Abstract
This report describes a spontaneous prostate fibromyxoid sarcoma with smooth muscle differentiation in an approximately 136-week-old intact male F344xBNF1 rat on a diet study for 2 weeks. At necropsy, the prostate was markedly distorted and enlarged by a firm white multinodular mass (6.0 × 4.5 × 3.5 cm). Histopathologically, the mass consisted of solid sheets of interlacing mesenchymal spindle cells with indistinct cell borders. Nuclei were separated by variable amounts of hyaline to fibrillar eosinophilic and/or myxomatous material. The extracellular myxomatous material tended to form whorls and stained positively with alcian blue. The mass stained strongly with Masson trichrome and vimentin throughout. Approximately 5% of the neoplastic cells were positive for smooth muscle actin, and none stained for desmin and pancytokeratin. To the authors’ knowledge, this fibromyxoid sarcoma with smooth muscle differentiation is the first such described prostatic sarcoma in a rat.
Adenocarcinomas of the prostate are one of the most commonly diagnosed cancers of adult men and are second only to lung cancer in cancer deaths. 9 Although dogs are the only companion animal species that develop spontaneous prostate cancer, 10 certain strains of laboratory rats, including ACI/Seg and Lobund-Wistar, develop spontaneous prostate tumors with some frequencies. 10,16 Reports indicate that approximately 70% of intact male ACI/Seg rats and 10% of intact male Lobund-Wistar rats over 20 months of age will develop carcinomas in the ventral prostate and in the dorsolateral prostate and seminal vesicle, respectively. 16 In contrast, the incidence of prostate carcinomas in aged F344 rats is approximately 4%. 16 While men with prostate cancer most frequently have adenocarcinomas, various sarcomas of the prostate have also been described, as well as nonmalignant stromal proliferations and prostatic stromal proliferations of uncertain malignant potential (PSPUMPs) . 2,4 –8,11 –13,15,17 To our knowledge, the only prostatic neoplasms that have been described in rats are adenocarcinomas; no stromal tumors or stromal hyperplasia have been described to date. Therefore, the case presented herein is the first case of a prostate sarcoma in a rat.
An intact F344xBNF1 rat (Harlan Laboratories, Inc., Indianapolis, IN) was acquired at 128 weeks of age as part of a group for a diet study. Animals came from colonies certified free of Hantaan virus, Kilham rat virus, lymphocytic choriomeningitis virus, mouse adenoviruses 1 and 2, pneumonia virus of mice, rat minute virus, rat parvovirus, rat Theiler virus, respiratory enteric virus III, Sendai virus, sialodacryoadenitis virus, Toolan H-1 parvovirus, cilia-associated respiratory bacillus, Citrobacter rodentium, Clostridium piliforme, Corynebacterium kutscheri, Helicobacter spp., Mycoplasma pulmonis, Pasteurella pneumotropica, Salmonella spp., Streptobacillus moniliformis, Streptococcus pnemoniae, as well as all dermatophytes, endoparasites, and ectoparasites. Before arrival, the rat was not subjected to any experimental procedures or used for breeding. Upon arrival, the rat was supplied ad libitum standard pelleted rodent chow (8640 Teklad 22/5, Harlan Laboratories) and individually housed in a Tecniplast individually ventilated cage, with autowaterer and corncob bedding, in the University Laboratory Animal Resources vivarium at The Ohio State University, which is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The rat was maintained on a 12:12 light:dark cycle and cared for in accordance with the Guide for the Care and Use of Laboratory Animals. All experimental procedures received approval from the Institutional Animal Care and Use Committee.
Six weeks after arrival, the rat was fed purified AIN-93M diet (5801-M, TestDiet, Richmond, IN) for 1 week, followed by an AIN-93M diet containing 9.35 g/kg of calcium β-hydroxy β-methyl butyrate monohydrate and 6.88 g/kg of curcumin until euthanasia. Rat feed weights were recorded daily. Although the rat was fed ad libitum, its food intake was approximately 3.8% to 38.4% that of the other rats, and its weight decreased from 524 g to 480 g over the course of 7 days (8.4% decrease in body weight). Three days before the planned study endpoint, the rat had moderate paresis of its hind limbs. At this time, the rat was bright, alert, and responsive, with a body condition score of 3 of 5. The following day, the rat exhibited an additional clinical sign of chromodacryorrhea, leading to the decision to euthanize immediately.
