Abstract
The so-called Schmallenberg virus (SBV), first detected in a German town of the same name in October 2011, is a novel emerging orthobunyavirus in Europe causing malformations and severe economic loss in ruminants. This report describes lesions in 40 sheep, 2 goats, and 16 cattle naturally infected with SBV as determined by real-time quantitative reverse transcription polymerase chain reaction. The most common macroscopic changes were arthrogryposis, vertebral malformations, brachygnathia inferior, and malformations of the central nervous system, including hydranencephaly, porencephaly, hydrocephalus, cerebellar hypoplasia, and micromyelia. Histologic lesions included lymphohistiocytic meningoencephalomyelitis in some cases, glial nodules mainly in the mesencephalon and hippocampus of lambs and goats, and neuronal degeneration and necrosis mainly in the brain stem of calves. Micromyelia was characterized by a loss of gray and white matter, with few neurons remaining in the ventral horn in calves. The skeletal muscles had myofibrillar hypoplasia in lambs and calves. The lesions of SBV-associated abortion and perinatal death are similar to those attributed to Akabane virus and other viruses in the Simbu group of bunyaviruses.
Keywords
Schmallenberg virus (SBV) is a novel emerging orthobunyavirus associated with reduced milk yield, inappetence, and diarrhea in adult ruminants, as well as malformations in aborted ruminants in Europe. 2,7 The virus was named after the German town in North Rhine-Westphalia, where it was first isolated. 4,6 SBV was first detected in October 2011 in Germany and subsequently in France, Belgium, the Netherlands, Luxembourg, the United Kingdom, Italy, and Spain (http://www.promedmail.org/; http://www.oie.int/our-scientific-expertise/specific-information-and-recommendations/schmallenberg-virus/). Nearly 3000 European animal holdings were infected with SBV. Germany (1120 farms), France (1048 farms), and the Netherlands (981 farms) were the most frequently affected countries as of April 2012 (http://www.promedmail.org/). Sheep and cattle herds were more frequently infected than goat herds. SBV is a negative-sense single-stranded RNA virus in the Simbu serogroup of the genus Orthobunyavirus, family Bunyaviridae. Akabane virus, which causes bovine arthrogryposis and hydranencephaly, also belongs to this family. The virus is transmitted by hematophagous insect vectors. 4 This article reports the major macroscopic and histologic lesions in SBV-infected fetal or perinatal sheep, goats, and cattle.
Cases and Methods
Fifty-eight animals (40 sheep, 2 goats, 16 cattle) with natural in utero infection by SBV were investigated. All animals were aborted, stillborn, or died during the perinatal period. Twenty-one sheep were female and 19 were male, with body weights of 1.5–5.5 kg (mean, 3.8 kg). The goats included a 5.2 kg-female and a 1.9 kg-male. There were 7 male and 9 female bovine cases; 12 of the 16 weighed 14.5–41.8 kg (mean, 24.1 kg). In 4 aborted bovine fetuses, crown–rump length was 59–80 cm (mean, 72.8 cm).
In all cases, SBV genomic and mRNA were detected using a real-time quantitative reverse transcription polymerase chain reaction developed at the Friedrich-Loeffler-Institute. 4 Brain was the specimen for all sheep and goats. In 11 bovine cases, SBV-specific transcripts were detected in brain and in 5 cases, only in meconium. All bovine cases were negative for bovine viral diarrhea virus (antigen ELISA, LDL-Labordiagnostik, Leipzig, Germany) and bluetongue virus (real-time quantitative reverse transcription polymerase chain reaction, LDL-Labordiagnostik), using blood samples.
Various brain regions (lobus frontalis, parietalis, temporalis, and occipitalis, hippocampus, brain stem, medulla oblongata, cerebellum, and mesencephalon) were evaluated histologically from all but 1 ovine and 3 bovine cases. Other organs were examined as available from 12 ovine, 1 caprine, and 15 bovine cases. For histology, all tissues were routinely processed in paraffin wax, cut at 3-μm thickness, and stained with hematoxylin and eosin. Cresyl echt violet, Luxol fast-blue, and von Kossa staining were performed as described. 1,8
Results
In the 40 ovine cases, arthrogryposis (n = 37; Fig. 1), brachygnathia inferior (n = 14), and curvature of the spine (torticollis, kyphosis, lordosis, or scoliosis; n = 15; Fig. 1) were the most common musculoskeletal defects. Malformations of the brain included unilateral or bilateral internal hydrocephalus (n = 26), characterized by dilated ventricles with loss of adjacent neuroparenchyma but still distinguishable gray and white matter and relative sparing of the paleopallium and basal nuclei; hydranencephaly (n = 6), characterized by segmental or complete loss of the neopallium without discernable gray and white matter and a nearly complete conversion of the neocortex into a transparent collapsed membrane; and cerebellar hypoplasia (n = 26). Porencephaly, macrocephaly, and a hypoplastic brain stem were found in 1 animal each. A domed skull was present in 3 sheep. Fetal mummification, cardiac ventricular septal defect, unilateral hydronephrosis, and colonic atresia were found in 1 case each.
