Abstract
An ongoing canine distemper epidemic was first detected in Switzerland in the spring of 2009. Compared to previous local canine distemper outbreaks, it was characterized by unusually high morbidity and mortality, rapid spread over the country, and susceptibility of several wild carnivore species. Here, the authors describe the associated pathologic changes and phylogenetic and biological features of a multiple highly virulent canine distemper virus (CDV) strain detected in and/or isolated from red foxes (Vulpes vulpes), Eurasian badgers (Meles meles), stone (Martes foina) and pine (Martes martes) martens, from a Eurasian lynx (Lynx lynx), and a domestic dog. The main lesions included interstitial to bronchointerstitial pneumonia and meningopolioencephalitis, whereas demyelination—the classic presentation of CDV infection—was observed in few cases only. In the brain lesions, viral inclusions were mainly in the nuclei of the neurons. Some significant differences in brain and lung lesions were observed between foxes and mustelids. Swiss CDV isolates shared together with a Hungarian CDV strain detected in 2004. In vitro analysis of the hemagglutinin protein from one of the Swiss CDV strains revealed functional and structural differences from that of the reference strain A75/17, with the Swiss strain showing increased surface expression and binding efficiency to the signaling lymphocyte activation molecule (SLAM). These features might be part of a novel molecular signature, which might have contributed to an increase in virus pathogenicity, partially explaining the high morbidity and mortality, the rapid spread, and the large host spectrum observed in this outbreak.
Keywords
Canine distemper virus (CDV) is a single-stranded, nonsegmented, negative-sense RNA virus of the genus Morbillivirus of the family Paramyxoviridae. CDV encodes for 6 structural proteins: matrix (M), fusion (F), hemagglutinin (H), nucleocapsid (N), polymerase (L), and phosphoprotein (P). Among these, the H protein has been shown to be pivotal for virus–host interaction through engagement of the viral cellular receptors on the host cells, for cytopathogenicity and cell-to-cell transmission. 25,41,52,55 Additionally, the H gene has been reported to frequently undergo mutations, which makes it a suitable target to assess genetic variations among different CDV strains. 4, 7, 18, 20, 22, 25, 28, 31, 32, 35, 41, 57
CDV has worldwide distribution and high pathogenic potential. It infects numerous wild and domestic carnivores, along with occasional infections in “unusual” hosts, such as nonhuman primates and collared peccaries (Tayassu tajacu). 1,3,48,56
Recent evidence of CDV infection in wild carnivores in Europe was reported in Spanish red foxes (Vulpes vulpes), genets (Genetta genetta), wolves (Canis lupus), stone marten (Martes foina), and Iberian lynx (Lynx pardinus). 26,27,33,42 Starting in 2006, an outbreak of canine distemper (CD) affecting red foxes, Eurasian badgers (Meles meles), and stone martens was detected in Italy. 9,29,36 Similarly, a CD outbreak was observed in 2008 in wildlife of southern Bavaria, Germany, affecting the same species. 46
In Switzerland, local CD epidemics were occasionally recorded in stone martens in the past. 40 The Centre for Fish and Wildlife Health (FIWI) at the University of Bern (Switzerland) has been performing general health surveillance on wildlife for decades, and isolated CD clinical cases were occasionally recorded in this species. Until 2008, no clinical cases had been diagnosed in red foxes and Eurasian lynx (Lynx lynx) despite seroprevalence of 26.4% and 24.3% in these 2 species, respectively. 34,37 In contrast, in the spring of 2009, a sudden emergence of clinical cases of distemper was observed in red foxes and badgers along the Swiss border with Liechtenstein and Austria. Since then and throughout 2010, the front of the epidemic has been moving westward within Switzerland. Numerous wild carnivores, including red foxes, Eurasian badgers, stone and pine martens (Martes martes), and a Eurasian lynx, with clinical signs consistent with neurologic and/or respiratory disease were submitted to the FIWI for complete diagnostic workup.
Differently from past local CDV outbreaks and isolated cases, the current epidemic was characterized by a nationwide territorial distribution, a larger spectrum of affected species, and high morbidity and mortality, especially in red foxes and badgers. These findings were suggestive of either a novel aggressive CDV strain recently arrived in the region or a lethally mutated resident one. In this study we tackled these questions through the characterization of the pathologic changes associated with this outbreak and the dissection of the molecular features of one of the Swiss CDV strain, in the attempt to link their molecular signature to their presumptive increased virulence. Finally, we formulated a hypothesis concerning the origin of the Swiss CDV strain.
Material and Methods
Animals
The first 50 animals confirmed CDV positive among those sent to the FIWI from January 2009 to mid-August 2010 were used for this study: 39 red foxes, 8 Eurasian badgers, 2 stone martens, and 1 pine marten. Additionally, a CDV-positive Eurasian lynx and a CDV-vaccinated dog from the 2009–2010 outbreak, as well as a stone marten from a 2002 CDV outbreak, were included in this study, but findings are reported separately in this article.
Pathology
Full necropsy and histologic examination was carried out on all wild carnivores included in this study, whereas for the domestic dog, only the brain was submitted for neuropathologic examination. Representative sections of brain, lung, heart, liver, spleen, kidney, stomach, duodenum, jejunum, ileum, cecum, colon, urinary bladder, tracheobronchial lymph nodes, adrenal glands, and skin were systematically collected along with any additional tissue showing abnormal changes. Tissues were fixed in 10% buffered formalin and embedded in paraffin. Five-micron-thick tissue sections were then obtained and stained with hematoxylin and eosin. Special stainings, including Gram, Giemsa, Ziehl-Neelsen, and Grocott, were applied to selected samples as needed.
Immunohistochemistry (IHC) was performed according to an established protocol 5 on the sections of brain and/or lung of 42 of 50 foxes and mustelids, including primarily those where the lesions were consistent, with but not specific of distemper virus infection (absence of convincing inclusion bodies). IHC was similarly performed on the lung of the Eurasian lynx and the brain of the CDV-vaccinated dog from the 2009–2010 CDV outbreak and on the lung of the stone marten from the 2002 CDV outbreak.
The remaining 8 cases, for which IHC was not performed, showed consistent lesions with obvious viral inclusion bodies.
An animal was considered positive for CDV infection when at least one of the following findings was observed: presence of characteristic intracytoplasmic or intranuclear eosinophilic inclusions, presence of viral antigen detected by IHC (CDV N protein), 5 or positive reverse transcription polymerase chain reaction (RT-PCR) for CDV N protein (with sequencing).
Cell Cultures and Virus Isolation
Vero and Vero-SLAM were grown as described elsewhere. 58 Tissue samples from 3 red foxes (W10/301/red-fox/Ch/2010-JF810106-lung, W10/593/red-fox/Ch/2010-JF810121-brain, W10/845/red-fox/Ch/2010-JF810108-brain) and a Eurasian lynx (W10/2733/Eurasian-lynx/Ch/2010-JF810109-brain) were selected for virus isolation on Vero cells expressing the signaling lymphocyte activation molecule (SLAM) receptor (universal Morbillivirus receptor; ie, Vero-SLAM cells) 50 as previously described. 23 Following inoculation of the monolayer, the cells were incubated at 37°C and checked daily for the presence of cytopathic effects.
Reverse Transcription Polymerase Chain Reaction
Three RT-PCRs were performed during the study. Total RNA extraction (from lung tissue unless differently specified) was carried out with the RNeasy Mini Kit (Qiagen, Hombrechtikon, Switzerland), while cDNA synthesis was performed using M-MLV reverse transcriptase (Promega, Madison, Wisconsin) according to the instruction of the manufacturer. The RT-PCR reactions were performed according to the following protocols (see Table 1)
Techniques Adopted for Investigating the 2009–2010 Canine Distemper Outbreak in Swiss Wildlifea
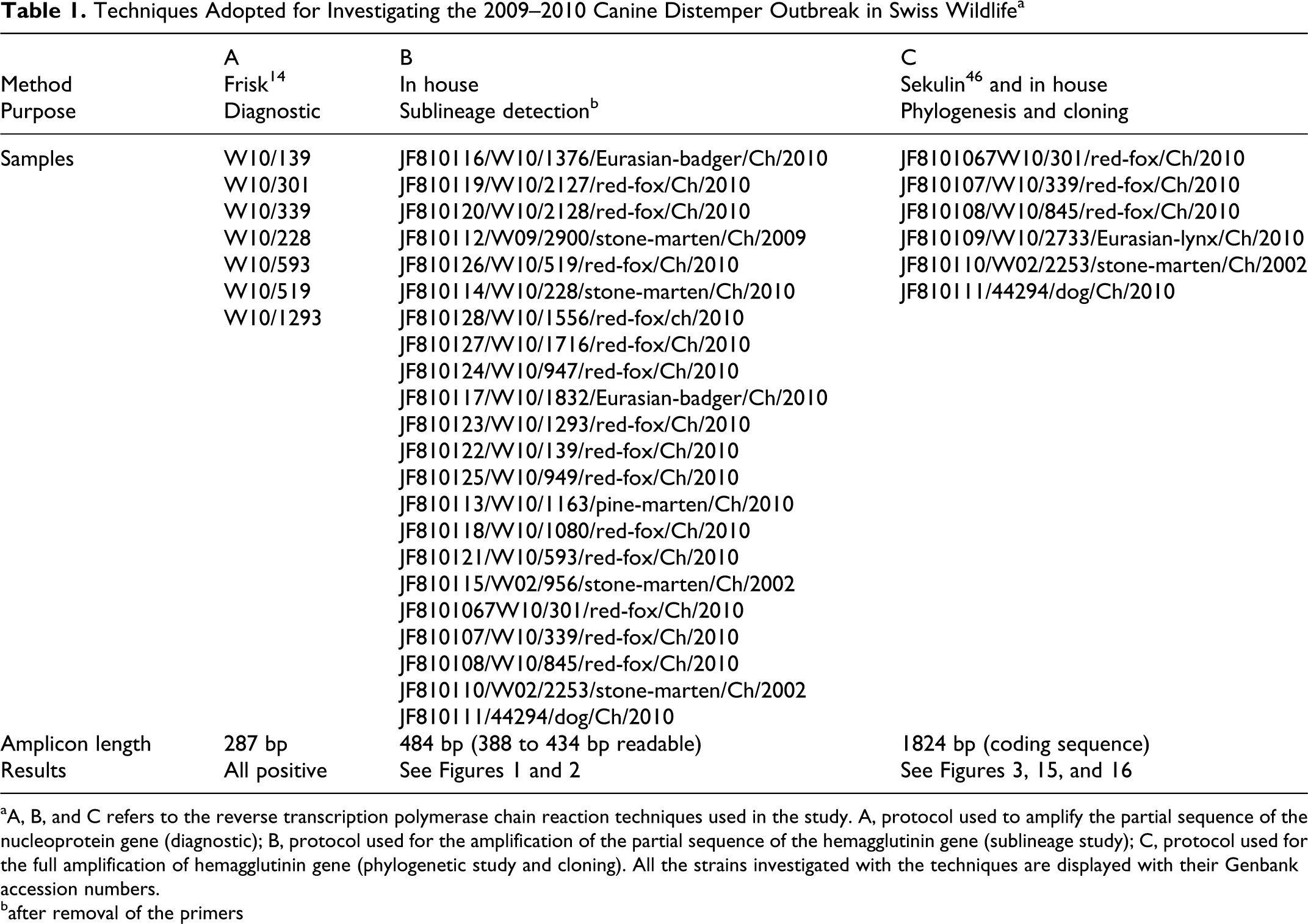
aA, B, and C refers to the reverse transcription polymerase chain reaction techniques used in the study. A, protocol used to amplify the partial sequence of the nucleoprotein gene (diagnostic); B, protocol used for the amplification of the partial sequence of the hemagglutinin gene (sublineage study); C, protocol used for the full amplification of hemagglutinin gene (phylogenetic study and cloning). All the strains investigated with the techniques are displayed with their Genbank accession numbers.