Following euthanasia via carbon dioxide asphyxiation, a complete necropsy was performed. The prostate contained a large, white, firm, multinodular mass measuring 6.0 × 4.5 × 3.5 cm (Figs. 1, 2). Distinct lobes of the prostate could not be identified due to obliteration by the mass. The dorsal surface of the mass contained a large area of hemorrhage. The seminal vesicles were red-purple with diffuse loss of lobulations. The urinary bladder was red-purple and enlarged, measuring 3.0 × 2.0 × 1.8 cm. The prostatic mass compressed the left ureter, which was also enlarged (hydroureter). There was a raised red lesion (0.5 × 0.5 × 0.2 cm) on the left kidney. All tissues were fixed in 10% neutral buffered formalin with the exception of the sternum, stifle, and vertebral column, which were fixed in Shandon TBD-2 Decalcifier (Thermo Fisher Scientific, Inc., Waltham, MA) for 72 hours. All tissues were processed by routine methods and embedded in paraffin wax, and 5-μm-thick sections were stained with hematoxylin and eosin. Histochemical and immunohistochemical staining was performed to better characterize the cells within the prostate tumor. These histochemical and immunohistochemical stains included alcian blue, Masson trichrome, vimentin (clone V9, catalog M0725, monoclonal mouse anti-rat, primary antibody dilution 1:100, secondary antibody dilution 1:500, Dako, Carpinteria, CA), smooth muscle actin (SMA; clone 1A4, catalog M0851, monoclonal mouse anti-human, primary antibody dilution 1:11000, secondary antibody dilution 1:500, Dako), desmin (catalog ab15200, polyclonal rabbit anti-human, primary antibody dilution 1:1000, secondary antibody dilution 1:1000, Abcam, Cambridge, MA), pancytokeratin (clone AE1/AE3, catalog M3515, monoclonal mouse anti-human, primary antibody dilution 1:100, secondary antibody dilution 1:500, Dako), and CD34 (clone C-18, catalog sc-7045, goat polyclonal anti-human, primary antibody dilution 1:75, secondary antibody dilution 1:200, Santa Cruz Biotechnology, Inc., Santa Cruz, CA). All immunohistochemistry staining protocols utilized the avidin–biotin complex method with DakoCytomation Target Retrieval Solution in a Decloaking Chamber (Biocare Medical) and the application of a protein block (DakoCytomation Serum-Free Protein Block).
