Abstract
Although abnormal sexual behavior, including boar-to-boar mounting with anal penetration, is recognized in pubescent pigs, reports of the pathologic consequences are scarce. A 7-month-old male minipig, housed with age-matched males, died within 1 day of the onset of lethargy and reluctance to rise. At necropsy, 2 rectal tears were identified as the cause for fibrinous peritonitis, and spermatozoa were identified in the pelvic and peritoneal cavity by light and transmission electron microscopy. According to DNA typing results, using 11 porcine microsatellites, the intraperitoneal semen was from at least 2 pen mates. The prohibition of castration of fattening pigs, implemented or planned in multiple European countries, could increase the risk of rectal perforation in co-housed pigs.
Keywords
Abnormal sexual behavior in pubescent pigs includes boar-to-boar mounting with anal penetration 11 ; however, the resulting anorectal trauma has not been characterized. In contrast, rectal tears from various causes are well documented in horses and cows. 3,6,13
DNA fingerprinting, employing microsatellite markers, is a forensic tool for paternity testing or assigning specimens to individuals 1 and can be applied to livestock and companion animals. 8 Microsatellites are short tandem repeats of 2 to 7 bases. The number of repeats varies among individuals, and microsatellite motifs are randomly distributed over the genome. 12 The length of both alleles can be assessed for each microsatellite employing polymerase chain reaction (PCR) technology and locus-specific primers flanking the tandem repeats. The combination of the allelic information of multiple microsatellites yields a unique pattern, the so-called genetic fingerprint, that identifies individuals. 1
Case History and Necropsy Findings