bafter removal of the primers
Protocol A
Amplification of the partial sequence of the N protein was performed for diagnostic purpose (according to the protocol by Frisk et al 14 ) to confirm the first case diagnosed as positive by histopathology and when histologic examination, including IHC, yielded questionable results (n = 6; Table 1).
Protocol B
In the attempt to determine if one or multiple sublineages of CDV were circulating during the Swiss outbreak, we amplified the partial sequence of the H gene of 22 CDVs strain from tissues of infected animals.
These animals were representative of all the species examined (except for the lynx) and, if available, of each Swiss canton where the disease was observed (Figs. 1, 2). Also included in the analysis were the vaccinated domestic dog (44294/dog/Ch/2010- JF810111) and the CDV-infected stone marten from the 2002 local CDV epidemic (W02/2253/stone-marten/Ch/2002-JF810110), along with an additional stone marten from the same epidemic (which was not included in the pathological study; W02/956/stone-marten/Ch/2002-JF810115). Specific forward (5′-CTTGCTTGCTATCACTGGAG-3′) and reverse (5′-TTTTGAAATCAAAGACATGG-3′) primers (7.5 μl each of a 0.15 μM solution) amplifying the partial sequence of the H gene were admixed to 25 μl of 2× HotStar Master Mix (Qiagen), 2 μl of cDNA from the above reverse transcription reaction and RNAse/DNAse–free water up to 50 μl. Following initial denaturation at 95°C for 15 minutes, the reaction comprised 35 cycles, including a denaturation step at 95°C for 1 minute, an annealing step at 52°C for 30 seconds, and an extension step at 72°C for 1 minute. A final extension step, at 72°C for 10 minutes to exhaust the polymerase, followed.
Protocol C
At least one representative sample from each of the detected sublineages (see protocol B, Fig. 1) along with that from the lynx (W10/2733/Eurasian-lynx/Ch/2010-JF810109) were selected for full amplification of the H gene for phylogenetic analysis (Fig. 3). The total RNA was extracted from infected Vero-SLAM cells, from lung, or from formalin fixed-paraffin embedded sections. Two microliters of the cDNA was used as template for the PCR amplification, which was performed according to Sekulin. 46
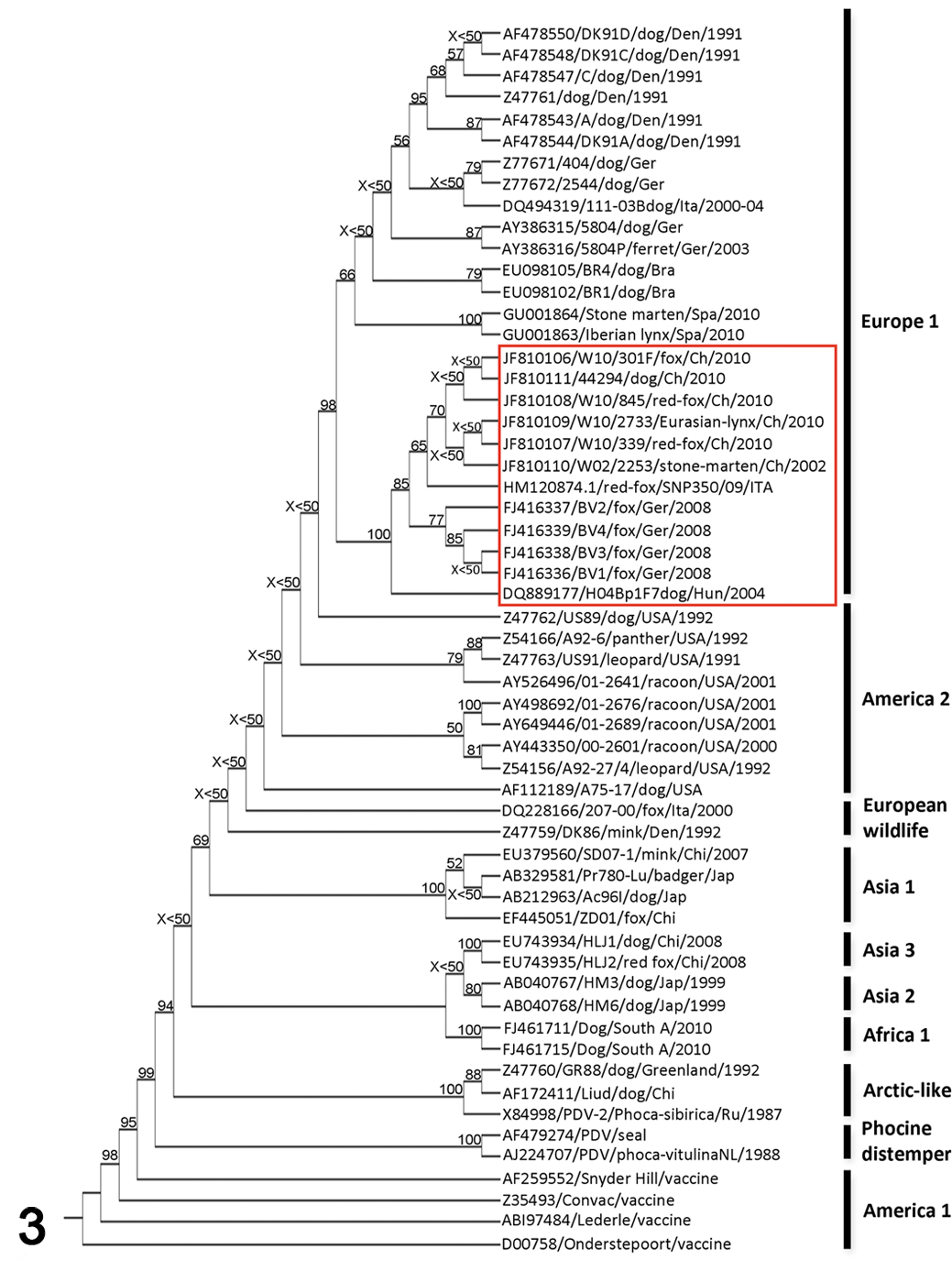
Phylogenetic analysis of 57 canine distemper virus (CDV) strains based on amino acid sequences of the H protein (derived from the nucleotide sequence accession numbers). In the phylogenetic tree (distance-matrix analysis), the CDV strains are indicated with their Genbank identifications and additional available information (see also supplemental file 2). A red box is highlighting the group of hemagglutinin sequences clustering with those from the canine distemper outbreak in wild carnivores in Switzerland in 2009–2010 (comprising the strains associated with recent CDV outbreaks in Italy and Germany and a Hungarian dog isolate). Geographic groups are reported on the right side of the tree, while the bootstrap values are reported at the intersections of the tree branches (bootstrap values lower than 50 are reported as X<50).
Sequencing, Sequence Comparison, and Phylogenetic and Glycosylation Sites Analysis
The RT-PCR products were submitted for full sequencing using an automated sequencer (capillary sequencer ABI3730XL) using BigDye chemistry. The partial nucleotide sequences of the hemagglutinin gene from the 27 CDV strain shown in Figure 1 (see also Fig. 2) were aligned and compared using Clustal W program (Biology Workbench 3.2, San Diego Supercomputer Center, http://workbench.sdsc.edu/), and nucleotide substitutions were analyzed. 44
The published amino acid sequences of the H protein of 51 CDV strain, along with those of 6 novel Swiss strain, were used for phylogenetic analysis (Fig. 3). Multiple alignments were performed with CLUSTAL W, 44 while the phylogenetic trees were constructed using software programs included in the PHYLIP program package (Phylogeny Inference Package 3.69). 13 For distance matrix analysis, the aligned amino acid sequences were first processed with PROTDIST (Jones-Taylor-Thornton model) and then the FITCH program (Fitch-Margoliash with global rearrangements and randomized input order). For bootstrap analysis, the SEQBOOT program was run before PROTDIST and FITCH. The most probable tree was calculated with the CONSENSE program. The trees were visualized with the TREEVIEW program. 39 For maximum likelihood analysis, the aligned sequences, following SEQBOOT, were analyzed with the program PROML, followed by the program CONSENSE and visualized with the program TREEVIEW. For parsimony analysis, the program PROTPARS was run following the SEQBOOT program. The most probable tree was then calculated with the program CONSENSE and visualized with TREEVIEW.
