Abstract
Respiratory disease is common in dolphins, primarily affecting pulmonary parenchyma and sparing large airways. Over a 10-year period, 4 captive adult bottlenose dolphins succumbed to chronic, progressive respiratory disease with atypical recurrent upper respiratory signs. All dolphins had severe, segmental to circumferential fibrosing tracheitis that decreased luminal diameter. Histologically, tracheal cartilage, submucosa, and mucosa were distorted and replaced by extensive fibrosis and pyogranulomatous inflammation centered on fungal hyphae. In 3 of 4 cases, hyphae were morphologically compatible with Aspergillus spp and confirmed by culture in 2 cases. Amplification of fungal DNA from tracheal tissue was successful in one case, and sequences had approximately 98% homology to Aspergillus fumigatus. The remaining case had fungi compatible with zygomycetes; however, culture and polymerase chain reaction were unsuccessful. Lesions were evaluated immunohistochemically using antibodies specific to Aspergillus spp. Aspergillus-like hyphae labeled positively, while presumed zygomycetes did not. These cases represent a novel manifestation of respiratory mycoses in bottlenose dolphins.
Mycotic diseases are well documented in captive and free-ranging bottlenose dolphins (Tursiops truncatus).5–7 Common sites of infection include the skin and respiratory tract, the latter typically in the form of pneumonia sparing large airways. 7
Pulmonary aspergillosis is the most commonly reported mycotic infection in cetaceans in general, with many cases having occurred in stranded bottlenose dolphins secondary to dolphin morbillivirus infection.5,7 In captive and free-ranging bottlenose dolphins, documented pulmonary mycoses include infections by opportunistic fungi (Aspergillus spp, zygomycetes, Candida spp) and endemic fungi (Blastomyces dermatitides, Histoplasma capsulatum, and Coccidioides immitis). 7 Second to aspergillosis, zygomycetous infections are the most commonly reported and can be devastating, with rapid dissemination and death. 7
Dolphins and other cetaceans with respiratory mycoses may present with obvious clinical signs of respiratory disease, or clinical signs may be nonspecific. Hematologic and biochemical alterations are similar to other infectious diseases. Clinical course can range from insidious to fulminating. 7 In some cases, fungal pulmonary diseases are diagnosed only on postmortem examination. Fungal identification is usually achieved through correlation of histopathology and culture and, more recently, molecular analysis. In chronic lesions, fungal hyphae may be rare or difficult to distinguish morphologically if degenerate, and in such cases, ancillary diagnostics are paramount for fungal identification.
This case series describes a novel manifestation of respiratory fungal disease in captive bottlenose dolphins. Additionally, molecular and immunohistochemical characterization of the fungal pathogens was attempted.
Materials and Methods
From 1998 through 2008, 4 adult captive Atlantic bottlenose dolphins (T. truncatus) with chronic upper respiratory disease were submitted to the University of Illinois Zoological Pathology Program for postmortem examination (Table 1). All dolphins had a similar prolonged (months to years) clinical history, including one or more of the following: episodic lethargy and inappetence, respiratory “wheezes,” abnormal “honking” behavior, and intermittent leukocytosis.
Signalment and Fungal Characteristics in 4 Captive Bottlenose Dolphins.