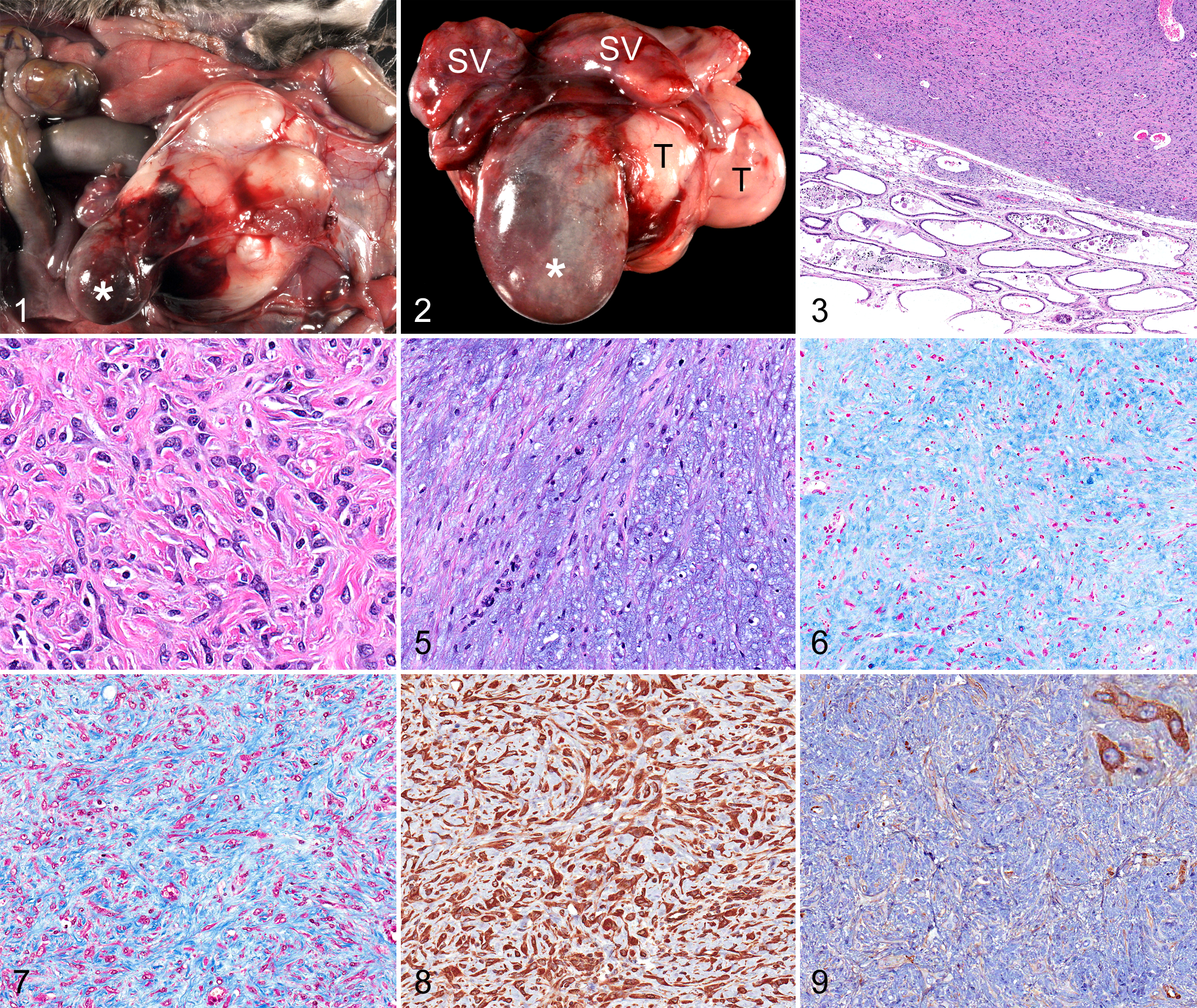
Microscopic examination of the hematoxylin and eosin–stained sections of the prostate revealed a partially encapsulated and well-circumscribed hypercellular expansile mass that obliterated almost all the normal parenchyma and extended into the adjacent seminal vesicles (Fig. 3). This mass was composed of sheets, fascicles, and bundles of interlacing mesenchymal spindle cells with indistinct cell borders. No glandular elements or distinct epithelial cells were present within the mass. The spindle cells contained oval to stellate, heterochromatic to euchromatic nuclei with occasional prominent nucleoli and eosinophilic cytoplasm (Fig. 4). Neoplastic cells were separated by variable amounts of extracellular hyaline to fibrillar eosinophilic and/or myxomatous material (Figs. 4, 5). The myxomatous material tended to form whorls and stained positively with alcian blue (Fig. 6). The majority of the matrix stained strongly with Masson trichrome (Fig. 7). Neoplastic cells throughout the mass were immunoreactive for vimentin (Fig. 8), whereas only 5% of cells were immunoreactive for SMA (Fig. 9) and all neoplastic cells were immunonegative for desmin and cytokeratin (not shown). CD34 immunoreactivity was restricted to endothelial cells in stromal blood vessels (not shown). Mild cytologic atypia characterized by anisocytosis and anisokaryosis was present, but mitotic figures were not. Few mast cells and lymphocytes were scattered throughout the mass. Much of the residual prostatic parenchyma had undergone coagulation necrosis and was infiltrated by neutrophils. Gram-positive coccobacilli were apparent within these areas of severe chronic necrosuppurative prostatitis.