Analysis of potential glycosylation sites was performed on the amino acid sequences of all the Swiss strain using the online software NetNGlyc 1.0 Server (http://www.cbs.dtu.dk/services/NetNGlyc).
Molecular Characterization
H cloning and other constructs
The full-length H protein of the strain W10/301/red-fox/Ch/2010-JF810106, named H301F, was obtained by PCR using the forward primer of the set 1 primers pair (72f) and the reverse primer of set 4 (2221r) published by Sekulin, 46 along with PFU ultra II fusion HS DNA polymerase (Agilent Technologies, Morges, Switzerland) according to the instructions of the manufacturer. A “Flag” (H tag-3′) was then added to the full-length H protein by PCR, using 2 primers (forward: 5′-ATATATCGGACCGATGCTCTCCTACCAAGAC-3′; reverse: Tag 5′-ATATATCGGTCCGTCATCACTTATCGTCGTCATCCTTGTAATCAGG
TTTTGAACGGTTACATGAGAATC-3′) containing each a RsrII restriction site. The PCR reaction was performed using PFU ultra II fusion HS DNA DNA polymerase (Agilent Technologies) according to the instruction of the manufacturer.
The H-Flag-tagged H301F was ligated into the mammalian expression vector pCI (Promega) 58 using T4 DNA ligase (Promega), following the instructions of the manufacturer. The ligation product was used to transform Top 10 (Agilent Technologies) competent cells according to the instructions of the manufacturer. Transformed cells were then plated, and positive colonies were selected through blue/white selection, expanded, and screened as previously described. 38 The selected positive plasmid named pCI-H301F, following confirmation of the expected sequence, was expanded and then stored at –20°C.
pCI-HWt (recombinant H protein from strain A75/17, which is considered a virulent and demyelinating strain), pCI-FWt (recombinant fusion protein from strain A75/17), and engineered pCI carrying the RsrII restriction site (empty vector) were already available. 58
Qualitative fusion
To assess the fusiogenicity of the hemagglutinin constructs, Vero cells (5 × 105) were transfected with pCI-H301F, pCI-HWt, or pCI (1 μg), along with pCI-FWt (2 μg) according to an established protocol. 58 The transfected cells (effector) were mixed with Vero-SLAM cells (target) (5.5 × 105) according to the protocol described by Zipperle. 58 Following incubation at 37°C overnight, qualitative fusion was assessed through the observation of syncytia under light microscopy. The fusion experiments were carried out in duplicate. In some experiments, phase-contrast pictures were taken at 24 hours posttransfection with a confocal microscope (Olympus Fluoview FV1000).
Quantitative fusion
To quantify the cellular fusion between the transfected Vero cells (with pCI-H301F, pCI-H-HWt, or pCI, along with pCI-FWt) and the target cells (Vero-SLAM), the fusion experiment was performed with the additional transfection of Vero cells with the construct pLuc (Luciferase), driven by a T7 RNA polymerase-dependent promoter according to the protocol described by Zipperle. 58 The quantitative fusion results, along with the standard deviation, were determined by calculating the mean of the duplicate samples within 3 successive experiments. In each experiment above, pCI-H301F expression was compared with that of the wild type (pCI-HWt) (positive control) and pCI (negative control). All the values were first normalized according to their FLAG expression (intrinsic normalization) and then according to the percentage of the values determined for the wild type (pCI-HWt) (extrinsic normalization), which was arbitrarily considered equal to 100%.
Cell surface expression and Western blotting
The cellular surface expression of the recombinant hemagglutinins in the transfected Vero cells were assessed by flow cytometric analysis using an anti-FLAG monoclonal antibody (MAb) (Sigma F3165, Buchs, Switzerland) according to an established protocol. 58 The FLAG values obtained for each sample analyzed were then used to normalize the results obtained for each sample in the quantitative fusion, SLAM binding, and MAb mapping experiments.
Western blotting was performed according to an established protocol 58 to determine the expression of the recombinant H protein following pCI-H301F transfection of Vero cells.
SLAM binding
The recombinant hemagglutinin interaction with a soluble SLAM recombinant protein was assessed as described by Zipperle et al. 58 Briefly, a soluble form of the N-terminally hemagglutinin-tagged SLAM molecule (HA-sSLAM) was engineered and expressed in 293 T cells. HA-sSLAM was then added to VERO cells previously transfected with either pCI-H301F, pCI-HWt, or pCI. The semiquantitative assessment of the occurrence of the HA-sSLAM-H binding was then performed by FACS analysis. Experiments were repeated twice in duplicate.
MAb mapping
To determine if mutations occurring within pCI-H301F might have induced some structural changes, transfected Vero cells expressing recombinant H301F were alternatively incubated with 5 MAbs (1C42, 3900, 1347, 2267, PDVH11) known to recognize either linear (1347) or conformational (1C42, 3900) epitopes around the SLAM binding site, although 2 (2267 and PDVH11) were only partially characterized. Of these, PDVH11 appears to bind in close association with the SLAM binding site as well (P. Plattet personal observation). The binding of the MAbs was assessed using a FACSCalibur flow-cytometer (Becton Dickinson, Franklin Lakes, New Jersey). Results, expressed as mean fluorescence intensities, were first normalized according to the specific recombinant hemagglutinin expression (FLAG values) and then compared to the binding efficiency of the MAbs to the wild-type hemagglutinin (pCI-HWt), considered arbitrarily equal to 100% binding efficiency. The empty vector pCI served as negative control.
Statistics
The 2-tailed Fischer exact test was applied to compare the occurrence of specific lesions between species. Analyses were performed with the NCSS 2007 software package (NCSS, Kaysville, Utah). Level of significance was set at P < .05.
Results
Clinical History and Signs
Red foxes and mustelids from the 2009–2010 epidemic
Infected wild carnivores included animals of both sexes and all age classes. Animals observed alive were reported to show abnormal behavior, including loss of fear for humans, disorientation, and/or respiratory distress.
Eurasian lynx and vaccinated dog from the 2009–2010 CDV epidemic and stone marten from the 2002 CDV outbreak
An adult male Eurasian lynx estimated to be at least 10 year old was found apathic on the side of the road. When approached, it neither attempted to escape nor showed aggressive behavior and seemed unaware of the surrounding environment. Eventually, the lynx was shot by the game warden. It was confirmed CDV positive by histopathology, IHC, and RT-PCR.
A 2-year-old female large mixed-breed domestic dog (44294/dog/Ch/2010-810111) from Laax (canton of Grisons; Fig. 2) that had undergone the standard anti-CDV vaccination protocol was presented to the veterinarian with central blindness and circling. The dog was euthanized because of poor prognosis. It was confirmed positive by histopathology, IHC, and RT-PCR.
An adult female stone marten (W02/2253/stone marten/Ch/2002-JF810110) was found in a private garden unable to move and was then shot by the game warden. It was confirmed positive by histopathology, IHC, and RT-PCR.
Pathology
Red foxes and mustelids from the 2009–2010 epidemic
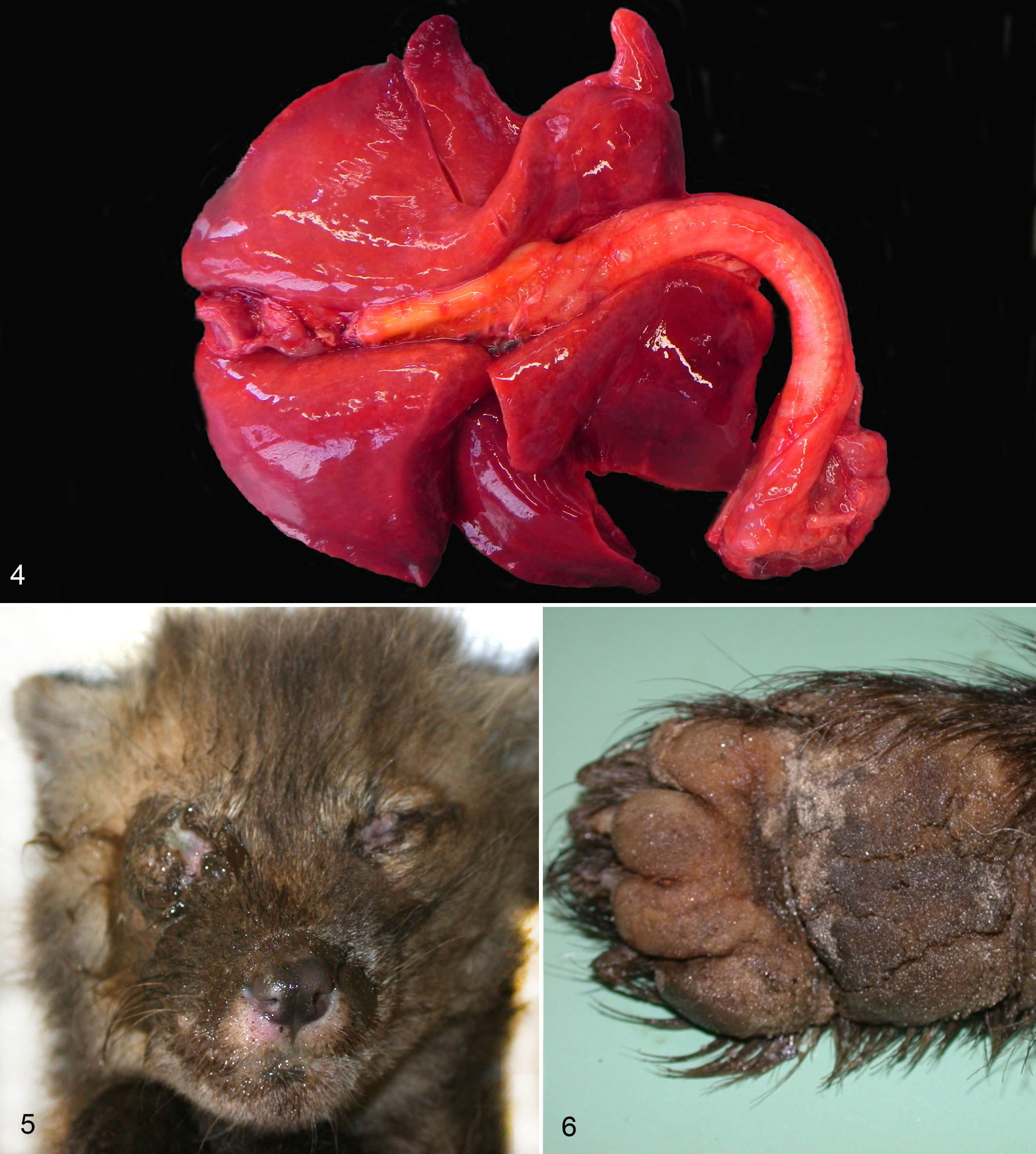
Gross findings included 26 animals (21 red foxes, 3 martens, 2 badgers; 52%) that were variably emaciated, with 6 (all foxes) showing serous atrophy of the coronary-associated adipose tissue. Lung lesions were seen in 28 cases (56%; 24 foxes, 2 badgers, 2 stone martens). They consisted of multifocal to coalescent, red to dark red areas of consolidation, more commonly affecting the margins and apices of the pulmonary lobes, although in the most severe cases they were expanding across entire pulmonary lobes (Fig. 4). Multifocal nodular consolidation of the lung (parasitic granulomas) and emphysematous areas were incidentally observed. Conjunctivitis with ocular discharge (1 case in a red fox puppy; Fig. 5) and hard pad disease (2 cases in badgers; Fig. 6) were rare findings. Pustular dermatitis was seen in 1 fox and 1 badger. Unrelated gross lesions observed in few CDV-positive animals comprised small intestine intussusception (1 fox) and sarcoptic mange (2 red foxes).
