Abstract
Papillomavirus-associated cervical cancer is the second most common neoplasm in women but has rarely been reported in animals. This report describes cervical and vaginal intraepithelial neoplasms identified in routine histologic specimens obtained from 20 (5.2%) of 385 female cynomolgus macaques (
Introduction
Cervical carcinoma is currently the second leading cause of cancer death in women, claiming over 200,000 lives per year worldwide. 12 The major risk factor for development of cervical cancer in women is infection with certain types of human papillomavirus (HPV). 3 Genital HPV infection is one of the most common sexually transmitted diseases, with a worldwide prevalence ranging from 20% to 46%. 1,4,9,10,16 Although most genital HPV infections are transient, specific types such as HPV 16 and 18 may persist and induce progressive neoplastic changes. 16,24 The causal role for HPV in cervical cancer is supported by extensive epidemiologic and mechanistic studies. In women, deoxyribonucleic acid (DNA) from the more oncogenic HPV types has been found consistently in 90% to 100% of cervical cancers, 27 and molecular studies in the past decade have confirmed the transforming properties of the primary papillomavirus oncoproteins, E6 and E7, which inhibit tumor-suppressor proteins p53 and retinoblastoma. 3,25 This evidence has established HPV as a leading model of viral carcinogenesis and sparked the claim that HPV is the first “necessary cause” to be identified for a human cancer.
In the veterinary literature, over 75 animal papillomaviruses have now been identified in approximately 20 different species. These viruses are generally associated with benign cutaneous or mucosal papillomas or fibropapillomas, and papillomavirus-associated genital dysplasias in animals have been documented in only a few reports. 20,22 In nonhuman primates, cervicovaginal dysplasias were originally described several decades ago, 14,15 although no viral association was investigated. Ostrow et al. 21,22 later identified and cloned papillomavirus DNA from a lymph node metastasis of a primary penile carcinoma in a male rhesus monkey. Subsequently, fragments of rhesus papillomavirus DNA were isolated from cervical samples of a breeding cohort of 30 female macaques, some of which had known mating contact with the index male. Six of these females exhibited dysplastic or invasive cervical intraepithelial lesions. In a more recent study, cervical smears obtained from 286 rhesus and 7 cynomolgus macaques yielded 12 types of genital rhesus papillomaviruses and one cynomolgus papillomavirus, 5 suggesting that the diversity of certain animal papillomaviruses may rival that of HPVs. In the present report, we document a previously undescribed range of spontaneous genital lesions in female cynomolgus macaques and investigate their association with papillomavirus infection.
Materials and Methods
Animal histories
Animals were obtained by Wake Forest University School of Medicine between March 1996 and May 2000 from the Institute Pertanian Bogor in Indonesia. The animals were adult female cynomolgus macaques in four independent studies investigating cardiovascular effects of various oral hormonal replacement treatments in the surgically postmenopausal monkey model. Of the total of 385 animals, 337 were ovariectomized within 3 months of arrival; the remaining animals were intact throughout the study periods. Hormonal treatments were given to 236 (61.3%) of the ovariectomized animals and included oral conjugated equine estrogens (CEE) alone (
Histopathology
After each experiment, animals were sedated with ketamine and euthanatized using sodium pentobarbital (100 mg/kg, intravenous), a method consistent with the recommendations of the Panel on Euthanasia of the American Veterinary Medical Association. Necropsies of all animals were performed between September 1999 and October 2001 at the Comparative Medicine Clinical Research Center of the Wake Forest University School of Medicine. For each animal, the reproductive tract was removed and the uterus separated by a transverse section through the uterine neck. The remaining tract was divided by a midsagittal section extending from the vaginal canal to the uterine cervix. Cervicovaginal tissues were fixed in 10% phosphate-buffered formalin (pH 7.0), embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE) according to standard procedures. Tissue collection procedures and sampling intensity were uniform for all 385 animals (one HE section per animal, as shown in Figs. 1, 2). All slides were reviewed by a board-certified veterinary pathologist (J. M. Cline) without prior knowledge of treatment status. Lesions were scored using criteria adapted from the cervical intraepithelial neoplasia (CIN) nomenclature system, 8,28 as represented in Table 1, and reviewed by a board-certified human reproductive pathologist (J. Cappellari) for consistency with human terminology.
Classification of vaginal and cervical neoplasms in cynomolgus macaques. Morphologic criteria based on the CIN system. 8

