Abstract
Canine osteosarcoma is an insidious disease with few effective treatment modalities; therefore, use of pharmacologic intervention to improve mortality or morbidity is constantly sought. The use of cyclooxygenase enzyme inhibitors has been an area of interest with limited efficacy based on retrospective examination of tumor expression and in vivo cell proliferation models. Recently, examination of dual cyclooxygenase and 5-lipoxygenase inhibitors in human and canine oncology suggests that 5-lipoxygenase inhibitors may be an effective approach in vitro and during tumor induction in rodent models. Therefore, the authors decided to examine 5-lipoxygenase expression in primary canine osteosarcoma samples and have shown that approximately 65% of osteosarcomas label positive for cytoplasmic 5-lipoxygenase. Further examination of a cell culture and xenograft model shows similar 5-lipoxygenase expression. Surprisingly, a canine 5-lipoxygenase inhibitor (tepoxalin) significantly reduced cell proliferation at physiologic doses in vitro and diminished xenograft tumor growth in nude mice, suggesting that further investigation is needed. Traditionally, 5-lipoxygense leads to production of lipid mediators, such as leukotriene B4 and 5-oxo-eicosatetraenoic acid, which, when added back to the media of tepoxalin-treated cells, did not recover cell proliferation. The lack of nuclear staining in primary and xenografted tumors and the lack of response to eicoasanoids suggest that lipid mediator production is not the primary means by which tepoxalin acts to alter proliferation. Regardless of the mechanisms involved in retarding cell proliferation, future investigation is warranted.
Osteosarcoma (OSA) in dogs accounts for 85% of all skeletal malignancies. 3,6,23 The disease primarily affects older, large-breed dogs, and 75% of disease localizes to the appendicular skeleton with only 25% located axially. 6,26,38 Primary tumors are locally aggressive, inducing bony lysis with pulmonary metastasis occurring via hematogenous spread in 90% of dogs, with less than 10% showing radiographically evident pulmonary nodules at the time of diagnosis. 2,3 Despite advancements in treatment, median survival times range from 3 months to 1 year, with less than 20% of dogs surviving >2 years following diagnosis. 3,39
Chemotherapeutic intervention, often used in conjunction with surgical intervention, involves combinations of several pharmacologic agents, including doxorubicin and/or the platinum analogues cisplatin, carboplatin, or lobaplatin. 6 Palliative treatment for pain associated with primary and secondary lesions often consists of nonsteroidal anti-inflammatory drug (NSAID). NSAIDs also have chemotherapeutic properties in slowing proliferation in certain human neoplastic diseases; for example, patients with colorectal cancer have shown either decreased incidence or increased survival time when administered aspirin therapy, and dogs with transitional cell carcinoma show increased survival time when administered the NSAID deraoxib as part of treatment protocol. 25,37 NSAIDs function by decreasing the production of various inflammatory mediators through inhibition of the enzyme cyclooxygenase (COX), of which there are 2 principal isoforms: constitutively expressed COX-1 and inducible COX-2. COX-2 has been implicated in proneoplastic behavior through the production of inflammatory prostaglandins, primarily PGE2, due to its promitogenic and prometastatic actions. 27,45
While the literature regarding expression of COX-2 in canine carcinomas is more numerous, the immunohistochemical expression of COX-2 in OSA exhibits a range of 23% and 77.3%, wherein the majority of cases demonstrated poor or minimal immunohistochemical staining for COX-2. 7,19 –21,24,28,29,30,33 In vitro and in vivo studies using the COX-2 inhibitors deracoxib and meloxicam have demonstrated efficacy against human and murine OSA cell lines; however, the doses needed were well beyond the upper end of physiologically achievable concentrations (>10 μM). 32,38 Furthermore, of the OSA cell lines examined to date, none show PGE2 production, and expression of COX-2 does not correlate with enzymatic activity. 13,27 Hence, follow-up clinical studies using COX inhibitors have not been performed in OSA since their utility is questioned.
More recently, the idea of dual inhibition of COX and 5-lipoxygenase (5-LOX) has received attention therapeutically, since 5-LOX has the ability to metabolize arachidonic acid much like the COX enzymes. Additionally, 5-LOX appears to be aberrantly expressed in human pancreatic, prostate, and bladder neoplasias. 9,11,15 Some studies show that inhibition of 5-LOX prevents production of 5-hydroxytetraenoic acid, a precursor to the bioactive molecules 5-oxo-eicosatetraenoic acid (5-oxo-ETE) and leukotriene B4 (LTB4) which may have promitogenic activities in certain neoplasms. 11,14,16,34 However, 5-LOX may have other cytoplasmic functions, and the use of 5-LOX inhibitors may exert antiproliferative effects via other mechanisms involving mitochondrial-induced apoptosis. 41,42
In veterinary medicine the use of tepoxalin as a dual COX/5-LOX inhibitor is primarily for pain relief. 1 There is little investigation into the use of these dual inhibitors in OSA, let alone other companion animal neoplasms; yet, recent work from our laboratory shows that in vitro tepoxalin is efficacious at inducing apoptosis and decreasing feline squamous cell carcinoma proliferation at presumed clinically achievable concentrations. 44 Furthermore, a recent examination of nude mice with canine OSA xenografts showed that tepoxalin could decrease tumor size, but examination of the incidence of 5-LOX expression in various canine neoplasms is lacking. 40
Here, we describe 5-LOX expression patterns in 60 clinically diagnosed canine OSA patients and the relative cytoplasmic versus nuclear expression pattern in both appendicular and axial OSAs. Additionally, 5-LOX expression was examined in 3 canine OSA cell lines with subsequent in vitro proliferation studies using the HMPOS cell line which overexpresses 5-LOX, to examine the in vitro efficacy of the 5-LOX inhibitor tepoxalin. Encouraging, in vitro results led us to perform nude mouse xenografts using the HMPOS cell line to induce tumors and examine their response or primary tumor growth and metastasis in response to oral tepoxalin treatment.
Materials and Methods
Case Inclusion Criteria, Histopathology, and Immunohistochemistry
Sixty canine OSA cases were identified from the Cornell University College of Veterinary Medicine anatomic pathology archives from 1996 to 2006. Thirty samples of each type, OSA of the appendicular or axial skeleton, were selected for sectioning. Histologic verification of tumor type and location were confirmed by a veterinary pathologist (JKP) after reviewing clinical history, gross description, and hematoxylin and eosin–stained tissue sections. All specimens were fixed in 10% neutral buffered formalin and embedded in paraffin blocks. Paraffin-embedded tissues that had did not require decalcification were chosen and sectioned at 4 μm and mounted onto positively charged slides for immunohistochemistry.
