Abstract
In December 2011, a previously unknown congenital syndrome of arthrogryposis and hydranencephaly in sheep and cattle appeared in the Netherlands as an emerging epizootic due to Schmallenberg virus (SBV). Gross lesions in 102 lambs and 204 calves included porencephaly, hydranencephaly, cerebellar dysplasia and dysplasia of the brainstem and spinal cord, a flattened skull with brachygnathia inferior, arthrogryposis, and vertebral column malformations. Microscopic lesions in the central nervous system showed rarefaction and cavitation in the white matter, as well as degeneration, necrosis, and loss of neurons in the gray matter. Brain and spinal cord lesions were more severe in lambs than in calves. Ovine and bovine cases examined early in the outbreak showed encephalomyelitis. SBV infection was confirmed by real-time quantitative reverse transcription polymerase chain reaction (RT-qPCR) in brain samples in 46 of 102 lambs (45%) and in 32 of 204 calves (16%). Immunohistochemistry, performed on tissue samples from 18 RT-qPCR–positive lambs, confirmed the presence of bunyaviral antigen in neurons of the brain in 16 cases. SBV antibodies were detected by enzyme-linked immunosorbent assay in fetal blood in 56 of 61 sampled ovine cases (92%). In a virus neutralization test, all tested dams of affected newborns, 46 ewes and 190 cows, were seropositive. Compared with other teratogenic viral infections, the pathogenesis and lesions of SBV in sheep and cattle fetuses are similar to those of other ruminant orthobunyaviruses. However, the loss of spinal ventral motor neurons and their tracts, resulting in micromyelia, distinguishes SBV infection from other viral central nervous system lesions in newborn ruminants.
Keywords
Schmallenberg virus (SBV), an arthropod-borne virus of the genus Orthobunyavirus, is related to the Simbu serogroup, including Shamonda virus, Akabane virus, and Aino virus, 9 which are known to cause teratogenic effects on the central nervous system (CNS) in cattle and small ruminant fetuses. 16 Viruses of this genus are widely distributed in Australasia, Africa, and the Middle East and are predominantly transmitted through Culicoides biting midges.
From December 2011 onward, many newborn malformed lambs born to dams that were negative to bluetongue virus, border disease virus (BDV), and bovine viral diarrhea virus (BVDV) were submitted for postmortem examination to GD Animal Health. After SBV was detected by quantitative reverse transcription polymerase chain reaction (RT-qPCR) in brain samples of affected lambs, 33 congenital malformations in newborn ruminants were made notifiable. Since that time, it had been compulsory for farmers to submit malformed lambs and calves for further diagnostic workup until July 2012. In the months following its first detection, SBV was also found in newborn malformed lambs, kids, and calves in Germany, Belgium, Luxembourg, France, the United Kingdom, Italy, and Spain (http://www.promedmail.org).
This study reports gross and microscopic lesions in lambs and calves due to the initial outbreak period of SBV in the Netherlands. The aim of this study was to compare these lesions with those reportedly caused by other teratogenic viruses in ruminants, including other viruses of the genus Orthobunyavirus.
Materials and Methods
Case Selection, Necropsy, and Histology
This study took place during the first 4 months of the SBV outbreak in the Netherlands, including 3 compulsory notification months. From 215 ovine and 421 bovine cases with congenital malformations belonging to the arthrogryposis and hydranencephaly syndrome, a selection of macroscopically and microscopically completely documented cases was compiled, totaling 102 lambs and 204 calves. The 102 selected lambs originated from 44 farms from different parts of the Netherlands and were obtained from 56 submissions, received between December 9, 2011, and March 26, 2012. Most lambs were stillborn, and some were born alive but died or were euthanized shortly after birth.
The 204 selected calves originated from 204 farms, also from different parts of the country, and were submitted between December 28, 2011, and March 29, 2012. Most calves were stillborn, but some were aborted in late gestation. No cases with spina bifida, cranium bifidum, congenital vertebral malformation (CVM), brachyspina-like lesions, chondrodystrophic lesions (“bulldog calves”), schistosomus reflexus, or perosomus elumbis lesions were included for this SBV investigation.
After all gross malformations, especially those of the brain and spinal cord, were recorded, cerebrum, cerebellum, brainstem, and the cervical and lumbar spinal cord intumescences, which many times included spinal ganglia, were fixed in buffered formalin. Samples of brainstem were also collected for RT-qPCR. In approximately 20% of the ovine and bovine cases, lung, liver, spleen, thymus, pancreas, skeletal muscle from arthrogrypotic limbs, and, when possible, placenta were formalin fixed and processed for routine, paraffin embedded, 4-μm sections and hematoxylin and eosin (HE) stained histology. From a few animals, the complete brain or 1 brain hemisphere was fixed in formalin, and serial coronal sections were prepared after fixation. Sections of the spinal cord from selected cases were also stained using Van Gieson’s method.
Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction
Brain tissue samples, including the brainstem, were tested at the Central Veterinary Institute, Lelystad, the Netherlands, using a newly developed RT-qPCR, kindly shared by the Friedrich Loeffler Institute. 1,3,6,15
Immunohistochemistry
Brain, spinal cord, lung, liver, spleen, thymus, pancreas, and skeletal muscle from 18 RT-qPCR–positive lambs, in 13 cases also including placenta, were stained with immunohistochemistry. An unconjugated monoclonal antibody raised against Tinaroo virus, a member of the Australian Simbu serogroup, generously provided by Dr P. Kirkbride, Australian Animal Health Laboratory, was used in the DAKO EnVision protocol (DAKO, Glostrup, Denmark), according to the manufacturer’s instructions. This monoclonal antibody, MAb 4H9/B11/F1, reacts strongly with all Australian members of Simbu serogroup reference viruses and was previously proposed for use as a serogrouping reagent for bunyaviruses of the Simbu serogroup. 2
After deparaffinization, heat-induced epitope retrieval was carried out on 4-μm tissue sections at 100°C for 20 minutes at pH 6; thereafter, sections were incubated with the monoclonal antibody against Tinaroo virus in a 1:500 dilution for 15 minutes at 37°C and incubated with a polymer-based immunoperoxidase (DAKO EnVision). 3,3′-Diaminobenzidine was used as chromogen, and the slides were counterstained with hematoxylin. As a negative control, mouse IgG1 was used as an isotype control in duplo-sections, replacing the monoclonal antibody against Tinaroo virus. The cerebellum of a SBV-infected lamb, which was strongly positive by RT-qPCR and immunohistochemistry, was used as a positive control mounted on the same slide. Specificity of the immunohistochemical data was investigated by excluding nonspecific binding of antibody to tissues of 2 lambs and 2 calves that died in 2009 of non-SBV-related diseases.