∗ VIN = vaginal intraepithelial neoplasms; CIN = cervical intraepithelial neoplasm.
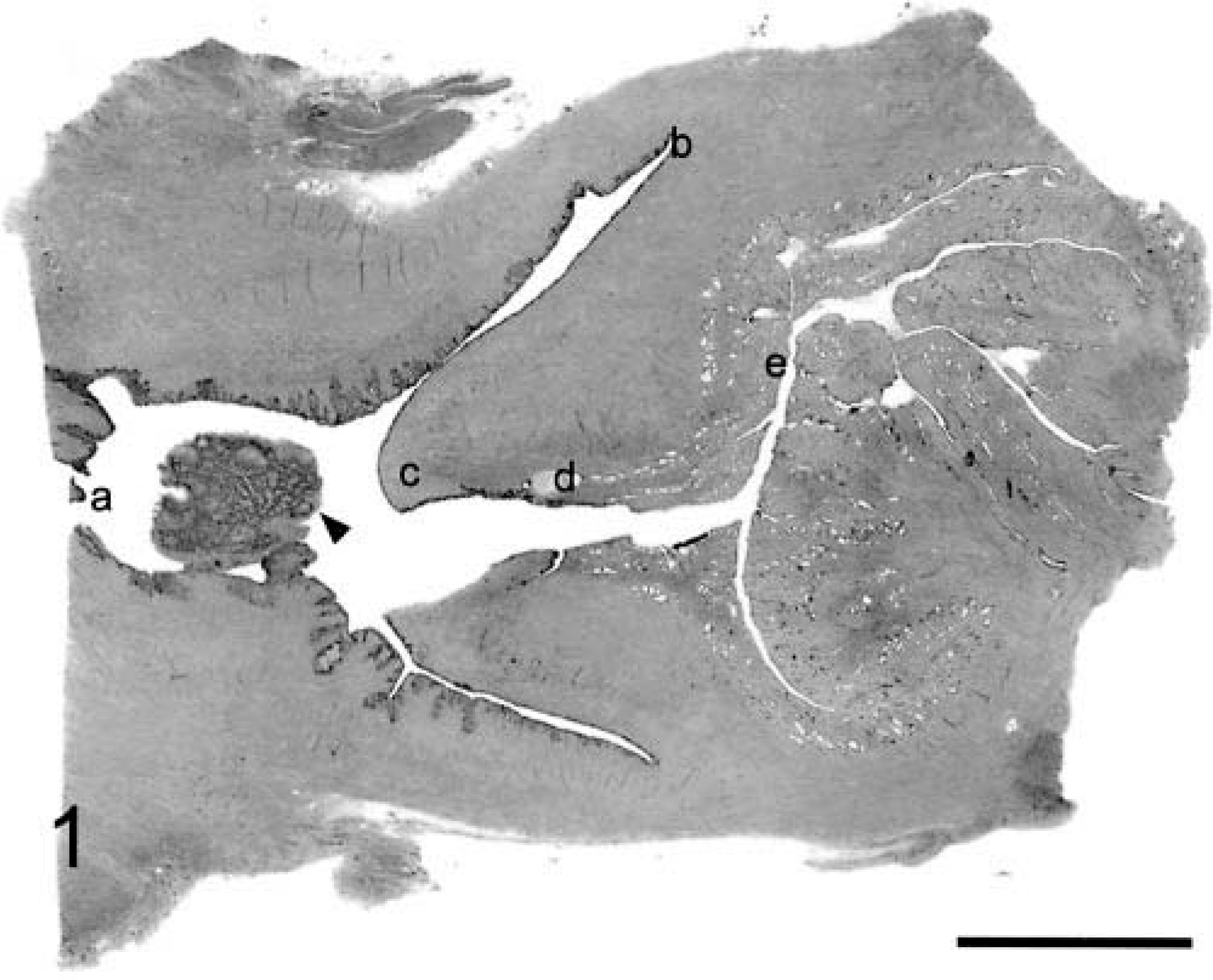
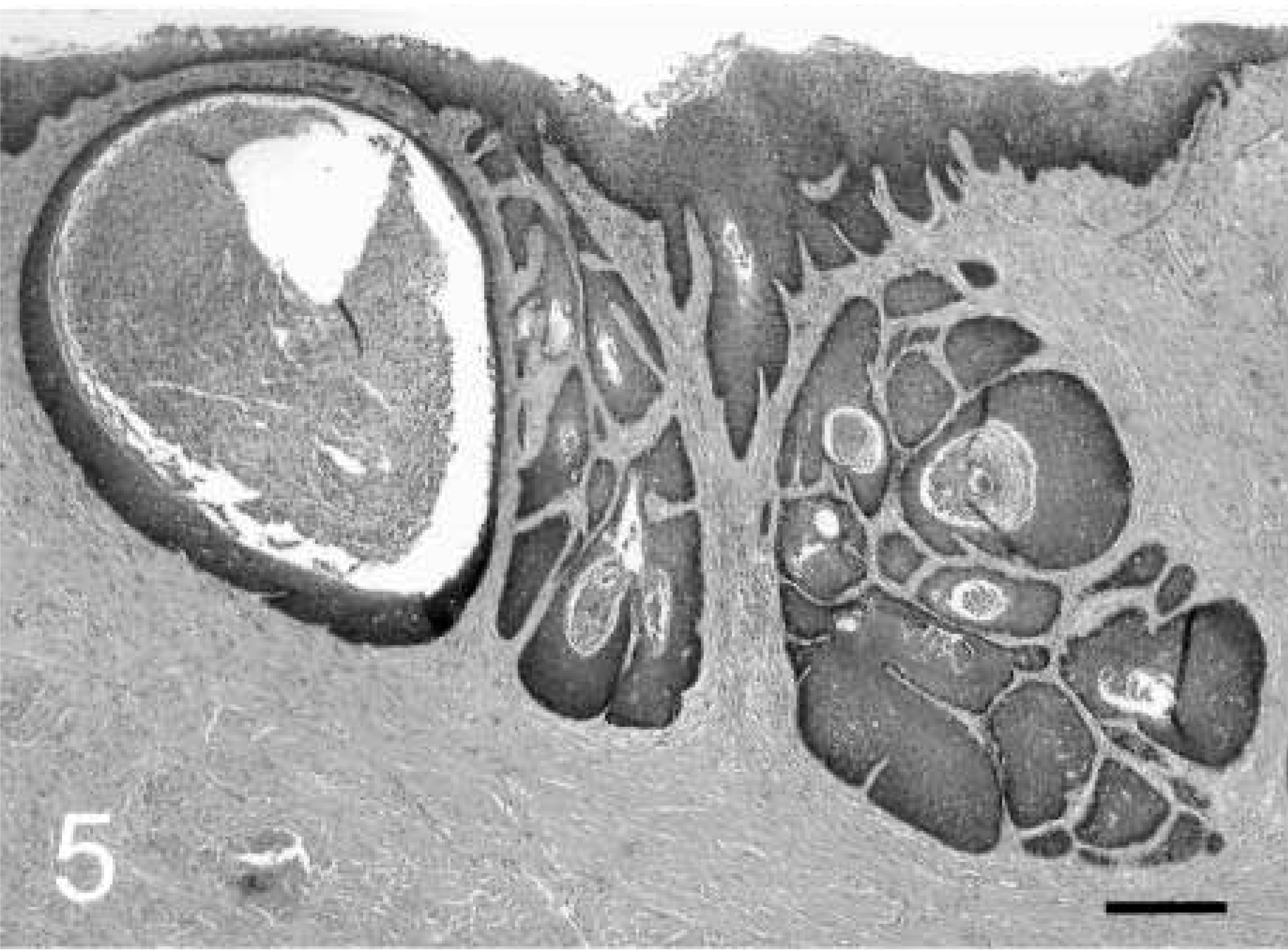
Cervix and vagina; cynomolgus macaques, animal Nos. 5295 and 5321. Subgross images of midsagittal sections through cervix and vagina. Letters indicate relevant anatomy: a = vagina; b = fornix; c = cervical os (ectocervix); d = squamocolumnar junction (endocervix); e = glandular or uterine cervix. Arrow denotes vaginal papilloma in
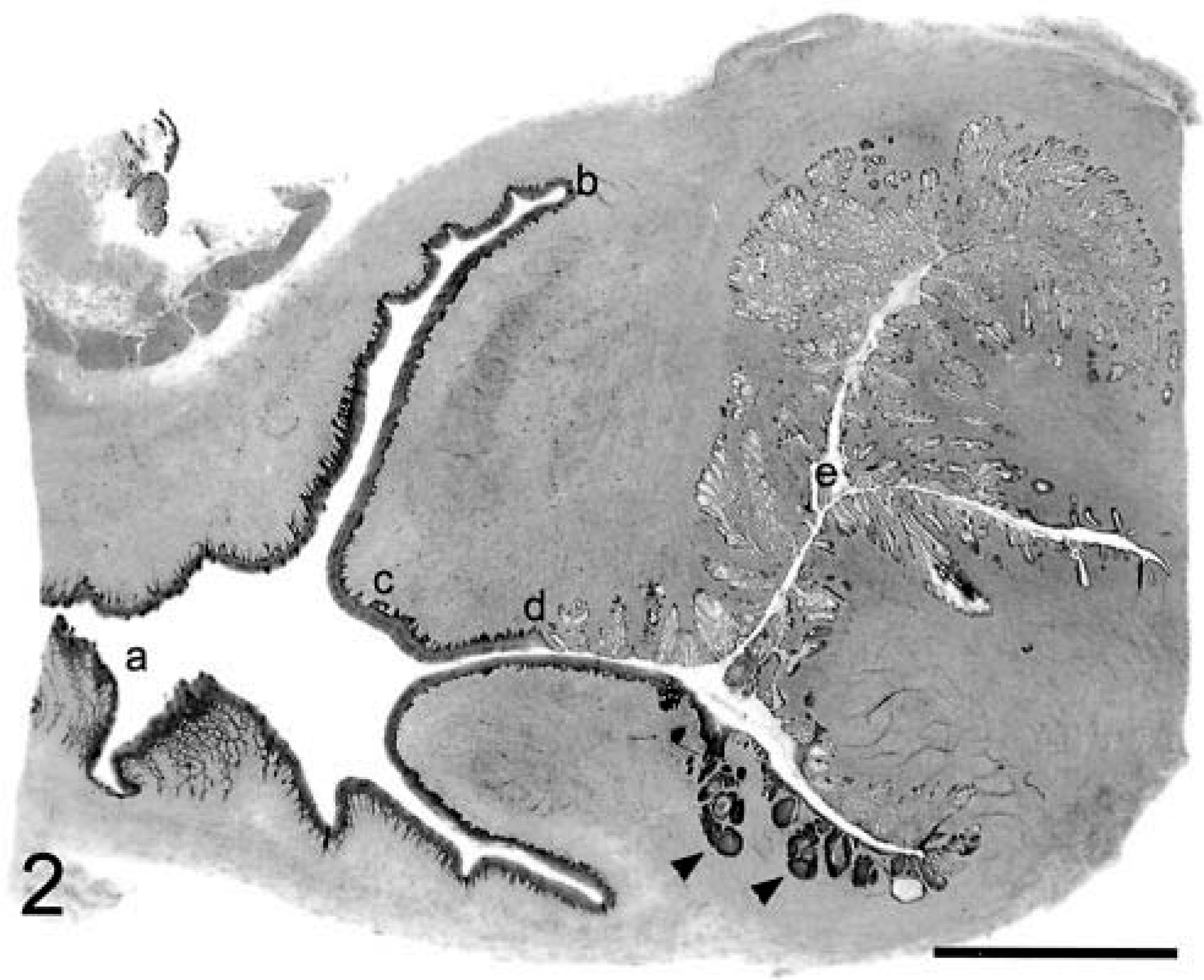
Cervix and vagina; cynomolgus macaques, animal Nos. 5295 and 5321. Subgross images of midsagittal sections through cervix and vagina. Letters indicate relevant anatomy: a = vagina; b = fornix; c = cervical os (ectocervix); d = squamocolumnar junction (endocervix); e = glandular or uterine cervix. HE. Bar = 5 mm.
Immunohistochemistry
Immunohistochemical staining for papillomavirus protein was performed on tissue sections from the 20 cases and four control tissues using the following primary antibodies: (A) rabbit polyclonal antibody raised to bovine papillomavirus type-1 antigens (NCL-PVp; Novacastra; Newcastle, UK), (B) mouse monoclonal antibody (IgG1) raised to E6 proteins from HPV types 16 and 18 (MAB874; Chemicon; Temecula, CA), and (C) mouse monoclonal antibody (IgG), specific for HPV types 1, 6, 11, 16, 18, and 31, raised to sodium dodecyl sulphate–disrupted bovine papillomavirus type 1 (MAB837; Chemicon). Primary antibodies were diluted to 1 : 50 (antibody A) or 1 : 100 (antibodies B and C) in Tris buffer, pH 7.4, containing 0.5% casein as blocking reagent. Staining was performed on representative tumor sections 4- to 5-µm thick that were deparaffinized and rehydrated in graded alcohols. For heat-induced epitope retrieval, slides to be treated with monoclonal antibodies (B and C) were incubated in 10 mM citrate buffer (pH 6.0) for 45 minutes in a steam bath. Biotinylated secondary Fc antibody was used as a linking reagent for all antibodies, and alkaline-phosphatase–conjugated streptavidin (Biogenex; San Ramon, CA) was applied for labeling. The chromogen used was Vector Red, obtained as Vector Red Substrate kit No. 1 (Vector Laboratories, Burlingame, CA), diluted in Tris (pH 8.2–8.5) and applied to slides for 5–10 minutes at 30–35 C. Negative staining controls were obtained by omitting the primary antibody and substituting nonimmune serum (BioGenex). For negative disease controls, cervical sections from cynomolgus macaques exhibiting no histologic lesions were immunostained as above. For positive disease controls, human cervical carcinoma sections were obtained from the Molecular Diagnostics Laboratory, Wake Forest University School of Medicine. All immunostaining procedures were performed without the knowledge of lesion status. The abundance and intensity of staining within lesions and in normal adjacent epithelium was subjectively graded on a scale of 0 to +4: 0, no staining; +1, minimal; +2, mild; +3, moderate; and +4, heavy. For detection of Epstein Barr–like virus proteins, immunostaining was performed as described above using a monoclonal mouse primary antibody (EBNA2, Clone PE2; DAKO; Glostrup, Denmark) diluted to 1 : 25 with Tris buffer. For a positive EBNA2 control, lymphoma tissue from a rhesus macaque (
DNA isolation and polymerase chain reaction
Polymerase chain reaction (PCR) analysis for detection of papillomavirus DNA was conducted using degenerate primers derived from multiple sequence alignment of rhesus macaque (type 1), pigmy and common chimpanzee, and bovine (type 4) and human (type 16) papillomavirus sequences, available on the NCBI nucleotide database (www.ncbi.nlm.nih.gov). HPV 16 was selected because of its association with invasive cervical carcinomas in women and bovine PV 4 because of its relative lack of species specificity. 19 Conserved sequences were identified using Clustal X MSA software program, and three pairs of homologous primers 17–21 bases in length were generated at the Wake Forest University DNA Core Laboratory, representing open reading frame regions from the viral E1, L2, and L1 genes. The degenerate primer pair in the L2 gene produced PCR products during preliminary studies and was used for all subsequent analyses. Sequences for this primer pair were 5′-RRTRKGBMGDGGWCAGCC-3′ and 5′-WMMCATDTCMCCATTCCTG-3′. DNA was isolated from formalin-fixed tissue sections using a TaKaRa DEXPAT kit (PanVera; Madison, WI), as described elsewhere. 13 Cervical tissues from animals with no histologic evidence of dysplasia were used as negative controls, and a pBR322 plasmid containing the full length genome of pHPV18 (ATCC 45152) was used as a positive control. PCR was performed using GeneAmp or GeneAmp Gold PCR Reagent kits from Applied Biosystems (Foster City, CA). Final thermal cycling conditions consisted of three temperature PCR, 95 C for 30 seconds, 60 C for 30 seconds, and 72 C for 30 seconds, for 25–45 cycles. A manual hot start at 60 C was used when standard Ampltaq polymerase was used.