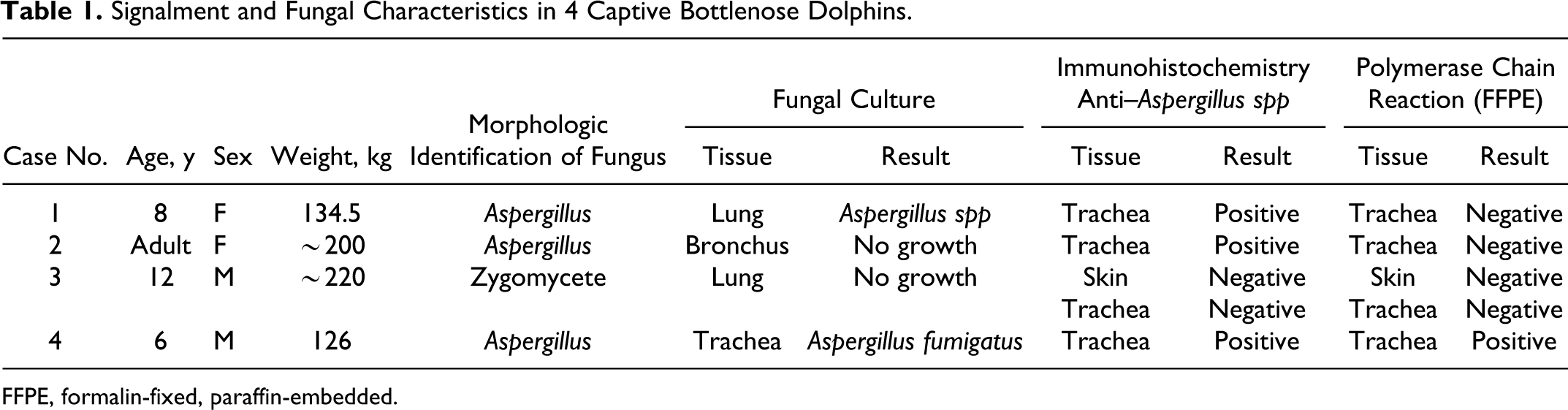
FFPE, formalin-fixed, paraffin-embedded.
Gross necropsies were performed, and a complete set of tissues was fixed in 10% neutral buffered formalin for histologic evaluation. Tissues were routinely processed, paraffin embedded, sectioned at 5 μm, and stained with hematoxylin and eosin. Gomori’s methenamine silver and Gram stains were utilized on selected sections. Fresh tissues from all cases were submitted for aerobic bacterial and fungal culture at time of necropsy.
Immunohistochemistry (IHC) was performed on sections of trachea (all cases) and skin (Case No. 3) using a modified streptavidin–biotin–peroxidase complex method. The primary antibody was mouse monoclonal anti-Aspergillus spp (WF-AF-1: AbD Sertotec, Oxford, UK) diluted 1:300 in 0.1 M phosphate-buffered saline solution. 2 Positive controls included sections of lung from a bird and a dolphin with polymerase chain reaction (PCR) and/or culture-confirmed aspergillosis (Aspergillus fumigatus).
DNA was extracted from 45-μm scrolls of paraffin-embedded blocks of trachea (all cases) and skin (Case No. 3) using a commercially available kit (RecoverAll Total Nucleic Acid Isolation Kit, Cat No. 1975) and amplified using 28S large subunit rRNA generic primers and MicroSeq D2 LSU rDNA primers (Applied Biosystems, Foster City, CA) per the manufacturer’s directions. 8 Both strands of successful amplicons were sequenced using an automated capillary sequencer (Applied Biosystems 3730XL) at the University of Chicago Cancer Sequencing Facility and compared for homology in GenBank genetic sequence database.
Results
Fungal morphologic, culture, molecular, and IHC data are summarized in Table 1. Consistent findings in all cases were moderate to severe, segmental to circumferential tracheal and/or bronchial stenosis and multifocal to regional mucosal ulceration with multiple yellow, green and tan, friable, adherent mats or plaques (Fig. 1). In Case Nos. 3 and 4, the entire trachea was affected, extending from the larynx to the carina. Tracheal and bronchial stenosis ranged from approximately 25% to almost complete occlusion primarily due to mural fibrosis and inflammation (Fig. 2), with lesser luminal accumulation of caseous material in some cases (Fig. 1). Histologic lesions were similar in all cases and included a range of changes: mucosal ulceration, necrosis, suppurative and pyogranulomatous inflammation, and exuberant immature to mature fibrous tissue with partial to complete effacement of tracheal cartilages. In most cases, extensive regions of tracheal mucosa and superficial submucosa were absent (ulcerated). Ulcerated regions were covered by layers