Serologic Testing
During the first 4 weeks of the epizootic, heart blood was sampled from 61 malformed lambs included in this study for detection of antibodies against SBV, using a newly developed in-house enzyme-linked immunosorbent assay (ELISA). 34
From 5 farms in different regions of the country that reported malformed lambs in the period between December 9 and 14, a maximum of 5 ewes per farm and their malformed offspring were blood sampled and tested for border disease and bovine viral diarrhea virus (Priocheck bvdv Ab [Prionics AG, Zurich, Switzerland]; IDEXX bvdv Ag/Serum Plus Test [IDEXX Laboratories, Westbrook, ME]) and bluetongue (IDEXX Bluetongue Competition Ab Test; IDEXX Laboratories). The malformed calves were not tested for bovine viral diarrhea virus or for bluetongue.
Blood samples from mothers of the ovine and bovine cases in this study were collected and tested for antibodies against SBV using a virus neutralization test (VNT). 3,23 Eighty-seven ewes from 27 farms were tested; the number of tested ewes per farm varied from 1 to 10. Blood samples of individual ewes could not be traced back to specific individual lambs but included 46 dams of at least 46 examined malformed lambs and a variable number of other ewes per flock, totaling 41 ewes. The total number of ewes involved per submission is unknown because information about siblings in case of multiple lambs per submission was not available. Ewes from the other 17 farms were not tested. From the 204 bovine cases, 190 dams were tested, and 14 dams were not tested.
Results
Macroscopic Findings in Lambs and Calves
Eighty-four of 102 lambs (82%) and 175 of 204 (86%) calves showed often multiple mild to marked malformations of the vertebral column: torticollis, scoliosis, kyphosis, and/or lordosis (Table 1). In lambs, scoliosis was most frequent (55 cases), often in combination with kyphosis (13 cases) or torticollis (10 cases); kyphosis was the second most frequent axial malformation (32 cases), with torticollis in 20 cases and lordosis in 4 cases. In calves, torticollis was most frequent (133 cases), often in combination with scoliosis (82 cases) and/or kyphosis (46 cases). The combination of torticollis, scoliosis, and kyphosis was seen in 28 cases; scoliosis was the second most frequent axial malformation (115 cases), followed by kyphosis (63 cases) and lordosis (6 cases). In both species, scoliosis, kyphosis, and kyphoscoliosis were mainly located in the thoracic vertebral column and were associated with a varying degree of malformation of the thorax, including a flattened ribcage, resulting in a reduced volume of the thoracic cavity.
Congenital Malformations in Appendicular and Axial Skeleton in 102 Lambs and 204 Calves Due to Schmallenberg Virus Infection.
aA lamb or calf can have multiple vertebral column malformations.
Ninety-nine of 102 lambs (97%) and 196 of 204 calves (96%) had arthrogryposis multiplex congenita (AMC) in various degrees (Figs. 1, 3; Table 1). In 87 lambs and in 148 calves, both forelimbs and hindlimbs were affected, mostly with bilaterally symmetric AMC (Figs. 1, 3). In 9 lambs and in 3 lambs, only forelimbs and only hindlimbs were arthrogrypotic, respectively. Lower parts of the arthrogrypotic limbs were sometimes broken or absent, supposedly as a consequence of obstetric intervention during parturition. In 29 calves and in 19 calves, only forelimbs and only hindlimbs were arthrogrypotic, respectively, and in 23 calves, arthrogryposis appeared unilaterally with either 1 forelimb (19 cases) or 1 hindlimb (4 cases) affected. From malformed calves in cases of dystocia, the distal parts of the limbs were sometimes taken off by fetotomy.
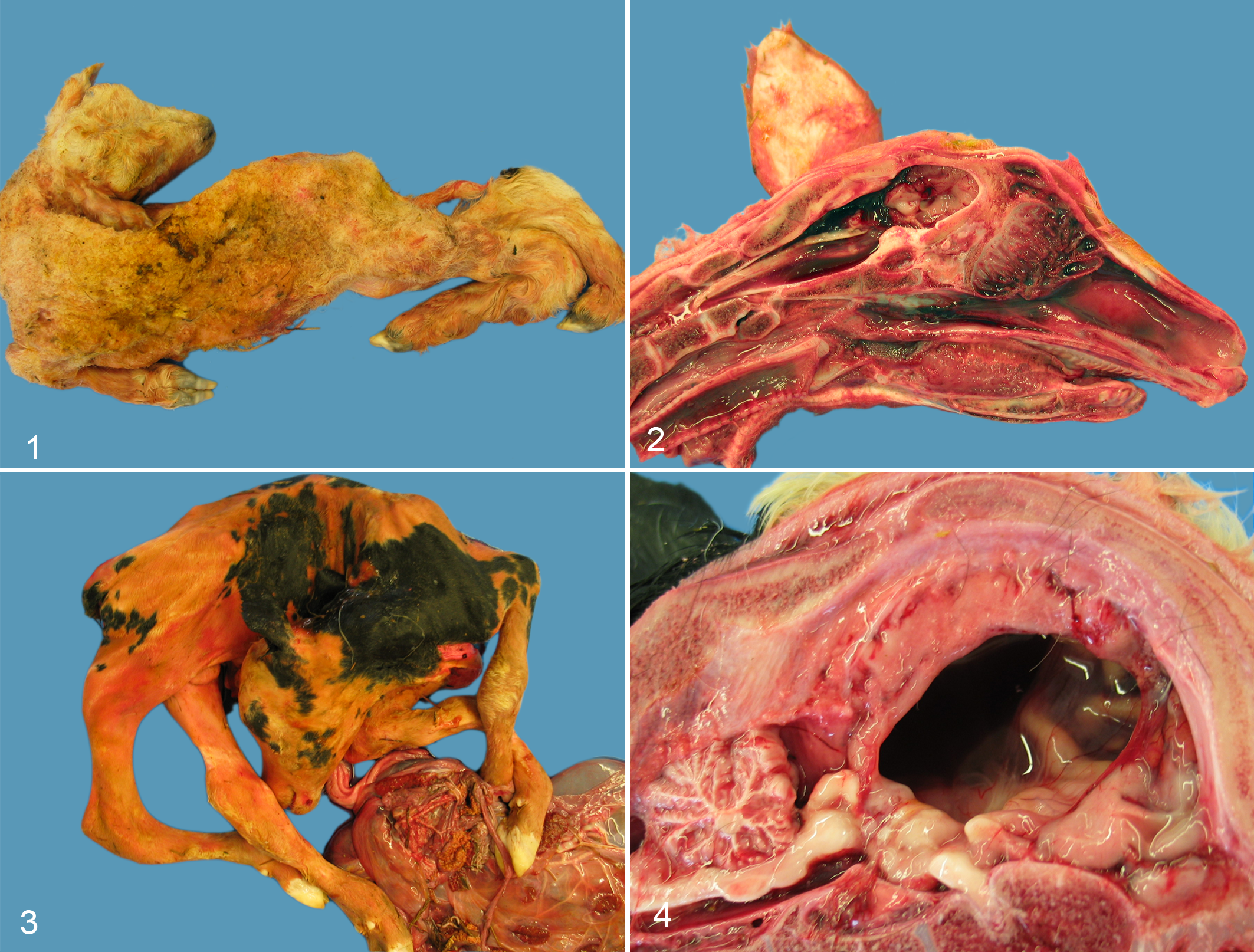
Schmallenberg virus (SBV) infection, stillborn lamb. Torticollis, scoliosis, arthrogryposis, and mild meconium staining.