Results
Histopathology
CIN and vaginal intraepithelial neoplasms (VIN) were observed in routine histologic specimens obtained from 20 (5.2%) of 385 cynomolgus macaques. The average age at death for case animals was 18.6 years (Table 2), similar to the mean age of 18.5 years for the 365 animals without lesions. Seven of the 20 case animals (35.0%) had received control diets, similar to the overall study population (38.7%). The hormonally treated cases (13 of 20) were receiving CEE alone (
Immunohistochemical scoring of cervical and vaginal lesions for papillomavirus antigen. Scores reflect the difference in immunostaining intensity between intraepithelial lesions and adjacent nonneoplastic epithelium. The following PPV Ab were used: (A) rabbit polyclonal antibody raised to bovine papillomavirus type 1 antigens; (B) mouse monoclonal antibody raised to E6 proteins from HPV types 16 and 18; and (C) mouse monoclonal antibody raised to bovine papillomavirus type 1.∗
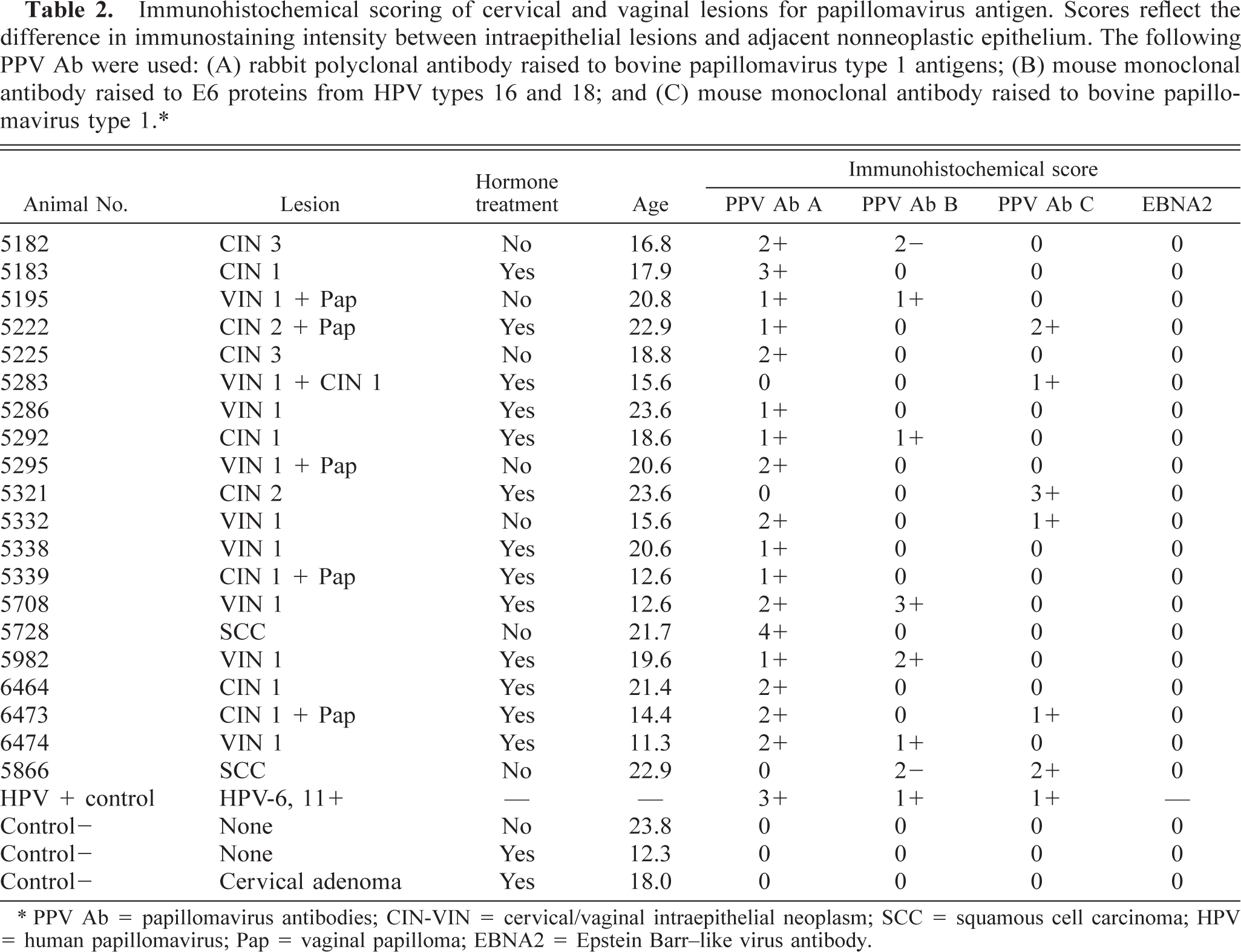
∗ PPV Ab = papillomavirus antibodies; CIN-VIN = cervical/vaginal intraepithelial neoplasm; SCC = squamous cell carcinoma; HPV = human papillomavirus; Pap = vaginal papilloma; EBNA2 = Epstein Barr–like virus antibody.