Histopathology
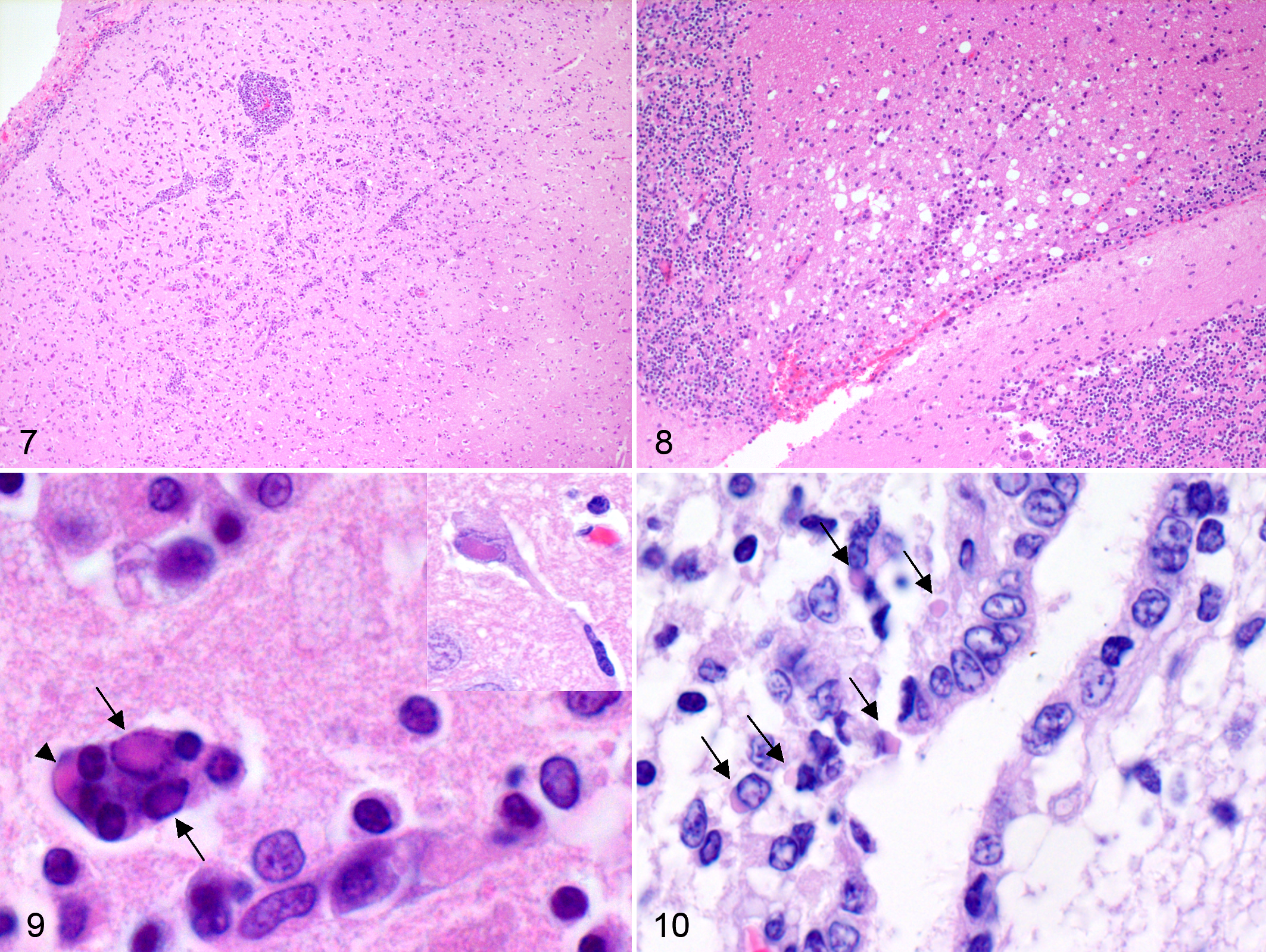
Microscopic lesions are summarized in Table 2. Brain and lung were the most commonly affected tissues. Thirty-three out of 39 foxes (84.6%), 8 out of 8 badgers, and 2 out of 3 martens had brain lesions. Glial nodules and multifocal to diffuse gliosis were the most common finding in the brain, while meningitis and encephalitis were less common and rarely severe (Fig. 7). The inflammatory infiltrate observed in the meninges was mostly composed of lymphocytes and histiocytes. Similar infiltrate was also observed in the perivascular cuffs (Fig. 7) scattered within the brain. The brain lesions were more commonly observed in the gray matter. The white matter was less frequently affected. Here, the most common lesion was multifocal gliosis, although frank demyelination was occasionally observed (5 out of 50 cases, all red foxes; Fig. 8). Intranuclear and, less frequently, intracytoplasmic inclusions (Fig. 9) were relatively common in neurons, while glial cells were less affected. Neuronal necrosis was observed in few individuals. The presence of syncytial cells (Fig. 9) was observed in badgers and foxes (3 red foxes and 2 badgers) but not in martens. Of interest was the presence of ependymitis and ependymal necrosis with or without associated intracytoplasmic inclusions (Fig. 10), which were more common in badgers than in red foxes (P = .002) (2 badgers and 1 fox with intracytoplasmic inclusions along with other 2 badgers with ependymitis but no inclusion bodies). Overall, brain lesions were more frequent in the cerebral cortex (n = 41) than in other locations, including the cerebellum (n = 8), thalamus (n = 7), hippocampus (n = 7), nucleus caudatus (n = 5), and periventricular areas (n = 4). Single observations of lesions were made in the optic tract, choroid plexus, corpus callosus, cerebellar pedunculi, internal capsule, and putamen.
Main Histopathologic Findings Observed in Red Foxes, Eurasian Badgers, and Martens Infected by Canine Distemper Virus in Switzerland During the 2009–2010 Outbreak
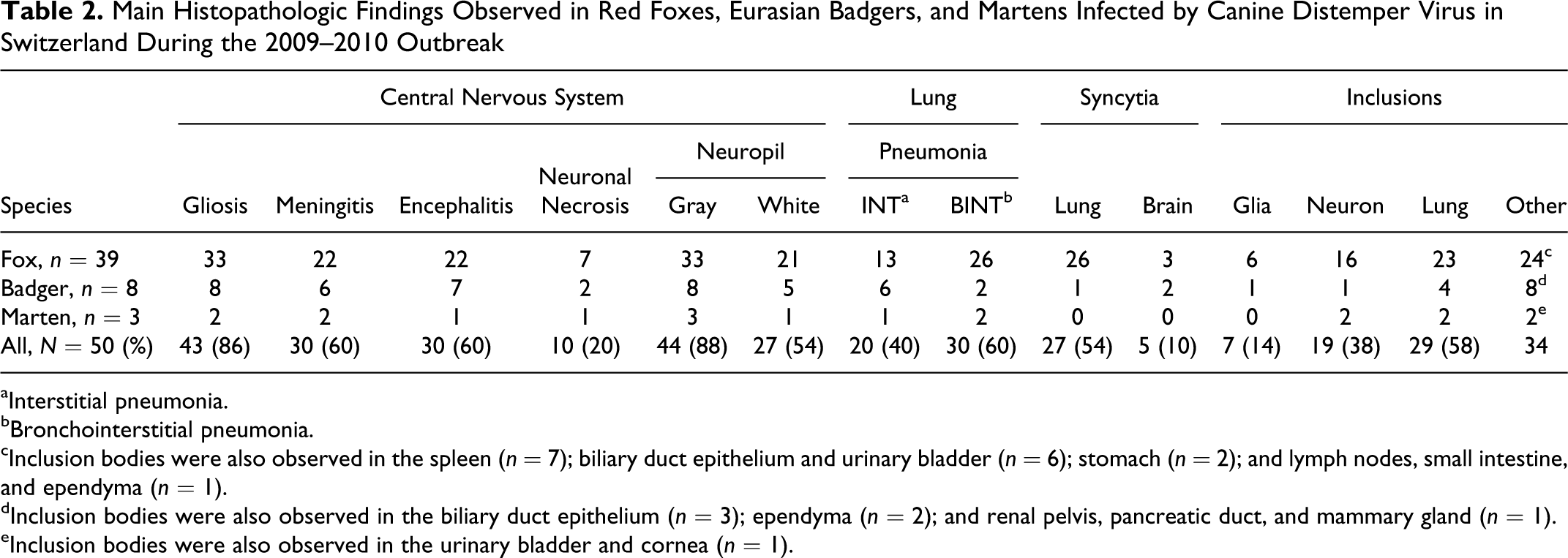
aInterstitial pneumonia.
bBronchointerstitial pneumonia.
cInclusion bodies were also observed in the spleen (n = 7); biliary duct epithelium and urinary bladder (n = 6); stomach (n = 2); and lymph nodes, small intestine, and ependyma (n = 1).
dInclusion bodies were also observed in the biliary duct epithelium (n = 3); ependyma (n = 2); and renal pelvis, pancreatic duct, and mammary gland (n = 1).
eInclusion bodies were also observed in the urinary bladder and cornea (n = 1).