In both species, skeletal muscles of affected limbs often showed severe hypoplasia. In 42 of 102 lambs (41%), an abnormally flattened skull was found at exterior inspection. The flattened skulls had a cranial vault reduced by thickened frontal, parietal, and occipital bones (Fig. 2). Twelve of 102 lambs (12%) had brachygnathia inferior. Ten lambs had severe hypoplasia of the masticatory muscles. Ten of 204 calves (5%) had a flattened skull, and 5 calves had brachygnathia inferior.
Defects in the cerebrum were seen in 85 of 102 lambs (83%) and in 108 of 204 (53%) calves, ranging from slight to moderate dysplasia, including microencephaly in a reduced cranium, cavitation of the cerebral hemispheres (porencephaly) in combination with the often dilated lateral ventricles (hydrocephalus), and lack of gyri (lissencephaly), to fully developed hydranencephaly within a normal or reduced cranium.
Various degrees of cerebellar dysplasia were seen in 96 of 102 lambs (94%) and in 92 of 204 (45%) calves, particularly in cases with cerebral defects (Table 2). In both species, the rostral brainstem (thalamus, mesencephalon) was observed to be markedly reduced in size in cases of hydrocephalus/hydranencephaly (Fig. 4), and in a few cases of microencephaly/porencephaly, small cavitations were seen in cross sections of the mesencephalon, laterally to the aqueduct.
Distribution and Severity of Gross and Microscopic Lesions in 204 Calves With Schmallenberg Virus Infection.
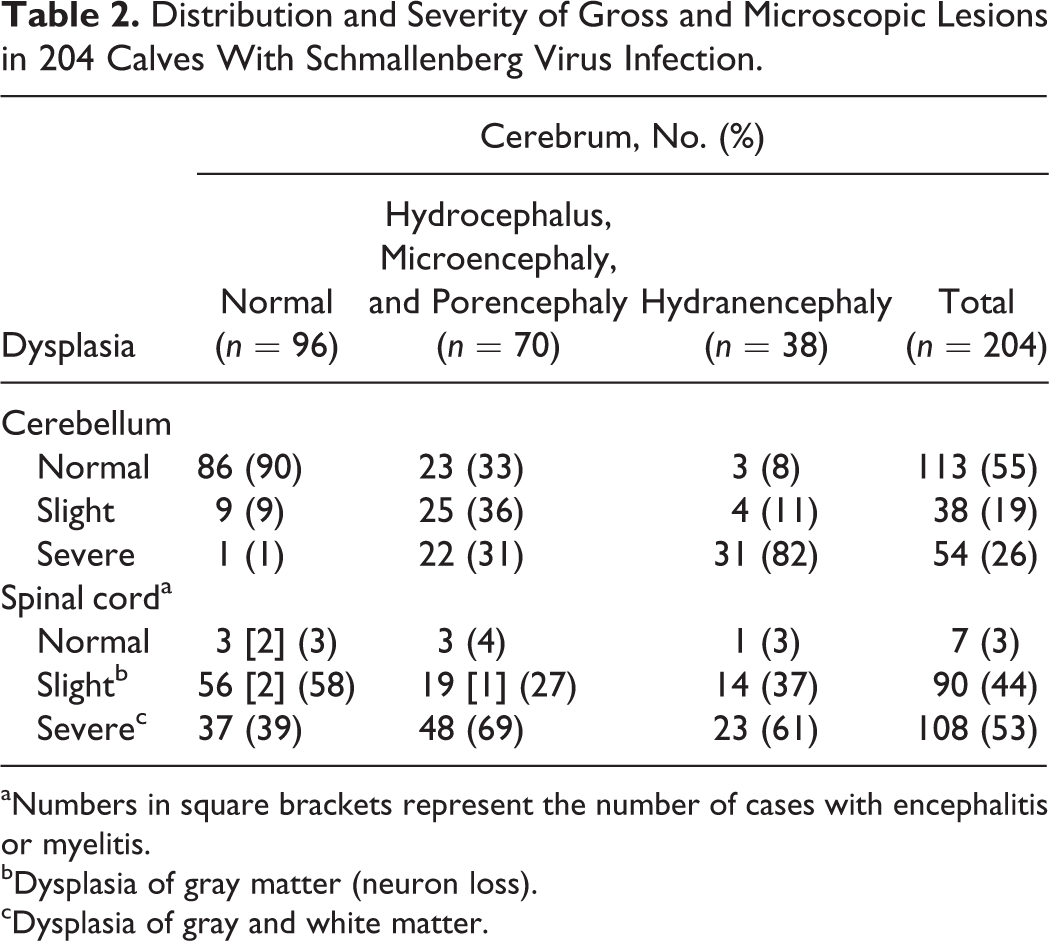
aNumbers in square brackets represent the number of cases with encephalitis or myelitis.
bDysplasia of gray matter (neuron loss).
cDysplasia of gray and white matter.
The medulla oblongata and spinal cord were macroscopically small in 77 of 102 lambs (75%) and in 108 of 204 calves (53%). The degree of reduction varied but was often more than 50% and was characterized by dorsoventral flattening.
The interrelationship between cerebral and cerebellar defects and between cerebral and spinal cord defects is shown in Tables 2 and 3 for calves and lambs, respectively. Thirteen of 17 lambs having a normal cerebrum nevertheless had cerebellar dysplasia. In 4 of these 13 lambs, no further defects of the CNS were observed, but the other 9 lambs had micromyelia as well (not specified in Table 3).
Distribution and Severity of Gross and Microscopic Lesions in 102 Lambs With Schmallenberg Virus Infection.