A standard immunohistochemistry protocol was used based on previous work in our laboratory. 44 Briefly, tissue sections were deparaffinized through a xylene and graded alcohol series. Endogenous peroxidases were quenched in 1% hydrogen peroxide in methanol for 10 minutes. Sections were rinsed in phosphate buffered saline (PBS) and incubated for 30 minutes in 1:10 diluted goat serum for 30 minutes. Primary polyclonal rabbit anti-human 5-LOX antibody (H-120, epitope 106-225; Santa Cruz Biologics, Santa Cruz, CA) was used at a 1:100 dilution, and control slides were treated in parallel with an equal concentration of control rabbit antibody (Vector Labs, Burlingame, CA) and treated identically to 5-LOX immunohistochemistry slides, incubating for 3 hours at room temperature in a humid chamber. Sections were rinsed with PBS-0.1% BRIJ solution 3 times and then incubated with a biotinylated anti-rabbit antibody (IgG H&L, BA-1000; Vector Labs) at room temperature for 30 minutes and then rinsed 3 times in PBS-0.1% BRIJ solution. Further incubation was performed in prediluted avidin–horse radish peroxidase solution (Invitrogen, Carlsbad, CA) for 30 minutes, followed by 3 rinses in PBS-0.1% BRIJ and exposed to diaminobenzidine tetrahydrochloride for approximately 60 to 90 seconds per slide. Slides were then washed in distilled water, counterstained with Gill’s hematoxylin, and cover-slipped. A veterinary pathologist (JPK) then examined each slide and its control and provided an overall intensity score as well as positive cellular compartmental staining as cytoplasmic, perinuclear, and/or nuclear. Staining was assessed as either positive or negative staining for all sections. An overall labeling intensity score was provided for every positive neoplasm: 0, no staining; 1, weak staining; 2, moderate staining; 3, marked intensity. Positive sections were also categorized for relative percentage of staining in the neoplasm as negative (0) if less than 10% of neoplastic cells were positive, patchy (1) if 11% to 40% of cells stained positive, multifocal (2) if 41% to 70%, and diffuse (3) if over 70% of the neoplastic cells labeled positive. This scoring system was adapted from previously published criteria. 12 Cell proliferation between the 2 groups was assessed through mitotic index (MI) scoring, counting cells only observed in prophase, metaphase, or telophase in 5 high-power fields (400×) and averaged. A χ 2 analysis was performed to examine positive versus negative labeling proportions to determine significant differences in staining across the 2 categories of OSA, appendicular versus axial locations. Labeling intensity and relative proportion of the neoplastic mass labeling positive were assessed using Wilcoxon ranked sum testing to determine differences between appendicular and axial tumors with α set at P < .05. To determine if labeling intensity was associated with cell proliferation, the MI was correlated to intensity and examined using a Spearman nonparametric correlation for significance in the relationship between staining intensity and MI with α set at P < .05.
Cell Culture
Growth medium consisted of RPMI 1640 (Invitrogen) with 10% fetal bovine serum (FBS; Invitrogen) with 1% antibiotic and antimycotic solution (Sigma, St. Louis, MO) for cell proliferation assays. OSA cells were obtained from 3 sources: OS 2.4 cells from Dr. Katrina Mealey at Washington State University, HMPOS cells from the Cornell University Comparative Cancer Program, and the D17 cell line from the American Type Culture Collection. NSAIDs were the 5-LOX inhibitor tepoxalin and the global COX inhibitor RWJ 20142 (tepoxalin active metabolite [TAM]; Schering Plough Pharmaceuticals, Kenilworth, NJ). All NSAIDs were reconstituted in dimethyl sulfoxide (DMSO) at 20 mM stock solution and stored at –80°C until used in vitro.
Cell Lysis and 5-LOX Western Blotting
OSA cells were passaged and grown for 48 hours without any treatment and lysed according to a previously described protocol adapted from mammary carcinoma cell culture lysis techniques. 2 Cell lysates were collected, and protein determination based on the Bradford technique was performed on each sample. Lysates were equilibrated to common μg/μl volume in lysis buffer and loading buffer. Western blot analysis was performed using 8% sodium dodecyl sulfate polyacrylamide gel electrophoresis, loading 30 μg of protein per well, followed by transfer to a polyvinylene difluoride membrane and immunoblotted. Lysates were immunoblotted for 5-LOX using a rabbit anti-human antibody (H-120, epitope 106-225; Santa Cruz Biologics) after membrane blocking with 10% nonfat dried milk, the cells were incubated with a primary antibody overnight at 4°C. Membranes were washed twice with TBST (tribuffered saline + 0.1% tween-20) and incubated at room temperature for 1 hour using a 1:5000 dilution of anti-rabbit (Cell Signaling, Danvers, MA). Blots were again washed 3 times with TBST and visualized using a Chemi-Doc XRS station and QuantiOne Software (Bio-Rad, Hercules, CA). A second immunoblot was performed using similar techniques utilizing a primary β-actin mouse antibody (Sigma) at a 1:5000 dilution to examine relative loading equality between lysates.
Tepoxalin/TAM MTT Proliferation Assays
All 3 cell lines were plated at a density of 5 × 103 cells per well in 96-well plates (BD Falcon, San Jose, CA) and incubated overnight. On the following day, cells were treated in quadruplicate with tepoxalin, TAM, or the vehicle control (DMSO) using a serial dilution ranging from 0.4 to 50 μM for 48 hours. (4,5-Dimethylthiazol-2-yl)-2-5-diphenyltetrazolium bromide (Sigma) assays were performed as previously described. 43 Each experiment with each NSAID was performed in triplicate and averaged. A 1-way analysis of variance was performed to compare each treatment to vehicle control–treated cells, with Tukey post hoc analysis to determine significance between groups, where P < .05 was considered statistically significant.
LTB4/5-oxo-ETE MTT Assays
HMPOS cells were plated at a density of 5 × 103 in 96 well plates and allowed to incubate in growth medium overnight. The following day, the cells were treated in quadruplicate with tepoxalin or vehicle control (DMSO) using a serial dilution ranging from 0.2 to 50 μM to for 48 hours. Immediately after addition of tepoxalin either LTB4 (Cayman Chemical, Ann Arbor, MI) or 5-oxo ETE (Cayman Chemical) or vehicle control (ethanol) was added to wells at either 500 nM or 100 nM. The following day treatment was repeated for a total of 48 hours of treatment. MTT assays were performed as previously described with each experiment being performed in quadruplicate on 3 separate occasions and averaged. Each lipid mediator treatment concentration (100 nM and 500 nM) was compared to the tepoxalin alone treatment at each concentration using a 1-way analysis of variance, where P < .05 was considered statistically significant.