Pneumonia was found in 100% of the 50 selected red foxes and mustelids, with a bronchointerstitial pattern being more frequent than the interstitial one in all species examined (Table 2; Fig. 11). Characteristic eosinophilic intracytoplasmic inclusions were commonly seen in the bronchiolar and bronchial epithelium (Fig. 12) and in the syncytial cells of epithelial and histiocytic origin (58% of the cases). Pulmonary syncytial cells (Fig. 13) were more frequent in red foxes than mustelids (P = .001); this difference was also present when mustelids species were considered separately (badgers, P = .007; martens, P = .048; Table 2). Multifocal single-cell necrosis of the bronchiolar and bronchial epithelium was also frequently observed.
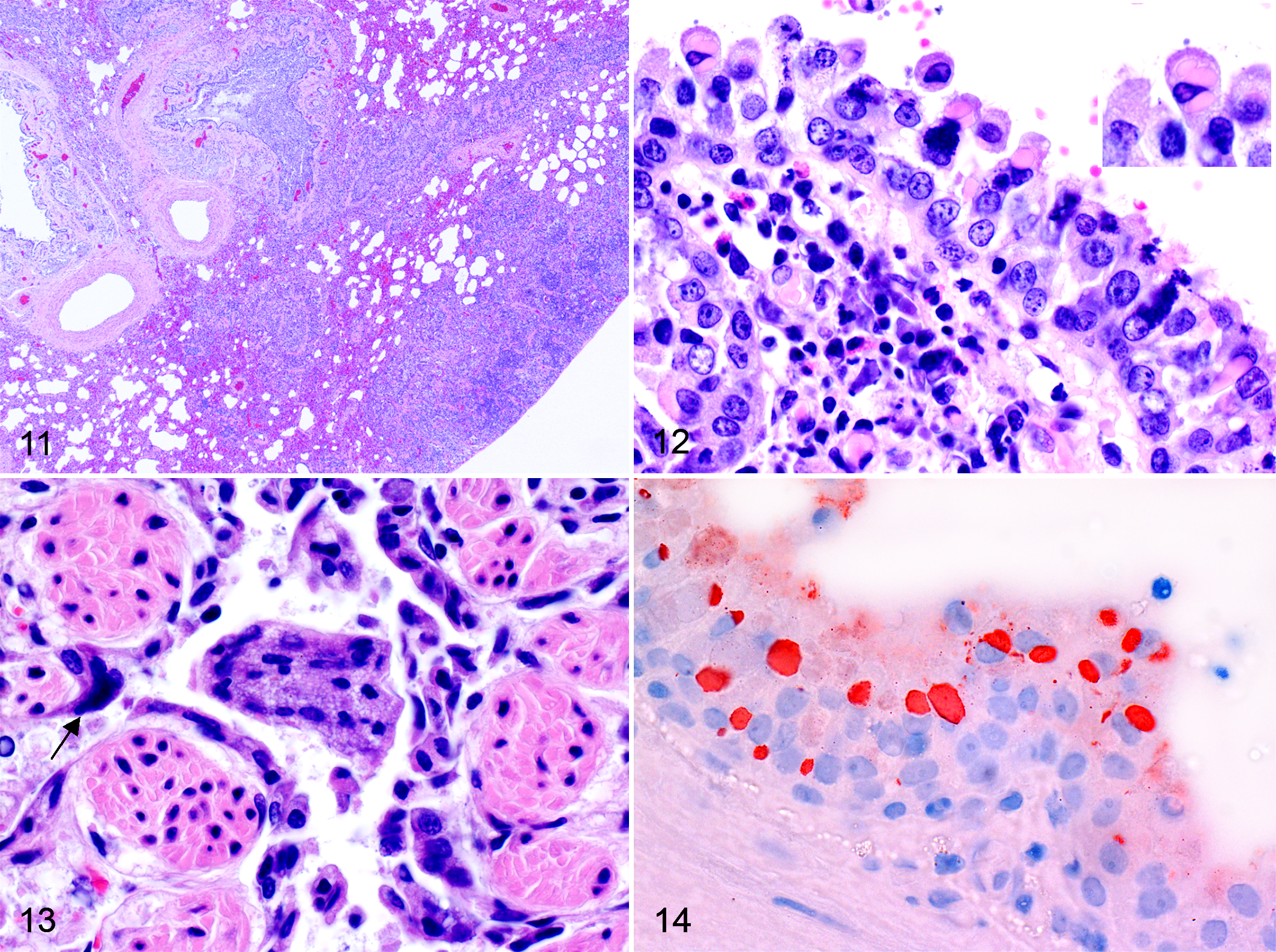
Frequently, the pulmonary interstitium was variably expanded by infiltrates of inflammatory cells comprising lymphocytes, plasma cells, and macrophages (Figs. 11, 12). Eosinophils were also often observed admixed to the primary lymphoid infiltrates. Transition from bronchointerstitial pneumonia to bronchopneumonia (Fig. 11) was observed in few animals (5 foxes and 1 badger). In these cases, polymorphonucleated neutrophils were seen collecting mostly in the alveoli, admixed with clusters of basophilic cocci. The presence of nematode larvae (Crenosoma vulpis and Angiostrongylus vasorum) and eggs was observed within the alveolar spaces and bronchiolar and bronchial lumina of 15 foxes and 2 badgers, surrounded by granulomatous inflammation. Multifocal smooth muscle hypertrophy and hyperplasia with thickening of the arteriolar wall and reduction of the vascular lumina were was commonly associated with the endoparasitism or rarely seen without contextual parasites (n = 2).
Eosinophilic intracytoplasmic inclusions were also occasionally seen in other tissues, including the biliary duct epithelium (n = 9), urinary bladder epithelium (n = 7), spleen (n = 7), ependymal epithelium (n = 3), stomach epithelium (n = 2), lymph nodes (n = 1), intestinal epithelium (n = 1), pancreatic duct epithelium (n = 1), mammary gland alveolar epithelium (n = 1), and corneal epithelium (n = 1) (Table 2). Lesions associated with these findings included lymphoid depletion (spleen, n = 7; lymph nodes, n = 1), cholangitis (n = 3), multifocal liver necrosis (n = 1), cystitis (n = 2), ependymitis (n = 2), and hydropic degeneration of the gastric mucosal epithelium (n = 1). In the remaining cases, the viral inclusions were observed in absence of cellular changes.
Additional changes observed in the CDV-infected animals included amyloidosis (2 foxes, 1 marten, and 1 badger), cardiomyopathy (6 foxes and 2 badgers), nephritis (9 foxes and 1 marten), toxoplasmosis (3 foxes), and cryptosporidiosis (1 marten).
Immunohistochemistry
Immunohistochemical staining of the brain highlighted the presence of numerous infected neurons, with only a limited number of glial cells stained positive. The number of positive neurons was highly variable depending on the tissue section, ranging from less than 10 to myriads of positive cells per section. Positive neurons were often arranged in clusters in the cortex. Positively stained mononuclear cells, mostly macrophages, were often observed within the meninges. Immunostaining was also observed in the lung sections. Here, the stain was rimming the alveolar margins and the epithelium of bronchioles and bronchi. Strong staining of the intracytoplasmic pulmonary inclusions was also observed (Fig. 14).
Inclusions were also positively stained in the other tissues along with the presence of granular positive pigment in the cytoplasm of the positive cells.
Pathology of the Eurasian Lynx and Vaccinated Dog From the 2009–2010 CDV Epidemic and Stone Marten From the 2002 CDV Outbreak
The lynx was emaciated and dehydrated, with pale mucous membranes. Brain lesions were consistent with a multifocal gliosis. No inclusions or obvious inflammation was observed in the central nervous system. In the lung, there was a minimal interstitial pneumonia, with multifocal hemorrhages and prominent intracytoplasmic eosinophilic inclusions observed in the bronchiolar and bronchial epithelium, along with numerous syncytial cells. Multifocal myofiber degeneration and necrosis with macrophage infiltration was observed in the heart, while moderate lymphoid depletion was observed in the spleen. Incidental moderate thyroid atrophy was also observed. IHC revealed the presence of CDV antigen in the lung.
Neuropathologic examination of the domestic dog vaccinated against CDV revealed a meningopolioencephalitis with mononuclear perivascular cuffs, neuronal degeneration, and necrosis and glial nodules mainly affecting the cortex, hippocampus, and midbrain. Numerous neurons contained intranuclear and intracytoplasmic eosinophilic inclusion bodies. The typical white matter changes of CD were unincisive with only few areas of mild axonal degeneration, white matter vacuolation, and gliosis. IHC revealed numerous positive neurons.
Necropsy of the stone marten from the 2002 CDV outbreak revealed lungs with mildly increased consistency. Microscopically, a lymphoplasmacytic to histiocytic mild interstitial pneumonia was observed along with occasional pyogranulomas with intralesional nematode larvae. IHC revealed the presence of CDV antigen within the lung. No brain tissue was available for this case.