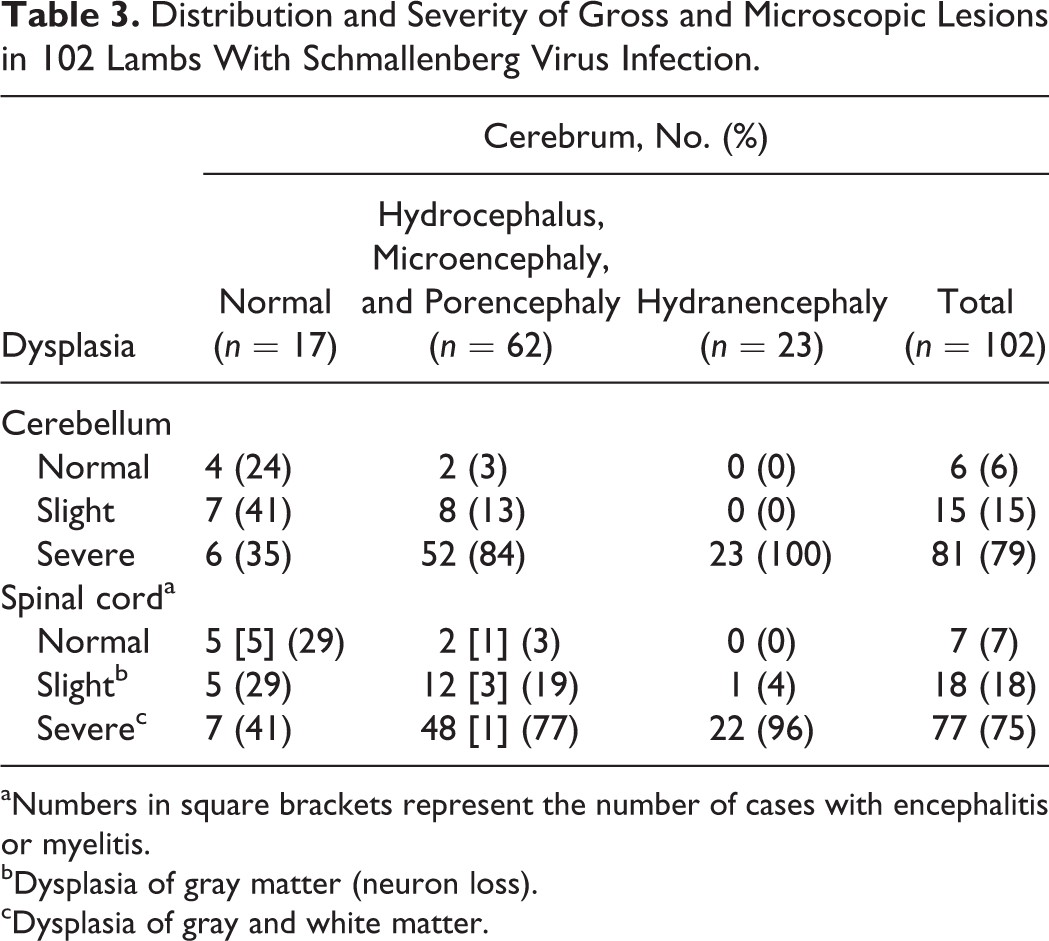
aNumbers in square brackets represent the number of cases with encephalitis or myelitis.
bDysplasia of gray matter (neuron loss).
cDysplasia of gray and white matter.
In cases with thoracic malformation, lung lobes in both species were small compared with congenitally atelectatic lungs. No other macroscopic lesions were detected. Except for postural changes, the axial and appendicular skeleton was normally developed.
Microscopic Findings in Lambs and Calves
Cerebrum
Microscopic cerebral lesions in lambs varied considerably in proportion with gross changes: in slight to moderate dysplastic cerebral hemispheres, microencephaly, lissencephaly, neuropil rarefaction, and cavitations (porencephaly) were found in the cerebral cortex. Cases of porencephaly were characterized by large cavitations dissecting the corona radiata. These cavities were often traversed by strands of neuroparenchyma with blood vessels and occasionally small fragments of disorganized neuropil (Fig. 5). The cerebral cortex showed variable degrees of dysplasia together with lissencephaly and dilatation of the ventricles (hydrocephalus). In cases of hydranencephaly, only a small remnant, if any, of the cerebral cortex remained. The hippocampus was generally spared.
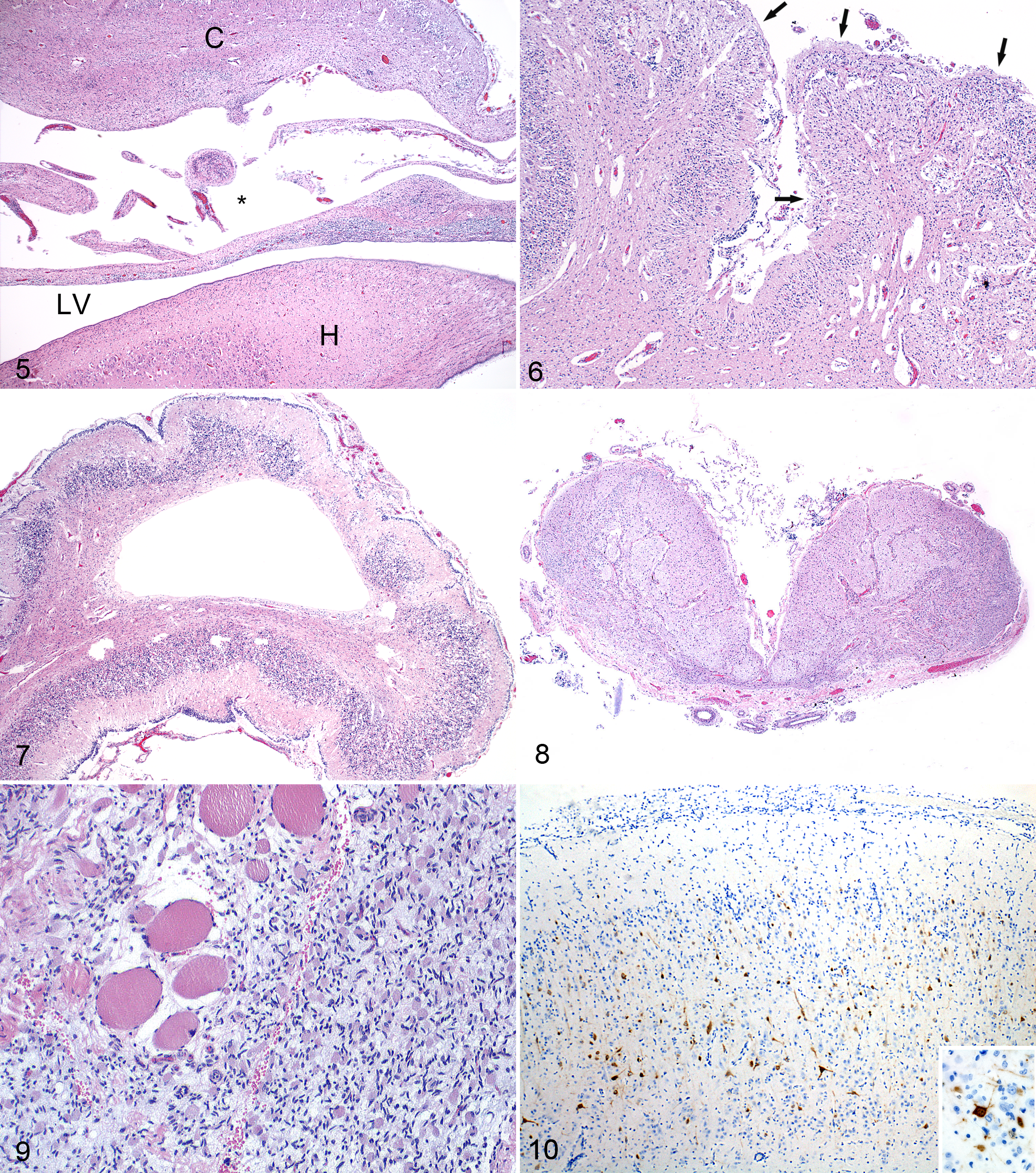
Schmallenberg virus (SBV) infection, brain, stillborn lamb.