In addition to identifying the lesions within the prostate, microscopic examination of other tissues revealed lesions within the urogenital tract that were presumed to be secondary to outflow obstruction from the prostatic mass, with subsequent urine stasis, bacterial infection, necrosis, and inflammation. There was unilateral, acute, marked, multifocal, necrosuppurative pyelonephritis and tubulonephritis with intralesional coccobacilli, intravascular fibrin thrombi, and mild hydronephrosis. Likewise, there was chronic, widespread, marked, transmural, necrosuppurative cystitis with many intralesional bacilli. The seminal vesicles showed chronic, multifocal, ulcerative, and suppurative seminal vesiculitis. Additional findings included lesions commonly seen in geriatric rats, such as moderate multifocal myocardial fibrosis of the left ventricular apex and papillary muscle, bilateral moderate widespread testicular degeneration and atrophy, and widespread mild unilateral thyroid C-cell hyperplasia. 1,14
At necropsy, a blood sample was collected via percutaneous cardiac puncture. A complete blood count was performed on a portion of EDTA anticoagulated whole blood (FORCYTE Autosampler 10, Oxford Science, Inc., Oxford, CT), and a biochemical profile was performed on serum (VetACE, Alfa Wasserman, West Caldwell, NJ). The hemogram was characterized by mild to moderate leukopenia at 1.5 K/μl (normal, 2.9–20.9 K/μl) with marked lymphopenia of 0.34 K/μl (normal, 3.8–15.3 K/μl), interpreted as a stress leukogram. Although the erythrocyte count was normal, the hematocrit and mean corpuscular volume were mildly decreased at 29.7% and 44.1 fl, respectively (normal, 34%–53% and 50–77.8 fl, respectively), while the mean corpuscular hemoglobin concentration was minimally increased at 36.7 g/dl (normal, 28.2–34.1 g/dl). The rat was also moderately thrombocytopenic, with 449 K/μl of platelets (normal, 685–1436 K/μl). Alkaline phosphatase was mildly to moderately elevated at 104.1 U/liter (normal, 15–45 U/liter), as was aspartate aminotransferase at 124.7 U/liter (normal, 10–46 U/liter), which was attributed to muscle wasting and the paresis. Albumin was mildly to moderately decreased at 2.2 g/dl (normal, 3.8–4.8 g/dl). The rat was moderately to markedly azotemic, with blood urea nitrogen markedly increased at 189 mg/dl (normal, 9–30 mg/dl) and creatinine moderately elevated at 3.7 mg/dl (normal, 0.5–2.2 mg/dl). Glucose was moderately decreased at 67 mg/dl (normal, 150–421 mg/dl). Phosphorous and potassium were both moderately elevated at 13.8 mg/dl and 7.0 mmol/l, respectively (normal, 4.2–8.5 mg/dl and 4.3–5.8 mmol/l, respectively). The moderate to marked azotemia and moderate hyperphosphatemia and hyperkalemia were consistent with acute renal failure.
Based on the histologic appearance, neoplastic cellular immunoreactivity (immunoreactive for vimentin throughout and multifocally immunoreactive for SMA), and histochemical characteristics (Masson trichrome positive throughout, multifocally alcian blue positive), this tumor is most consistent with a prostatic fibromyxoid sarcoma exhibiting smooth muscle differentiation. This neoplasm has not been described in the veterinary literature. The histologic appearance and clinical behavior of this tumor most closely resembles that of an atypical fibromyxoid tumor of the prostate described in a 1984 case report in an adult man. 4 In that case report, the neoplastic cells were spindle, demonstrated nuclear atypia and anisocytosis, and resided in a myxoid stroma. No mitotic figures were observed, and the cells were diffusely positive for Masson trichrome. This tumor was described as benign with potential for locally aggressive behavior and invasion. 4 Likewise, the tumor in the rat had areas where the stroma was myxoid, there were no mitotic figures, and some atypia was observed. Behaviorally, the rat prostate tumor was similar to the atypical fibromyxoid tumor in that it was locally invasive but not metastatic.