of clumped fibrin with enmeshed karyorrhectic (necrotic) cellular debris, intact and necrotic neutrophils, hemorrhage, variable numbers of fungal hyphae, and multiple aggregates of bacteria (Case Nos. 1 and 4) (Fig. 3). Adjacent intact mucosal epithelium often had squamous metaplasia (Case Nos. 1 and 3). The submucosa contained nodular accumulations of intact and necrotic neutrophils and cellular debris centered on small numbers of variably apparent fungal hyphae and surrounded by concentric bands of epithelioid macrophages and few multinucleated giant cells (pyogranulomas). Few sections had evidence of cartilage invasion by fungal hyphae with associated inflammation (Fig. 4). In Case Nos. 1, 2, and 4, hyphae were generally abundant, 6–15 μm in diameter, septate, and parallel walled with acute-angle, dichotomous branching, consistent with Aspergillus spp (Fig. 5). In Case No. 3, fungal tracheitis was more chronic with extensive mural fibrosis and markedly decreased (approximately 55%) luminal diameter (Fig. 2) and a fistulous tract with rare remnant intralesional fungal hyphae. Hyphae were 4–9 μm in diameter, nonparallel walled, aseptate with acute- to right-angle branching, most compatible with a zygomycete. Also in Case No. 3, acute dissemination was evident with severe cerebral and cutaneous necrotizing vasculitis and infarction with large numbers of similar intralesional, intravascular fungal hyphae. Fungal hyphae were positively stained with Gomori’s methenamine silver in all cases (Fig. 5).
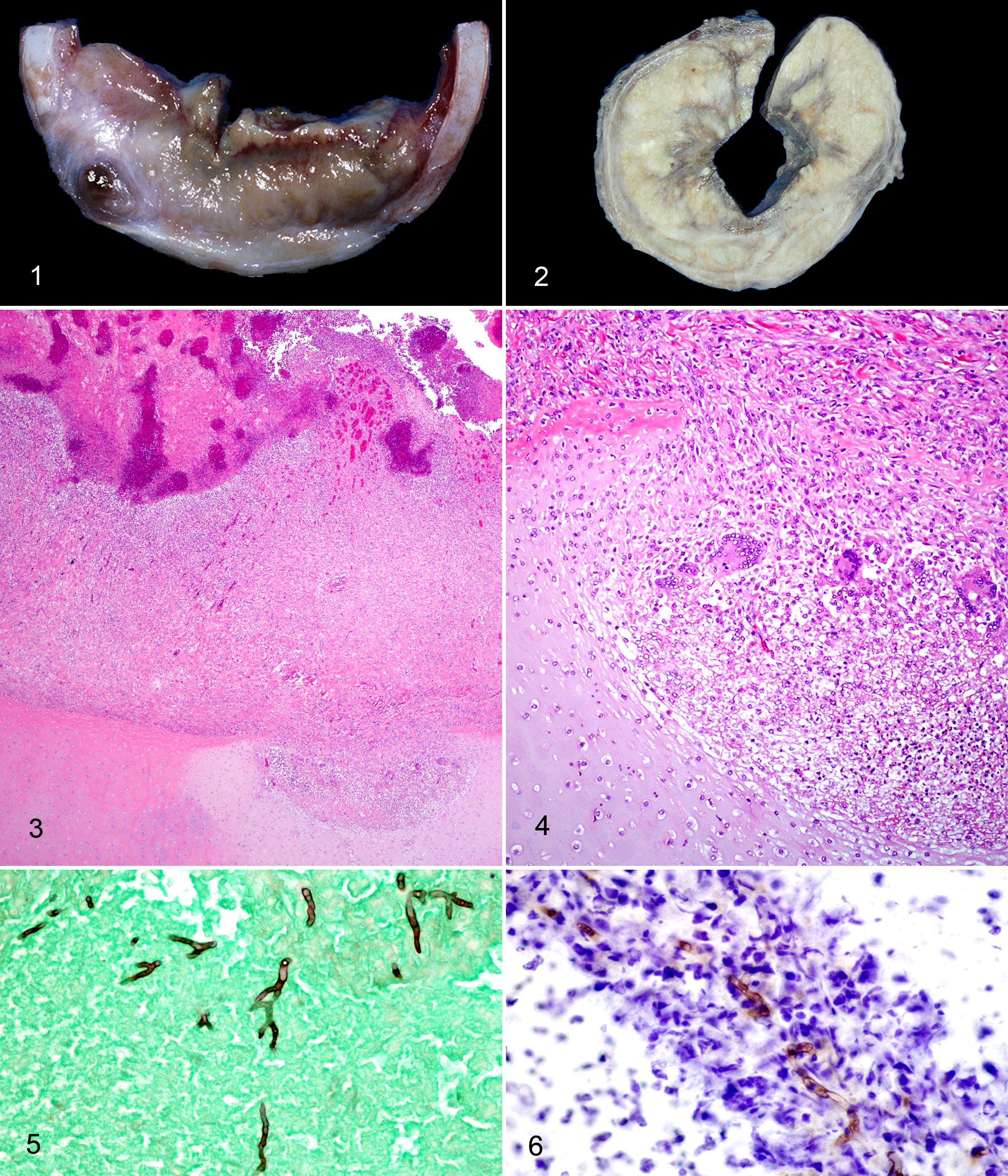
Case Nos. 1 and 4 also had pyogranulomatous and necrotizing bronchopneumonia with intralesional fungal hyphae and Gram-positive cocci. In Case No. 4, bacteria were confirmed as Staphylococcus intermedius by culture. Culture of lung tissue from Case No. 1 yielded only contaminants. Bronchopneumonia in Case No. 4 was subacute. Bronchopneumonia in Case No. 1 was chronic, with fibrosis surrounding regions of necrosis and inflammation, though fibrous tissue was of lesser quantity and less mature than in the trachea. Case No. 2 lacked significant pneumonia but did have multifocal alveolar accumulations of proteinic fluid with linear aggregates of fibrin and scattered hemorrhage and neutrophils, suggestive of acute respiratory distress syndrome. Case No. 3 did not have pneumonia but did have histologic changes reminiscent of the condition chronic obstructive pulmonary disease, with interstitial and peribronchiolar mononuclear infiltrates and fibrosis with emphysema and abundant intra-alveolar mucus.