Inflammation consisted of foamy macrophages (gitter cells), gliosis, and mild perivascular infiltration of mononuclear cells. Nonsuppurative meningoencephalitis and neuronophagia were seen in 20 cases, with all but one being submitted between December 13, 2011, and January 4, 2012, during the first 3 weeks of the epizootic. The case with inflammation submitted later (February 2) had porencephaly, micromyelia, and polioencephalomyelitis.
Microscopic cerebral lesions in calves were in line with those in lambs but were usually less severe. Most cases classified as cerebral dysplasia had dilated lateral ventricles (hydrocephalus) with slight reduction of the cerebral gray and white matter (microencephaly and lissencephaly) and gliosis in the periventricular white matter. Porencephalic lesions were less frequently observed, and hydranencephaly appeared similar compared with lambs.
Cerebellum
Cerebellar dysplasia was similar in lambs and calves and was characterized by disorganization of the cerebellar cortex with depletion of the granular layer and loss of Purkinje cells. The cerebellar lesions were irregularly distributed between the folia. Focally, the leptomeninges were entrapped between collapsed cerebellar folia (Fig. 6). Cavities in the cerebellar white matter were seen in few cases (Fig. 7).
Brainstem
Brainstem lesions were similar in lambs and calves. The following changes were observed: loss of neurons, calcified neurons and areas of gliosis in the thalamus, areas of rarefaction and porencephalic cavities in the mesencephalon with a dilated aqueduct, and absent or defective corticospinal tracts (crura cerebri, pyramidal tracts) in cases of hydranencephaly and porencephaly. Dysplasia of the medulla oblongata was predominantly caused by the absence of the ventral structures such as the olivary nuclei and pyramidal tracts and by neuronal loss in the reticular formation. Loss of neural tissue made extant connective tissue around leptomeninges prominent (Fig. 8).
Spinal Cord
Spinal cord lesions were present in all lambs except one, which had slight porencephaly and cerebellar dysplasia but no malformation of the vertebral column and no arthogryposis. All but 4 calves had microscopic lesions in the spinal cord. These 4 calves had no arthrogryposis in forelimbs or hindlimbs but showed varying degrees of cerebral and/or cerebellar dysplasia.
Microscopic lesions varied considerably in both lambs and calves. In 6 lambs (6%) and 3 calves (1%) without macroscopic evidence of micromyelia, there was a mild degree of neuronal loss in all parts of the gray matter, particularly in the ventral horns. In cases with undersized spinal cords at gross examination, there was variable but mostly severe loss of neurons in the dorsal and ventral gray columns along the spinal cord. Gray matter areas lacking neurons were reduced in size. In the white matter, the ventral and lateral funicular tracts were small and sometimes completely absent. In contrast, the dorsal spinal funicular tracts were unaffected in all cases (Fig. 12). Similarly, ventral spinal nerve roots showed loss of nerve fibers, but dorsal spinal nerve roots and spinal ganglia, when seen, were unaffected. In cases of severe micromyelia, loss of parenchyma made the amount of connective tissue in the ventral membranes and blood vessels appear increased (relative to fibrosis) (Figs. 13, 14).
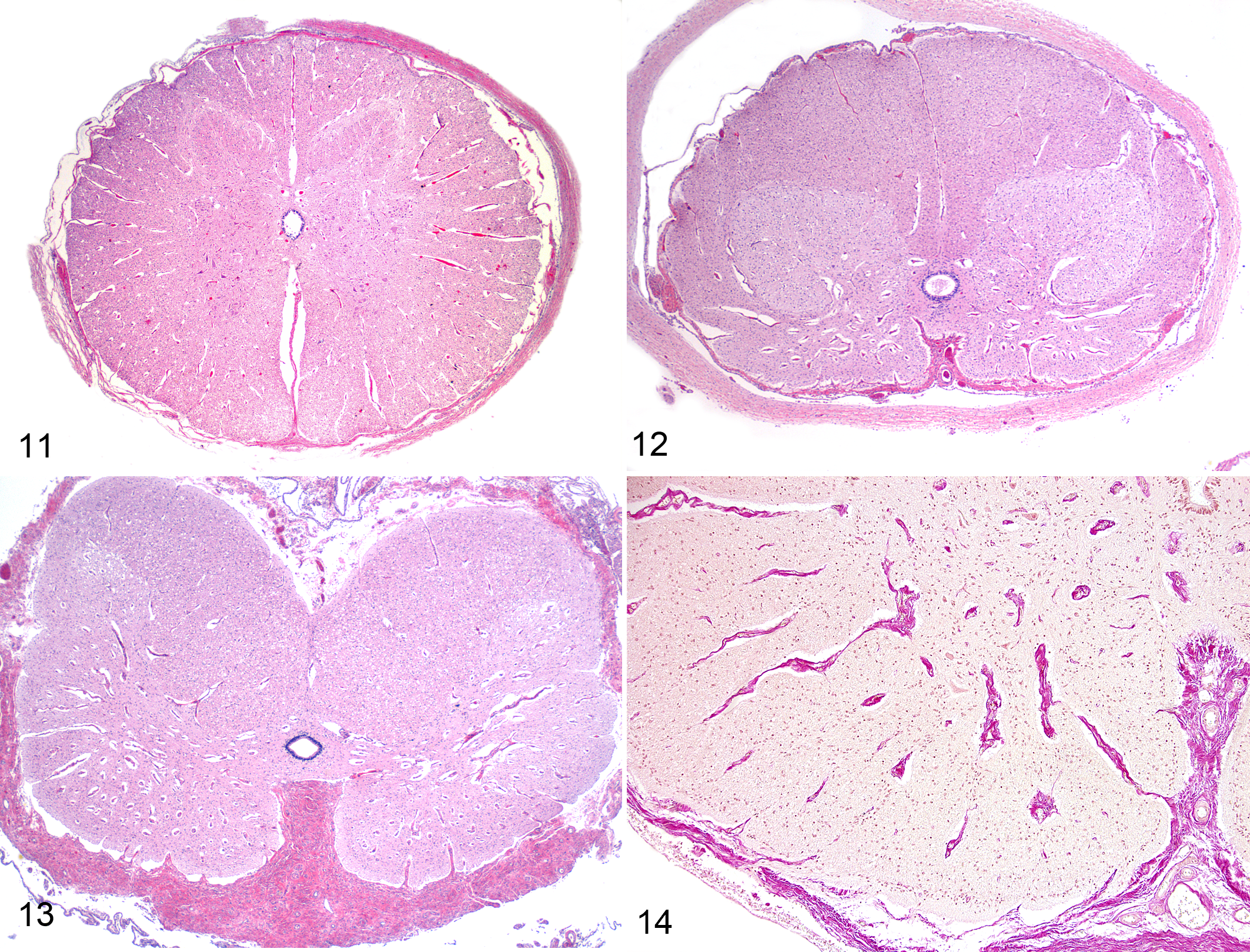
Control spinal cord within dura mater, cervical intumescence, full-term lamb. Hematoxylin and eosin (HE).