OSA Xenograft Model
Our xenografting protocol was approved by standards set forth by the Cornell University Institutional Animal Care and Use Committee. Twelve 4-month-old female CByB6F1/J-Foxn1nu (nu/nu) mice were purchased from Jackson Laboratories and irradiated with 1.4 Gy. Two days later, HMPOS cells, grown to 80% confluence, were trypsinized with 0.05% trypsin and immediately centrifuged at 2500 g for 6 minutes; the cells were then gently washed in sterile phosphate buffered saline (PBS) and re-centrifuged. Cells were then suspended in PBS and counted using a manual hemocytometer; cells were suspended to equate to 25 million cells per milliliter, and 0.3 ml of cells were injected subcutaneously into the right flank at the level of the hip and allowed to form tumors over the next 4 days. Day 4 postinjection mice were randomly divided into 3 groups of 4 mice. Group A received 0.2 ml of 0.9% sterile saline; group B received 1 mg/ml of solution at a dosing equivalent of 50 mg/kg of tepoxalin, while group C received 0.2 mg/ml of solution at a dosing equivalent to 10 mg/kg of tepoxalin. All treatments were administered via oral gavage every 24 hours between 8 a.m. and 9 a.m. Powdered tepoxalin was solublized in 0.9% saline and frozen in aliquots at –80°C, and freshly thawed aliquots were used daily for treatments. Tumor measurements started from baseline (4 days postinjection) and were measured twice a week. Mice were euthanized if the tumor reached 2.5 × 2.5 cm or at the end of the 28-day protocol. The relative tumor volume was measured using calipers in 2 dimensions, and volume was assessed based on previous calculations by Kadosowa et al. 17 The equation takes into account the length (L) and width (W) in millimeters as measured by calipers, assuming that the depth cannot be measured well, thereby utilizing the following equation: (mm3) = L (mm) × W 2 × 0.5. 17 Additional data collected was live days, final tumor weights, and final tumor volumes. At the time of euthanasia, the tumors were then dissected from the body wall and weighed before a complete necropsy was performed, with all tissues being fixed in 3.7% buffered formalin. Due to the differences in growth between the three groups histologic assessment of the xenografted tumors were assessed for MI, which was done by counting the number of mitotic figures in 10 random areas of viable tumor tissue at 400×. The fields were then averaged for each mouse, and the mean and standard deviation of each group were determined. Slides were prepared with hematoxylin and eosin staining, and all tissues were examined by a boarded veterinary pathologist (JPK), assessing the tissues for toxicologic changes and the lung tissues (left lobe, right inferior lobe, and right superior lobe) for metastasis of the primary mass. All xenograft data from the 3 groups of mice was assessed using a 1-way analysis of variance and Tukey post hoc comparison at specific time points with a P value set at .05.
Results
Immunohistochemistry