Representative sections of trachea in Case Nos. 1, 2, and 4 demonstrated positive and specific labeling of many intact and some fragmented fungal hyphae with minimal background staining using murine monoclonal anti–Aspergillus spp (wall fraction) antibodies (Fig. 6). Positive controls had consistent and specific labeling of fungal hyphae using primary antibody. Sections of trachea and skin (Case No. 3) and negative controls had no immunolabeling and negligible background staining.
In Case No. 4, extraction and amplification of fungal DNA was successful using real-time PCR on fresh-frozen tissues and traditional PCR on formalin-fixed paraffin-embedded tissues. A 228–base pair amplicon sequence had approximately 98% homology to the 28S large subunit rRNA of A. fumigatus. Amplification of fungal DNA was not successful in Case Nos. 1-3 using formalin-fixed, paraffin-embedded tissues.
Discussion
Gross and histologic findings were similar in all cases and unique in comparison to previously described lesions of bottlenose dolphins with mycotic respiratory disease. In contrast to previous reports, fungal organisms invaded, effaced, and distorted essential components of the trachea and bronchi, most notably the cartilaginous rings. This destruction and distortion of the airways resulted in significant reduction of luminal diameter and disturbances in ventilation. Similar lesions have been documented in humans and attributed to Aspergillus spp and rarely zygomycetes.1,2,10 Recently, invasive Aspergillus infection limited to the tracheobronchial tree in humans has been recognized as a distinct syndrome designated isolated invasive Aspergillus tracheobronchitis. This disease is believed to occur most commonly in patients with impaired local defense mechanisms of the airways, regardless of systemic immune status.4,10
In the 4 dolphins presented in this series, there were no clinical or histologic abnormalities suggestive of immune compromise. Similar to isolated invasive Aspergillus tracheobronchitis in humans, alterations of local mucosal immune function could be an important factor in the development of fungal tracheitis in these dolphins. Though the immunology of dolphins and other cetaceans has been studied, several aspects of immune functions remain unclear, in particular localized immunity in organ systems such as the respiratory tract. 3 In situ defense mechanisms are important factors in the development of respiratory disease, and these mechanisms in aquatic mammals likely differ to some degree from those of terrestrial mammals. 7 Furthermore, the unique anatomical features of the dolphin respiratory tract, such as the lack of nasal sinuses and protective hairs, may play a role in their susceptibility to respiratory infections, including mycoses.