Ten of 102 lambs (10%) and 5 of 204 calves (2%) had poliomyelitis and meningoencephalitis as described above (Tables 2 and 3). In these cases, neuronal loss was accompanied with perivascular infiltration of mononuclear cells, and the presence of glial nodules suggested neuronophagia. These cases of poliomyelitis were particularly seen during the beginning of the epizootic.
Spinal cord lesions in calves were similar to those in lambs, but the number of calves with severe micromyelia (108 of 204 [53%]; Table 2) was fewer than that of lambs with severe micromyelia (77 of 102 [75%]; Table 3). In most calves, the degree of dysplasia was equal between the cervical and lumbar intumescences, but in approximately 20% of the cases, marked differences were noticed. In these cases, neuronal loss was mild or even absent in either the cervical or the lumbar intumescence.
Skeletal Muscles
Skeletal muscles of arthrogrypotic limbs had a severely reduced diameter of muscle fibers (Fig. 9), and muscle masses often consisted primarily of fibrous and adipose tissue.
Organs
In lung, liver, spleen, thymus, pancreas, and placenta, no significant histopathologic lesions were present.
RT-qPCR
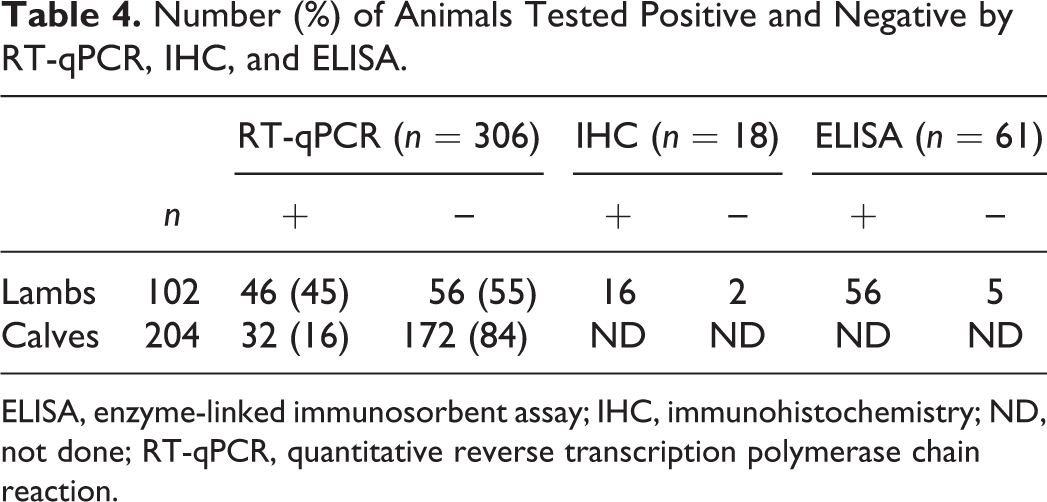
In 46 of the 102 lambs, RT-qPCR testing of brain samples, including the brainstem, yielded a positive result (45%) (Table 4). Sixteen of 20 lambs with evidence of a nonsuppurative meningoencephalitis and/or poliomyelitis were RT-qPCR positive. The brain samples of 32 of the 204 calves (16%) (Table 4) were RT-qPCR positive. Three of 5 calves with evidence of poliomyelitis were RT-qPCR positive.
Number (%) of Animals Tested Positive and Negative by RT-qPCR, IHC, and ELISA.
ELISA, enzyme-linked immunosorbent assay; IHC, immunohistochemistry; ND, not done; RT-qPCR, quantitative reverse transcription polymerase chain reaction.
Immunohistochemistry
Immunoreactive viral antigen was seen in neurons and neuronal processes in the brain and/or spinal cord in 16 of 18 tested lambs from the 46 RT-qPCR–positive ovine cases 24 (Table 4, Fig. 10). Eight of these 16 immunohistochemistry (IHC)–positive cases showed a nonsuppurative meningoencephalitis and/or poliomyelitis. The other 8 cases showed immunoreactivity of neurons in dysplastic brain tissue, while the spinal cord was IHC negative. Immunoreactive neurons were located in the cerebral and cerebellar cortex and included Purkinje cells. In the midbrain and brainstem, immunoreactive neuroblasts were in the ventricular zone. Skeletal muscle, lung, liver, spleen, thymus, pancreas, and placenta showed no immunoreactivity.
Serologic Testing
Fifty-six of 61 samples from lambs tested positive for SBV antibodies by ELISA (Table 4). Two ELISA-negative lambs had IHC-positive nonsuppurative meningoencephalomyelitis, and 3 ELISA-negative cases had mild to severe dysplasia of the brain and spinal cord.
Ewes and their malformed offspring were seronegative for border disease virus, bovine viral diarrhea virus, and bluetongue virus. All 87 tested ewe dams, including 46 ewes of at least 46 examined malformed lambs, and all 190 tested cow dams had SBV serum neutralizing antibodies (Table 5).
Distribution of VNT Titers From 87a Ewes and 190b Cows.