Immunohistochemical analysis of axial and appendicular OSA showed that both locations had many 5-LOX positive neoplasms (see Table 1). All OSAs had exclusive cytoplasmic staining with only 1 neoplasm showing both nuclear and cytoplasmic labeling. High magnification shows the typical homogeneous cytoplasmic labeling for all 3 intensities (Figs. 1 –3). Negative staining using rabbit control antibody showed no labeling across all sections (Fig. 4). Twenty-three of the 30 appendicular OSAs labeled positive with an average intensity of 1.53, while 18 of 30 axial tumors labeled positive with an average intensity of 1.03. Chi-square analysis of positive and negative tumors between axial and appendicular was not significant, while intensity of labeling based on the 3-point scale showed no significant difference with a possible trend (P = .075) for higher intensity in appendicular tumors (Table 1). Average MI for both appendicular and axial tumors was not significantly different, with average MI being approximately 2 mitoses per high-power field. There was no significance difference between mitotic index and tumor location, nor was there any correlation between MI and labeling intensity (Table 1). Further examination of no, focal, patchy, and diffuse tumor labeling was examined, with average labeling being between 41% and 70% of the tumor for both axial and appendicular tumors (Table 1).
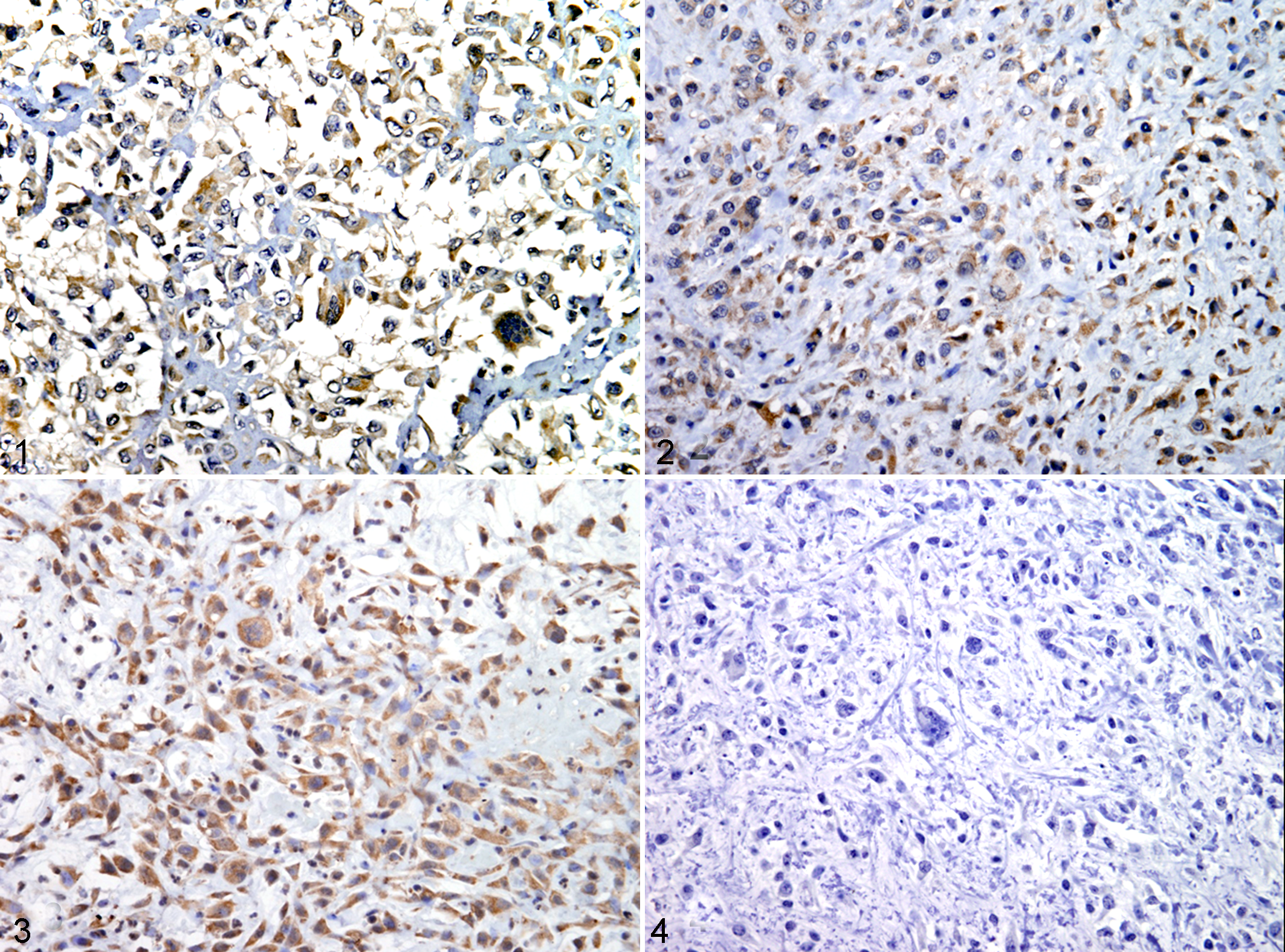
Osteosarcoma; canine, No. 1, osteosarcoma cells immunohistochemically labeled 5-LOX (intensity score 1) counterstained with Gill’s hematoxylin.
Osteosarcoma Relative 5-LOX Labeling Intensity, Mitotic Index, Percentage Positive Tumors and Mitotic Index/Labeling Intensity Correlation
NSAID MTT proliferation assays and Western blot for 5-LOX
Basal expression of 5-LOX in the 3 OSA cell lines showed that both OS2.4 and HMPOS cells displayed easily identifiable bands for 5-LOX (76 kDa) when compare to histidine-tagged commercially available 5-LOX control protein (80 kDa, Cayman Chemical; Fig. 5). Examination of COX inhibition showed no significant decrease in cell viability within physiologically relevant concentrations of RWJ 20142 (< 10 μM) for all 3 cell lines. There was mild cell viability differences observed at 25 μM in the HMPOS and OS2.4 cell line and at 50 μM in the HMPOS cell line when compared to vehicle control treated cells (DMSO). The 5-LOX inhibitor tepoxalin showed significant reduction in cell viability for all 3 cell lines (Fig. 6). The OS2.4 cell line seemed to be most sensitive, showing a significant decrease in cell viability at concentrations of 1.67 and higher (P < .05; Fig. 6), with the HMPOS cells showing significantly decreased viability at concentrations of 3.13 μM and higher (P < .05; Fig. 6). However, neither cell line achieved over 50% inhibition until treatment with 12.5 μM. The least sensitive cell line was the D17 cell line where 50% growth inhibition was only achieved with the highest dose provided (50 μM).
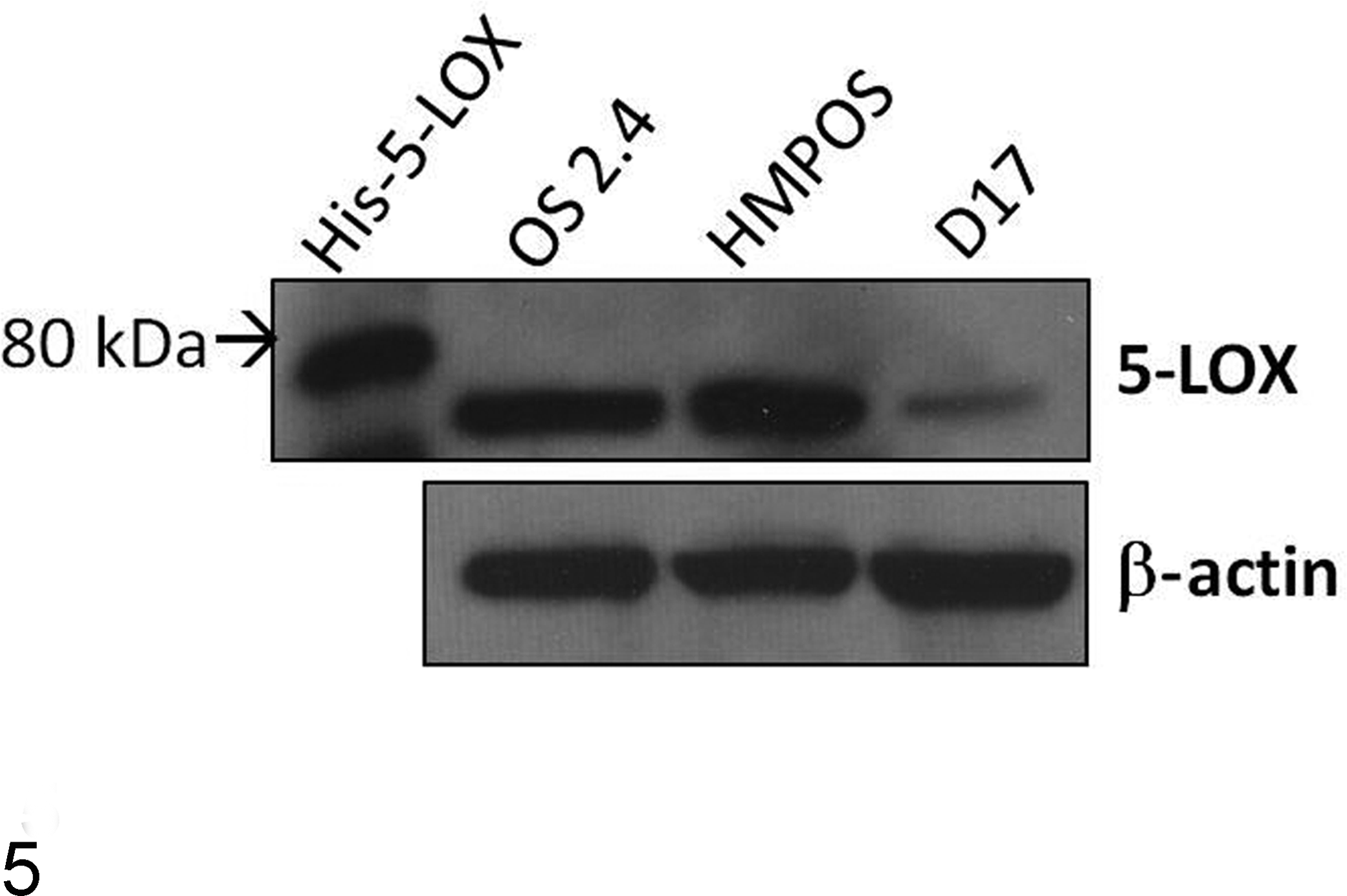
Western blot analysis of 3 canine osteosarcoma cell lines, including OS2.4, HMPOS, and D17 cells, as compared to commercially available his-tagged 5-LOX. Both HMPOS and OS2.4 cells show pronounced 5-LOX expression. All cells were loaded equally as represented by the uniform β-actin immunoblot.
