Moreover, the Galectin-9/Tim-3 pathway of CD4 T cell was blocked or activated by anti-Tim-3 or Galectine-9 molecule, respectively. As shown in Figure 4(a), the proportion of IL17A secreting CD4 T cells accelerated significantly after Tim-3 blocking. Furthermore, Galectin-9/Tim-3 pathway activation led to less IL17A production in CD4 T cells (Figure 4(b)). These results are consistent with the IL-17A concentration of CD4 T cell supernatant detected by ELISA (Figure 4(c) and (d)).

The Galectin-9/Tim-3 pathway promotes the differentiation of Th17 cell. Polarizing cytokines and neutralizing antibodies were added to C57/BL6 splenic CD4+ T cells to promote the Th17 differentiation. The anti-Tim-3 antibody, Galectin-9, or related isotype control antibody were added to the supernatant for 7 days. The proportion of IL17A secreting CD4 T cells stimulated by anti-Tim-3 antibody (a) or Galectin-9 (b) were investigated by flow cytometry. The concentrations of IL17A stimulated by anti-Tim-3 antibody (c) or Galectin-9 (d) were detected by ELISA. Data are presented as the mean ± SEM; n = 4 samples/group. Levels of significance for comparison between two groups were determined by the Student
Discussion
The aim of current MS therapies is to minimize symptoms and improve function. These regimens are dependent on disease-modifying drugs, which have failed as treatments for progressive multiple sclerosis. 20 Thus, further studies investigating the underlining mechanism and developing novel treatments are needed. The research progresses of immune checkpoint and clinical application in cancer treatment suggest a potential approach for MS treatment. 5
Recent studies have identified several immune checkpoint molecules, including CTLA-4, PD-1, Lag-3, Tim-3, TIGIT, and VISTA. 9 Tim-3 is one of the checkpoint receptors that is expressed on helper T cells, regulatory T cells, and innate immune cells. 21 Multiple studies showed that Tim-3 signal suppression led to the inhibition of immune cell responses upon interaction with its ligand, Galectin-9.22,23 In our study, we demonstrated the repressive role of Tim-3 in the pathology of EAE consistent with a previous study by Lee and Goverman 24 Because the Tim-3 shows complicated functions in different immune cells and tissues, the detailed mechanism of Tim-3 is still elusive. Previous studies suggested regulatory effects of Tim-3 on Th1 immunity.25,26 Moreover, Liu et al. 27 showed that Tim-3 enhanced the regulatory function of Treg cells. Meanwhile, Hou et al.’s 28 results indicated that Tim-3 promotes Th2 polarization and Th2-biased immunity. Our data indicated the expression of Tim-3 in EAE mice and the negative effect of Tim-3 on Th17 polarization and proliferation. These data show a potential approach to MS therapy.
Application of monoclonal antibodies is already a reliable therapy in cancer therapy in combination with standard chemotherapy regimens. 29 As shown in our research, anti-Tim-3 blocking antibody treatment significantly increased the clinical score and the proportion of Th17 cells in EAE mice. This result suggests that Tim-3 stimulation, such as Galectin-9, is a potent therapy for further MS therapy. Meanwhile, further studies should be conducted to investigate the side effects of anti-Tim-3 antibodies in EAE therapy in vivo.
In this study, we observed the effect of the Galectin-9/Tim-3 pathway on Th17 cell polarization and apoptosis. Tim-3+ lymphocytes exhibit the exhausted phenotype and co-express PD-1 in anti-tumor immunity. 30 Our results confirmed these findings on Th17 cells and illustrated a further effect on the polarization of Th17 cells. Cytokine IL-23 and transcriptional factor RORγt have been targeted as checkpoints in the development and function of Th17 cells. 31 These molecules play critical roles in Th17 differentiation. But, as intracellular molecules, they are not properly regulated in vivo. Our results suggested that Tim-3 might play a related role and could be accessible in autoimmune treatments.
In this study, the anti-Tim3 antibody was applied to block the Galectin-9/Tim3 pathway in vivo. While Tim3 is expressed on several immune cells including CD4 T cells. The effect of the anti-Tim3 antibody is derived from the combined effect of the immune cells expressed Tim3. Thus, to investigate the Tim3 pathway in CD4 T cells, a specific knockout of Tim3 in CD4 T cells (Tim3fl/fl CD4Cre) would be a more accurate model. The lack of comprehensive investigation of Tim3 in the CNS is another limitation of this article, we would process more intensive studies on microglia, macrophage, and neuron in the future. Furthermore, studies on the molecular mechanism of Tim-3 and other checkpoints that affect Th17 differentiation and function are needed in the future.
Conclusion
This study confirmed the expression of Tim-3 in CD4 T cells in the mouse EAE model. We then showed that the anti-Tim-3 blocking treatment accelerates the clinical severity of EAE and promotes IL17A production of CD4 T cells. In vitro results indicated that the Galectin-9/Tim-3 pathway induces the apoptosis of CD4 T cells and inhibits the polarization of Th17 cells. Additional studies are needed to disclose the detailed mechanism of the immune checkpoint in EAE pathology and the therapeutic effect of the Galectin-9/Tim-3 pathway in EAE and other autoimmune diseases.
Footnotes
Author contributions
QG carried out the experiment and drafted the manuscript. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Institutional Animal Care and Use Committee of the Jilin University.
Animal welfare
The present study followed Institutional Animal Care and Use Guideline of the Jilin University for humane animal treatment and complied with relevant legislation.