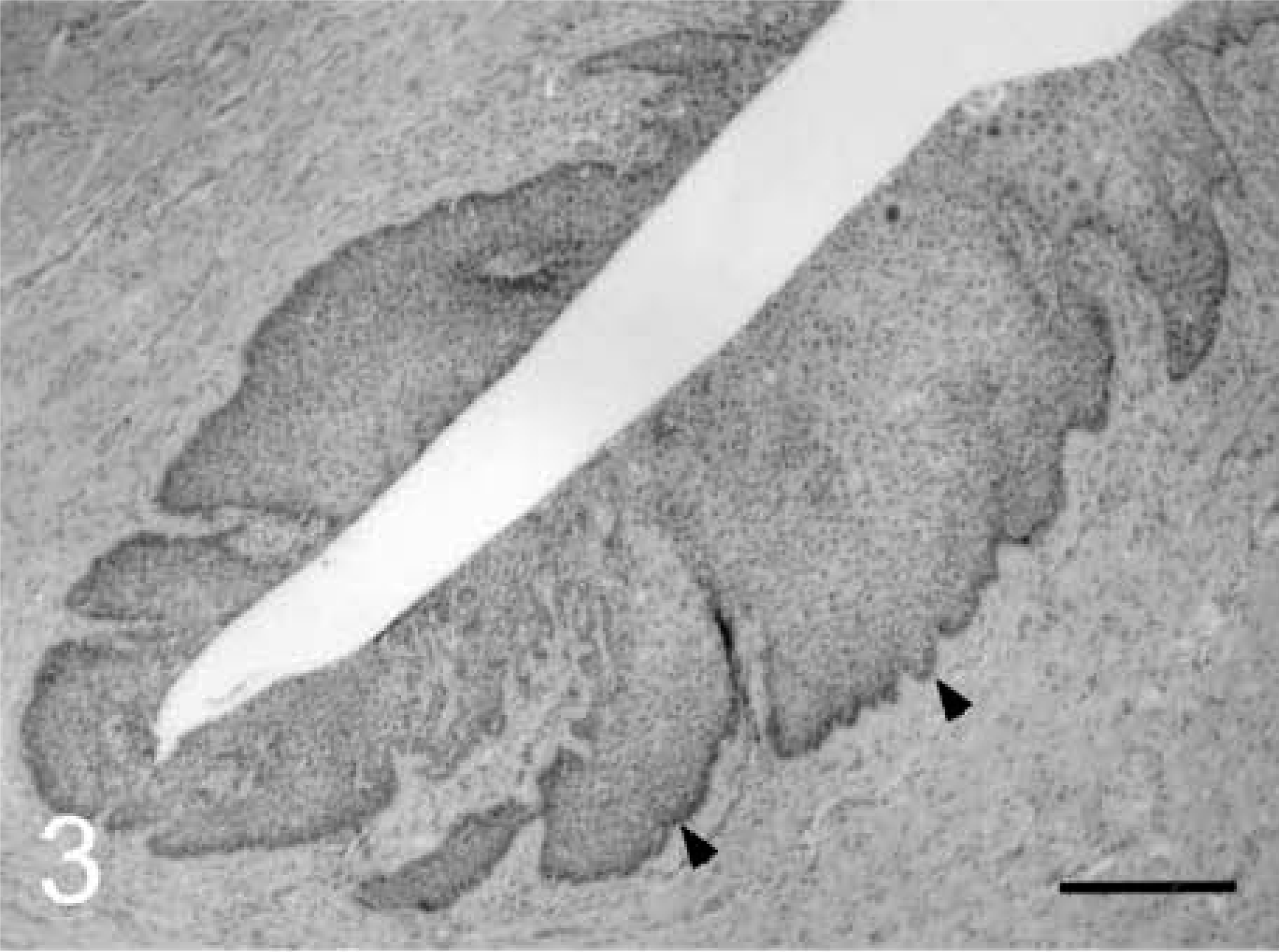
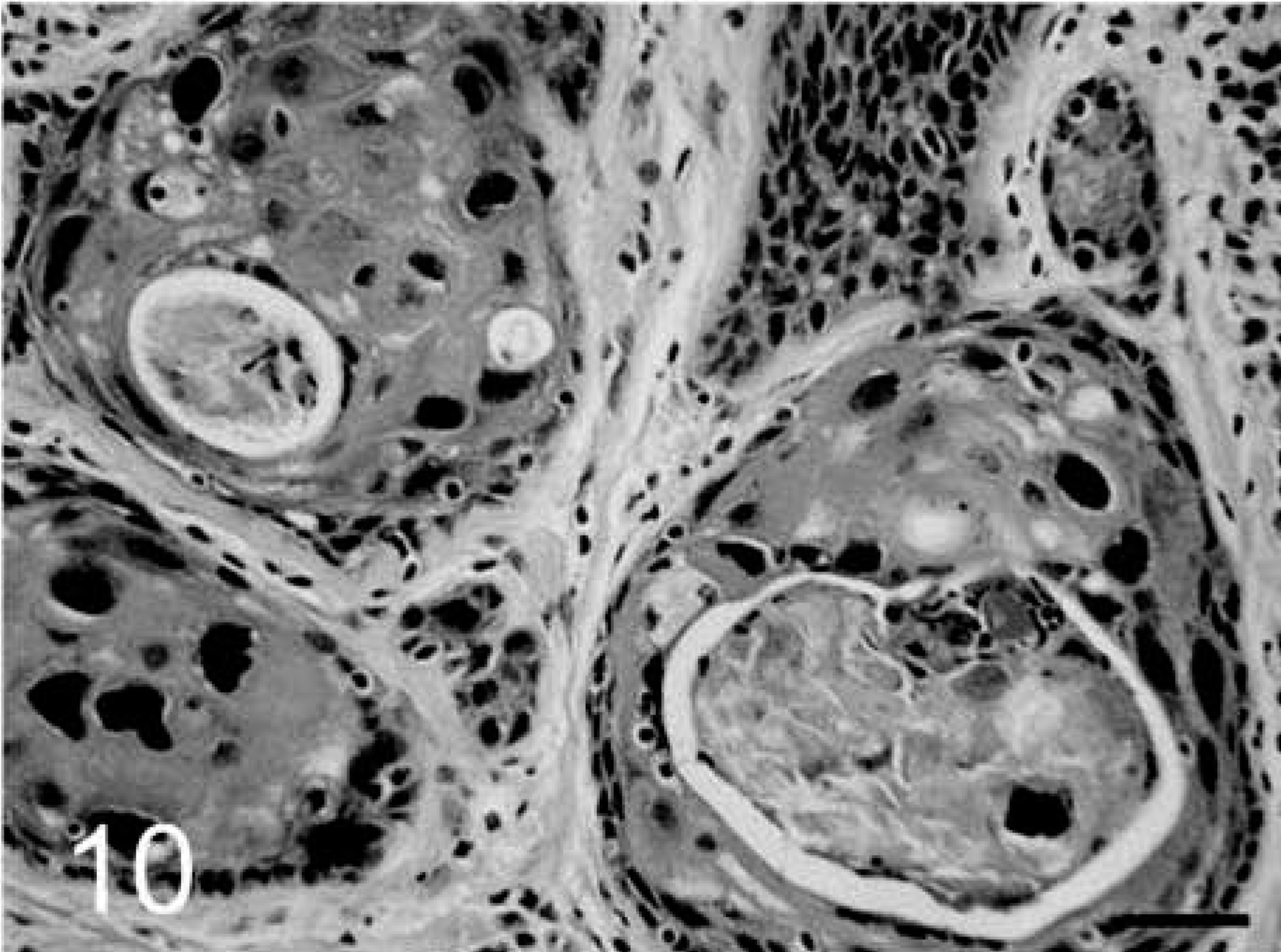
A spectrum of cervicovaginal lesions was represented, including 14 CIN-VIN 1, two CIN 2, two CIN 3, and two squamous cell carcinomas (Table 1, Figs. 3–10). Normal vaginal mucosa is shown in Figs. 11, 12 for comparison. A range of dysplastic changes was also present within individual cases as lesions grade CIN 2 and higher were often associated with lesser degrees of dysplasia. Common morphologic features of the intraepithelial neoplasms included mild to marked epithelial dysplasia with nuclear atypia, anisokaryosis, loss of polarity, formation of epithelial pearls, expansion of the basal epithelium, and multifocal perinuclear vacuolation (koilocytosis). Koilocytic changes were more common in the CIN-VIN 1 lesions. Lesions grade CIN 2 and higher had an increased mitotic index with atypical mitoses and mitotic figures above the basal layer. The two invasive carcinomas exhibited epithelial invasion of the underlying stroma with multifocal disruption of the basement membrane and submucosal aggregates of densely packed, disorganized, hyperchromatic basal epithelial cells (Figs. 9, 10). All lesions were found only on histologic evaluation; therefore, regional lymph nodes were not evaluated for metastases. All lesions graded CIN 2 and higher involved the cervical squamocolumnar junction (6 of 6) (Fig. 2), whereas CIN-VIN 1 lesions were located in the vaginal epithelium (10 of 14), fornix (2 of 14), ectocervix (3 of 14), and squamocolumnar junction (1 of 14). No viral inclusions were observed in any of the sections.
Cervicovaginal intraepithelial neoplasm (CIN/VIN); cynomolgus macaque, animal No. 5292. CIN 1 lesion at the fornix.
Ectocervical epithelium with atypical nuclei and surrounding koilocytes in the spinosum. HE. Bar = 20 µm.
Cervical intraepithelial neoplasm (CIN); cynomolgus macaque, animal No. 5321. CIN 2 lesion near the squamocolumnar junction.
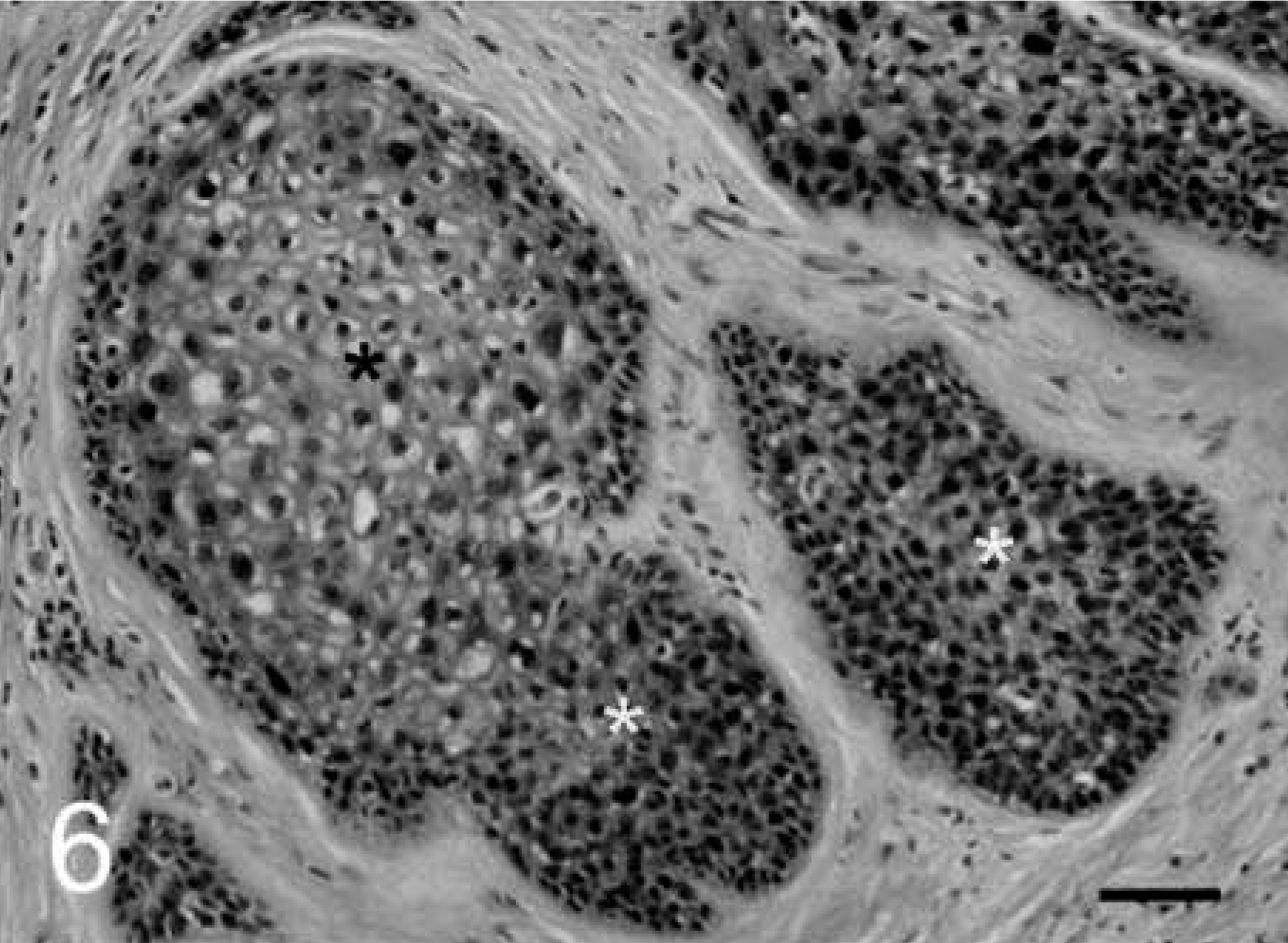
Aggregates of proliferative ectocervix with expanded zones of basal epithelium (white asterisks) and central areas of koilocytotic atypia (black asterisk). HE. Bar = 25 µm.