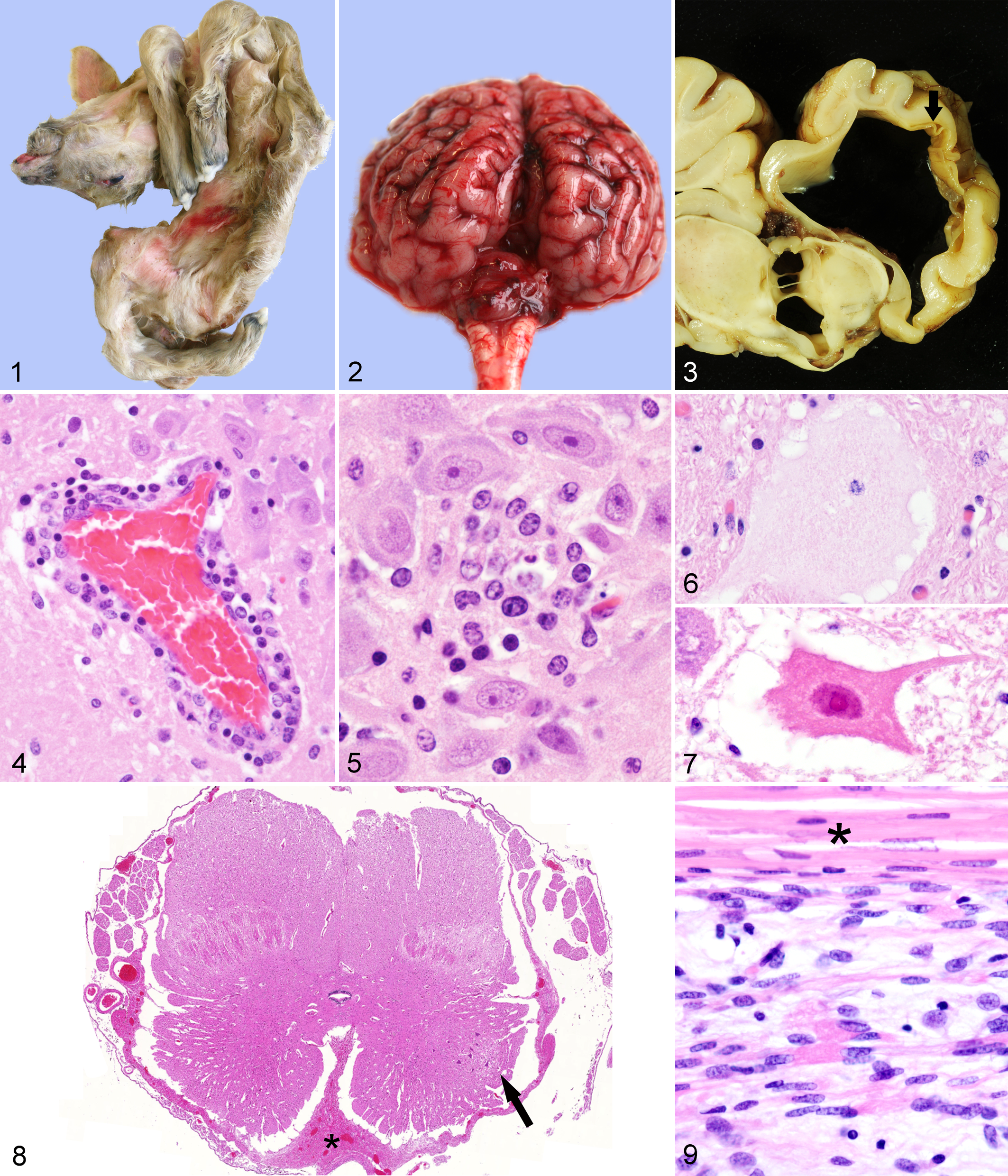
The male goat had arthrogryposis, vertebral deformities, and pulmonary hypoplasia. The female goat had arthrogryposis and porencephaly.
Of the bovine cases, 6 had cerebellar hypoplasia (Fig. 2); 3, hydrocephalus associated with destruction of adjacent neuroparenchyma (Fig. 3); 2, hydranencephaly; 3, brain stem hypoplasia; and 2, micromyelia. Skeletal defects included 12 cases with arthrogryposis, 10 cases with curvature of the spine (torticollis, kyphosis, lordosis, or scoliosis), 8 cranial malformations, and 3 cases with brachygnathia inferior. Ectopia cordis, pulmonary hypoplasia, and a ventricular septal defect were found in 1 bovine case each.