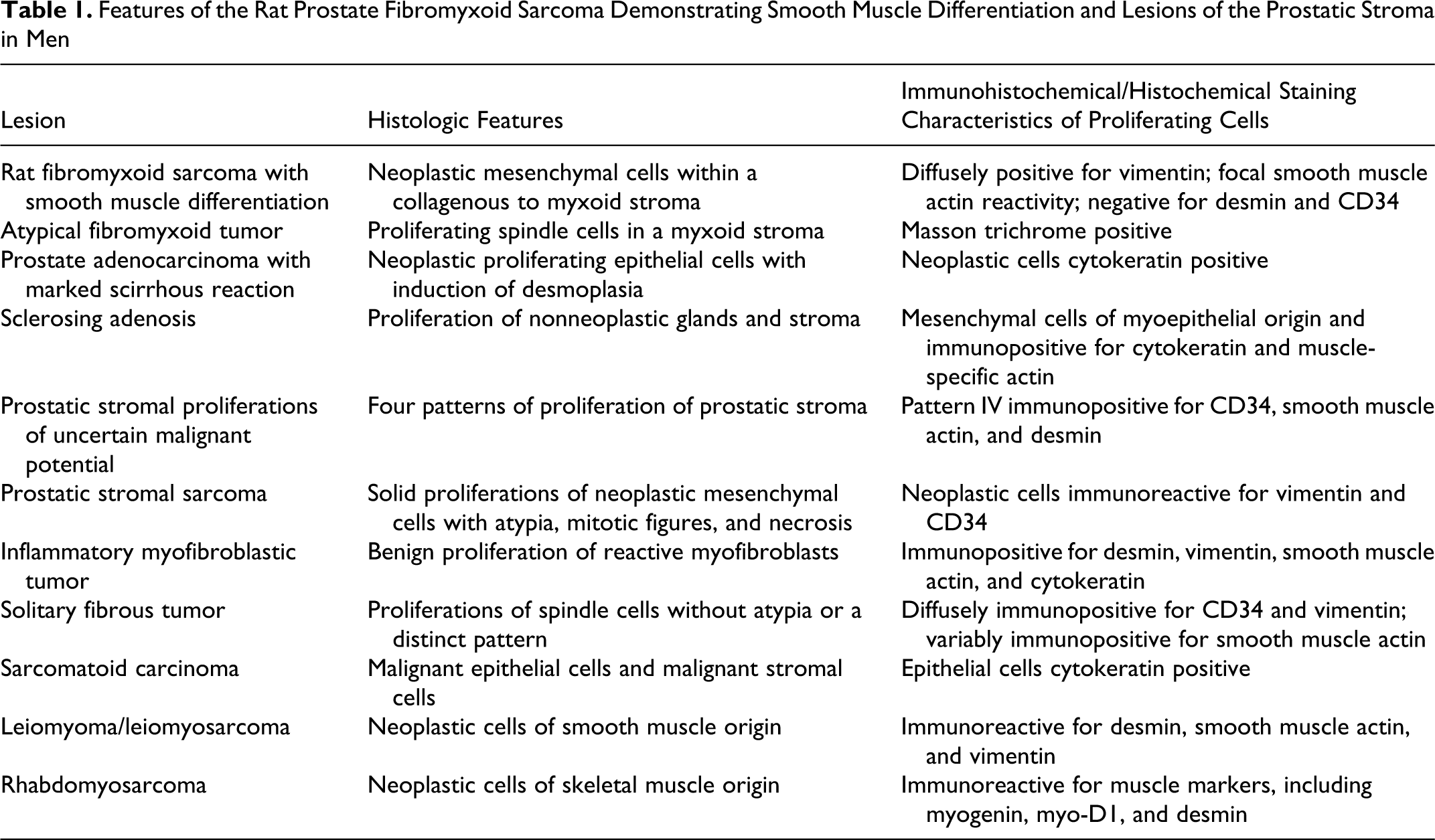
Although the most common prostate tumors in men are adenocarcinomas, a review of the available literature on human prostate pathology reveals a number of other tumors and prostate stromal proliferations that have been described. A review of these conditions provides us with a comprehensive list of differential diagnoses for the tumor we described in this rat (Table 1), including prostate carcinoma with a marked scirrhous reaction, sclerosing adenosis, PSPUMP, prostatic stromal sarcoma, inflammatory myofibroblastic tumor (IMT), solitary fibrous tumor (SFT), sarcomatoid carcinoma, leiomyoma, leiomyosarcoma, and rhabdomyosarcoma.