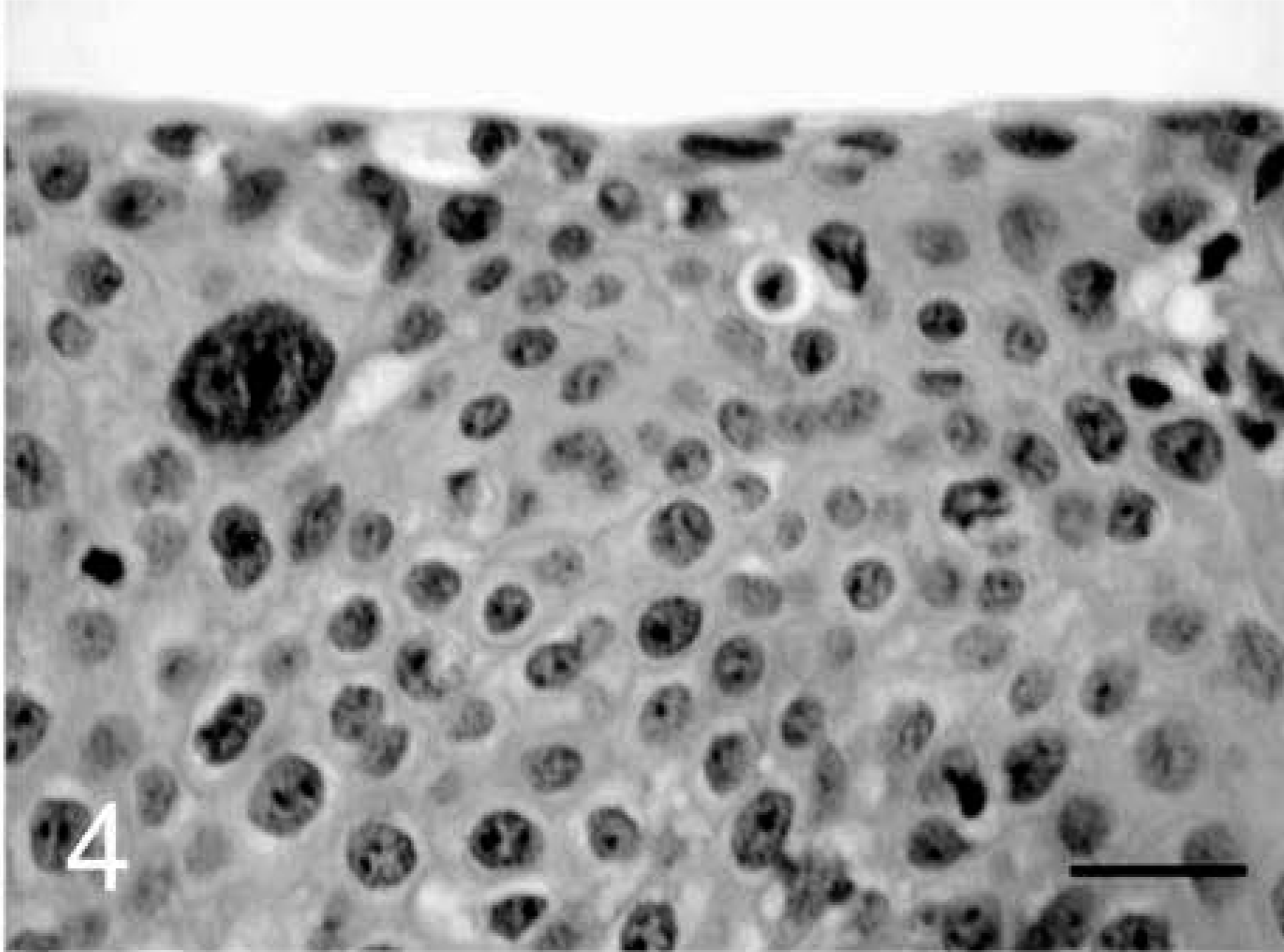
Cervical intraepithelial neoplasm (CIN); cynomolgus macaque, animal No. 5225. CIN 3 lesion near the fornix.
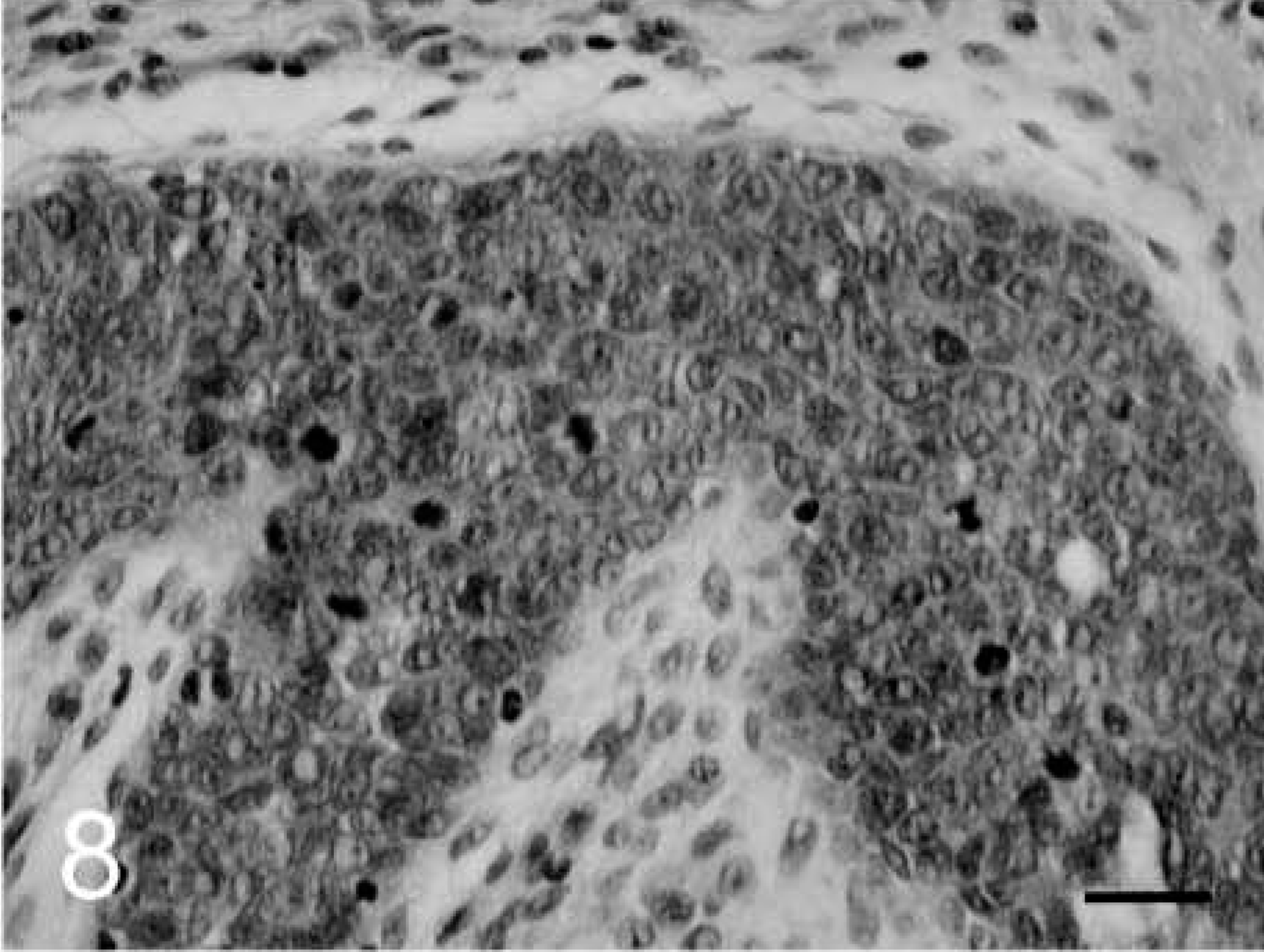
Full-thickness nonmaturative cervical epithelium with numerous mitoses. HE. Bar = 25 µm.