In 2 cases (Case Nos. 2 and 3), upper airway disease led to secondary pulmonary syndromes in the absence of severe pneumonia. In Case No. 2, intra-alveolar edema and fibrin strands were noted and analogous to acute respiratory distress syndrome, which is reported commonly in humans undergoing mechanical ventilation. Speculatively, mechanical alveolar stress from prolonged forceful breathing may have led to these changes in this individual. Histologic pulmonary changes noted in Case No. 3 were reminiscent of chronic obstructive pulmonary disease, which likely contributed significantly to respiratory compromise. Though cerebral infarction was considered the ultimate cause of death, the chronic tracheal and lung lesions served as a source of fungal dissemination and intracranial fungal infection.
In Case Nos. 1 and 4, upper airway disease was accompanied by necrotizing and pyogranulomatous bronchopneumonia with intralesional fungi and Gram-positive cocci. In both cases, bronchopneumonia, while related to tracheitis, was of lesser chronicity based on morphologic features and thus considered secondary to tracheal disease. Intralesional bacteria were considered secondary colonizers but contributory to severity and progression of pneumonia.
Generally, culture and/or molecular characterization are considered essential to fungal identification, as there are similarities and overlap of histologic morphologies. Despite this, appropriate samples for fungal culture or molecular analysis are not always collected. Cultures can also be time-consuming and unreliable, as some fungal species do not grow readily in the laboratory. 9 Fungal culture was successful in only 2 of the 4 cases; therefore, additional methods were employed to identify fungal pathogens. Unfortunately, amplification of fungal DNA from paraffin-embedded tissues in Case Nos. 1-3 was unsuccessful. One plausible factor influencing PCR was that in these chronic lesions, the relative concentration of intralesional organisms was low, so initial quantities of fungal DNA were also likely very low. Additionally, tissues from Case Nos. 1-3 spent longer periods in formalin, and subsequent DNA fragmentation and degradation may have also influenced unsuccessful PCR amplification.
Case No. 4 had PCR- and culture-confirmed aspergillosis and served as an internal control for immunohistochemical analysis. Fungal DNA was amplified from both fresh-frozen and paraffin-embedded tissues. Notably, tissues were immersion fixed in formalin for only a short period. Case Nos. 1, 2, and 4 had positive immunolabeling of intralesional fungal hyphae using antibody to the wall fraction of Aspergillus spp. This demonstrates that in situations in which histologic morphology is not definitive and fungal culture and molecular diagnostics are unavailable or inconclusive, IHC may be utilized to characterize fungal pathogens. Though immunohistochemical cross-reactivity has been reported, specifically between Aspergillus spp and zygomycete species, fungal hyphae consistent with zygomycetes in Case No. 3 had no positive labeling with the primary antibody. 9 At this time, however, antibodies specific to common fungal pathogens are not universally available at veterinary diagnostic laboratories.
Fungal pneumonia is the most common respiratory mycotic disease in dolphins; however, this case series demonstrates that fungal organisms may localize to invade and obliterate normal components of the upper respiratory tract, leading to airway obstruction and respiratory compromise. Complete postmortem examinations including histologic evaluation and ancillary diagnostics (culture, PCR, IHC) have and will continue to provide valuable insight into respiratory mycoses of dolphins and other cetaceans. From this case series, it is recommended that fungal tracheitis be included as a differential in captive individuals with recurrent upper respiratory signs. Additionally, determining further correlations between clinical and postmortem findings is critical to better understanding pathogenesis and manifestations of fungal diseases in dolphins.
Footnotes
Acknowledgements
We would like to thank the veterinarians and support staffs at respective institutions for providing case materials; Dr. Jennifer Landolfi and Stacy Schultz for their technical assistance; and the Histology Lab of the University of Illinois Veterinary Diagnostic Laboratory for slide preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