Features of the Rat Prostate Fibromyxoid Sarcoma Demonstrating Smooth Muscle Differentiation and Lesions of the Prostatic Stroma in Men
Given that all of the previously described reports of prostate tumors in rats to date have been carcinomas, the possibility must be considered that this tumor is in actuality a carcinoma that has induced a marked scirrhous reaction that obscures the epithelial component. However, as previously stated, none of the cells within the tumor were immunoreactive for cytokeratin. If this tumor were a carcinoma with marked desmoplasia, the sections examined would contain at least 1 neoplastic epithelial cell identifiable by its immunoreactivity. Given that none of the tumor cells within any of the sections of prostate were immunoreactive for cytokeratin, we can conclude that this tumor is not a carcinoma with marked scirrhous reaction. Likewise, the absence of identifiable neoplastic epithelial cells allows one to rule out the possibility that this tumor represents a carcinoma with portions undergoing epithelial-to-mesenchymal transition, a phenomenon that has been described in men with prostate cancer. 3
Sclerosing adenosis is a nonneoplastic proliferation of the glands and stroma of the human prostate, observed in approximately 2% of biopsy specimens. Sclerosing adenosis may be mistaken for carcinoma, since epithelial cells within the proliferative glands may exhibit cell crowding and cytologic atypia. However, because these glands retain distinct basal cells, they are nonneoplastic, and the lesion is considered proliferative only. 5,11 Although the stroma of a sclerosing adenosis lesion may be myxoid with mucin deposition similar to our case, the mesenchymal cells are nonneoplastic, and cytologic atypia is rarely seen. These mesenchymal cells are believed to be derived from myoepithelial cells and are frequently immunoreactive for high molecular weight cytokeratin and muscle-specific actin. 5,11 This is in contrast to the neoplastic cells within the rat prostate tumor, all of which were immunonegative for cytokeratin, with only a small percentage demonstrating positivity for SMA. Additionally, in almost all cases of sclerosing adenosis, glandular elements are present. 5,11 Our rat prostate tumor contained no identifiable glandular elements or epithelial cells.
PSPUMPs are rare proliferations of the prostatic stroma in men. 2,5,7,15 These stromal proliferations are described as having uncertain malignant potential because they may recur after surgical excision and may progress to sarcomas. 2,5,7 Further information regarding the malignant potential of these lesions is lacking because of their rarity and the dearth of long-term case follow-up. 7 Also called stromal tumors of uncertain malignant potential, 5,7 4 patterns of PSPUMPs have been described in men. These patterns differ as to whether glands of nonmalignant prostate epithelial cells are present within the lesion (patterns I, II, and III), what appearance the glands have (normal in patterns I and II compared with compressed and proliferative in pattern III), and whether the proliferative stromal cells exhibit any cytologic atypia (patterns I and III). 2,5,7,15 With its expansile growth, lack of glandular elements, and absence of mitotic figures, the lesion within the prostate of the rat is most similar to PSPUMP pattern IV. However, cytologic atypia is reportedly absent in PSUMP pattern IV, whereas we describe mild cytologic atypia in the rat prostate lesion. Additionally, the immunohistochemical profile of the lesion in the rat does not match that of PSUMP pattern IV. The proliferating stromal cells in PSPUMP pattern IV are reported to be immunoreactive (at least focally) for CD34 in all the cases described, immunoreactive for SMA in approximately 60% of cases, and immunoreactive for desmin in 80% of cases. 2,15 This is in contrast to the rat prostate tumor in which the neoplastic cells were immunonegative for CD34 and desmin and only a small percentage of cells within the myxomatous areas were immunoreactive for SMA.
A prostate stromal sarcoma is another rare lesion that has been described in the human prostate. Prostate stromal sarcomas are reportedly composed of solid proliferations of mesenchymal cells forming fascicles or bundles. These neoplastic mesenchymal cells may entrap nonneoplastic glands. 2,5,7,15 Occasionally, the stroma may be focally myxoid. 7 Prostate stromal sarcomas are differentiated from PSPUMPs based on the presence of cytologic atypia, frequent mitotic figures, and necrosis. 2,5,7,15 The neoplastic cells are immunoreactive for vimentin and CD34. Occasionally, cells may be immunopositive for the progesterone receptor. The neoplastic cells are reportedly immunonegative for SMA, desmin, and S100. 2,5,15 Although the rat prostate tumor was immunoreactive for vimentin and immunonegative for desmin and multifocally contained myxoid stroma, the neoplastic cells were immunonegative for CD34 with evidence of SMA immunoreactivity, and no mitotic figures were visualized.