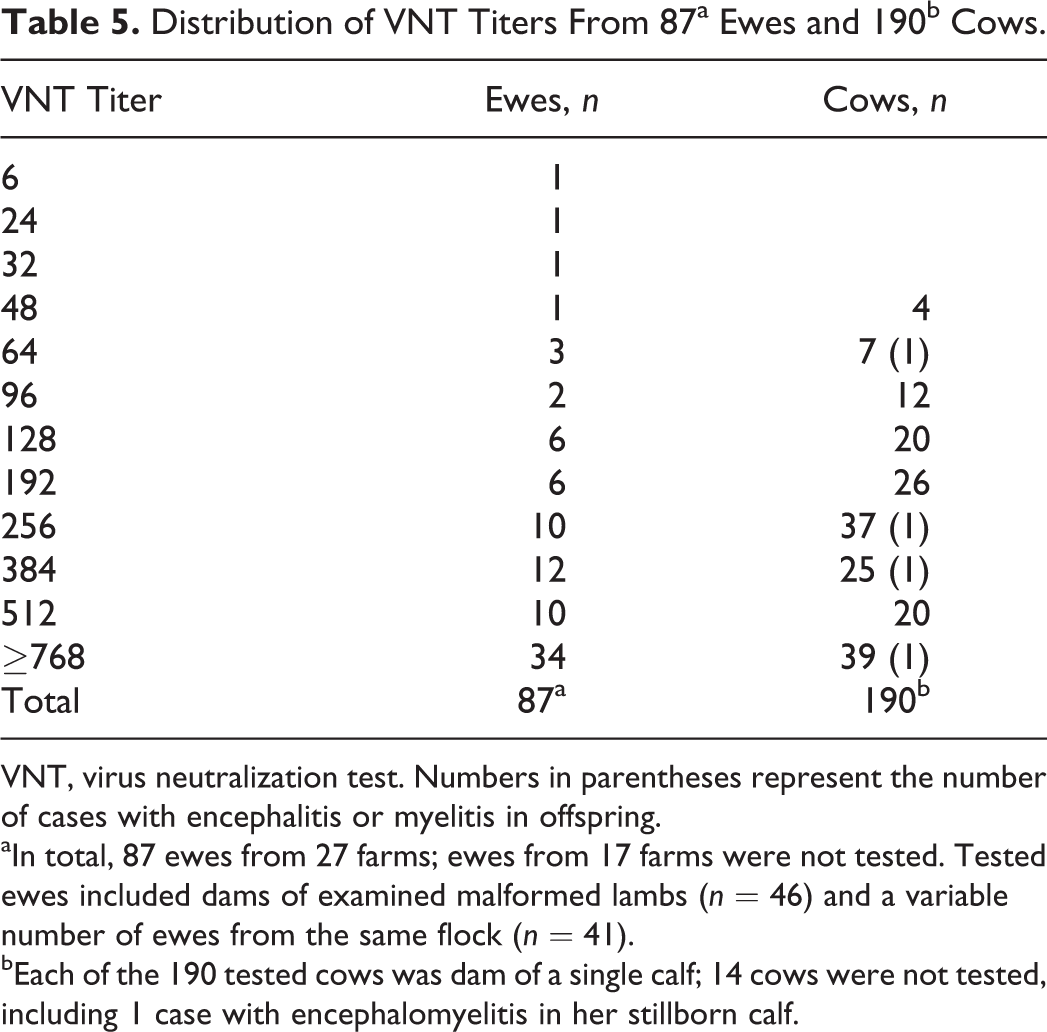
VNT, virus neutralization test. Numbers in parentheses represent the number of cases with encephalitis or myelitis in offspring.
aIn total, 87 ewes from 27 farms; ewes from 17 farms were not tested. Tested ewes included dams of examined malformed lambs (n = 46) and a variable number of ewes from the same flock (n = 41).
bEach of the 190 tested cows was dam of a single calf; 14 cows were not tested, including 1 case with encephalomyelitis in her stillborn calf.
Discussion
Since 2002, a system for monitoring and surveillance of animal diseases has been developed in the Netherlands to rapidly detect emerging diseases, 7,27 which is of utmost importance to government and livestock policy makers as well as the livestock industry. 6,22 The sudden, frequent occurrence of stillborn malformed lambs and calves in December 2011 eventually resulted in the association between the arthrogryposis-hydranencephaly syndrome and the recently discovered SBV. Until that moment, SBV was associated with an unidentified disease, affecting dairy cattle in the Netherlands and Germany, clinically characterized by milk yield drop, diarrhea, and fever. 15,26 After December 20, 2011, the arthrogryposis-hydranencephaly syndrome became notifiable, which allowed us to study a large number of cases. A range in severity and extent of lesions was observed in the CNS of malformed lambs and calves, concurrent with reports from Germany. 10,12 Brain and spinal cord malformations were more severe and more extensive in lambs than in calves. This may be associated with the shorter duration of the gestation period of sheep, requiring a more rapid organogenesis compared with cattle. In most cases, CNS lesions were seen without evidence of inflammation, presumably resulting from SBV infection early in gestation (ie, at the end of the first or during the second trimester of gestation in calves 11,36 and during the second month of gestation in lambs). Severe CNS lesions were characterized by rarefaction and cavitation in white matter areas with extensive neuron loss in the cerebral and cerebellar cortices, the brainstem nuclei, and the gray columns of the spinal cord. Loss of spinal cord descending tracts, particularly the ventrolateral funiculi, is thought to be secondary to this neuronal loss or failure of neurons to develop their axonal processes. 11 The small ventral spinal nerve roots reflect the loss or failure to develop axonal processes of ventral horn motor neurons. The spinal ganglia and dorsal funiculi of the spinal cord were not affected in any case. The neuronal cell bodies of these proprioceptive pathways (fasciculus gracilis and fasciculus cuneatus) are in the spinal ganglia and pass to the gracile and cuneate nuclei in the caudal medulla oblongata. 5 Apparently, SBV has no tropism for neurons in the spinal ganglia, originating from neural crest cells.
Fibrous thickening of the leptomeningeal areas and around blood vessels in the ventral aspect of the spinal cord parenchyma in severe cases of micromyelia is probably related to parenchymal collapse.
The musculoskeletal malformations were regarded to be secondary to the lesions in the CNS. Most cases in both species had significant neuronal loss in the spinal cord, particularly in the ventral horns, with associated deficiency of ventral spinal nerve roots. This pathogenesis would logically result in the development of arthrogryposis and vertebral column malformations in these animals. Skeletal muscle motor units deprived of their innervating lower motor neuron fail to develop normally and become hypoplastic. 36 Denervation of appendicular and axial muscles would result in arthogryposis of joints and vertebral column malformation. Particularly in calves, neuronal loss in the cervical and lumbar intumescences was associated with arthrogryposis in forelimbs and/or hindlimbs. Unilateral neuronal loss was also noted in cases with unilateral arthrogryposis. Severe atrophy of masticatory muscles noticed in a few lambs may have been related to motor neuron loss in brainstem nuclei. Lung, liver, spleen, thymus, pancreas, and placenta were not affected.