A 7-month-old, 26.8-kg, intact male minipig, in a pen of 23 age-matched boars, died within 24 hours of signs of lethargy and reluctance to move. At necropsy, the peritoneum was coated with fibrinous exudate, and about 25 ml of a colorless, transparent, gelatinous substance was in the pelvic and peritoneal cavity. The rectum, 10 to 20 cm orad to the anus, was dilated to 2.9 cm (in comparison to 1.8 cm diameter in a grossly normal rectum nearer the anus). Sheets of fibrinous exudate adhered to the rectal serosa. An 8-mm-diameter round tear in the ventral rectal mucosa was approximately 9.5 cm orad to the anus (Fig. 1). A second crescentic and, in its orad half, transmural tear (6.3 cm long and up to 1.2 cm wide) was in the floor of the rectum about 15.5 cm orad to the anus. Mild colonic impaction was observed.
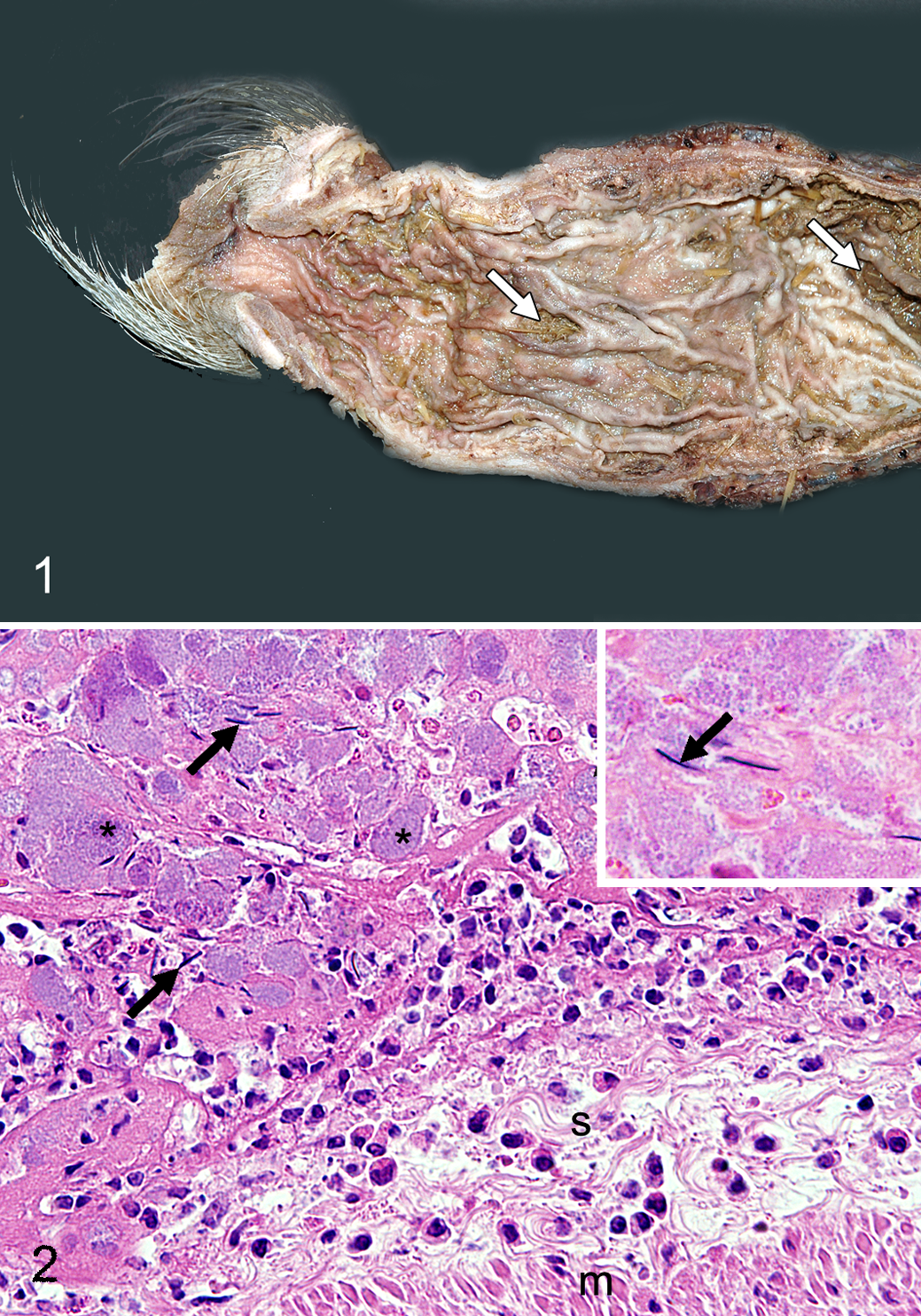
Tissue samples were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin. Histologically, the peritoneal and rectal serosal fibrinous exudate included neutrophils, bacterial colonies, and structures interpreted as spermatozoal heads (Fig. 2). The rectal tears were associated with intramural hemorrhage, fibrin deposition, and moderate neutrophilic infiltration. The number of neutrophils was also increased in sinuses of the mesenteric lymph nodes. Multifocal to laminar hemorrhage was observed in the adrenal cortices.
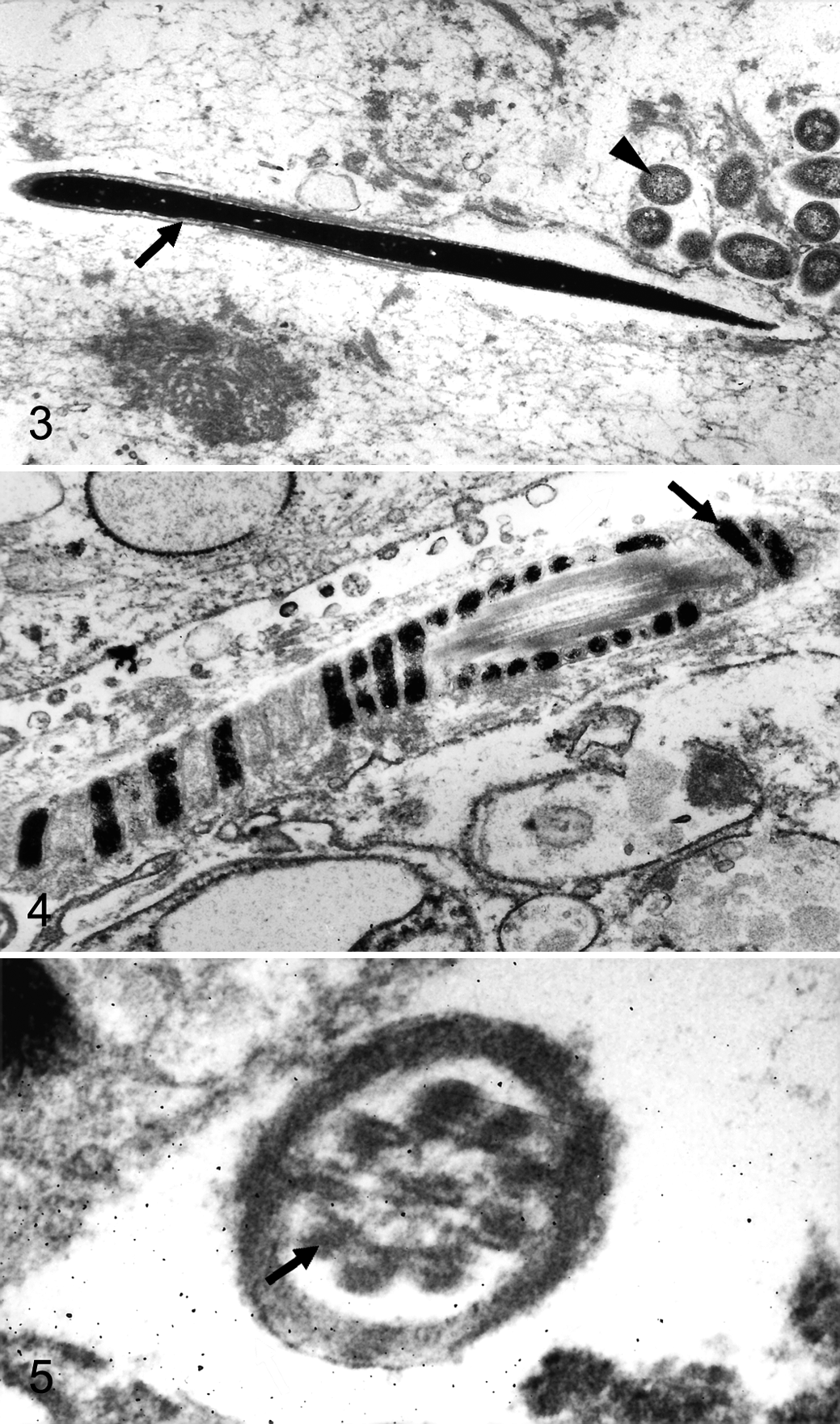
For electron microscopy, tissue samples were postfixed in 5% glutaraldehyde followed by 1% osmium tetroxide and embedded in epoxy resin. Ultrathin sections were contrasted with uranyl acetate and lead citrate and analyzed with a transmission electron microscope (EM10C; Zeiss, Oberkochen, Germany). Ultrastructurally, the peritoneal exudate contained spermatozoa in a meshwork of fibrin. Spermatozoal heads had a rod-shaped (about 7 × 0.3 μm) heterochromatic nucleus covered by the acrosome (Fig. 3). Middle pieces had central axial filaments surrounded by helically arranged mitochondria (Fig. 4). Principal pieces had central axonemal microtubules surrounded by 9 dense fibers, creating a 9 + 9 × 2 + 2 cross-sectional pattern, and a peripheral submembranous fibrous sheath (Fig. 5). Coccobacillary bacteria had a diameter of ~0.5 μm and a trilaminar membrane (Fig. 3).