Inflammation in the central nervous system (CNS)—characterized by a lymphohistiocytic, mainly perivascular, infiltration of the gray and white matter (Fig. 4) in all investigated brain regions, including meninges—was found in 10 of 40 ovine cases. Perivascular cuffs ranged from 1 to approximately 15 layers of mononuclear cells. Glial nodules occurred predominantly in the mesencephalon and hippocampus (Fig. 5). Hydranencephaly was associated with neuroparenchymal necrosis, gitter cell accumulation, edema, and vacuolization, as well as deposition of von Kossa–positive, extracellular mineral in the cerebral cortex. Neuronal changes included chromatolysis and necrosis (Fig. 6). The male goat had histologic lesions like those in the sheep, plus a predominantly perivascular lymphohistiocytic poliomyelitis. The female goat lacked inflammatory changes.
Only 1 of the 13 evaluated bovine brains had mild, diffuse, lymphohistiocytic meningitis without perivascular or parenchymal infiltration of the brain. Neuronal changes of necrosis and chromatolysis (Figs. 6, 7) were mainly in the brain stem. Micromyelia in 2 of the 9 examined spinal cords presented as severe bilateral reduction of the gray matter as well as circumferential reduction of white matter with severely diminished spinal cord diameter. In particular, the ventral horns were small with few neurons (Fig. 8). The ventral median fissure was expanded by collagen-rich fibrous tissue.
All examined brains in all 3 species had mild to severe, diffuse astrogliosis and/or microgliosis. Demyelination in areas with and without inflammation was evident in selected sections stained with Luxol fast blue. Cerebellar hypoplasia in all 3 species was characterized by a multifocal to diffuse, mild to moderate, reduced thickness of the molecular layer with variable reduction of Purkinje neurons, whereas the granular cell layer seemed to be less affected.
Skeletal muscles had severe reduction of myofibrils with diminished myofiber diameter in 6 of 13 sheep and 1 of 8 bovine cases (Fig. 9). Few mature myofibers were present in the hypoplastic muscles. One sheep had Morgagnian globules in both lenses. The bone marrow of one sheep had reduced hematopoietic cellularity. Lymphoid depletion was evident in lymph node or spleen of 3 sheep and in thymus and lymph node of 1 bovine case.
The following organs and tissues lacked significant histologic lesions: urinary bladder, thyroid gland, liver, heart muscle and vessels, uterus, ovaries, testes, peripheral nerves, placenta, esophagus, abomasum, small and large intestine, adrenal glands, celiac ganglion, trachea, skin, tongue, and adipose tissue.
Summary
This study provides a description of macroscopic and histologic lesions in domestic ruminants naturally infected with SBV. The most frequently observed malformations were arthrogryposis, brachygnathia inferior, torticollis, kyphosis, lordosis, scoliosis, cerebellar and cerebral hypoplasia, hydranencephaly, porencephaly, and hydrocephalus. Because the ventricular dilation was usually eccentric and associated with loss of adjacent brain tissue, this was probably, in most cases, hydrocephalus ex vacuo, secondary to viral-induced destruction of neuroparenchyma. Some cases had lymphohistiocytic meningoencephalomyelitis in addition to neuroparenchymal degeneration and necrosis. Some bovine cases had micromyelia. Some ovine and bovine cases had skeletal muscle hypoplasia.
This pattern of lesions in domestic ruminants resembles that attributed to other orthobunyaviridae, especially the Simbu-group viruses, including Akabane and Aino virus. 3,5,10 Infection with these viruses is usually clinically inapparent in nonpregnant ruminants, whereas fetal infection can lead to CNS and musculoskeletal malformations. 9 Simbu-group viruses are neurotropic in fetuses, which may account for the neuroparenchymal loss, muscle hypoplasia, and arthrogryposis. Arthrogryposis and hydranencephaly are typical lesions in natural and experimental infection with Akabane or Aino viruses. 9,10 Inflammation of the CNS occurs in fetuses that survive infection early in gestation. 9 Interestingly, only 11 of the 43 ruminants (9 of 32 sheep, 1 of 2 goats, and 1 of 9 bovine fetuses) with gross malformations had lymphohistiocytic inflammation of the CNS, indicating that the timing of infection with Schmallenberg virus determined lesion development as in Akabane viral infections. Further studies should investigate the correlation between lesions and viral protein or RNA distribution.
Footnotes
Acknowledgements
We gratefully acknowledge the help of Dirk Schaudien, PhD, Dipl ACVP, Fraunhofer-Institut für Toxikologie und Experimentelle Medizin, Hannover, for scanning the spinal cord section. We thank Caroline Schütz and Bettina Buck for their excellent technical work.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