The observed gross and microscopic lesions are very similar to lesions caused by other viruses of the genus Orthobunyavirus: Akabane virus, 21,29 Aino virus, 32 and Cache Valley virus. 8
Cases of polio(encephalo)myelitis occurred during the beginning of the epizootic, which is presumably related to late-gestation infection and corresponds with the introduction of SBV-infected Culicoides spp mid-August 2011 and exposure of naive pregnant ruminants. 37 Late gestational bunyaviral infections cause nonsuppurative inflammation in the brain and spinal cord. 4 Cases of polioencephalomyelitis due to Akabane virus have also been reported in neonatal calves, suggesting late intrauterine infection. 15,17 Noteworthy, a 10-day-old calf (not included in this study) showed recumbency from birth and polioencephalomyelitis due to SBV. This calf was born in April 2012, implicating that SBV was still circulating after the initial outbreak period. 30
Polioencephalomyelitis was noted in 16 of 20 and 3 of 5 RT-qPCR–positive lambs and calves, respectively. However, 56 of 102 (55%) malformed lambs and 172 of 204 (84%) calves tested RT-qPCR negative. Most lambs and calves with severe CNS lesions were presumed to have been infected early in gestation and were virus negative because they had more time to develop an effective immune response and clear the virus. 6 The large difference in RT-qPCR positivity between lambs (45%) and calves (16%) also may be explained by the longer gestational period of calves compared with lambs, giving calves more time to clear the virus. 11,36
Immunohistochemistry demonstrated SBV antigen in neurons and neuroblasts in the CNS; however, SBV was not detected in other organs, including skeletal muscle tissue and placenta. These findings are similar to those of other studies of spontaneous SBV in utero-infected lambs and calves. 10 SBV neurotropism has been reported in an experimental mouse infection model. 36 At the moment of the SBV outbreak in the Netherlands, the genus-specific monoclonal antibody, MAb 4H9/B11/F1, was available only for our immunohistochemical investigations, 2 and there was no access to a SBV N protein polyclonal rabbit antiserum, 36 generated in a later phase of the SBV epidemic. The immunohistochemical data showed concordance with results of RT-qPCR, and nonspecific binding of this monoclonal antibody was excluded.
Fifty-six of 61 lambs tested positive with SBV ELISA. This further validates the use of this ELISA test for diagnostic purposes. 35 Two SBV ELISA-negative ovine cases demonstrated polioencephalomyelitic lesions, and both tested RT-qPCR and IHC positive. The 3 remaining SBV ELISA-negative ovine cases showed gross and microscopic lesions, compatible with the arthrogryposis hydranencephaly syndrome, but were also RT-qPCR negative (IHC not tested). All blood samples collected from ovine and bovine dams were SBV VNT positive, which supports the hypothesis that congenital malformations were related to SBV infection. 3 No statistically significant association was found between VNT titer values of the bovine dams and lesion severity in the CNS of their malformed calves.
From experimental infection studies with Akabane and Aino virus, using chick embryos 19,20,25 or primary cultures of fetal bovine brain, 18 it is known that these viruses preferentially infect neuronal and astroglial cells. Interestingly, vascular wall thickening in brain tissue was seen to be associated with necrotizing encephalitis in chickens at 7 days postinfection to form granulomatous lesions at 10 and 13 days postinfection. 19,20 Akabane virus–induced congenital malformations in ruminants revealed that the earlier in gestation that fetal infection occurs, the more severe lesions are, reflecting a large population of vulnerable cells and lack of fetal immunocompetency at earlier stages of gestation. 4 In a prospective study of an Akabane virus outbreak in cattle in Australia, it was observed that hydranencephaly and porencephaly developed after infection between 76 and 104 days of gestation, whereas arthrogryposis developed after infection between 103 and 174 days of gestation. 17 In our cases, no such time-related distinction in appearance occurred. Almost all lambs, with or without cerebral defects, had spinal cord lesions with corresponding arthrogryposis. The proportion of lambs with cerebral defects was high (83%) and remained constant during the period of submissions. The proportion of calves with cerebral defects (53%) was lower, and affected calves usually had additional spinal cord defects, which tended to be more severe compared with calves with an intact cerebrum (Table 2).
Spinal cord lesions caused by intrauterine bunyaviral infections are distinctive and of great value in the differential diagnosis with other teratogenic viruses in ruminants. Hydranencephaly and porencephaly, sometimes combined with cerebellar dysplasia, may be caused by intrauterine infection with BTV. However, fetal BTV infection does not result in spinal cord lesions with associated arthrogryposis and vertebral column malformations. 38,39 Pestiviruses are another virus group with teratogenic potential in ruminants. 13,14 Intrauterine BVDV and BDV infections cause mainly cerebellar dysplasia but may be associated with hydranencephaly, internal hydrocephalus, microencephaly, or porencephaly. 13 Spinal cord lesions, caused by BVDV in fetal calves, are relatively uncommon compared with infected fetal lambs and, if present, are limited to diffuse neuraxial hypomyelination and dependent on viral strain and timing of infection. 28,31 Arthrogryposis and vertebral column malformation have not been reported in cases of fetal pestivirus infection.
Our report exemplifies early recognition of a new emerging syndrome through surveillance of samples for pathologic analysis. SBV infection in ovine and bovine fetuses early in gestation resulted in severe dysplastic CNS lesions, including a distinctive micromyelia, and viral clearance in seropositive offspring, whereas late gestational SBV infections present as an encephalomyelitis with detectable viral RNA and antigen.
Footnotes
Acknowledgements
We thank Dr M. Beer and Dr B. Hoffmann, Friedrich-Loeffler-Institute, Germany, for kindly sharing their RT-qPCR assay for detection of the Schmallenberg virus. We thank Dr W. van der Poel and Dr R. Bouwstra, Central Veterinary Institute, the Netherlands, for carrying out the SBV RT-qPCR and the VNT diagnostic tests. We are grateful to Dr P. Kirkbride, Australian Animal Health Laboratory CSIRO, for generously providing monoclonal antibodies raised against Tinaroo virus. We thank Dr G. Grinwis, Faculty of Veterinary Medicine, Utrecht University, for photographic assistance and Dr S. Kimpfler, GD Animal Health, for critical comments. Results of our findings concerning this emerged orthobunyaviral disease in small and large ruminants were presented at the 2012 Joint Meeting of the European Society of Veterinary Pathology, the European College of Veterinary Pathologists, and the Spanish Society of Veterinary Pathology in León, Spain.
Declaration of Conflicting Interests
The author(s) declared that they had no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Monitoring of animal diseases in the Netherlands is financially supported by the Dutch Ministry of Economic Affairs, Agriculture and Innovation and the Product Board for Livestock and Meat. Pathological examination in this study was financed by the Dutch Ministry of Economic Affairs, Agriculture and Innovation.