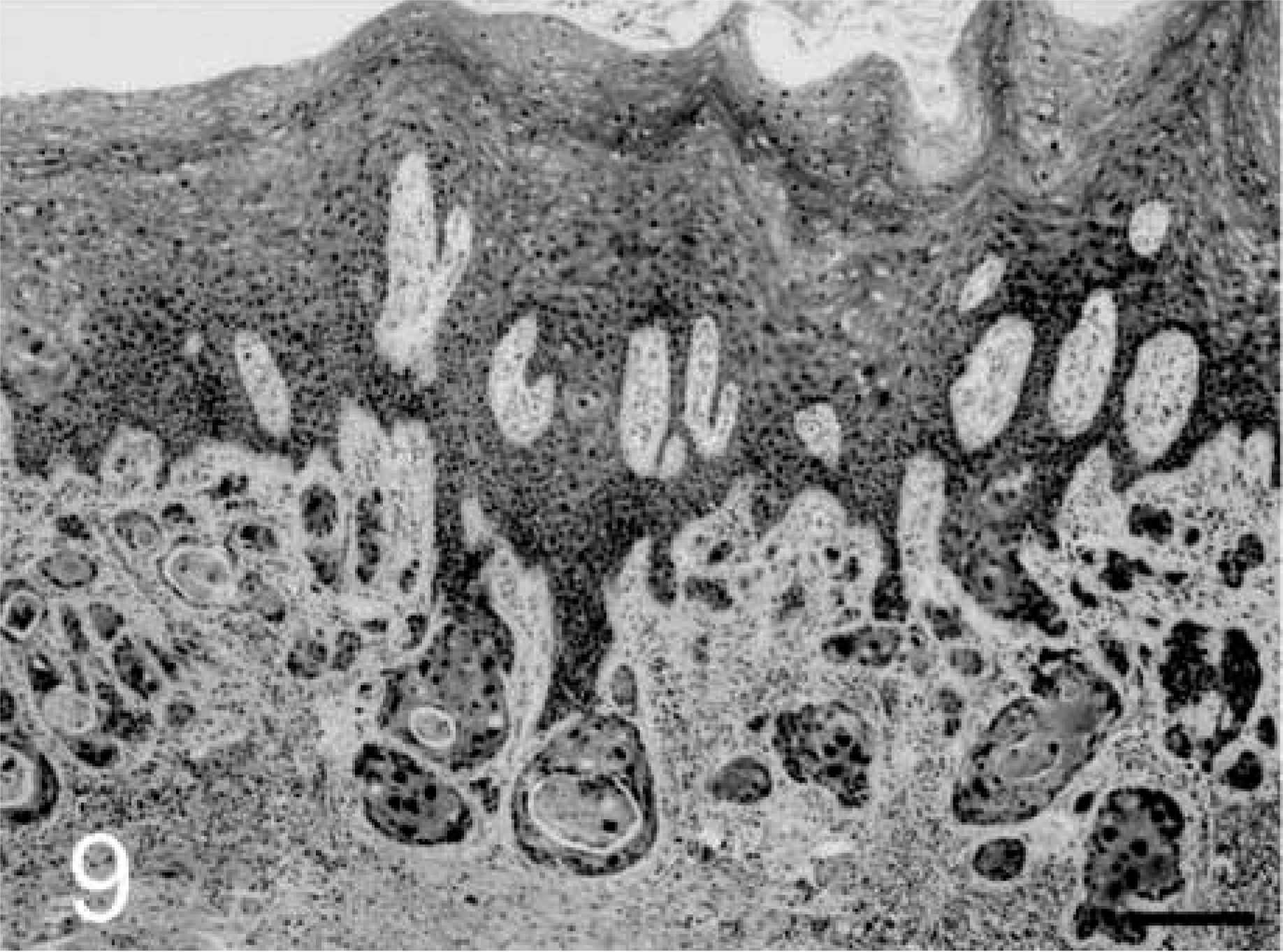
Invasive cervical carcinoma; cynomolgus macaque, animal No. 5728. Squamous cell carcinoma near the squamocolumnar junction.
Malignant squamous cells with large atypical nuclei and keratin accumulation. HE. Bar = 25 µm.
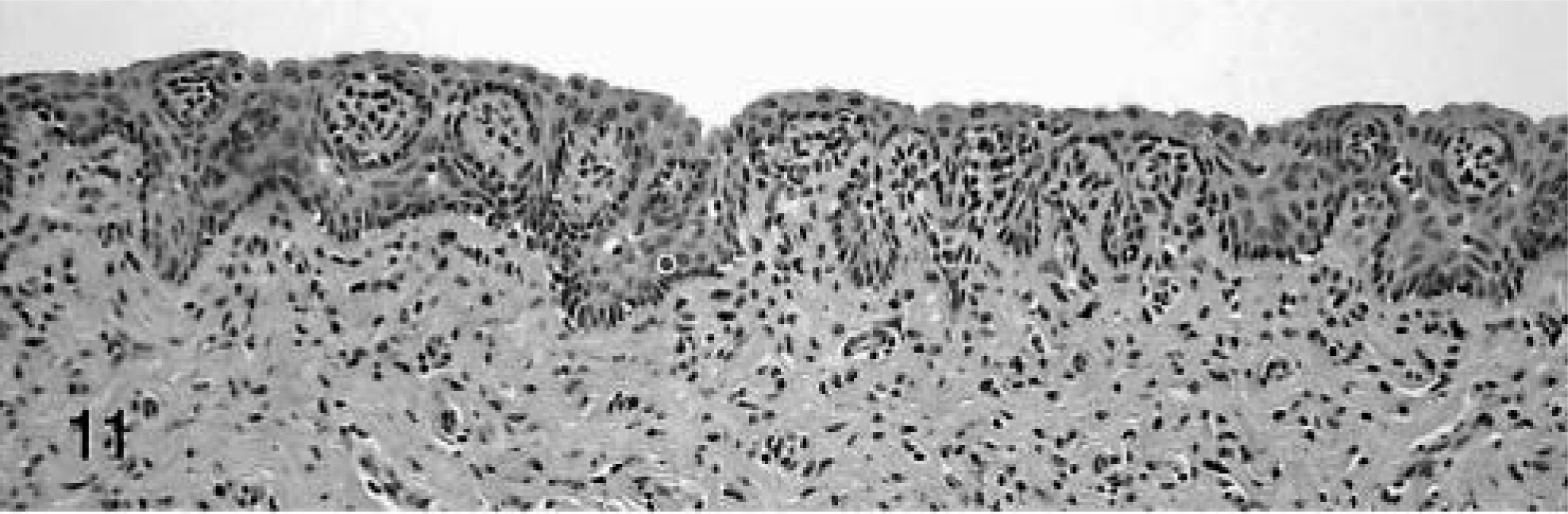
Normal vaginal mucosa; cynomolgus macaques.