IMTs are benign proliferations of reactive myofibroblasts and may occur in the prostate or bladder. 5 IMTs have been described as postoperative spindle cell nodules found in prostates that have undergone surgical manipulation and as pseudosarcomatous fibromyxoid tumors found in prostates with no history of surgical manipulation. 5,15 IMTs are most often small, measuring less than 1 cm, and contain a relatively monomorphic population of reactive myofibroblasts that lack atypia. Admixed with the myofibroblasts are variable numbers of inflammatory cells. Although the mitotic index of these lesions varies widely, 5,15 from approximately 1 to 25 mitotic figures in 10 high-powered fields, 15 no abnormal mitoses are apparent. 5,15 The proliferating cells within the lesion are strongly immunoreactive for vimentin and SMA 15 and are commonly immunoreactive for desmin and pancytokeratin. 5 Although the proliferating cells within the rat prostate tumor were strongly positive for vimentin, only a small percentage of cells were immunoreactive for SMA, and none stained for cytokeratin and desmin. In addition, no mitotic activity was noted.
SFTs are uncommon tumors in the prostate of men, 5,12,15 with only approximately 32 cases reported in the literature. 5 SFTs are tumors that are behaviorally benign and arise on serosal surfaces, most commonly the visceral pleurae. However, extraserosal locations have been reported, including the urinary bladder, kidney, prostate gland, and spermatic cord. These tumors are composed of spindle cells without atypia that do not exhibit a distinct pattern (“patternless pattern”). The histology of some tumors may multifocally resemble that of a hemangiopericytoma and may contain areas of myofibroblastic differentiation. The proliferating spindle cells are usually diffusely immunopositive for CD34 and vimentin, may be variably immunoreactive for SMA, and are usually immunonegative for pancytokeratin and S-100. 5,12,15 Although the proliferating spindle cells described in the rat tumor showed minimal atypia and were diffusely positive for vimentin, the rat tumor is not likely to be a SFT, because the neoplastic cells were immunonegative for CD34. Additionally, there was no gross or histologic evidence that the tumor in the rat was arising from the adventitial surface of the prostate.
Sarcomatoid carcinomas are rare tumors of the prostate in men and are composed of malignant epithelial cells forming glands and malignant stromal cells. Within these tumors, the epithelial component typically resembles that of an acinar adenocarcinoma, although these cells may alternatively resemble a more rare form of prostate carcinoma, such as small cell or adenosquamous carcinoma. The mesenchymal component may make up approximately 5% to 99% of the mass; these cells typically exhibit nuclear atypia, frequent mitotic figures, and regions of necrosis and may form giant cells. 5,17 However, in approximately 10% of cases, the mesenchymal portion of the neoplasm exhibits only mild atypia. Furthermore, the mesenchymal component may show differentiation toward an osteosarcoma, chondrosarcoma, or rhabdomyosarcoma in approximately 30% of cases. 5 Although the rat prostate tumor is a potential example of a sarcomatoid carcinoma with minimal atypia in the mesenchymal component, there was no evidence of neoplastic epithelial cells within the tumor.
Tumors originating from muscle, either smooth (leiomyoma and leiomyosarcoma) or skeletal (embryonal rhabdomyosarcoma), are described to occur rarely in the human prostate. 5,15 Leiomyosarcomas are differentiated from leiomyomas based on the presence of atypia, mitotic figures, necrosis, local invasion, and/or increased cellularity in the former. Spindle cells composing these smooth muscle tumors show a typical profile of immunoreactivity, with cells being immunopositive for desmin, vimentin, and SMA. 5,15 Rhabdomyosarcomas of the human prostate are most frequently described in children, although they may also occur in adults. In children, these tumors most commonly exhibit the embryonal pattern 5,13,15 and exhibit immunoreactivity against myogenin 5,13 and myo-D1, as well as other muscle immunohistochemical markers, such as desmin. 5 As stated previously, the tumor in the rat prostate was immunonegative for desmin.
The rat prostate tumor presented in this report represents the first nonepithelial tumor of the rat prostate that we are aware of in the veterinary literature. As discussed, various nonneoplastic and neoplastic lesions of the prostatic stroma have been described in men, with an atypical fibromyxoid tumor most closely resembling the tumor described in this rat. We hope that this report and the review of comparative lesions that occur in the prostate stroma of men have served to inform readers about the variety of lesions, in addition to adenocarcinomas, that can occur in the aged prostate.
Footnotes
Acknowledgements
We thank members of the Comparative Pathology and Mouse Phenotyping Shared Resource for assistance with the necropsy and histology slide preparation, as well as Tim Vojt for image processing.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