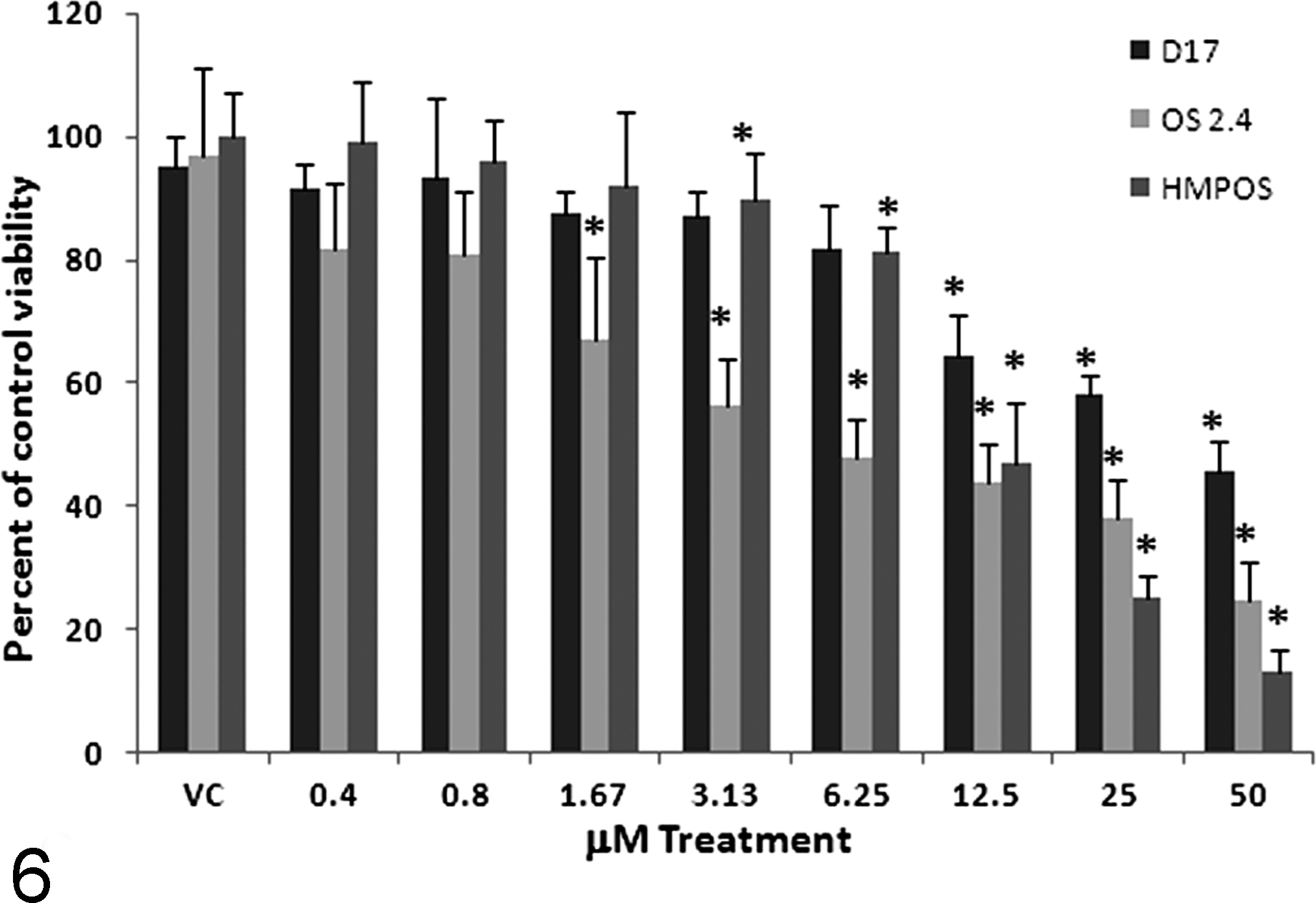
Canine HMPOS, OS2.4, and D17 cell viability (mean ± standard deviation) with tepoxalin treatment. Cells were plated at 5000 cells per well and treated with a serial dilution of tepoxalin for 48 hours. MTT viability was assessed as a percentage of control cell viability, revealing that tepoxalin treatment causes a significant retardation of cell growth for all 3 cell lines when compared to control and vehicle control–treated cells. Asterisk (*) indicates significant reduction in cell growth at these concentrations (P < .05).
LTB4 and 5-oxo-ETE/tepoxalin MTT assays
Cell viability was assessed using concentrations of tepoxalin that caused a decrease in cell viability (0.2–50 μM) in a serial dilution with the addition of promitogenic lipid mediators back into the culture media to determine if the proliferative capacity could be recovered after tepoxalin-induced 5-LOX inhibition (Fig. 7). Addition of 100 and 500 nM of LTB4 or 5-oxo-ETE, at previously used concentrations from successful lipid mediator experiments, 11 did not cause an increase in cell viability regardless of the concentration of tepoxalin used.