Viral Isolation
The inocula obtained from the lung of 3 red foxes and the brain of the lynx were seeded in Vero-SLAM cells, and viral isolation was successful in all 4 attempts performed. Classic cytopathic effects, including large syncytia, were observed after 18 to 20 hours from the seeding of the inoculum. Isolate W10/301/red-fox/Ch/2010-JF810106 was passed twice and then frozen, while the other 3 isolates were frozen unpassed.
RT-PCRs and Sequencing
Protocol A
Amplification of a 287-bp fragment (N gene) was obtained from the cDNA of all the tested samples (7) using the A primer set (Table 1). Sequencing confirmed the amplicon to be part of CDV N gene.
Protocol B
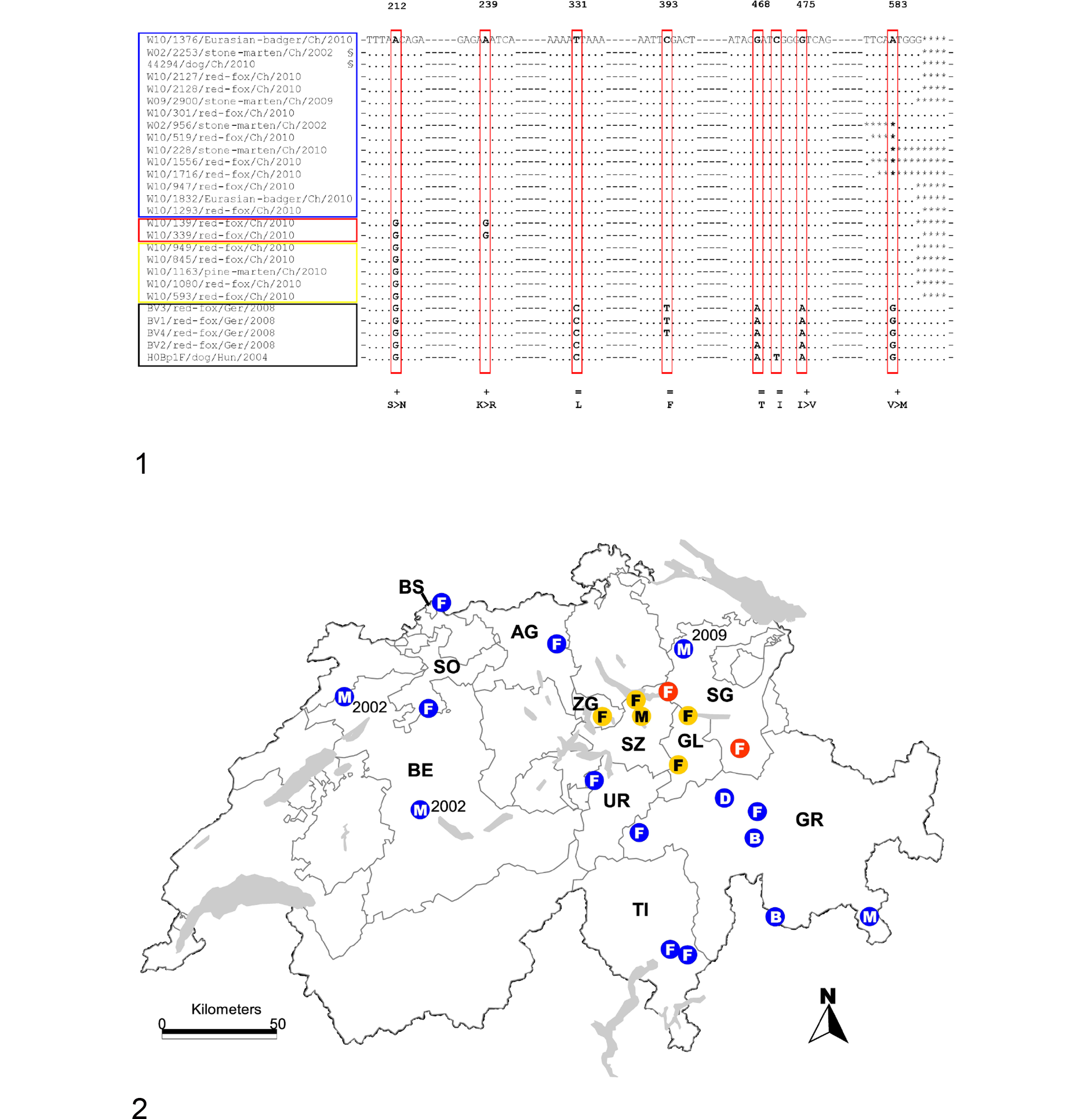
A 484-bp fragment (with fully readable 388 to 415 nucleotides-after removal of the primer) was obtained with the B primer set on all the tested samples (Table 1). The sequencing of this fragment of the H gene revealed the potential existence of at least 3 distinct Swiss sublineages (Figs. 1, 2; Table 1). The largest of them (blue box; Figs. 1, 2) comprised animals examined during the 2009–2010 outbreak and originating from neighboring and distant cantons (Aargau, Basel, Bern, Grisons, St Gallen, Solothurn, Ticino, and Uri), the vaccinated domestic dog (44294/dog/Ch/2010-JF810111), and 2 stone martens from a 2002 CDV outbreak (W02/2253/stone-marten/Ch/2002-JF810110; W02/956/stone-marten/Ch/2002-JF810115). The second-largest sublineage (yellow box; Figs. 1, 2) comprised animals from the neighboring cantons of Glarus, Schwyz, and Zug. The third sublineage (red box; Figs. 1, 2) comprised animals from the canton of St Gallen only.
Protocol C
The H genes from selected CDV strain from each sublineage above were fully sequenced using the C primer sets (Table 1). The previously observed nucleotide differences (Fig. 1) were confirmed with the exception of the vaccinated dog and 1 of the 2 stone martens from 2002 (W02/2253/stone-marten/Ch/2002-JF810110), which showed the alternative occurrence of 2 nucleotides at position 212. An A-to-G (twice A, once G) substitution in the dog and a G-to-A (twice G, once A) substitution in W02/2253/stone-marten/CH/2002-JF810110 were seen in 3 sequencing runs. Analysis of the chromatogram was consistent with the presence of a double peak for both isolates, most likely secondary to the presence of a mixture of both sequences (presumptive single nucleotide polymorphism nucleotide). Additional details of the fully sequenced H genes are summarized in Table 3.
Nucleotide and Amino Acid Substitutions Between the Swiss Canine Distemper Virus Strains From the 2009–2010 Outbreak and Their More Distant Related Strain
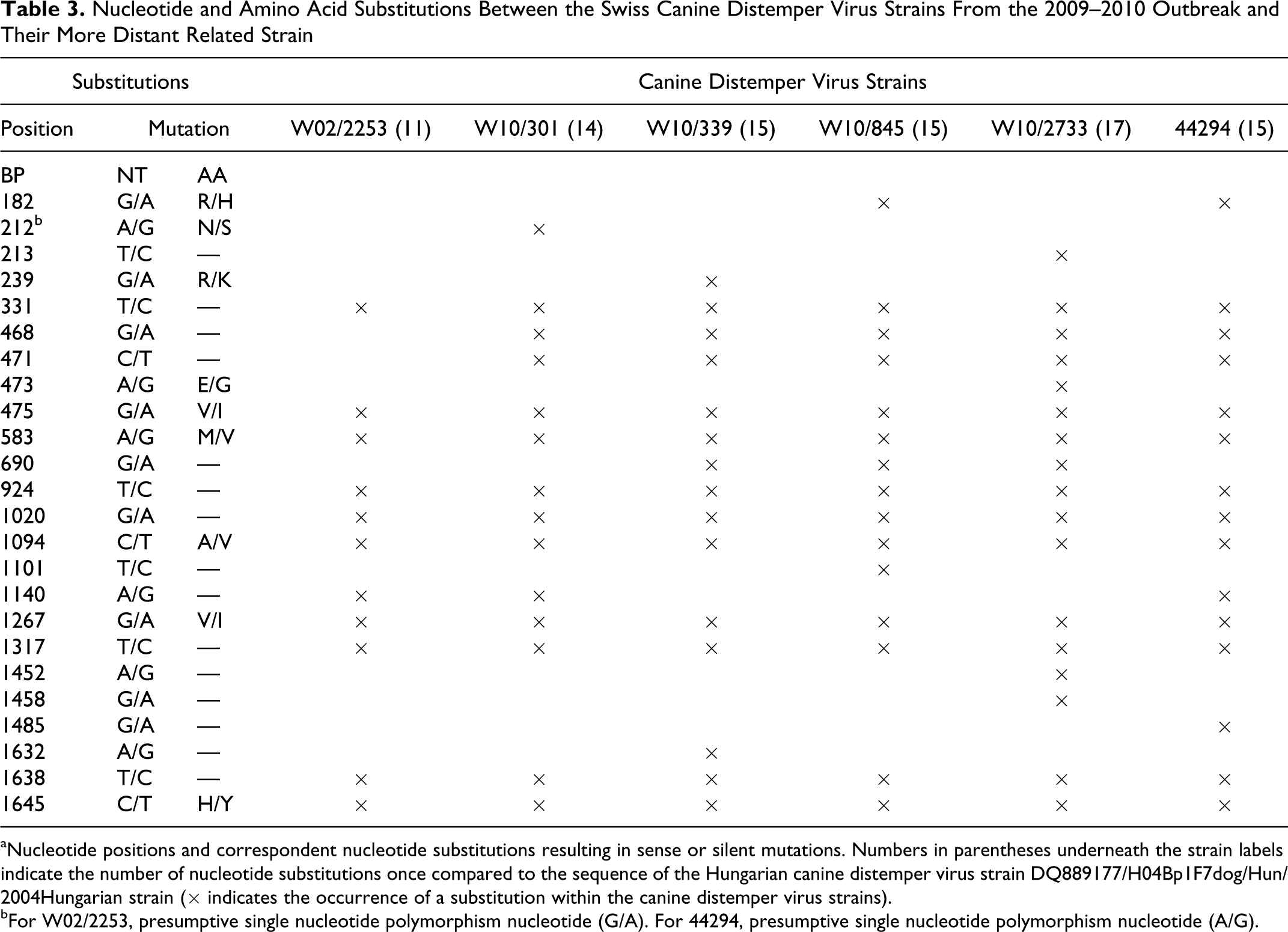
aNucleotide positions and correspondent nucleotide substitutions resulting in sense or silent mutations. Numbers in parentheses underneath the strain labels indicate the number of nucleotide substitutions once compared to the sequence of the Hungarian canine distemper virus strain DQ889177/H04Bp1F7dog/Hun/2004Hungarian strain (× indicates the occurrence of a substitution within the canine distemper virus strains).
bFor W02/2253, presumptive single nucleotide polymorphism nucleotide (G/A). For 44294, presumptive single nucleotide polymorphism nucleotide (A/G).