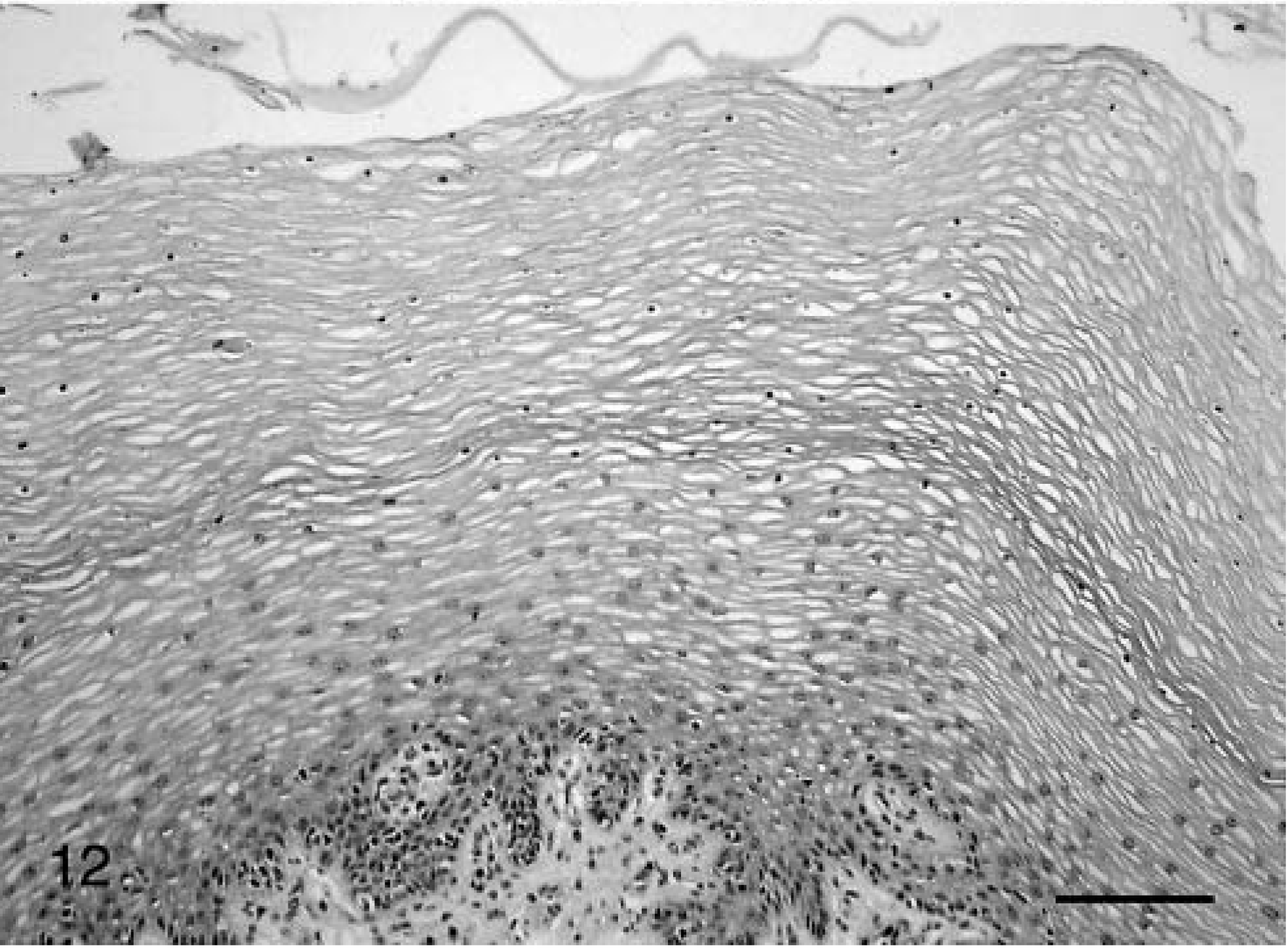
Hyperkeratotic vaginal epithelium from an ovariectomized animal treated with estrogen. HE. Bar = 100 µm.
Nine of the cases had multiple intraepithelial lesions; and of these, five animals exhibited vaginal papillomas, also known as warts or condylomata acuminata (Fig. 2). These exophytic verrucous neoplasms are associated with lower risk papillomaviruses in women (HPV types 6 and 11) and are generally considered benign. 11 Changes associated with the papillomas included pedunculation, expansion of the spinosum with koilocytosis and an arborizing epithelial pattern, and minimal to no expansion of the basal epithelium. Other genital lesions included lymphoplasmacytic cervicitis/vaginitis (5 of 20), squamous metaplasia of the glandular cervix (2 of 20), and cystic dilation of cervical mucous glands (3 of 20).
Immunohistochemistry
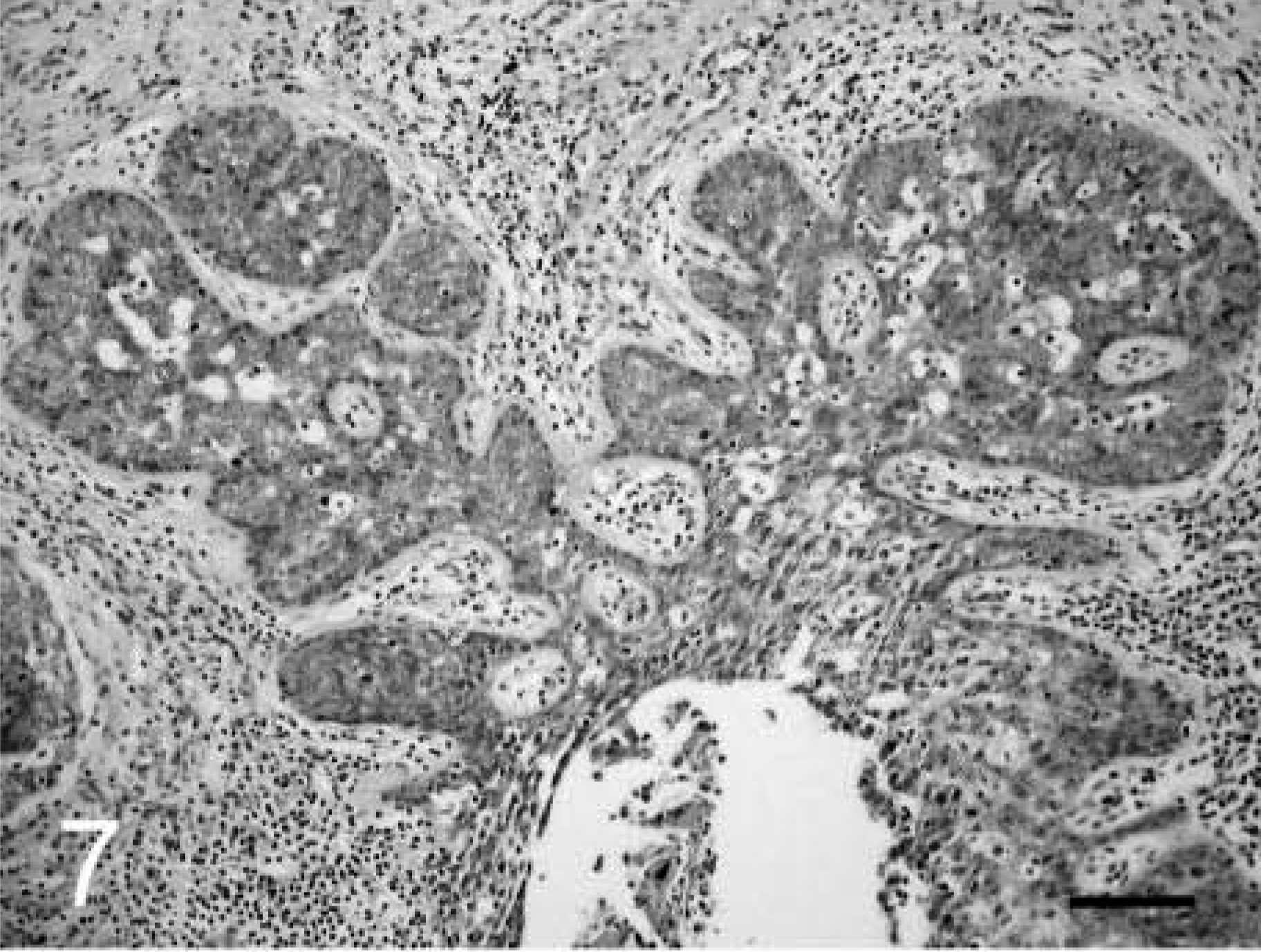
Tissues from all CIN-VIN cases were evaluated for the presence of papillomavirus and Epstein Barr–like virus antigens using immunohistochemistry. Selective lesional staining was observed with at least one of the three papillomavirus antibodies in all 20 cases: 17 of 20 (85%) cases for antibody A, five of 20 (25%) cases for antibody B, and six of 20 (30%) for antibody C (Table 2). In addition, four of the five papillomas tested stained positively for papillomavirus antigen. Immunoreactivity with antibodies A and B was cytoplasmic and located predominantly in the more superficial epithelial layers (Figs. 13, 14), whereas antibody C staining was both nuclear and cytoplasmic and included basal epithelium (Fig. 15). Sporadic staining of adjacent morphologically normal vaginal or cervical basal epithelium was present in several cases. No stromal staining was noted for any of the antibodies. Human cervical carcinoma control tissue stained positively with all three antibodies.
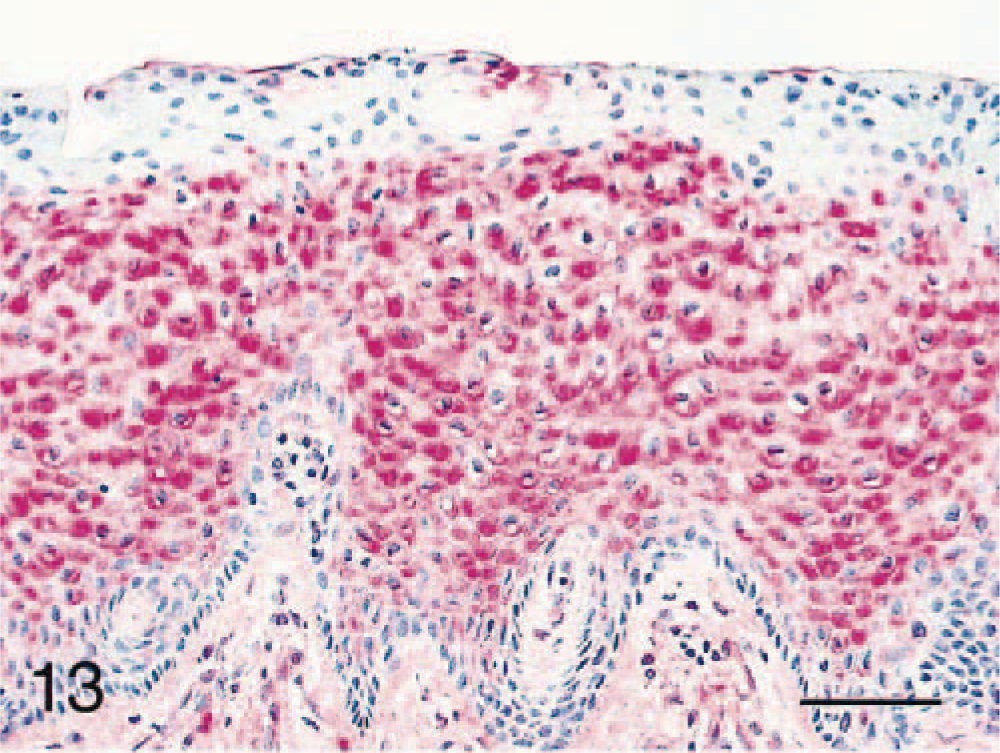
Cervical intraepithelial neoplasm (CIN); cynomolgus macaque, animal No. 5728. Positive immunostaining of CIN 1 lesion (adjacent to a cervical carcinoma) using a polyclonal antibody to bovine papillomavirus type 1 (antibody A). Bar = 50 µm.