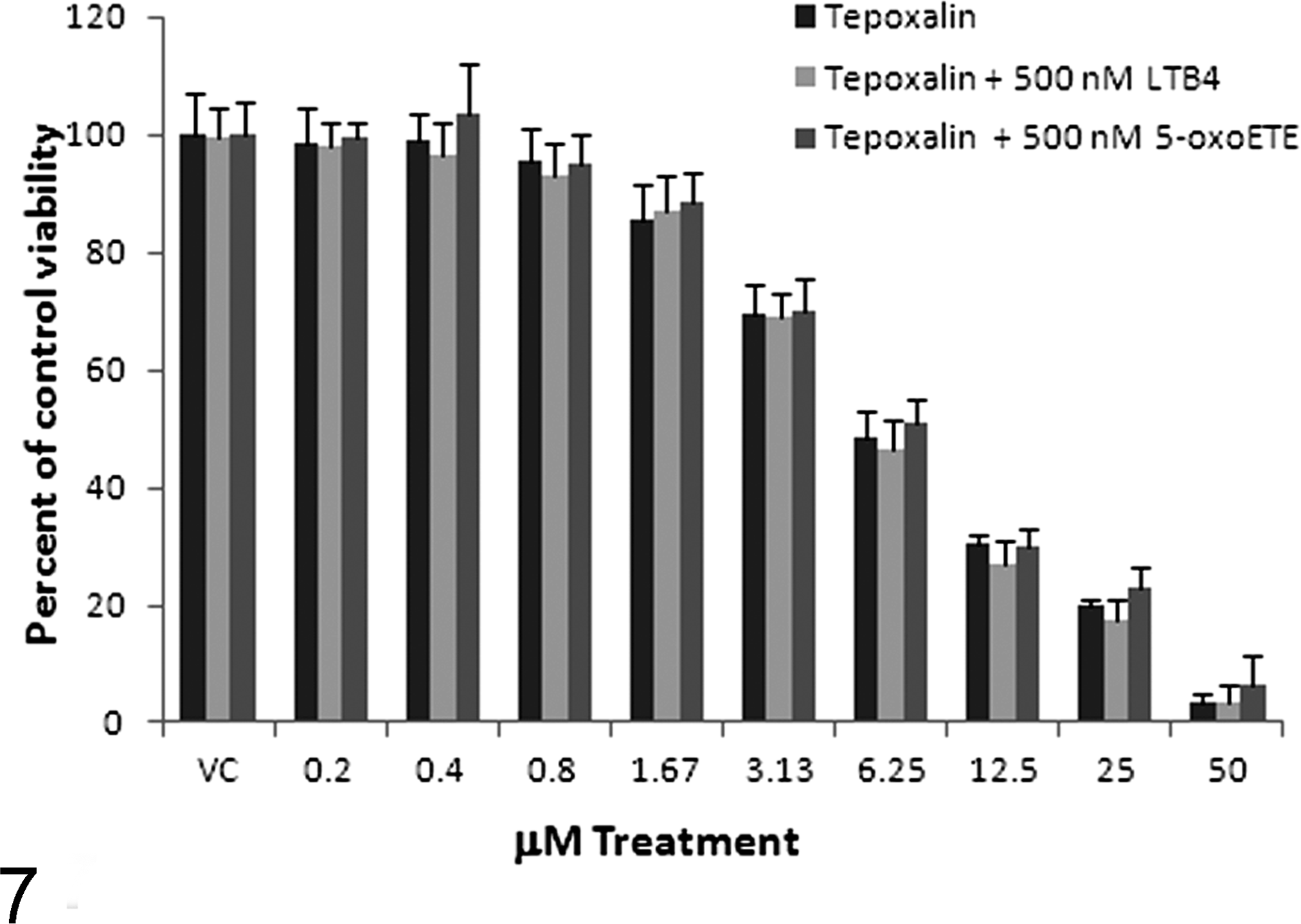
Canine HMPOS MTT assay percentage viability (mean ± standard deviation) with 5-LOX inhibition and promitogenic lipid mediators (LTB4 and 5-oxo-ETE). Cells were treated with varying concentrations of vehicle control, tepoxalin alone, or tepoxalin with LTB4 or tepoxalin plus 5-oxo-ETE for 48 hours. There were no significant differences noted among the 3 treatment groups at any concentration of tepoxalin when compared to viability of as a percentage of control cell viability.
Xenografted mice
All mice grew measureable tumors along the right flank, and all xenograft tumors labeled positive for 5-LOX (data not shown). Unfortunately, 1 mouse from group B aspirated during oral gavage and had to be euthanized by day 3 of the experiment, leaving only 3 mice in group C. Examination of the mice histologically showed that organ tissues were grossly and histologically normal with no signs of degeneration or toxic changes associated with the tepoxalin treatment in any of the treated mice. Lung sections examined on all mice showed evidence of pulmonary metastasis histologically (Fig. 8), while on gross necropsy only 2 of the xenografted tumor mice showed visual metastatic tumor burden in the lung tissue (one 10 mg/kg and one 50 mg/kg).
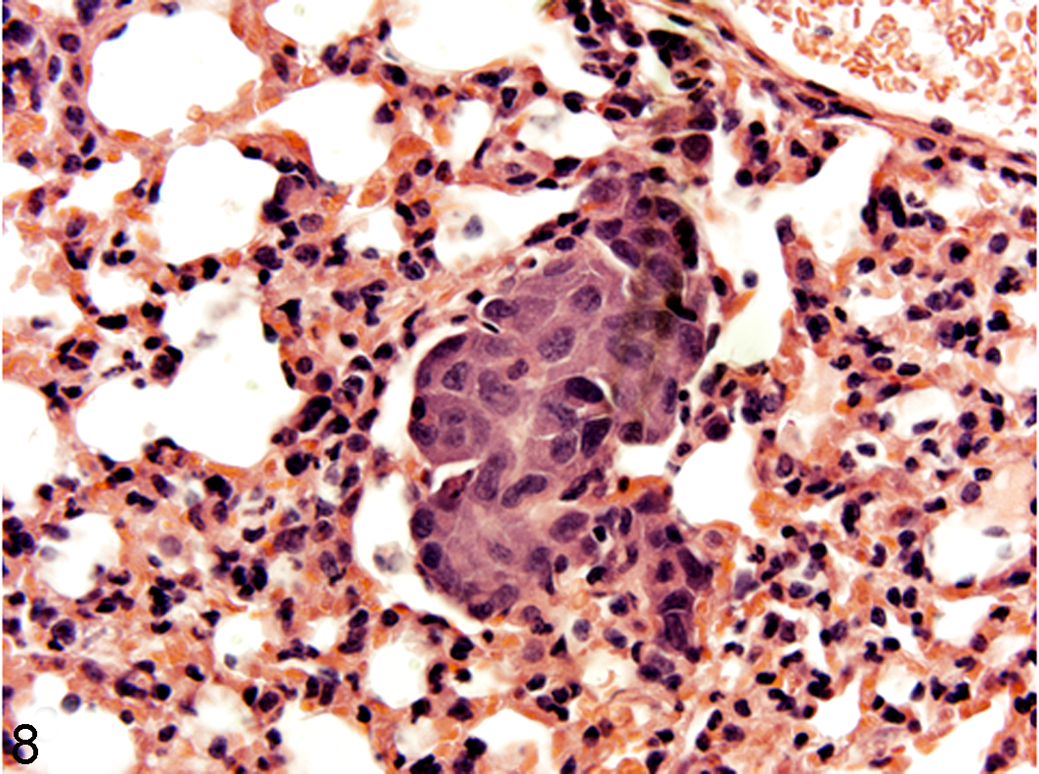
Lung; CByB6F1/J-Foxn1nu mouse; group B, No. 1, micrometastasis of the original HMPOS canine osteosarcoma xenografted tissue, hematoxylin and eosin staining.