Phylogenetic Analysis and Strain Comparison
The phylogenetic analysis unambiguously clustered the 6 novel Swiss CDV strain (W10/301/red-fox/Ch/2010-JF810106, W10/845/red-fox/Ch/2010-JF810108, W10/339/red-fox/Ch/2010-JF810107, W10/2733/Eurasian-lynx/Ch/2010-JF810109, W02/2253/stone-marten/Ch/2002-JF810110, 44294/dog/Ch/2010-JF810111) with the CDV strain detected during the German 46 and Italian 29 CDV outbreaks with a convincing bootstrap value (100) (Fig. 3). Similar to other recently detected CDV strain in Europe, 29,36,46 all the Swiss strain, including W02/2253/stone-marten/Ch/2002-JF810110 from the 2002 outbreak, appeared to be closely related to a Hungarian CDV isolate, H04Bp1F7, originally detected in a Vizla dog in Budapest that had been vaccinated for CDV. 12 The separation of the strain belonging to different geographic areas is consistent with those observed in previous publications. 18,19,24,32,54,57 Maximum likelihood and maximum parsimony trees yielded similar results, with the only exception seen in the parsimony tree, where W10/301/red-fox/Ch/2010-JF810106 clustered separately from W10/845/red-fox/Ch/2010-JF810108 and 44294/dog/Ch/2010-JF810111 but still within the Swiss lineage (data not shown). Phylogenetic analysis was also performed using the nucleotide sequences of the H genes, and overlapping results were observed (data not shown).
The Swiss strain that showed least nucleotide mutations when compared to the Hungarian isolate H04Bp1F7 was the 2002 stone marten isolate W02/2253/stone-marten/Ch/2002-JF810110, with 11 nucleotide mutations (99.3% nucleotide identities) (Table 3, Fig. 3). In contrast, the strain showing most nucleotide differences was the lynx isolate (W10/2733/Eurasian-lynx/Ch/2010-JF810109) (99.06% nucleotide identities). The stone marten strain W02/2253/stone-marten/Ch/2002-JF810110 and that of the lynx showed 99.6% nucleotide identities. Additional features of the Swiss strain are summarized in Table 3.
All the Swiss strain examined, including those from 2002, showed the Y-to-H amino acid mutation at position 549, similar to the CDV strain from the German and Italian outbreaks. 29,32,36,46 Additionally, all the Swiss strain revealed the presence of 7 potential N-glycosylation sites (ectodomain), overlapping with those detected in the sequence of H04Bp1F7.
Molecular Characterization
Hemagglutin cell surface expression, fusion support activity, SLAM binding, and quantitative fusion
Indirect evidence of the expression and cell surface targeting of H301F was first obtained through microscopic observation of syncytia formation when the double-transfected Vero cells (with pCI-H301F and pCI-FWt) were admixed with Vero-SLAM cells 24 hours posttransfection. Direct evidence of recombinant H301F total expression was confirmed through Western blot analysis (data not shown), while cell surface expression was assessed through flow-cytometric detection of a FLAG-tag epitope c-terminally fused (Figs. 15, 16). 58 Qualitative fusion assay revealed the presence of a slightly higher number and size of cell syncytia than those observed when the cells were transfected with pCI-HWt. No syncytia were observed in the Vero cells transfected with the empty plasmid (pCI) when admixed with the Vero-SLAM cells. Flow cytometry detection of cellular surface expression of the different H recombinant proteins (FLAG) is shown in Figures 15 and 16. Cell surface expression of pCI-H301F in transfected Vero cells, revealed an increase of approximately 20% (119.5%) when compared to that observed for HWt (100%) by FLAG labeling. No surface expression was observed with the empty plasmid pCI (Fig. 15).
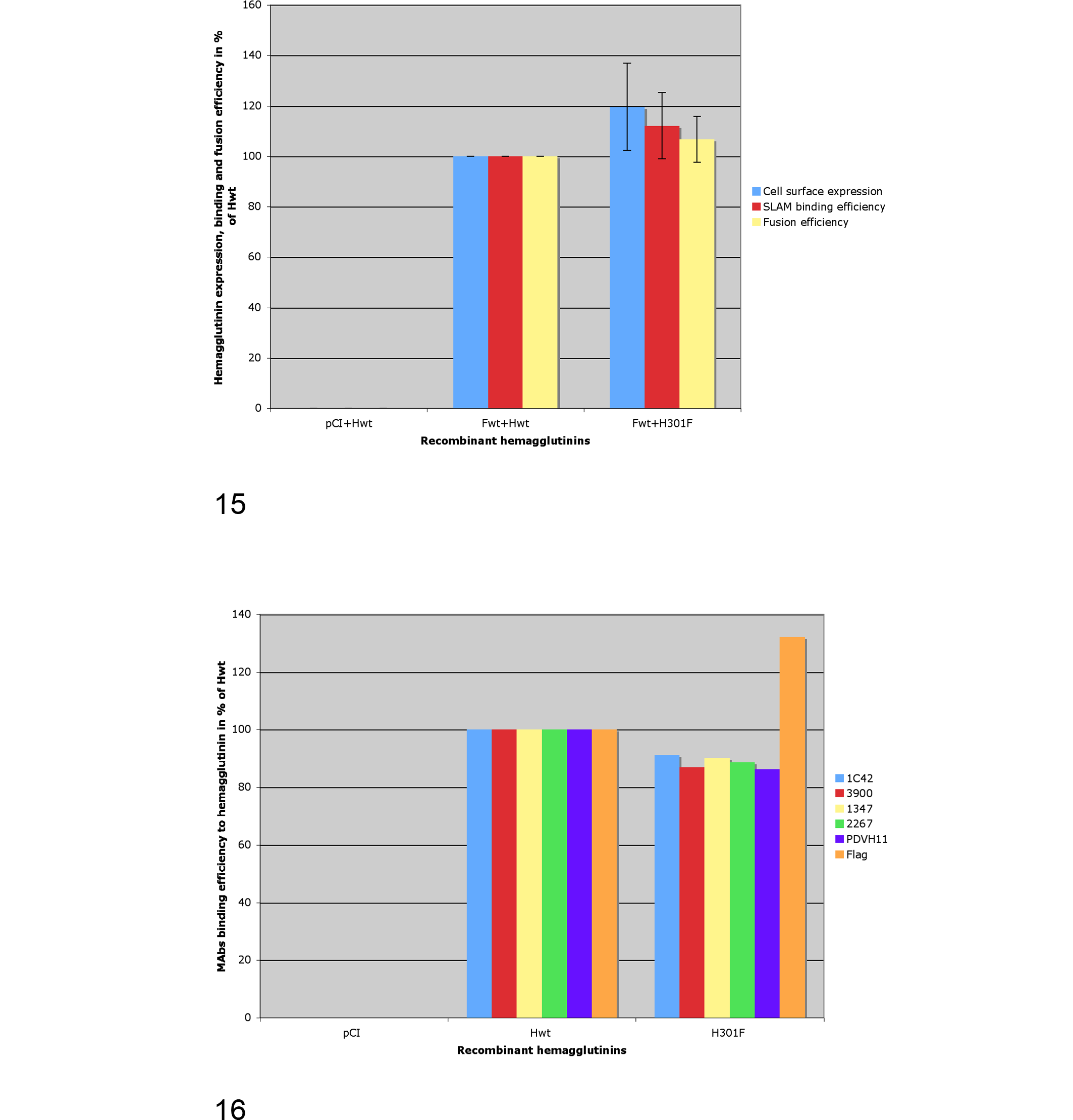
When fusogenicity of pCI-H301F was analyzed according to a quantitative assay, the recorded values showed that pCI-H301F induced a slightly higher (106.62%) fusion than that observed with pCI-HWt (100%) (Fig. 15).
The assessment of the ability of the recombinant hemagglutinins to interact with the universal Morbillivirus SLAM receptor was performed in vitro using a soluble HA-tagged recombinant canine SLAM molecule (HA-sSLAM). 58 Using an indirect fluorescence-based semiquantitative H/receptor-binding assay, 58 the results showed a slight increase (112.1%) in SLAM-binding efficiency for H301F once compared to HWt (100%) (Fig. 15).
Structural conformation
To determine if H301F was bearing significant structural differences with HWt, we used a series of MAbs known to recognize either linear (1347) or conformational (1C42, 3900) epitopes along with other MAbs whose H protein binding site is still not well characterized (2267 and PDVH11) (Fig. 16). When the binding abilities of these MAbs to H301F and HWt were compared, we observed that all the MAbs were binding to H301F with a 9% to 14% lower efficiency (91.2 for 1C42, 86.9 for 3900, 90.2 for 1347, 88.7 for 2267, and 86.17 for PDVH11) than when binding to HWt (100%), suggesting the existence of subtle structural variations between the 2 recombinant hemagglutinins.