Blood was collected from the jugular vein of the dead boar and its pen mates. Genomic DNA was extracted from blood leukocytes and from a sample of the semen from the pelvic and peritoneal cavity. PCR was carried out in a 12-μl reaction volume containing 10 ng DNA, 1.5 mM MgCl2, 100 μM deoxynucleoside triphosphates, 5% dimethylsulfoxide, 5 pmol of each primer, and 1 unit Taq polymerase (QBiogene, Heidelberg, Germany). The pigs were genotyped employing primer pairs for 11 microsatellite markers (SW174, SO217, SO097, SW24, SW632, SO068, SW902, SW787, SW1023, SW295, SO225) distributed on 9 chromosomes. The sequences, annotations, and specific annealing temperatures of these primer pairs were derived from the database of the swine genome mapping project (Agricultural Research Service, United States Department of Agriculture; http://www.marc.usda.gov/genome/swine/swine.html). 12 Cycling was performed using the following conditions: (1) initial denaturation at 94°C for 5 minutes; (2) amplification over 35 cycles with 94°C denaturation for 30 seconds, annealing at the primer pair–specific annealing temperature for 30 seconds, and elongation at 72°C for 60 seconds; and (3) final extension at 72°C at for 10 minutes. The resulting DNA fragments were size fractionated on an automatic LI-COR sequencer (LI-COR, Lincoln, Nebraska), and the genotypes were assigned by visual examination. 10 Six of the 11 microsatellite markers (SW632, SO068, SW902, SW787, SW295, SO225) had alleles in the semen sample that were not detected in the blood leukocytes of the dead pig (Suppl. Table S1, found online at http://vet.sagepub.com/suppelemental).
Notably, marker SW787 had 3 alleles in the semen, all of which differed from those in the blood of the dead boar (Fig. 6). For 3 other microsatellites (SW174, SO097, SW1023), only alleles like those of the dead boar were observed in the semen. Valid results were not obtained for 2 microsatellites (SO217, SW24).