Two of the mice in group A were euthanized due to tumor size (greater than 2.5 × 2.5 cm) by day 7 of treatment. The remaining 2 mice in group A had to be euthanized by day 21. One mouse from group B was euthanized on day 17 of treatment, and 2 mice from group C were euthanized on day 21 of treatment. All early euthanasia was due to xenografts reaching the upper limits of tumor size according to our institutional care and use committee protocol; therefore, data were censored at each of these points due to euthanasia. When examining the groups of mice for mean number of living days, the mean for each group showed a significant difference between groups A and C (P < .05; Table 2). Final tumor weights and final tumor volumes were not significantly different between groups, while tumor volume assessed at each time point was significantly different between groups A and C at days 7, 10, 14, and 21 (P < .05; Fig. 9). Additionally, group B tumor volumes were significantly greater than that group C at days 24 and 28 (P < .05). MI comparing all 3 groups showed that group C had a lower MI than group A (P < .05; Table 2).
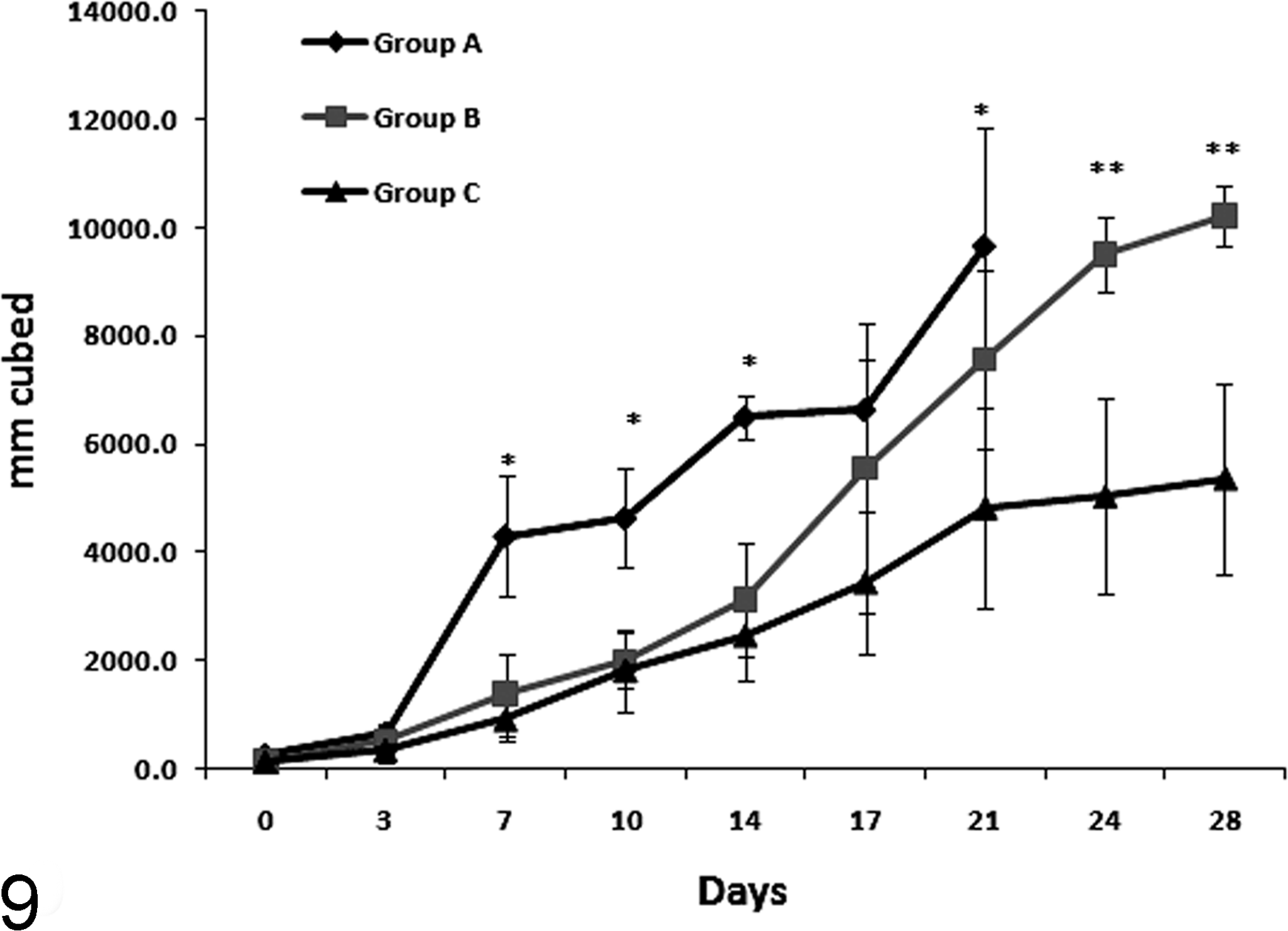
Mean (± standard error) tumor volume for all groups (A, placebo daily; B, 10 mg/kg of tepoxalin daily; C, 50 mg/kg of tepoxalin daily) over the 28-day trial. Asterisk (*) indicates a significant difference was observed at days 7, 11, 14, and 21 between group A (placebo) and group C (50 mg/kg) only. Asterisks (**) indicate significant differences between group B (10 mg/kg) and group C (50 mg/kg).