Discussion
Pathology
One of the main goals of this study was to characterize the pathologic changes associated with the 2009–2010 CDV outbreak in wild carnivores of Switzerland. While gross pathology findings were similar to those described in the literature for animals infected with CDV, 3,11 histopathology clearly showed that the strain involved in the Swiss outbreak are characterized by an accentuated neuronotropism, with only mild involvement of the white matter similar to what has been described by Summers for Snyder-Hill-like strain. 47 The most convincing demyelinating lesions were observed in the cerebellum, where Summers also describes them to sporadically occur in dogs infected with the Snyder-Hill strain. 47 Another similarity between Snyder-Hill and the Swiss CDV strain was the more common presence of intranuclear (and/or intracytoplasmic) eosinophilic inclusions in neurons rather than in glial cells, while demyelinating CDV strain are reported to infect primarily astrocytes. 53,55 Additionally, the cortex was the portion of the brain mostly affected, consistent with what was described by Summers for neuronotropic strain. 47
The inflammatory response observed in the large majority of the cases examined was mild, suggesting that death occurred almost invariably during the early stage of the disease. 49 Interestingly, although ependymal lesions were generally uncommon, they were significantly more frequent in badgers than red foxes. It is possible that this lesion reflects a different manner (or kinetic) of viral spread within the central nervous system between badgers and other carnivores.
The second-most significant lesion was observed in the lung, consistent with what has been observed in other CD outbreaks in wild carnivores. 16 Interstitial pneumonia or, more frequently, bronchointerstitial pneumonia was observed in all animals. A distinctive feature of CDV-associated pneumonia was the presence of syncytial cells often containing intracytoplasmic eosinophilic inclusions, similar to what was observed in CDV-infected raccoons in the United States. 25 Interestingly, syncytial cells in the lung were significantly more numerous in red foxes than in badgers, while not observed in martens. This finding might indicate differences at the receptor level between the respiratory epithelium of red foxes and mustelids.
During this study, we conclusively diagnosed the first case of CDV infection in a Eurasian lynx through histopathologic examination, molecular detection, and viral isolation. Different from what has been reported in the literature for CDV infection in large felids, 1,45 Canadian lynx (Lynx canadensis), bobcats (Lynx rufus), 11 and the other carnivores affected by the current Swiss CDV outbreak, neither inflammatory infiltrates nor inclusions were observed in the brain of the lynx, but only multifocal gliosis. However, histopathologic findings in the lung of the lynx were similar to those described in large felids by Appel, 2 except for type 2 alveolar pneumocyte proliferation, which was not observed in our lynx. Mild heart lesions were observed in the Eurasian lynx, similar to what was observed in a bobcat and a Canadian lynx infected with CDV 11 and in a number of foxes and badgers examined during the current Swiss outbreak.
In contrast to wild felids, domestic cats do not develop disease following CDV infection. 18 Interestingly, the strain isolated from the Eurasian lynx showed the most numerous nucleotide mutations (along with the most numerous unique ones among the Swiss strain) compared to the closest CDV “ancient relative” (Hungarian isolate from 2004) (Table 3). This might suggest that felids represent an “inconvenient” host for CDV, which requires substantial adaptation to productively infect these carnivores.
Among the animals examined, one was a domestic dog, which, despite having been vaccinated against CDV, became fatally ill with the disease. Pathologic changes observed in this dog were overlapping with those observed in wild carnivores examined in the current Swiss outbreak. The results of the sequence analysis are consistent with a likely transmission of CDV from wild carnivores to this dog. This hypothesis is strongly supported by the very high similarity of the nucleotide and amino acid sequences of the dog strain and those of the strain from the wild carnivores examined in this study (Fig. 1; Table 3) and, consequently, by their close phylogenetic relationship (Fig. 3). Phylogenetic results are relevant to rule out that clinical disease was caused by residual virulence of the vaccine strain. 30 The reason for the vaccination failure is not known, but given that the appropriate vaccination protocol was applied, individual immunity-associated factors or lack of cross-reactivity between the vaccine and the wild-type strain are considered possible causes.
Investigation Into the Origin of the Swiss CDV Strain (Phylogenetic Analysis)
During the past 2 years, distemper outbreaks have been recorded in several countries bordering the Swiss territory, including Germany 46 and Italy. 9,29,36 Phylogenetic analyses performed by Sekulin, 46 Martella, 29 and Monne 36 are supporting the stemming of the CDV strain detected in the German and Italian outbreaks together with a Hungarian isolate. Our results are consistent with these findings and suggest that the Swiss, German, and Italian CDV strain might be all related and likely part of a single large ongoing transnational European CDV outbreak, spreading from Eastern to Western continental Europe, priming the current Swiss outbreak and hitting on a large scale most of the susceptible wild carnivores. Although this hypothesis is the most likely, we obtained a surprising result suggesting that the epidemiologic scenario might be partially different. While analyzing the nucleotide sequence of the H gene of a CDV strain (W02/2253/stone-marten/Ch/2002-JF810110) from the 2002 epidemic in stone martens in Switzerland, we observed striking similarities between its sequence and that of the homologous gene of the 2009–2010 CDV isolates (Table 3). The hypothesis of a laboratory contamination was considered unlikely because (1) repeated sequencing yielded consistent results, (2) the H gene from a second strain from 2002 was partially amplified (1300 nt; data not shown) and gave similar results, and (3) reextraction of the RNA and new sequencing of the RT-PCR product were consistent with the initial findings. According to these findings, it is then possible that a CDV strain related to the 2004 Hungarian one has spread through Europe and reached Switzerland far before 2009, although remaining virtually undetected except for few local outbreaks in martens. This hypothesis is supported by the following observations: (1) the closer relatedness of the 2002 marten’s CDV strain to the Hungarian strain H04Bp1F7 among all the Swiss strain available; (2) serologic evidence of CDV exposure of Swiss red foxes and Eurasian lynx previous to the current outbreak; 34,37 and (3) the notorious long-term persistence of CDV strain in wildlife shown to occur for decades 6 and not necessarily associated with fatal consequences. 16 The occurrence of earlier CD outbreaks in Switzerland in martens only might be explained by the reported higher sensitivity of martens to CDV infection than red foxes. 15,21 The high mortality newly observed in foxes and badgers, together with the lynx case, are however suggestive of a recent significant change in the epidemiologic scenario, such as an increased virulence of the etiologic agent.
Sublineages
The strain comparison revealed the existence of distinct sublineages, which appears to have close territorial association, since distinctive mutations are apparently associated with specific territories (Figs. 1, 2). However, we cannot exclude that this distribution might have been influenced by a sampling bias. No host species–specific strain association was observed, consistent with what is reported in the literature. 8,11,17,18,25,31,57
Molecular Signature Characterization
We were interested in determining if a specific “molecular signature” was present in the 2009–2010 Swiss CDV strain, which might have been associated with the pathogenicity features described above. Our results suggest that the surface expression of H301F and the binding efficiency to SLAM were moderately enhanced compared to that of A75/17, while the fusion ability of 301F was minimally increased compared to that of A75/17 (Fig. 15). Although the magnitude of the standard deviations is unlikely to grant statistical significance to these data (Fig. 15), we think that they might reflect a “trend” actually occurring. Several studies have speculated about the possible effect of specific mutations occurring within the H protein in correspondence of SLAM binding site (the only known receptor for virulent CDV up to date), 43,51 in particular those occurring at sites 530 and 549. 32,46 Similar to what was observed in the strain from the German and Italian outbreaks, H301F carried a Y-to-H mutation at site 549 along with all the other Swiss isolates, including W02/2253/stone-marten/Ch/2002-JF810110 and 44295/dog/Ch/2010-JF810111, when compared to the Hungarian isolate. 29,36,46 Interestingly, this mutation is not present in the homologous site of the H protein of A75/17. Given the overall results of our in vitro experiments, this mutation might concur to confer to the mutated CDV an increased cell surface expression with secondary increase of binding efficiency to the SLAM receptor. Interestingly, the enhanced binding to SLAM has been observed using a recombinant canine SLAM receptor and not a fox-derived SLAM, which H301F might have been more adapted to. This may suggest that our results actually represent only an underestimation of the potential benefit secondary to the SLAM-binding site mutation and indirectly help to explain why this strain successfully infected a CDV-vaccinated dog. However, other factors may have contributed to functional differences potentially leading to increased virulence of the Swiss strain, including the additional differences existing between the H genes of A75/17 and W10/301/red-fox/Ch/2010-JF810106 and those between the other genes within their genomes (Fig. 16).
In this article, we provided the partial characterization of the pathologic changes and the viral strain associated with the current CDV epidemic in Switzerland. The current outbreak represents a clearly different clinical, epidemiologic, and pathologic entity from the former local outbreaks that have occurred in Switzerland. 40 The spectrum of clinically affected species was larger than in the past, and we report the first documented case in a Eurasian lynx. Importantly, we documented also a CDV spillover to a domestic dog. Furthermore, we showed that the CDV strain involved in this outbreak caused a polioencephalitis instead of the more common demyelinating form of CDV-associated central nervous system disease. All these findings were suggestive either of a novel (nomad) 10 virulent CDV strain only recently arrived in the Swiss territory or the emergence of one of the presumptive Swiss resident strain as a lethal-mutated virus. In light of these results, substantial contributing factors to the high morbidity and mortality observed in the Swiss outbreak might have been (1) the presence of a Snyder-Hill-like CDV, whose pathogenetic features are consistent with severe acute disease and high mortality rate; (2) the possible combination of the activity of both novel and recirculating CDV strain; and (3) the existence of functional and structural differences of the H proteins of the CDV Swiss strain from that of a more conventional demyelinating CDV strain.
Footnotes
Acknowledgements
We thank Professor Ernst Peterhans (Institute of Veterinary Virology, Vetsuisse Faculty, University of Bern) for making available laboratory facilities, Dr Frank Steffen (Neurologie, Kleintierklinik, University of Zurich) for having provided the case of the domestic dog vaccinated for canine distemper virus (44294/dog/Ch/2010-JF810111), and Professor Marc Vandevelde for a critical review of the manuscript. We also thank all the technical personnel of the FIWI and Institute of Animal Pathology (Vetsuisse Faculty, University of Bern) for their valuable support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work has been partially supported by a grant (1.11.q) from the Swiss Federal Veterinary Office (BVET).