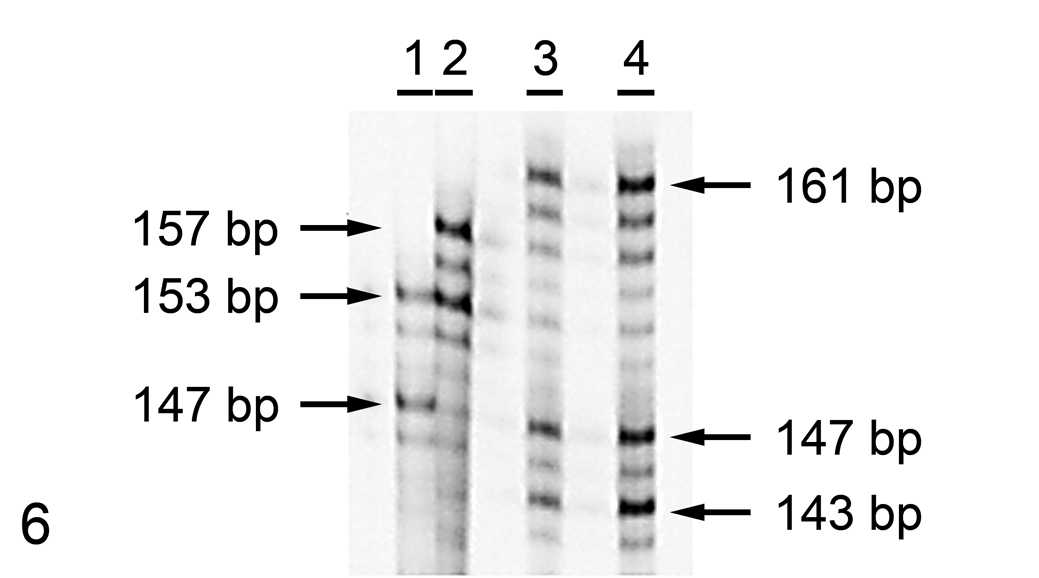
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis gel of microsatellite SW787 in semen from the peritoneal cavity. Three alleles (143, 147, and 161 base pairs [bp]) differ from those of the blood of the boar (153 and 157 bp). Lane 1 = reference sample with 2 alleles of 147 and 153 bp; lane 2 = blood; lanes 3 and 4 = semen from the peritoneal cavity (2 independent extractions). The length of the alleles from the reference sample and the blood of the boar is indicated on the left; that of alleles in the semen is on the right.
Discussion
The causes for peritonitis in pigs include ascending bacterial infections from castration wounds, perforating gastric ulcers, rectal strictures, Glässer’s disease (Haemophilus parasuis), Mycoplasma hyorhinis, Streptococcus suis type 2, intestinal anthrax, tuberculosis, and Stephanurus dentatus larva. 2 In this boar, death was attributed to circulatory failure secondary to septic shock, resulting from rectal perforation. The detection of semen in the pelvic and peritoneal cavity was consistent with anal penetration and rectal perforation from boar-to-boar mounting. The detected spermatozoa were morphologically consistent with those of boars. 5,9 The location of the rectal tear ~15.5 cm orad to the anus is in agreement with the penile length of boars. 14 Furthermore, the location of both tears on the ventral side of the rectum is typical in human anorectal trauma due to sexual assault. 7 In contrast, equine rectal tears, which are most commonly iatrogenic (associated with rectal examination), usually develop on the dorsal side of the rectum at a species-specific weak point in the tunica muscularis. 6 The adrenocortical hemorrhage was attributed to sepsis (Waterhouse-Friedrichsen syndrome).
Genetic fingerprinting and DNA typing methods have been rarely used in veterinary pathology because of cost and lack of standardized laboratory protocols for species, individual, and sex identification. 8 However, comprehensive porcine genomic maps and related marker sets are available, and DNA typing is routinely used in livestock breeding programs. 12 In this case, the identification of alleles within the semen that differed from those in blood leukocytes of the pig proved that the ejaculate was derived from at least 2 different boars. This report highlights that microsatellite marker technology can fail to identify the individual source of the semen in the case of mixed samples. Furthermore, anal penetration and rectal perforation could occur without ejaculation.
In summary, the pathologic and molecular findings are reported in a porcine case of fatal anorectal trauma with semen in the pelvic and peritoneal cavity. Pathologists should include penile anal penetration with rectal trauma and perforation in the differential diagnosis for peritonitis in pigs. The implemented or planned prohibition of castration of fattening pigs in multiple European countries may increase the prevalence of this condition in pigs housed with boars. 4
Footnotes
Acknowledgements
The authors thank Bettina Buck and Kerstin Rohn for technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Finding
The authors received no financial support for the research, authorship, and/or publication of this article.