HMPOS Canine Osteosarcoma Xenograft Model: Mean Final Tumor Weight, Mean Tumor Volume, Mean Living Days a

aAll data expressed as mean ± standard deviation.
bSignificant difference from control mice.
Discussion
Inhibitors of arachidonic acid metabolism have been used clinically to treat or prevent some neoplasias with COX inhibition being the primary target. 21,31,32,37 Traditionally, in veterinary medicine, COX inhibitors have been utilized to treat transitional cell carcinomas. 26 In canine OSA, the clinical utility for COX inhibitors has been scrutinized due to only mild COX-2 expression in neoplastic cells and the need for supraphysiologic concentrations to inhibit OSA cell proliferation or migration. 31,37 Growing evidence in humans shows that dual COX/LOX inhibitors may demonstrate greater efficacy in treating certain types of cancers. 5,9,11 In veterinary medicine, the dual COX/5-LOX inhibitor tepoxalin is an established analgesic for orthopedic disease and has recently been shown to be mildly efficacious in slowing tumor growth in a canine OSA xenograft model. 40
The relative expression of 5-LOX in canine or human OSA was unknown; therefore, we examined the expression of 5-LOX through immunohistochemistry on 30 appendicular and 30 axial OSAs. Results showed equal relative numbers, percentage of tumor labeling and intensity across the 2 anatomic locations, revealing just over 65% of canine OSAs labeled positive; however, this staining was only mild to moderate in most cases, making its overall relevance questionable. The pattern of labeling was primarily cytoplasmic, which might have biologic relevance, as nuclear localization of 5-LOX is necessary for production of the lipid mediators associated with pro-proliferative activities, such as LTB4 and 5-oxo-ETE. 10,35,36 Previous literature has shown that human OSA cells do not secrete lipid mediators such as LTB4 or 5-hydroxytetraenoic acid when overexpressing 5-LOX. 18 Only after treatment with calcium ionophores will 5-LOX localize to the nucleus and induce eicosanoid production from arachidonic acid metabolism. 18 Further examination of the HMPOS cells in vitro suggests that 5-LOX is not metabolizing arachidonic acid into eicosanoids under normal growth conditions. Use of 5-LOX inhibitors in vitro has been associated with an increased apoptotic rate and slowing in cell proliferation in various cancer cell lines. 4,8 The underlying mechanisms are thought to be multifactorial and may include enhanced tumor suppressor phosphatase and tensin homologue function, induction of tumor necrosis factor-related apoptosis-inducing ligand receptors and death receptor 5, and increased mitochondrial dysfunction. 4,5,8,41,42,47 Regardless, of the mechanism(s) involved, our results suggest that these nontraditional activities may be involved and should be fruitful areas of investigation based on our HMPOS, OS2.4, and D17 viability data.
Previous investigations into the viability of OSA cells with tepoxalin treatment or RWJ20142 (TAM) showed that even at high concentrations (above 100 μM) there was no measurable decrease in cell proliferation. 40 Our MTT proliferation assays demonstrated growth inhibition using tepoxalin concentrations ranging from 3.13 to 50 μM, with the low end of the concentration range being physiologically achievable when administered to dogs at 100 to 300 mg/kg of body weight daily. 22 No inhibitory effects on growth were noted when RWJ20142 (TAM) was used in the same assays, further validating previous findings where meloxicam and deracoxib were unsuccessfully used on OSA cells at similar concentrations. 32,38,46 The disparity of our results using HMPOS cells versus the results of Sottnik and colleagues and their Abrams xenograft model of OSA are perplexing and might be related to the cell lines used in the experiments or possibly the growth conditions used in vitro. We typically use a very simple growth medium containing only 10% FBS and an antibiotic and antimycotic solution to maintain cells and perform experiments; however, Sottnik and colleagues use a high glucose solution, increased amounts of FBS, multiple buffers, and other amino acid and vitamin additives to promote growth that may mask the effects of tepoxalin or alter tepoxalin availability in the media. Additionally, the effects of tepoxalin on tumor growth appear to be more dramatic than in the previous study that used the Abrams OSA xenograft model, which may be due to multiple factors. These include differences in HMPOS cell sensitivity or overall increased aggressiveness (shorter study time due to rapid tumor growth) of our xenograft model.
We used the HMPOS cells in a xenograft model to confirm whether tepoxalin could slow tumor formation due to our viability results and the known 5-LOX expression, as well as it being established as a fast-growing metastatic model where we could examine relative growth rate and metastasis during tepoxalin treatment. 17 Our dosing regimen falls within the safe dosing regimen where dogs showed no negative consequences during a 6-month toxicity study. 22 However, it must be noted that the mice appear to metabolize tepoxalin much quicker than dogs or rats, 40 achieving very high serum concentrations of the tepoxalin metabolite RWJ20142, a COX inhibitor. COX inhibition has been associated with a decreased metastatic potential in canine in vitro and xenograft studies. 32 This disparity brings into questions whether our oral dosing and the decreased tumor size and MI was due to the 5-LOX inhibitory capability or the COX inhibitory capabilities. Due to the previous pharmacokinetic data in mice, we postulate that RWJ20142 could be the more important metabolite as it relates to tepoxalin treatment and may be the primary antiproliferative metabolite. Regardless of the mechanism of action, these positive xenograft the results are encouraging and warrant further investigation into tepoxalin as an antiproliferative drug. Use in dogs might deliver higher doses of tepoxalin to tumor tissue based on the species differences in pharmacokinetics, and the combination of tepoxalin and RWJ20412 may prove to be efficacious in retarding primary tumor growth, as well as potential metastasis.
To conclude, we have demonstrated expression of 5-LOX in a population of canine appendicular and axial OSAs, suggesting that 5-LOX may be a potential target for antineoplastic therapy; however, it does not appear that antiarachidonic metabolism is the primary means by which 5-LOX inhibitors influence neoplastic cell proliferation. We have yet to elucidate the exact mechanism for anti-5-LOX activity, but these inhibitors appear to be effective in decreasing cell proliferation at potentially clinically achievable concentrations. Further, we have shown that tepoxalin treatment using a canine OSA xenograft model is effective at retarding tumor growth. Thus, in an era of multimodal therapies, further examination of tepoxalin in OSA models and potentially even clinical patients is warranted.
Footnotes
Acknowledgements
The retrospective tumor analysis and in vitro work were supported by a grant from the Morris Animal Foundation, with special appreciation to the American Animal Hospital Association Foundation for their support of this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Morris Animal Foundation competitive grant for the work represented.