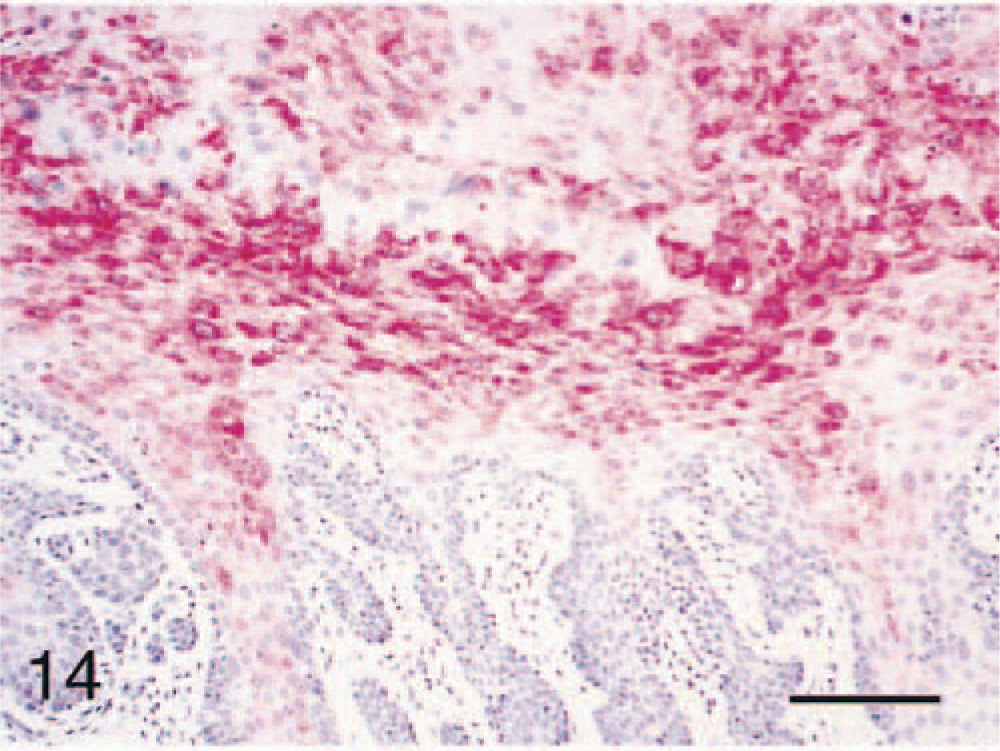
Vaginal intraepithelial neoplasm (VIN); cynomolgus macaque, animal No. 5708. Positive immunostaining of VIN 1 lesion using a monoclonal antibody to human papillomavirus antigens (antibody B). Bar = 40 µm.
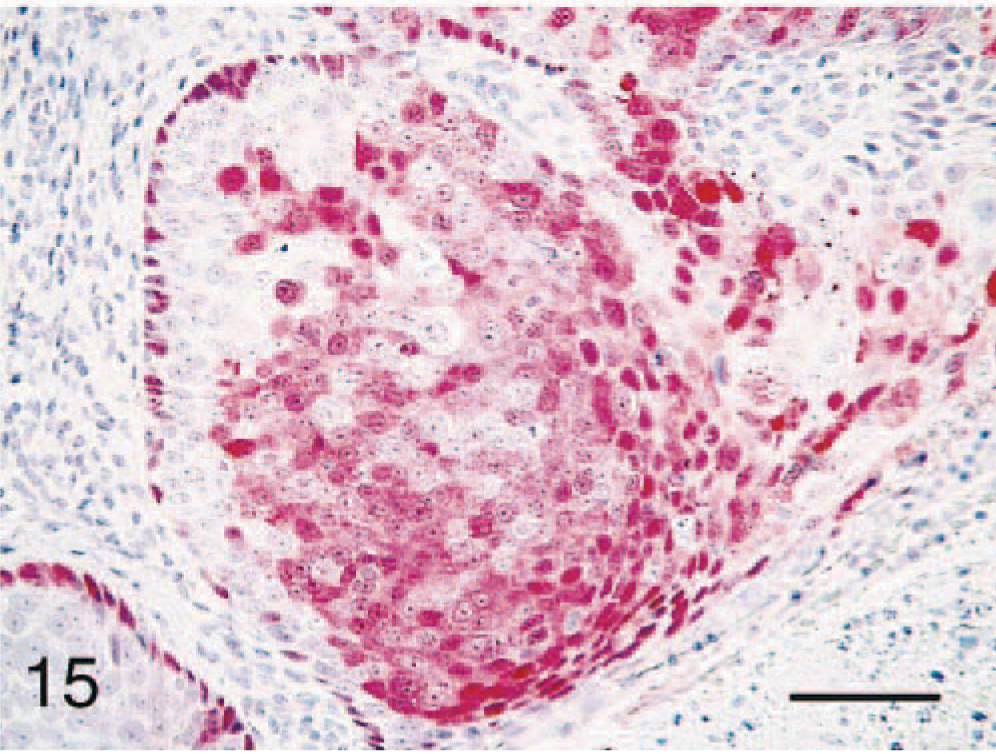
Cervical intraepithelial neoplasm (CIN); cynomolgus macaque, animal No. 5866. Positive immunostaining of CIN 2 lesion (adjacent to a cervical carcinoma) using polyclonal antibody to bovine papillomavirus type 1 (antibody C). Bar = 40 µm.
A recent study of genital intraepithelial neoplasms in sea lions found an association with an Epstein-Barr–related gammaherpesvirus but not papillomavirus. 17 Accordingly, we used immunohistochemistry to detect the presence of Epstein Barr–like virus proteins. Immunostaining was present in positive control tissue but not in any of the CIN-VIN cases (zero of 20) (Table 2).
Polymerase chain reaction
PCR amplification of DNA extracted from slides produced DNA products between 200 and 300 bp for nine of 16 cases and one of three negative controls. Several attempts to sequence these products for verification of product identity were unsuccessful, perhaps because of insufficient amounts of product or the degeneracy of the primers, or both. Because of these technical limitations, the PCR results do not conclusively confirm the presence of papillomavirus genetic material in the assessed lesions.
Discussion
Papillomaviruses comprise a diverse group of small epitheliotropic double-stranded DNA viruses, best known for causing benign spontaneously regressing papillomas on skin and mucosal surfaces. Recently, certain types of HPVs have been confirmed as causal agents of progressive cervical neoplasia. 3 In this report, we describe a range of CIN and VIN in aged cynomolgus macaques that are similar morphologically to those observed in women. Individual cases also included a range of dysplastic lesions, and it is speculated that this could suggest a progression toward malignancy. The observed lesions exhibited well-described morphologic characteristics of genital HPV infections such as koilocytosis, epithelial atypia, and loss of basal cell maturation. In addition, all higher-grade lesions involved the squamocolumnar junction, the predominant target site for oncogenic HPVs.
Papillomavirus protein was identified by immunohistochemistry in all lesions from these macaques. Each lesion stained selectively with at least one of three commercial papillomavirus antibodies tested although different specificity patterns were observed among lesions and among antibodies (Table 2). Interlesional staining variation (with the same antibody) suggests either the presence of different types of macaque papillomaviruses or changing patterns of viral protein expression relative to duration of infection, immunologic status, and hormonal influences on keratinocyte maturation. The presence of vaginal papillomas in five cases is suggestive of potential coinfection with different macaque papillomavirus types.
Despite the prevalence of cervical carcinoma in women, few suitable animal models currently exist for the study of oncogenic papillomaviruses. Widely used models such as cottontail rabbit papillomavirus and canine oral papillomavirus differ from oncogenic HPVs in terms of genetic organization, life cycle dynamics, genital mucosal tropism, and persistence, 23 limiting use of these systems for therapeutic and vaccine development. Even among genital HPVs, specific genotypic variants of the E6 and E7 genes are required for transformation and immortalization of primary genital keratinocytes in culture, 18 reflective of the exacting conditions required for genital HPV carcinogenesis. In this study, cervicovaginal neoplasms were observed in approximately one of every 20 animals examined from at least four distinct populations, suggesting that papillomavirus-associated genital dysplasias are a common lesion in older adult female cynomolgus macaques. In this case, cynomolgus macaques may offer a valuable model for the study of cervical cancer in women.
Footnotes
Acknowledgements
We thank Gerald Perry, Kendra Finley, Jean Gardin, and Diana Swaim for technical support. We also thank Dr. Jim Cappellari for his help in evaluating lesions and Dr. Sherry Klumpp for providing positive control lymphoma tissue. Support for molecular diagnostics was provided in part by the Comprehensive Cancer Center of Wake Forest University.
